Abstract
Tree diversity may buffer the negative impact of drought events according to the diversity insurance hypothesis. During the extreme pan-European drought of 2018, we tested whether tree species richness modulated drought impacts on communities of a young tree diversity experiment in Freiburg, Germany. We utilized drone-based hyperspectral images to capture early symptoms of drought stress indicated by variation in Normalized Difference Vegetation Index (NDVI), and ground-based measures that are indicative of tree canopy function, including Plant Area Index (PAI) and leaf chlorophyll content (Chl). Measures were taken the fifth growing season after planting on six broadleaved and six conifer species originating from Europe and North America in monocultures, two-, four- and six-species mixtures. NDVI decreased successively in response to the summer drought. In contrast to our expectation, tree species richness did not reduce declines in NDVI at the plot level. NDVI values were generally lower in six-species mixtures, compared to monocultures or two-species mixtures, which may be indicative of antagonistic tree species interactions in mixtures of high diversity. Changes in PAI in response to the drought were similar to changes in NDVI; however, modeled differences in PAI between mixtures were not statistically significant. Chl initially decreased, but recovered within a period of two months following the drought. Remotely sensed NDVI facilitated monitoring of the drought response of our large field experiment and could aid in monitoring canopy health in response to extreme drought events. Our observations suggest that NDVI responses are likely more strongly related to leaf shedding within tree canopies than decreases in Chl. Tree stands with a high abundance of deciduous trees may therefore be especially sensitive in triggering NDVI changes in response to drought during the stages leading up to tree mortality. Future studies of drought-related canopy responses should include monitoring of leaf shedding as well as the Chl of shed leaves.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Extreme drought events have increased in frequency and severity in recent years, with large-scale impact on forests around the globe (Shukla et al. 2019). During the extreme summer drought that affected Central Europe in 2018, significantly elevated tree damage, reduced primary production, and partial canopy die-back became obvious in many regions (Buras et al. 2018; Brun et al. 2020; Schuldt et al. 2020). The impacts of severe drought on forest canopies, e.g., individual death and die-back, can thereby fundamentally affect various forest functions and ecosystem services, such as forest community and ecosystem dynamics (Anderegg et al. 2013), biodiversity (Maron et al. 2015), ecohydrology (Adams et al. 2012), and feedbacks to the global earth system (Bonan 2008; Adams et al. 2010). However, although it is posited that an increased tree diversity in forest stands could positively affect the resistance to several environmental disturbances, such as extreme drought (Jactel & Brockerhoff 2007; Guyot et al. 2016; Jactel et al. 2021), evidence to date remains mixed (Grossiord et al. 2014).
Recent impacts of severe drought on European forests have highlighted the importance of understanding the linkages between tree diversity, community composition, and forest drought resistance. Forests’ species composition is expected to change dramatically due to large increases in tree mortality triggered by recent extreme drought events (Schuldt et al. 2020; Hajek et al. 2022). In order to re-establish forest cover on drought-affected sites and facilitate drought-adapted forests, species composition and species selection is crucial. Understanding the potential benefits or drawbacks of species richness in order to predict the consequences of climatic changes on forest ecosystems is an emerging task and one of the grand challenges for forest ecologists and managers.
Increasing forest tree diversity may buffer the negative impact of drought events according to the diversity insurance hypothesis (Yachi & Loreau 1999). Yet the effects of altered species composition on forest ecosystems in response to global warming is still debated (Grossiord 2019). A number of observational studies have examined if more species rich or structurally diverse forests exhibit higher resistance to drought (Lloret et al. 2007; Grossiord et al. 2014; Forrester et al. 2016), but evidence from experimental studies is scarce (Sousa-Silva et al. 2018; Schnabel et al. 2021). The diversity of functional traits, specifically physiological traits related to water (hydraulic) transport, play an essential role mediating ecosystem resilience to drought (Anderegg et al. 2018). Biodiversity ecosystem functioning (BEF) experiments manipulating tree diversity facilitate detailed investigations of drought impacts on various species composition in young stands (Grossman et al. 2018; Hajek et al. 2022). The controlled mixing design allows for the investigation of drought sensitivity including functional traits of co-occurring species exhibiting contrasting water-use strategies, and thus, if mixing increases forest resistance to drought.
Monitoring of forest ecosystem responses to drought is often achieved by collection of empirical data using in situ ground-based methods which can be used to track forest drought response at a variety of scales, from the leaf, to the canopy (West et al. 2019). Canopy properties monitored often include leaf area index, or the closely related measure Plant Area Index (PAI; which includes the contribution of both leaf area and stems) because of its sensitivity to drought stress and close link to productivity. PAI declines across planted sites as water stress increases (Battaglia et al. 1998), declines in response to experimentally imposed water stress (c. 20% LAI reduction, Asner et al. 2004), and is observed to decline in response to growing season drought (Jump et al. 2017). At the leaf scale, drought can have transient effects on photosynthesis by causing stomatal closure. More lasting effects on leaf function resulting from water stress, such as a reduction in leaf chlorophyll content (Chl), may be useful for describing forest responses to water stress (Allen et al. 2010; Zarco-Tejada et al. 2019).
To date, few studies have quantified how PAI and Chl vary across experimental gradients in tree diversity and composition, and still fewer, how diversity may modulate the effects of strong droughts on the canopy (Grossman et al. 2018; Pardos et al. 2021). Methods for estimating PAI include direct methods such as destructive harvest (Breda 2003), line-intercept methods (Nock et al. 2008), as well as indirect methods based on predicting PAI from the amount of light transmitted through the canopy (Breda 2003). Of these ground-based methods, estimation of PAI from measurements of light passing through the canopy is popular as it is fairly rapid, although can become challenging when attempting to capture temporal variation in PAI as well as how this varies across gradients in forest conditions because of the large number of measurements that need to be made directly by the researcher in situ.
Multispectral indices have been widely applied in drought monitoring, including the Normalized Difference Vegetation Index (NDVI; Tucker 1979; Orth et al. 2016, West et al. 2019). Near canopy remote sensing using Remotely Piloted Aircraft Systems (RPAS; “drones”) represents a promising approach to collecting remotely sensed data on the response of plants to spatial and temporal variations in environmental conditions. In recent decades, lightweight multi- and hyperspectral sensors have been developed specifically as RPAS payloads (Zhong et al. 2018). RPAS systems with sensor payloads have a great potential to assist in monitoring field experiments with trees, because the scale of tree diversity experiments (i.e., size of the experimental unit or plot size) matches well with the resolution of the sensor, and the option that researchers can repeat sampling campaigns at desired intervals presents a significant benefit. However, to date, studies correlating remote-sensed data with physiological parameters connected to in situ measurements are lacking (Marusig et al. 2020).
In this study, we combined remotely sensed and ground-based data on tree canopy status in response to drought along experimentally controlled gradients of tree diversity. The experiment, established in 2013 using saplings belonging to twelve tree species (six deciduous and six conifer species from Europe and North America), was designed to test relationships between tree diversity and ecosystem function by manipulating tree species richness and functional diversity. During summer 2018, the site was exposed to record dry and hot conditions, which can be regarded as a global-climate-change-type drought (Breshears et al. 2005). Mean growing season temperature, precipitation as well as hydroclimate pattern such as mean vapor pressure deficit and climatic water balance (CWB; precipitation potential evapotranspiration) were indicative for a severe drought in this region (Buras et al. 2020; Schuldt et al. 2020). This setting resulted in a unique opportunity to monitor drought responses in various temperate forest tree species mixtures, and to provide evidence for the severe impact of the 2018 drought on young tree stands. The field-based experimental setting allows to test the diversity insurance hypothesis (Yachi and Loreau 1999) and the utility of remote-sensed Normalized Difference Vegetation Index (NDVI) recordings for monitoring forest canopy responses to drought.
The NDVI was thereby compared to ground-based measures of canopy status, such as the Plant Area Index (PAI) and leaf chlorophyll content (Chl). Thereby, the following hypotheses were tested:
-
1.
Plot-level NDVI measures decline in response to the drought event and—if so—the effect is weaker at higher levels of tree species richness (SR).
-
2.
Similar patterns should be observed in ground-based measures of canopy status. Both, PAI and leaf chlorophyll should decline in response to the drought event, which may be buffered in stands of high tree species richness.
Materials and methods
Study site
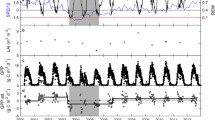
The study was carried out at the Freiburg experimental site of the International Diversity Experiment Network with Trees (IDENT) and is part of the global Tree Diversity Network “TreeDivNet” which shares the common aim of exploring relationships between tree species diversity and ecosystem functioning (Paquette et al. 2018; Verheyen et al. 2016). The site is located in Freiburg in southwestern Germany (48°01′10″N/7°49′37″E) at an elevation of about 240 m above sea level. IDENT-Freiburg was installed on shallow sandy-loamy cambisols (40 cm) with a high gravel content subsoil (below 100 cm). The climate in the region is classified as temperate oceanic (Cfb Köppen-Geiger climate classification) and Freiburg ranges among the warmest cities in Germany with a mean annual temperature of 11.57 °C and a mean annual precipitation of 881.4 mm (averages from 1991 to 2019) (Data available from https://www.dwd.de, station 1443—Freiburg). All months are humid, and the maximum precipitation is recorded during the summer months (May: 94.8 mm–August: 81.95 mm). The maximum temperatures are recorded in July and August (mean temperature 20.5 °C); the coldest month is January (mean temperature 2.9 °C). During the summer of 2018, the IDENT-Freiburg experiment was subject to a prolonged drought and heatwave (Fig. 1a, b). The temperatures between June and August were 0.96 °C above the long-term mean (19.98 °C) and precipitation between June and August was 55% below normal levels (264.5 mm vs. 118.2 mm) (DWD-CDC 2021). The extremely high air temperatures, e.g. 27.4 °C on August 4th and 7th 2018, coincided with anomalously low precipitation during much of the growing season (Fig. 1a, b) and caused low relative air humidity, especially during July and August (Fig. 1c). We measured average plot-level soil moisture content (%) with an HH2 moisture meter (HH2, DeltaT, Burwell, Cambridge, UK) at 10 cm soil depth at three dates during the summer season, using the mean of two measurements along the short distances toward the central tree of each plot. Measures revealed a decrease in soil moisture levels from the early growing season in June to the peak growing seasons in July and showed an increase in October, coinciding with higher recorded precipitation levels (Fig. 1d).
Climatic condition at the IDENT-Freiburg site. Mean growing season air temperature (MGT) (a) and mean growing season precipitation (MGP) (b) are shown for the normal period 1991–2020. The horizontal gray lines in (a, b) represent the data for the drought year 2018. Relative air humidity (RH) (c) is shown for the growing season (April–October) 2018 (DWD-CDC 2021). Vertical lines indicate our observation periods, defined along the 2018 growing season (pre-drought (PreDr), during peak drought in July 2018 (Dr), in September 2018 (PostDr I) and October 2018 (PostDr II). The volumetric soil moisture content (d) was recorded in 10 cm soil depth, error bars represent one standard error
Experimental design
For our study, we selected 196 plots at the IDENT site in Freiburg along a species richness (SR) gradient including monocultures (SR 1) of twelve species (six exotic species originating from North America and six native species from Europe, Table 1), and additionally diverse cultures with combinations of two- (SR 2), four- (SR 4), or six-species (SR 6) mixtures.
The experiment as a whole includes two sub-experiments, one with manipulation of species richness and functional diversity with tree species originating from Europe and North America (SR = 1, 2, 6), and a second with a manipulation of species richness and functional diversity using only European species (SR = 1, 2, 4). Four-species mixtures only included European-, but no North American tree species (Table 2).
Further details on the experiment are provided in Wein et al. (2016). In this study, all unfertilized mixtures were measured, which then included a slight imbalance of EU vs. NA tree species overall (No NA at SR = 4); however, analyses did not indicate that this unbalance caused any confounding with species richness effects in our study (see statistical analyses below). The realized proportion of deciduous vs. coniferous trees was balanced along the diversity gradient (Wein et al. 2016). The overall mortality in early 2018 was 3.38% and not systematically related to the experimental treatments but differed among species.
All trees in the experiment were planted in 2013 in a factorial randomized block design with four blocks (1.13 ha in total) containing 415 nested plots of 13 m2 in total. Within each plot, seven rows and seven columns (total of 49 trees) of trees were planted into a square plot with a planting distance of 0.45 m which is utilized to foster interactions among individuals. It is important to note that the resulting low light regime due to this dense planting strategy led to closed canopy-cover and the absence of an herb layer within the plots. Within each plot, an even tree species abundance was realized, whereby multiple species were planted, but their relative proportions were maintained equal. A surrounding buffer zone of 0.9 m is separating the neighboring plots (Wein et al. 2016). In 2018, field measurements were carried out during three sampling campaigns. To account for temporal discrepancies in dates of recording remote and ground-based data, we defined the main observation periods along the 2018 growing season, representing the time during the drought event in July (Dr), and two post-drought periods, i.e., the time shortly after drought in September (PostDr I) and later after the drought in October (PostDr II).
Normalized Difference Vegetation Index (NDVI)
We monitored the seasonal leaf dynamics (changes in greening, foliage cover or defoliation) on the 196 plots selected across the IDENT-Freiburg experiment as one means of assessing the vegetation response to the 2018 drought and heatwave (aerial image, Fig. 2).
Hyperspectral images were taken of the IDENT site during three remote sensing campaigns, carried out on August 7, September 20, and October 10, 2018 using a drone (Octo8XL, Mikrokopter—HiSystems GmbH, Moormerland, Germany) flown at approximately 38 m above the ground and equipped with an optical hyperspectral sensor (UHD185, Cubert GmbH, Ulm, Germany). Overlap for the images was greater than 90% forward and side lap. To standardize measurements of canopy reflectance, flights were conducted as close to solar noon as possible and only on days with clear skies. In addition, before and after each flight, measurements of a white reference were carried out using a Spectralon panel (Spectralon, SphereOptics, Herrsching am Ammersee, Germany). We also collected reflectance measurements of the spectralon panel during the flights to verify that sky conditions remained stable and visual inspection of the data indicated this was the case. Utilizing the sequence of images, an orthomosaic of the whole experiment for each of the sampling dates was created using Agisoft Metashape (Metashape, Agisoft LLC, St. Petersburg, Russia). Spectra were extracted from the hyperspectral orthomosaic for the central area of the plot (excluding the outermost row of trees; 0.5 m). The Normalized Difference Vegetation Index (NDVI) was then calculated and then averaged per plot (Tarpley et al. 1984) using the reflectance of red light (ρred) at 625 nm and the reflectance of near infrared light (ρNIR) at 802 nm (Eq. 1)
Plant Area Index (PAI) and chlorophyll measurements
We also used ground-based assessments to quantify drought effects on tree canopy health. We estimated the Plant Area Index (PAI, total surface area, m2 m−2) from diffuse radiation transmittance measured at the plot level using two LAI-2000 plant canopy analyzers (Li-Cor, Inc., Lincoln, NE). LAI-2000 measurements were carried out between August 6th and November 12th 2018 in the selected 196 plots and on three occasions corresponding to the NDVI measurement in August, September, and October. The PAI measurements require some time to complete for the 1568 samples (eight measures × 196 plots), so were collected within a week of NDVI measurements (except September which was two weeks). In each plot, we sampled eight positions (diffuse radiation below canopy; DRb) at ~ 10 cm above the ground. Measurements were positioned around the tree closest to plot center and in between this central tree and the eight nearest neighboring trees surrounding it. Corresponding diffuse radiation above canopy measurements (DRa) are used by the LAI-2000 software to calculate PAI for each DRb measurement. To collect the set of DRa measurements, data were logged at 15 s intervals at 4.5 m above the ground at an open field next to the site using a second identical LAI-2000 instrument. The data were processed to calculate PAI from DRa and DRb measurements using the FV2200 software (version 2.1.1, LI-COR Biosciences Inc., Lincoln, NE, USA). The DRb measurements were paired with the DRa measurements by matching records closest in time. The outermost view angle of the five optical sensor rings (central zenith angle 68°) was excluded to avoid interference of adjacent plots. Values for the eight measurements were averaged to represent one integrated plot-level PAI value.
Leaf chlorophyll content (Chl, mg m leaf−2) was measured on three dates (July 31, September 12, and October 3, 2018) on three fully developed leaves, or needles, that were selected randomly from the upper third canopy of one tree individual of each species, growing in the inner 5 × 5 tree rows of each plot. We used a portable fluorescent ratio chlorophyll content meter (CCM-300, Opti-Sciences, Hudson, NH, USA). The average of all individual Chl measurements per plot was used as average plot-level chlorophyll content.
Quantification of tree species richness effects
We used the proportional deviation index “D” (Hector et al. 2002) to compare observed and expected NDVI values across plots varying in diversity, calculated as follows:
where O is the observed NDVI of species mixtures and E is the expected NDVI of the corresponding monocultures or mixtures.
Expected NDVI values were based on weighted average plot-level NDVI values that were calculated based on the average of the NDVI within monocultures (mean of four plots) of the respective species at the same measurement date, weighted by the proportion of each species represented within a mixed culture, see analogous calculations of “Dweight” in Palmborg et al. (2005). The D index reflects a net diversity effect, where positive D values indicate positive diversity effects, i.e., that mixtures have a higher NDVI value than expected based on the performance of the corresponding monocultures; negative D values indicate lower performance in mixtures than in monocultures, e.g., due to negative species interactions.
Statistical analysis
All statistical analyses were performed with the software R, R version 3.6.0 (2019-04-26)—“Planting of a Tree” (R Core Team 2019). We first assessed whether the design-related confounding between tree species origin (North America vs. Europe) and the different species richness levels (SR 4 with only European species) could cause any bias in terms of species richness effects in our study. A priori to all major analyses, we therefore fitted fixed effects models in R, using the lm function. We included the categorical variables species origin (North America vs. Europe), plot-level tree species richness (SR, with four levels: monoculture, SR 2, SR 4 and SR 6), and their two-way interaction as fixed factors in the model. ANOVA tests indicated that species origin did not have any significant impact on NDVI, PAI or chlorophyll measures (Supplementary material Table S1). We therefore used the entire dataset (including plots with North American species) in the following analyses.
We tested for differences in NDVI in relation to the time periods when we recorded data during peak drought in July 2018 (Dr), in September 2018 (PostDr I), and October 2018 (PostDr II) of the vegetation period. We used the lme function of the nlme package to fit linear mixed effects models (Pinheiro et al. 2021). As NDVI was not normally distributed we transformed it using an Order-Norm transformation prior to modeling. The time period was set as a three-level fixed factor (Dr, PostDr I, and PostDr II). We additionally included the plot-level tree species richness as a fixed factor with four levels (SR 1, SR 2, SR 4, SR 6). According to our hypotheses, we also included the two-way interaction between time period and tree species richness. To account for the spatial hierarchy of the experimental design, we defined the experimental block and plot as nested random effects. To test whether the two-way interaction of time period and species richness explained a significant proportion in the models, we used the ANOVA command comparing models including the interaction or not with an analysis of deviance (Zuur et al. 2009). ANOVA tests showed that the two-way interaction did not explain a significant proportion of variance in NDVI (based on a 5% Type I error rate), hence we reduced the model to include the time period and tree species richness as additive fixed effects. The difference between the two models in deviance was ~ 6.95 following a Chi-square distribution with 6 degrees of freedom (ΔAIC = 5, SR*time term P value = 0.325). We followed the exact same modeling approach to model changes in PAI and chlorophyll, in relation to time period and tree species richness. Similar to results for NDVI, none of the two-way interactions were statistically significant, so we reduced the models to only include additive fixed effects. The difference in deviance was 4.32 following a Chi-square distribution with 6 degrees of freedom for the PAI model, and 0.49 with 2 degrees of freedom for the chlorophyll model (PAI: ΔAIC = 7.7, P value = 0.633, Chlorophyll: ΔAIC = 3, P value = 0.781).
All final models fulfilled model assumptions in terms of homoscedasticity and normality of residuals, which we checked visually (Figure S1a–c). For all three response variables, we assessed ANOVA outputs (F Tests) to test the statistical significance of time period and tree species richness. We additionally extracted and visualized the estimated marginal means from all models, including their 95% confidence intervals, as a measure of uncertainty. Pairwise comparisons between estimated means were generated using the emmeans function in R (package emmeans), which implements the Tukey method to adjust p values for multiple comparisons, while assessing differences between individual factor levels (Lenth et al. 2019).
Results
Changes in canopy status related to drought and tree species richness
Remote-sensed NDVI values gradually declined (P < 0.0001; Table 3) from initial values recorded during the drought (beginning of August 2018), and reached their minimum at the period PostDr II (mid October 2018, Fig. 3a) and (Figure S2). Since the interactions of time period and the species richness level were not statistically significant, the species richness effects were generalized across all time stages. The NDVI values differed significantly between the species richness levels (SR) of the studied plots (P < 0.03; Table 3). Post hoc comparisons showed that six-species mixtures had significantly lower NDVI in general, as compared to monocultures and two-species mixtures (Fig. 3b).
Estimated marginal means of NDVI (a, b), PAI (c, d) and leaf chlorophyll (e, f). Estimated means, including their 95% confidence intervals, are presented for different times in relation to drought 2018: peak drought in July (Dr), post-drought shortly after drought in September (PostDr I) and later after the drought in October (PostDr II). As there were no significant interactions between time in relation to drought and plot-level tree species richness (SR), the SR effects are separately presented in the right figure panel. Different lower case letters indicate statistically significant differences among group means, based on Tukey post hoc comparisons (P < 0.05)
Ground-based measures of monocultures showed variation among tree species in patterns of PAI throughout the growing season (Figure S3). The ground-based measured PAI decreased significantly over time in relation to the defined phases of drought (Dr, PostDr I and PostDr II) during the growing season 2018 (P < 0.0001, Table 3). The highest PAI values were recorded during the drought event in July 2018. As of August 2018, the PAI decreased significantly PostDr I and PostDr II (Fig. 3c). Similar to the NDVI signal, the interaction of time period and the species richness level was absent. However, differences between levels of tree diversity for the PAI could not be observed (Table 3, Fig. 3d).
The Chl levels differed significantly over time (P < 0.0001, Table 3) and reached their minimum PostDr I in September. Leaf Chl increased again significantly in October 2018, PostDr II (Fig. 3e) and (Figure S4). There were no significant differences in chlorophyll content between stands of different tree species richness (Table 3, Fig. 3d).
Proportional deviation index (D), significantly decreased in relation to plot-level tree species richness (SR) (R2 = 0.41, Fig. 4). For communities of either four species or six species, antagonistic effects on NDVI were observed, indicated by D values below zero. In 2018, many tree species featured early leaf coloration and senescence, among Betula and Larix. The relationship between plot-level tree species richness and NDVI were explored further by examining the relationship between the proportion of the most drought-affected species Betula and Larix in a plot and NDVI (Fig. 5). As the fraction of Betula and Larix species increased in a plot, ranging from total absence in mixed cultures to 100 percent in respective monocultures, the plot-level NDVI decreased from 0.86 to 0.76. The relationship between species fraction and NDVI was supported by an R2 = 0.72.
Proportional deviation index (D), in relation to plot-level tree species richness (SR). D values below zero indicate antagonistic species effects on NDVI. The regression model is based on linear mixed effects models, including block and plot as nested, random effects. The shadow behind the regression line represents its 95% CI
Discussion
We initially expected that negative drought responses in Normalized Difference Vegetation Index (NDVI), Plant Area Index (PAI), and chlorophyll would be buffered in species mixtures of high diversity following the diversity-stability hypothesis (Tilman et al. 2006; Isbell et al. 2015). However, drought responses at the plot level were independent of tree species richness at our site even though canopy NDVI varied significantly with drought and was also significantly negatively related with species richness.
Drought-induced canopy changes
The NDVI decline reflected a substantial response in tree canopies triggered by the 2018 drought at IDENT-Freiburg (Fig. 3) and represented a reliable indicator of drought-induced defoliation (Camarero et al. 2015). Among the twelve investigated tree species, we observed that early leaf loss occurred primarily in Betula spp. which are broadleaved pioneer species, and also Larix (Figure S2). In contrast, Acer species as well as Quercus rubra were mostly unaffected by drought. We assume that a direct response of both Betula species may indicate a higher sensitivity to water deficit, and that they reach their physiological limits earlier than the late successional species, e.g., Acer, Fagus or Quercus (Meyer et al. 2020). This is an example of plants mitigating drought stress by “sacrificing” their terminal organs. Crown defoliation varied considerably across species which matches species-specific patterns of drought response observed in other studies (Michel and Seidling 2016). Our observations follow the pattern along a gradient of decreasing hydraulic safety margins (HSM) as determined for the respective species (Hajek et al. 2022). Those are reflecting the mechanisms of xylem embolism resistance of the species investigated. Betula, and also Larix, displays an isohydric strategy according to their stringency of stomatal limitation to transpiration in drying soils. In contrast, Quercus and Acer species are characterized by anisohydric regulation, allowing leaf water potential to drop when the soil or the air are dryer.
The peak defoliation with a maximal reduction in the photosynthetic active surface was reached after three months of drought exposure and coincides with the trend observed in other drought-affected sites in Central Europe (Rohner et al. 2021; Walthert et al. 2021). The mismatch between fluctuations in water deficit and temperature and the slower lagged canopy responses of trees can be attributed to the ability of trees to buffer their water status from seasonal variations in water availability through mechanisms such as deep rooting, and the capacity of individual trees to accommodate short-term changes in water stress without losing their leaves (Jump et al. 2017). Although it is widely recognized that leaf coverage status (PAI) is sensitive to climate variables (Iio et al. 2014), the long-term lags in tree drought recovery and their significant implications for forest ecosystems remain uncertain (Kannenberg et al. 2020). The significant decrease in PAI during the main growing season of 2018 also mirrors NDVI patterns at our site. We therefore assume that the NDVI trend not only reflects the process of leaf loss in response to the 2018 drought event, but also includes variability due to changes in Chl at our site. Further measurements of drought stress-related physiological parameters (e.g., vegetation water content) would allow calibrating remote-sensed observations accordingly (Konings et al. 2021). As a limitation to this approach, it is important to note that the relationship between Plant Area Index (PAI) and NDVI saturates above a threshold around a PAI of six (Gamon et al. 1995), which was not reached in our experiment. However, consequently, NDVI may exhibit limited sensitivity to changes in canopy structure when PAI values are higher. Additional indices such as the photochemical reflectance index (PRI) should be considered in those cases (Mänd et al. 2010).
Parallel to the decline in NDVI, the leaf chlorophyll content also decreased significantly during the months of drought, as supported by the NDVI anomalies observed by Buras et al. (2020). However, after reaching a minimum, the leaf chlorophyll content increased again during the end of the vegetation period, likely resulting from regrowth of leaves or needles observed for some species subsequent to a precipitation event in September. The rain event was indicated by the peak in relative air humidity (Fig. 1c) and corresponds well with the declining soil moisture content which reached a minimum of 5% in July and later increased up to 8.5% (Fig. 1d). This short-term tree response induced by a precipitation event enabling some species to regrow previously shed leaves and partly recover their photosynthetic capacity. The fluctuations in NDVI throughout the drought also follow the trend of decreasing chlorophyll levels, suggesting that foliar pigments are being tracked by NDVI (Wu et al. 2010), as expected.
Tree diversity effects on canopy response to drought
Tree diversity at the plot level had a significant negative effect on NDVI, with the lowest levels in the most diverse plots (SR 6), also reflected by the decreasingly negative D values with higher tree species richness. Tree diversity was also observed to negatively affect PAI, although the effect was not statistically significant (Table 3). These results suggest that more diverse tree communities were not more resistant to the effects of the drought, contrasting results from syntheses on grassland responses to drought (Isbell et al. 2015). The mechanism resulting in higher drought resistance and resilience of diverse grassland communities is described by the insurance hypothesis (Yachi & Loreau 1999) stating that if under certain conditions, e.g., drought, the failure of the whole community is less likely with increasing number of species. As observed by Tilmann et al. (2006), the species’ abundances and their contribution to biomass productivity and stability varied with environmental changes in their long-term grassland experiment. This mechanism cannot be easily applied to woody ecosystems subject to a severe drought, like in our experiment, for two reasons. Firstly, failure in trees is in most species more final, meaning death of the individual, and recovery under favorable conditions is impossible. And second, because the turnover of individuals is much slower. Hence, the temporal and spatial scales of any insurance mechanisms would be much larger in woody systems.
Mechanisms like resource complementarity or positive species interactions among co-occurring tree species, like those often described for diversity effects on biomass production or resource use (e.g., complementarity or facilitation, Scherer-Lorenzen 2014), are more likely to buffer resource shortages in woody species, because they can become stronger with longer times of interactions (Sousa-Silva et al. 2018, Juncker et al. 2020). However, it is not clear yet, whether they can be applied to any resource, stress level and ecosystem processes such as leaf stress responses. Vice versa, they can even contribute to the observed sensitivity to drought via the positive diversity effect on biomass production or canopy packing that has been reported in other tree diversity studies (e.g., Jucker et al. 2020; Shovon et al. 2022). If such effects were already present in high diversity plots in our experiment, we would have expected the additional biomass and leaf area, to reduce soil water availability faster and increase the risk of stress during a drought event. From our observations in a young tree experiment, we can conclude that niche partitioning or facilitation processes regulating water accessibility among the interacting species were not expressed at the early tree growth status.
On the other hand, the proportion of drought-tolerant species within a tree community can explain NDVI anomalies during a drought episode, as argued by Lloret et al. (2007). They observed lower NDVI values with higher number of species during a severe drought in 2003 resulting from a selection effect. Given the similar proportion of coniferous and broadleaved species along the diversity gradient at our site, we assume that the proportion of trees from intensively drought-affected species, specifically Larix species (L. laricina and L. decidua) and Betula (B. papyrifera and B. pendula), contribute disproportionately to low NDVI in plots of high tree diversity. Indeed, we observed a decrease in NDVI with increasing fraction of the most drought-affected species Betula and Larix at the plot level (Fig. 5). This is in accordance with the observations of De Cáceres et al. (2021) that drought stress primarily manifests in size-asymmetric competition for water induced by differences in key functional traits of the interacting species (Forrester 2019), and is also a strong indicator of the importance of the influence of particular mean trait values in influencing community-wide responses to drought (Anderegg et al. 2018). Indeed, Grossiord et al. (2014) and Jactel et al. (2021) also suggested that managing forest ecosystems for high tree species diversity (simply by increasing the number of species) does not necessarily assure improved resistance to the more severe and frequent drought events predicted for the future. Instead, intermixing trees of different levels of susceptibility to a broad range of hazards may reduce the risk of total failure.
Conclusion
Remote-sensed NDVI, as well as ground-based PAI and leaf chlorophyll content, showed significant drought responses at the IDENT tree diversity experiment, Freiburg. The monitoring of NDVI thereby constitutes a reliable tool for monitoring canopy health responses in relation to drought. Contrary to our expectation, high tree species richness did not buffer drought effects on canopy status. This can be partly explained by selection effects. Simply increasing the number of tree species may not be beneficial for all ecosystem services provided by tree plantations, and species should be selected based on their functional roles and based on the main management purposes. However, since we did not observe any significant effects of tree species richness on biomass or canopy packing in our study, further research is needed to determine if this potential mechanism played a role in our findings. Future studies could also cover mature forests on a landscape scale, for example by extending measurements to permanent monitoring sites. These assessments are timely, and essential to calibrate drought model estimates which are specifically relevant to predict long-term ecosystem responses to drought experiencing natural stress conditions.
References
Adams HD, Macalady AK, Breshears DD, Allen CD, Stephenson NL, Saleska SR, Huxman TE, McDowell N (2010) Climate-induced tree mortality: earth system consequences. EOS Trans Am Geophys Union 91:153–154. https://doi.org/10.1029/2010EO170003
Adams HD, Luce CH, Breshears DD, Allen CD, Weiler M, Hale VC, Smith AMS, Huxman TE (2012) Ecohydrological consequences of drought- and infestation-triggered tree die-off: insights and hypotheses. Ecohydrology 5:145–159. https://doi.org/10.1002/eco.233
Allen CD, Macalady AK, Chenchouni H, Bachelet D, McDowell N, Vennetier M, Kitzberger T, Rigling A, Breshears DD, Hogg ET (2010) A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For Ecol Manag 259:660–684. https://doi.org/10.1016/j.foreco.2009.09.001
Anderegg WRL, Kane JM, Anderegg LDL (2013) Consequences of widespread tree mortality triggered by drought and temperature stress. Nat Clim Change 3:30–36. https://doi.org/10.1038/nclimate1635
Anderegg WRL, Konings AG, Trugman AT, Yu K, Bowling DR, Gabbitas R, Karp DS, Pacala S, Sperry JS, Sulman BN, Zenes N (2018) Hydraulic diversity of forests regulates ecosystem resilience during drought. Nature 561:538–541. https://doi.org/10.1038/s41586-018-0539-7
Asner GP, Nepstad D, Cardinot G, Ray D (2004) Drought stress and carbon uptake in an Amazon forest measured with spaceborne imaging spectroscopy. Proc Natl Acad Sci 101:6039–6044. https://doi.org/10.1073/pnas.0400168101
Battaglia M, Cherry ML, Beadle CL, Sands PJ, Hingston A (1998) Prediction of leaf area index in eucalypt plantations: effects of water stress and temperature. Tree Physiol 18:521–528. https://doi.org/10.1093/treephys/18.8-9.521
Bonan GB (2008) Forests and climate change: forcings, feedbacks, and the climate benefits of forests. Science 320:1444–1449. https://doi.org/10.1126/science.1155121
Bréda NJ (2003) Ground-based measurements of leaf area index: a review of methods, instruments and current controversies. J Exp Bot 54:2403–2417. https://doi.org/10.1093/jxb/erg263
Breshears DD, Cobb NS, Rich PM, Price KP, Allen CD, Balice RG, Romme WH, Kastens JH, Floyd ML, Belnap J, Anderson JJ, Myers OB, Meyer CW (2005) Regional vegetation die-off in response to global-change-type drought. Proc Natl Acad Sci 102:15144–15148. https://doi.org/10.1073/pnas.0505734102
Brun P, Psomas A, Ginzler C, Thuiller W, Zappa M, Zimmermann NE (2020) Large-scale early-wilting response of Central European forests to the 2018 extreme drought. Glob Change Biol 26:7021–7035. https://doi.org/10.1111/gcb.15360
Buras A, Schunk C, Zeiträg C, Herrmann C, Kaiser L, Lemme H, Straub C, Taeger S, Gößwein S, Klemmt H-J, Menzel A (2018) Are Scots pine forest edges particularly prone to drought-induced mortality? Environ Res Lett 13:025001. https://doi.org/10.1088/1748-9326/aaa0b4
Buras A, Rammig A, Zang CS (2020) Quantifying impacts of the 2018 drought on European ecosystems in comparison to 2003. Biogeosciences 17:1655–1672. https://doi.org/10.5194/bg-17-1655-2020
Buras A, Rammig A, Zang CS (2021) The European Forest Condition Monitor: using remotely sensed forest greenness to identify hot spots of forest decline. Front Plant Sci 12:689220. https://doi.org/10.3389/fpls.2021.689220
Camarero JJ, Franquesa M, Sangüesa-Barreda G (2015) Timing of drought triggers distinct growth responses in holm oak: implications to predict warming-induced forest defoliation and growth decline. Forests 6:1576–1597. https://doi.org/10.3390/f6051576
De Cáceres M, Mencuccini M, Martin-St.Paul N, Limousin J-M, Coll L, Poyatos R, Cabon A, Granda V, Forner A, Valladares F (2021) Unravelling the effect of species mixing on water use and drought stress in Mediterranean forests: A modelling approach. Agric for Meteorol 296:108233. https://doi.org/10.1016/j.agrformet.2020.108233
DWD-CDC (2021) Daily station observations precipitation height in mm, mean temperature at 2 m above ground in °C and mean of relative humidity at 2 m above ground in % at the Freiburg climate station (data-source: Station 1443—Freiburg)
Forrester DI (2019) Linking forest growth with stand structure: Tree size inequality, tree growth or resource partitioning and the asymmetry of competition. For Ecol Manag 447:139–157. https://doi.org/10.1016/j.foreco.2019.05.053
Forrester DI, Bonal D, Dawud S, Gessler A, Granier A, Pollastrini M, Grossiord C (2016) Drought responses by individual tree species are not often correlated with tree species diversity in European forests. J Appl Ecol 53:1725–1734. https://doi.org/10.1111/1365-2664.12745
Gamon JA, Field CB, Goulden ML, Griffin KL, Hartley AE, Joel G, Penuelas J, Valentini R (1995) Relationships between NDVI, canopy structure, and photosynthesis in three Californian vegetation types. Ecol Appl 5:28–41. https://doi.org/10.2307/1942049
Grossiord C (2019) Having the right neighbors: how tree species diversity modulates drought impacts on forests. New Phytol 228:42–49. https://doi.org/10.1111/nph.15667
Grossiord C, Granier A, Ratcliffe S, Bouriaud O, Bruelheide H, Chećko E, Forrester DI, Dawud SM, Finér L, Pollastrini M, Scherer-Lorenzen M, Valladares F, Bonal D, Gessler A (2014) Tree diversity does not always improve resistance of forest ecosystems to drought. Proc Natl Acad Sci 111:14812–14815. https://doi.org/10.1073/pnas.1411970111
Grossman JJ, Vanhellemont M, Barsoum N, Bauhus J, Bruelheide H, Castagneyrol B, Cavender-Bares J, Eisenhauer N, Ferlian O, Gravel D, Hector A, Jactel H, Kreft H, Mereu S, Messier C, Muys B, Nock C, Paquette A, Parker J, Perring MP, Ponette Q, Reich PB, Schuldt A, Staab M, Weih M, Zemp DC, Scherer-Lorenzen M, Verheyen K (2018) Synthesis and future research directions linking tree diversity to growth, survival, and damage in a global network of tree diversity experiments. Environ Exp Bot 152:68–89. https://doi.org/10.1016/j.envexpbot.2017.12.015
Guyot V, Castagneyrol B, Vialatte A, Deconchat M, Jactel H (2016) Tree diversity reduces pest damage in mature forests across Europe. Biol Let 12:20151037. https://doi.org/10.1098/rsbl.2015.1037
Hajek P, Link RM, Nock CA, Bauhus J, Gebauer T, Gessler A, Kovach K, Messier C, Paquette A, Saurer M, Scherer-Lorenzen M, Rose L, Schuldt B (2022) Mutually inclusive mechanisms of drought-induced tree mortality. Glob Change Biol 28:3365–3378. https://doi.org/10.1111/gcb.16146
Hector A, Loreau M, Schmid B, Beierkuhnlein C, Caldeira MC, Diemer M, Dimitrakopoulos PG, Finn JA, Freitas H, Giller PS (2002) Biodiversity manipulation experiments: studies replicated at multiple sites. Biodiversity and ecosystem functioning: synthesis and perspectives. Oxford University Press, Oxford, pp 36–46
Iio A, Hikosaka K, Anten NPR, Nakagawa Y, Ito A (2014) Global dependence of field-observed leaf area index in woody species on climate: a systematic review. Glob Ecol Biogeogr 23:274–285. https://doi.org/10.1111/geb.12133
Isbell F, Craven D, Connolly J, Loreau M, Schmid B, Beierkuhnlein C, Bezemer TM, Bonin C, Bruelheide H, De Luca E (2015) Biodiversity increases the resistance of ecosystem productivity to climate extremes. Nature 526:574–577. https://doi.org/10.1038/nature15374
Jactel H, Brockerhoff EG (2007) Tree diversity reduces herbivory by forest insects. Ecol Lett 10:835–848. https://doi.org/10.1111/j.1461-0248.2007.01073.x
Jactel H, Moreira X, Castagneyrol B (2021) Tree diversity and forest resistance to insect pests: patterns, mechanisms, and prospects. Annu Rev Entomol 66:277–296. https://doi.org/10.1146/annurev-ento-041720-075234
Jucker T, Koricheva J, Finér L, Bouriaud O, Iacopetti G, Coomes DA (2020) Good things take time—diversity effects on tree growth shift from negative to positive during stand development in boreal forests. J Ecol 108:2198–2211. https://doi.org/10.1111/1365-2745.13464
Jump AS, Ruiz-Benito P, Greenwood S, Allen CD, Kitzberger T, Fensham R, Martínez-Vilalta J, Lloret F (2017) Structural overshoot of tree growth with climate variability and the global spectrum of drought-induced forest dieback. Glob Change Biol 23:3742–3757. https://doi.org/10.1111/gcb.13636
Kannenberg SA, Schwalm CR, Anderegg WRL (2020) Ghosts of the past: how drought legacy effects shape forest functioning and carbon cycling. Ecol Lett 23:891–901. https://doi.org/10.1111/ele.13485
Konings AG, Saatchi SS, Frankenberg C, Keller M, Leshyk V, Anderegg WR, Humphrey V, Matheny AM, Trugman A, Sack L (2021) Detecting forest response to droughts with global observations of vegetation water content. Glob Change Biol 27:6005–6024. https://doi.org/10.1111/gcb.15872
Lenth R, Singmann H, Love J, Buerkner P, Herve M (2019) Package ‘emmeans’
Lloret F, Lobo A, Estevan H, Maisongrande P, Vayreda J, Terradas J (2007) Woody plant richness and NDVI response to drought events in Catalonian (northeastern Spain) Forests. Ecology 88:2270–2279. https://doi.org/10.1890/06-1195.1
Mänd P, Hallik L, Peñuelas J, Nilson T, Duce P, Emmett BA, Beier C, Estiarte M, Garadnai J, Kalapos T, Schmidt IK, Kovács-Láng E, Prieto P, Tietema A, Westerveld JW, Kull O (2010) Responses of the reflectance indices PRI and NDVI to experimental warming and drought in European shrublands along a north–south climatic gradient. Remote Sens Environ 114:626–636. https://doi.org/10.1016/j.rse.2009.11.003
Maron M, McAlpine CA, Watson JE, Maxwell S, Barnard P (2015) Climate-induced resource bottlenecks exacerbate species vulnerability: a review. Divers Distrib 21:731–743. https://doi.org/10.1111/ddi.12339
Marusig D, Petruzzellis F, Tomasella M, Napolitano R, Altobelli A, Nardini A (2020) Correlation of field-measured and remotely sensed plant water status as a tool to monitor the risk of drought-induced forest decline. Forests 11:77. https://doi.org/10.3390/f11010077
Meyer BF, Buras A, Rammig A, Zang CS (2020) Higher susceptibility of beech to drought in comparison to oak. Dendrochronologia 64:125780. https://doi.org/10.1016/j.dendro.2020.125780
Michel A, Seidling W (2016) Forest condition in Europe: 2016 technical report of ICP forests: report under the UNECE convention on long-range transboundary air pollution (CLRTAP). https://doi.org/10.13140/RG.2.2.35983.38566
Nock CA, Caspersen JP, Thomas SC (2008) Large ontogenetic declines in intra-crown leaf area index in two temperate deciduous tree species. Ecology 89:744–753. https://doi.org/10.1890/07-0531.1
Orth R, Zscheischler J, Seneviratne SI (2016) Record dry summer in 2015 challenges precipitation projections in Central Europe. Sci Rep 6(1):1–8. https://doi.org/10.1038/srep28334
Palmborg C, Scherer-Lorenzen M, Jumpponen A, Carlsson G, Huss-Danell K, Högberg P (2005) Inorganic soil nitrogen under grassland plant communities of different species composition and diversity. Oikos 110:271–282. https://doi.org/10.1111/j.0030-1299.2005.13673.x
Paquette A, Hector A, Castagneyrol B, Vanhellemont M, Koricheva J, Scherer-Lorenzen M, Verheyen K (2018) A million and more trees for science. Nat Ecol Evol 2:763–766. https://doi.org/10.1038/s41559-018-0544-0
Pardos M, del Río M, Pretzsch H, Jactel H, Bielak K, Bravo F, Brazaitis G, Defossez E, Engel M, Godvod K, Jacobs K, Jansone L, Jansons A, Morin X, Nothdurft A, Oreti L, Ponette Q, Pach M, Riofrío J, Ruíz-Peinado R, Tomao A, Uhl E, Calama R (2021) The greater resilience of mixed forests to drought mainly depends on their composition: Analysis along a climate gradient across Europe. For Ecol Manag 481:118687. https://doi.org/10.1016/j.foreco.2020.118687
Pinheiro J, Bates D, DebRoy S, Sarkar D, Team RC (2021) nlme: linear and nonlinear mixed effects models. R package version 31-153. https://svn.r-project.org/R-packages/trunk/nlme/
R Core Team (2019) R: a language and environment for statistical computing (Version 3.6. 0)[Computer software]. Vienna, Austria: R Foundation for Statistical Computing, 2019. https://www.r-project.org/
Rohner B, Kumar S, Liechti K, Gessler A, Ferretti M (2021) Tree vitality indicators revealed a rapid response of beech forests to the 2018 drought. Ecol Ind 120:106903. https://doi.org/10.1016/j.ecolind.2020.106903
Scherer-Lorenzen M (2014) The functional role of biodiversity in the context of global change. In: Burslem D, Coomes D, Simonson W (eds) Forests and global change. Cambridge University Press, Cambridge, pp 195–238. https://doi.org/10.1017/CBO9781107323506.011
Schnabel F, Liu X, Kunz M, Barry KE, Bongers FJ, Bruelheide H, Fichtner A, Härdtle W, Li S, Pfaff C-T (2021) Species richness stabilizes productivity via asynchrony and drought-tolerance diversity in a large-scale tree biodiversity experiment. Sci Adv 7:abk1643. https://doi.org/10.1126/sciadv.abk1643
Schuldt B, Buras A, Arend M, Vitasse Y, Beierkuhnlein C, Damm A, Gharun M, Grams TEE, Hauck M, Hajek P, Hartmann H, Hiltbrunner E, Hoch G, Holloway-Phillips M, Körner C, Larysch E, Lübbe T, Nelson DB, Rammig A, Rigling A, Rose L, Ruehr NK, Schumann K, Weiser F, Werner C, Wohlgemuth T, Zang CS, Kahmen A (2020) A first assessment of the impact of the extreme 2018 summer drought on Central European forests. Basic Appl Ecol 45:86–103. https://doi.org/10.1016/j.baae.2020.04.003
Shovon TA, Kang S, Scherer-Lorenzen M, Nock CA (2022) Changes in the direction of the diversity–productivity relationship over 15 years of stand development in a planted temperate forest. J Ecol 110:1125–1137. https://doi.org/10.1111/1365-2745.13855
Shukla PR, Skea J, Calvo Buendia E, Masson-Delmotte V, Pörtner HO, Roberts DC, Zhai P, Slade R, Connors S, Van Diemen R (2019) IPCC, 2019: climate change and land: an IPCC special report on climate change, desertification, land degradation, sustainable land management, food security, and greenhouse gas fluxes in terrestrial ecosystems. https://www.ipcc.ch/srccl/download/
Sousa-Silva R, Verheyen K, Ponette Q, Bay E, Sioen G, Titeux H, de Peer TV, Meerbeek KV, Muys B (2018) Tree diversity mitigates defoliation after a drought-induced tipping point. Glob Change Biol 24:4304–4315. https://doi.org/10.1111/gcb.14326
Tarpley JD, Schneider SR, Money RL (1984) Global vegetation indices from the NOAA-7 meteorological satellite. J Appl Meteorol Climatol 23:491–494. https://doi.org/10.1175/1520-0450(1984)023%3c0491:GVIFTN%3e2.0.CO;2
Tilman D, Reich PB, Knops JMH (2006) Biodiversity and ecosystem stability in a decade-long grassland experiment. Nature 441:629–632. https://doi.org/10.1038/nature04742
Tucker CJ (1979) Red and photographic infrared linear combinations for monitoring vegetation. Remote Sens Environ 8:127–150. https://doi.org/10.1016/0034-4257(79)90013-0
Verheyen K, Vanhellemont M, Auge H, Baeten L, Baraloto C, Barsoum N et al (2016) Contributions of a global network of tree diversity experiments to sustainable forest plantations. Ambio 45:29–41. https://doi.org/10.1007/s13280-015-0685-1
Walthert L, Ganthaler A, Mayr S, Saurer M, Waldner P, Walser M, Zweifel R, von Arx G (2021) From the comfort zone to crown dieback: Sequence of physiological stress thresholds in mature European beech trees across progressive drought. Sci Total Environ 753:141792. https://doi.org/10.1016/j.scitotenv.2020.141792
Wein A, Bauhus J, Bilodeau-Gauthier S, Scherer-Lorenzen M, Nock C, Staab M (2016) Tree species richness promotes invertebrate herbivory on congeneric native and exotic tree saplings in a young diversity experiment. PLoS ONE 11:e0168751. https://doi.org/10.1371/journal.pone.0168751
West H, Quinn N, Horswell M (2019) Remote sensing for drought monitoring & impact assessment: Progress, past challenges and future opportunities. Remote Sens Environ 232:111291. https://doi.org/10.1016/j.rse.2019.111291
Wu C, Wang L, Niu Z, Gao S, Wu M (2010) Nondestructive estimation of canopy chlorophyll content using Hyperion and Landsat/TM images. Int J Remote Sens 31:2159–2167. https://doi.org/10.1080/01431161003614382
Yachi S, Loreau M (1999) Biodiversity and ecosystem productivity in a fluctuating environment: The insurance hypothesis. Proc Natl Acad Sci 96:1463. https://doi.org/10.1073/pnas.96.4.1463
Zarco-Tejada PJ, Hornero A, Beck PSA, Kattenborn T, Kempeneers P, Hernández-Clemente R (2019) Chlorophyll content estimation in an open-canopy conifer forest with Sentinel-2A and hyperspectral imagery in the context of forest decline. Remote Sens Environ 223:320–335. https://doi.org/10.1016/j.rse.2019.01.031
Zhong Y, Wang X, Xu Y, Wang S, Jia T, Hu X, Zhao J, Wei L, Zhang L (2018) Mini-UAV-borne hyperspectral remote sensing: from observation and processing to applications. IEEE Geosci Remote Sens Mag 6:46–62. https://doi.org/10.1109/MGRS.2018.2867592
Zuur AF, Ieno EN, Walker NJ, Saveliev AA, smith, G. M. (2009) Mixed effects models and extensions in ecology with R, vol 574. Springer, New York, p 574. https://doi.org/10.18637/jss.v032.b01
Acknowledgements
This research was funded by the German Research Foundation (DFG 384026712, DFG 316733524) and the establishment of the field site was supported by University of Freiburg (Innovationsfonds Forschung) in cooperation with Jürgen Bauhus. We acknowledge the support of Christian Messier and Alain Paquette and thank all student helpers and field assistants involved in this project for their valuable contributions to establish this dataset under extreme field conditions during the drought and heatwave 2018. We highly acknowledge the revisions of two anonymous reviewers on an earlier version of this manuscript which significantly improved our work.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
CAN, LR, and MS-L conceived the original idea. MM, KG, and KRK analyzed the data and prepared the figures with input from PH. PH performed ground-based measurements with support from LR and KRK contributed remote sensing data. PH, MM, and KRK wrote the first draft of the manuscript with support from CAN. All authors critically reviewed and approved the final version for submission.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by Marta Pardos.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hajek, P., Mörsdorf, M., Kovach, K.R. et al. Quantifying the influence of tree species richness on community drought resistance using drone-derived NDVI and ground-based measures of Plant Area Index and leaf chlorophyll in a young tree diversity experiment. Eur J Forest Res 143, 141–155 (2024). https://doi.org/10.1007/s10342-023-01615-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-023-01615-3