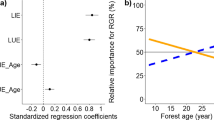
Abstract
Maximum rooting depth is a key functional trait to increase the fitness of trees and also influences terrestrial ecosystem processes. Despite its importance, the drivers of the interspecific variation of maximum rooting depth or its relation to other plant traits and plant age are not well understood. In this study, we aimed to clarify the drivers of the interspecific variation of maximum rooting depth with special reference to its relation to plant leaf traits and age. We analyzed how maximum rooting depth of single individuals of 227 tree species planted in the same common garden in the temperate region of central Japan is correlated to commonly measured leaf functional traits (specific leaf area (SLA), leaf dry matter content (LDMC), leaf nitrogen (N) concentration) extracted from the TRY database. We did this by employing the phylogenetic comparable method and included the age of all target trees. When excluding the effect of phylogenetic signals from the relationships between rooting depth and leaf traits, SLA was negatively correlated with maximum rooting depth in deciduous, but not evergreen species. Further, rooting depth and leaf N concentration were negatively correlated in evergreen trees, a pattern driven by young trees. These results implicate that the relationship between maximum rooting depth and leaf traits differed depending on the leaf habits and age of the tree species.




Similar content being viewed by others
Data availability
The data of rooting depth of the trees are available in Karizumi (2010) and the latest illustrations of tree roots in Seibundo Shinkosha Publishing. ISBN978-4–416-41,005–9. The data of the leaf functional traits are available in the following database: Kattge J, Bönisch G, Díaz S et al. (2020), TRY plant trait database—enhanced coverage and open access, Glob Change Biol 26:119–188. https://doi.org/10.1111/gcb.14904
References
Adams DC, Collyer ML (2018) Multivariate phylogenetic comparative methods: evaluations, comparisons, and recommendations. Syst Biol 67:14–31. https://doi.org/10.1093/sysbio/syx055
Alvarez-Clare S, Kitajima K (2007) Physical defence traits enhance seedling survival of neotropical tree species. Funct Ecol 21:1044–1054. https://doi.org/10.1111/j.1365-2435.2007.01320.x
Bartušková A, Lubbe FC, Qian J et al (2022) The effect of moisture, nutrients and disturbance on storage organ size and persistence in temperate herbs. Funct Ecol 36:314–325. https://doi.org/10.1111/1365-2435.13997
Burton JI, Perakis SS, Brooks JR, Puettmann KJ (2020) Trait integration and functional differentiation among co-existing plant species. Am J Bot 107:628–638. https://doi.org/10.1002/ajb2.1451
Canadell J, Jackson RB, Ehleringer JR et al (1996) Max rooting depth of veg types at global scale. Oecologia 108:538–595. https://doi.org/10.1007/s10705-016-9812-z
Casper BB, Jackson RB (1997) Plant competition underground. Annu Rev Ecol Syst 28:545–570. https://doi.org/10.1146/annurev.ecolsys.28.1.545
Chamberlain SA, Hovick SM, Dibble CJ et al (2012) Does phylogeny matter? Assessing the impact of phylogenetic information in ecological meta-analysis. Ecol Lett 15:627–636. https://doi.org/10.1111/j.1461-0248.2012.01776.x
Cohen D, Schwarz M (2017) Tree-root control of shallow landslides. Earth Surf Dyn 5:451–477. https://doi.org/10.5194/esurf-5-451-2017
Cornelissen JHC, Cerabolini B, Castro-Díez P et al (2003) Functional traits of woody plants: Correspondence of species rankings between field adults and laboratory-grown seedlings? J Veg Sci 14:311–322. https://doi.org/10.1111/j.1654-1103.2003.tb02157.x
Crozier MJ (2010) Deciphering the effect of climate change on landslide activity: a review. Geomorphology 124:260–267. https://doi.org/10.1016/j.geomorph.2010.04.009
de la Riva EG, Villar R, Pérez-Ramos IM et al (2018) Relationships between leaf mass per area and nutrient concentrations in 98 Mediterranean woody species are determined by phylogeny, habitat and leaf habit. Trees - Struct Funct 32:497–510. https://doi.org/10.1007/s00468-017-1646-z
Fan Y, Miguez-Macho G, Jobbágy EG et al (2017) Hydrologic regulation of plant rooting depth. Proc Natl Acad Sci U S A 114:10572–10577. https://doi.org/10.1073/pnas.1712381114
Felsenstein J (1985) Phylogenies and the comparative method. Am Nat 125:1–15. https://doi.org/10.1086/284325
Fort F, Jouany C, Cruz P (2013) Root and leaf functional trait relations in Poaceae species: Implications of differing resource-acquisition strategies. J Plant Ecol 6:211–219. https://doi.org/10.1093/jpe/rts034
Fujimaki R, Tateno R, Hirobe M et al (2004) Fine root mass in relation to soil N supply in a cool temperate forest. Ecol Res 19:559–562. https://doi.org/10.1111/j.1440-1703.2004.00669.x
Gastauer M, Meira-Neto JAA (2017) Updated angiosperm family tree for analyzing phylogenetic diversity and community structure. Acta Bot Brasilica 31:191–198. https://doi.org/10.1590/0102-33062016abb0306
Ge J, Xie Z (2017) Geographical and climatic gradients of evergreen versus deciduous broad-leaved tree species in subtropical China: Implications for the definition of the mixed forest. Ecol Evol 7:3636–3644. https://doi.org/10.1002/ece3.2967
Henn JJ, Damschen EI (2021) Plant age affects intraspecific variation in functional traits. Plant Ecol 222:669–680. https://doi.org/10.1007/s11258-021-01136-2
IPCC (2021) Summary for policymakers. In: Climate Change 2021: The physical science basis. Cambridge University Press, Cambridge
Jackson RB, Canadell J, Ehleringer JR et al (1996) A global analysis of root distributions for terrestrial biomes. Oecologia 108:389–411. https://doi.org/10.1007/BF00333714
Karizumi N (2010) The latest illustrations of tree roots. Seibundo Shinkosha Publishing. ISBN978–4–416–41005–9
Kattge J, Díaz S, Lavorel S et al (2011) TRY - a global database of plant traits. Glob Chang Biol 17:2905–2935. https://doi.org/10.1111/j.1365-2486.2011.02451.x
Kattge J, Bönisch G, Díaz S et al (2020) TRY plant trait database – enhanced coverage and open access. Glob Chang Biol 26:119–188. https://doi.org/10.1111/gcb.14904
Lachaise T, Bergmann J, Rillig MC, van Kleunen M (2021) Below- and aboveground traits explain local abundance, and regional, continental and global occurrence frequencies of grassland plants. Oikos 130:110–120. https://doi.org/10.1111/oik.07874
Leuschner C, Meier IC (2018) The ecology of Central European tree species: trait spectra, functional trade-offs, and ecological classification of adult trees. Perspect Plant Ecol Evol Syst 33:89–103. https://doi.org/10.1016/j.ppees.2018.05.003
Li FL, Bao WK (2015) New insights into leaf and fine-root trait relationships: Implications of resource acquisition among 23 xerophytic woody species. Ecol Evol 5:5344–5351. https://doi.org/10.1002/ece3.1794
Li H, Si B, Wu P, McDonnell JJ (2019) Water mining from the deep critical zone by apple trees growing on loess. Hydrol Process 33:320–327. https://doi.org/10.1002/hyp.13346
Locatelli B, Catterall CP, Imbach P et al (2015) Tropical reforestation and climate change: beyond carbon. Restor Ecol 23:337–343. https://doi.org/10.1111/rec.12209
Maréchaux I, Saint-André L, Bartlett MK et al (2020) Leaf drought tolerance cannot be inferred from classic leaf traits in a tropical rainforest. J Ecol 108:1030–1045. https://doi.org/10.1111/1365-2745.13321
Paradis E, Claude J, Strimmer K (2004) APE: Analyses of phylogenetics and evolution in R language. Bioinformatics 20:289–290. https://doi.org/10.1093/bioinformatics/btg412
Pérez-Harguindeguy N, Díaz S, Garnier E et al (2013) New handbook for standardised measurement of plant functional traits worldwide. Aust J Bot 61:167–234. https://doi.org/10.1071/BT12225
Pierret A, Maeght JL, Clément C et al (2016) Understanding deep roots and their functions in ecosystems: an advocacy for more unconventional research. Ann Bot 118:621–635. https://doi.org/10.1093/aob/mcw130
Poorter H, Lambers H, Evans JR (2014) Trait correlation networks: a whole-plant perspective on the recently criticized leaf economic spectrum. New Phytol 201:378–382. https://doi.org/10.1111/nph.12547
Reich P, Kloeppel B, Ellsworth D, Walters M (1995) Different photosynthesis-nitrogen relations in deciduous hardwood and evergreen coniferous tree species. Oecologia 104:24–30
Sandel B, Monnet AC, Vorontsova M (2016) Multidimensional structure of grass functional traits among species and assemblages. J Veg Sci 27:1047–1060. https://doi.org/10.1111/jvs.12422
Seiwa K (1999) Changes in leaf phenology are dependent on tree height in Acer mono, a deciduous broad-leaved tree. Ann Bot 83:355–361
Siefert A, Violle C, Chalmandrier L et al (2015) A global meta-analysis of the relative extent of intraspecific trait variation in plant communities. Ecol Lett 18:1406–1419. https://doi.org/10.1111/ele.12508
Stone EL, Kalisz PJ (1991) On the maximum extent of tree roots. For Ecol Manage 46:59–102. https://doi.org/10.1016/0378-1127(91)90245-Q
Tobner CM, Paquette A, Messier C (2013) Interspecific coordination and intraspecific plasticity of fine root traits in North American temperate tree species. Front Plant Sci 4:1–11. https://doi.org/10.3389/fpls.2013.00242
Tomlinson KW, Poorter L, Sterck FJ et al (2013) Leaf adaptations of evergreen and deciduous trees of semi-arid and humid savannas on three continents. J Ecol 101:430–440. https://doi.org/10.1111/1365-2745.12056
Vargas GG, Kunert N, Hammond WM et al (2022) Leaf habit affects the distribution of drought sensitivity but not water transport efficiency in the tropics. Ecol Lett. https://doi.org/10.1111/ele.14128
Webb CO, Ackerly DD, Kembel SW (2008) Phylocom: software for the analysis of phylogenetic community structure and trait evolution. Bioinformatics 24:2098–2100. https://doi.org/10.1093/bioinformatics/btn358
Weemstra M, Kiorapostolou N, Ruijven J et al (2020) The role of fine-root mass, specific root length and life span in tree performance: a whole-tree exploration. Funct Ecol 34:575–585. https://doi.org/10.1111/1365-2435.13520
Weigelt A, Mommer L, Andraczek K et al (2021) An integrated framework of plant form and function: the belowground perspective. New Phytol 232:42–59. https://doi.org/10.1111/nph.17590
Wikström N, Savolainen V, Chase MW (2001) Evolution of the angiosperms: calibrating the family tree. Proc R Soc B Biol Sci 268:2211–2220. https://doi.org/10.1098/rspb.2001.1782
Zhou Y, Wigley BJ, Case MF et al (2020) Rooting depth as a key woody functional trait in savannas. New Phytol 227:1350–1361. https://doi.org/10.1111/nph.16613
Acknowledgements
We sincerely acknowledge Dr. Karizumi Noboru for his outstanding illustration of woody roots in Japan.
Funding
This study was financially supported by JSPS Grant in aid basic study B (19H02986 to MK).
Author information
Authors and Affiliations
Contributions
KM conceived the idea of the study, KM collected the data, RK conducted the analysis and KM, RK and GBW contributed to the discussion and writing of the paper.
Corresponding author
Ethics declarations
Competing interest
The authors declare that they have competing interest.
Additional information
Communicated by Marta Pardos.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Makoto, K., Kitagawa, R. & Blume-Werry, G. How do leaf functional traits and age influence the maximum rooting depth of trees?. Eur J Forest Res 142, 1197–1206 (2023). https://doi.org/10.1007/s10342-023-01585-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-023-01585-6