Abstract
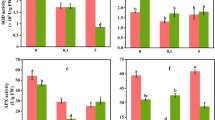
Ectomycorrhizal fungi (EMF) can enhance drought resistance of host plants by increasing nutrient and water absorption. Calcium (Ca) plays a central role in drought stress adaptation, yet it remains unclear how EMF influence Ca uptake of plants under drought stress. We carried out a pot experiment in which Quercus acutissima seedlings were inoculated with or without the ectomycorrhizal (EM) fungus Xerocomus chrysenteron under control and water stress soil moisture conditions in order to investigate how soil Ca availability and plant Ca uptake are modulated by EMF and how this may contribute to drought resistance of the seedlings. We found that soil water-extractable Ca was significantly reduced by drought in non-ectomycorrhizal treatment, while EM treatment significantly increased soil Ca availability and Ca uptake of Quercus acutissima seedlings under drought conditions. Furthermore, seedling water use efficiency (WUE) was positively correlated with soil Ca availability and plant Ca content. Calcium uptake promotion seems to be involved in the mechanisms of drought tolerance enhancement by EMF.



Similar content being viewed by others
References
Ahmadi B, Ahmadalipour A, Tootle G, Moradkhani H (2019) Remote sensing of water use efficiency and terrestrial drought recovery across the contiguous United States. Remote Sens 11:731. https://doi.org/10.3390/rs11060731
Alva AK, Gascho GJ, Guang Y (1991) Soil solution and extractable calcium in gypsumamended coastal plain soils used for peanut culture. Commun Soil Sci Plant Anal 22:99–116. https://doi.org/10.1080/00103629109368398
Alvarez M, Huygens D, Fernandez C, Gacitúa Y, Olivares E, Saavedra I, Alberdi M, Valenzuela E (2009) Effect of ectomycorrhizal colonization and drought on reactive oxygen species metabolism of Nothofagus dombeyi roots. Tree Physiol 29(8):1047–1057. https://doi.org/10.1093/treephys/tpp038
Anderegg WRL, Plavcová L, Anderegg LDL, Hacke UG, Berry JA, Field CB (2013) Drought’s legacy: multiyear hydraulic deterioration underlies widespread aspen forest die-off and portends increased future risk. Global Change Biol 19:1188–1196. https://doi.org/10.1111/gcb.12100
Beer C, Ciais P, Reichstein M, Baldocchi D, Law BE, Papale D, Soussana JF, Ammann C, Buchmann N, Frank D, Gianelle D, Janssens IA, Knohl A, Köstner B, Moors E, Roupsard O, Verbeeck H, Vesala T, Williams CA, Wohlfahrt G (2009) Temporal and among-site variability of inherent water use efficiency at the ecosystem level. Glob Biogeochem Cycle 23:GB2018. https://doi.org/10.1029/2008GB003233
Bigelow SW, Canham CD (2007) Nutrient limitation of juvenile trees in a northern hardwood forest: calcium and nitrate are preeminent. For Ecol Manag 243(2):310–319. https://doi.org/10.1016/j.foreco.2007.03.027
Birhane E, Sterck FJ, Fetene M, Bongers F, Kuyper TW (2012) Arbuscular mycorrhizal fungi enhance photosynthesis, water use efficiency, and growth of frankincense seedlings under pulsed water availability conditions. Oecologia 169:895–904. https://doi.org/10.2307/23260122
Blum JD, Klaue A, Nezat CA, Driscoll CT, Johnson CE, Siccama TG, Eagar C, Fahey TJ, Likens GE (2002) Mycorrhizal weathering of apatite as an important calcium source in base-poor forest ecosystems. Nature 417:729–731. https://doi.org/10.1038/nature00793
Bogeat-Triboulot M-B, Bartoli F, Garbaye J, Marmeisse R, Tagu D (2004) Fungal ectomycorrhizal community and drought affect root hydraulic properties and soil adherence to roots of Pinus pinaster seedlings. Plant Soil 267:213–223. https://doi.org/10.1007/s11104-005-5349-7
Bottero A, D’Amato AW, Palik BJ, Bradford JB, Fraver S, Battaglia MA, Asherin LA (2017) Density-dependent vulnerability of forest ecosystems to drought. J Appl Ecol 54:1605–1614. https://doi.org/10.1016/10.1111/1365-2664.12847
Brownlee C, Duddridge JA, Malibari A, Read DJ (1983) The structure and function of mycelial systems of ectomycorrhizal roots with special reference to their role in forming inter-plant connections and providing pathways for assimilate and water transport. Plant Soil 71:433–443. https://doi.org/10.1007/BF02182684
Burke EJ, Brown SJ, Christidis N (2009) Modeling the recent evolution of global drought and projections for the twenty-first century with the Hadley Centre climate model. J Hydrometeorol 7(5):1113–1125. https://doi.org/10.1175/JHM544.1
Bücking H, Kuhn AJ, Schröder WH, Heyser W (2002) The fungal sheath of ectomycorrhizal pine roots: an apoplastic barrier for the entry of calcium, magnesium, and potassium into the root cortex. J Exp Bot 53:1659–1669. https://doi.org/10.1093/jxb/erf011
Cernusak LA, Winter K, Aranda J, Virgo A, Garcia M (2009) Transpiration efficiency over an annual cycle, leaf gas exchange and wood carbon isotope ratio of three tropical tree species. Tree Physiol 29:1153–1161. https://doi.org/10.1093/treephys/tpp052
Chen DH, Liu HP, Li CL (2019) Calcium-dependent protein kinase CPK9 negatively functions in stomatal abscisic acid signaling by regulating ion channel activity in Arabidopsis. Plant Mol Biol 99:113–122. https://doi.org/10.1007/s11103-018-0805-y
Ciais Ph, Reichstein M, Viovy N, Granier A, Ogée J, Allard V, Aubinet M, Buchmann N, Chr B, Carrara A, Chevallier F, De Noblet N, Friend AD, Friedlingstein P, Grünwald T, Heinesch B, Keronen P, Knohl A, Krinner G, Loustau D, Manca G, Matteucci G, Miglietta F, Ourcival JM, Papale D, Pilegaard K, Rambal S, Seufert G, Soussana JF, Sanz MJ, Schulze ED, Vesala T, Valentini R (2005) Europe-wide reduction in primary productivity caused by the heat and drought in 2003. Nature 437:529–533. https://doi.org/10.1038/nature03972
Clarkson DT (1984) Calcium transport between tissue and its distribution in the plant. Plant Cell Environ 7:449–456. https://doi.org/10.1111/j.1365-3040.1984.tb01435.x
Cromack K, Sollins P, Graustein WC, Speidel K, Todd AW, Spycher G, Li CY, Todd RL (1979) Calcium oxalate accumulation and soil weathering in mats of the hypogeous fungus Hysterangium crassum. Soil Biol Biochem 11:463–468. https://doi.org/10.1016/0038-0717(79)90003-8
Dahlqvist R, Benedetti MF, Andersson K, Turner D, Larsson T, Stolpe B, Ingri J (2004) Association of calcium with colloidal particles and speciation of calcium in the Kalix and Amazon rivers. Geochim Cosmochim Acta 68:4059–4075. https://doi.org/10.1016/j.gca.2004.04.007
Danielsen L, Polle A (2014) Poplar nutrition under drought as affected by ectomycorrhizal colonization. Environ Exp Bot 108:89–98. https://doi.org/10.1016/j.envexpbot.2014.01.006
Dauer JM, Perakis SS (2013) Contribution of calcium oxalate to soil-exchangeable calcium. Soil Sci 178(12):671–678. https://doi.org/10.1097/SS.0000000000000029
Drouet T, Herbauts J, Demaiffe D (2005) Long-term records of strontium isotopic composition in tree rings suggest changes in forest calcium sources in the early 20th century. Glob Change Biol 11:1926–1940. https://doi.org/10.1111/j.1365-2486.2005.01034.x
dos Santos MAd, Valadares RV, Neves JCL, Silva IRd, Tótola MR, Costa MD (2020) Ammonium nitrogen increases Ca uptake from non-exchangeable reservoirs by eucalypt plants. For Ecol Manag 465:118062. https://doi.org/10.1016/j.foreco.2020.118062
Gebauer R, Volařík D, Urban J (2012) Quercus pubescens and its hemiparasite Loranthus europaeus: nutrient dynamics of leaves and twigs. Acta Physiol Plant 34:1801–1809. https://doi.org/10.1007/s11738-012-0978-y
Gradowski T, Thomas SC (2008) Responses of Acer saccharum canopy trees and saplings to P, K and lime additions under high N deposition. Tree Physiol 28(2):173–185. https://doi.org/10.1093/treephys/28.2.173
Haber MF, Young E, Faust M (1983) Effects of PEG-induced water stress on calcium uptake in peach seedlings. J Am Soc Hortic Sci 108:737–740
Han WX, Fang JY, Reich PB, Ian WF, Wang ZH (2011) Biogeography and variability of eleven mineral elements in plant leaves across gradients of climate, soil and plant functional type in China. Ecol Lett 14:788–796. https://doi.org/10.1111/j.1461-0248.2011.01641.x
Hepler PK (2005) Calcium, a central regulator of plant growth and development. Plant Cell 17:2142–2155. https://doi.org/10.1105/tpc.105.032508
Hirschi KD (2004) The calcium conundrum. Both versatile nutrient and specific signal. Plant Physiol 136:2438–2442. https://doi.org/10.1104/pp.104.046490
Hobbie EA, Colpaert JV (2010) Nitrogen availability and mycorrhizal colonization influence water use efficiency and carbon isotope patterns in Pinus sylvestris. New Phytol 164:515–525. https://doi.org/10.1111/j.1469-8137.2004.01187.x
Holzmueller EJ, Jose S, Jenkins MA (2007) Relationship between Cornus florida L. and calcium mineralization in two southern Appalachian forest types. For Ecol Manag 245:110–117. https://doi.org/10.1016/j.foreco.2007.04.004
Hornbeck JW, Kropelin W (1982) Nutrient removal and leaching from a whole-tree harvest of northern hardwoods. J Environ Qual 11:309–316. https://doi.org/10.2134/jeq1982.00472425001100020032x
Hynicka JD, Julie C, Pett-Ridge PSS (2016) Nitrogen enrichment regulates calcium sources in forests. Glob Change Biol 22(12):1–13. https://doi.org/10.1111/gcb.13335
Jaleel CA, Manivannan P, Sankar B, Kishorekumar A, Gopi R, Somasundaram R, Panneerselvam R (2007) Water deficit stress mitigation by calcium chloride in Catharanthus roseus: effect on oxidative stress, proline metabolism and indole alkaloid accumulation. Colloid Surf B-Biointerfaces 60:110–116. https://doi.org/10.1016/j.colsurfb.2007.06.006
Johnson AH, Siccama TG (1983) Acid deposition and forest decline. Environ Sci Technol 17:294A-305A. https://doi.org/10.1021/es00113a717
Jones HG, Tardieu F (1998) Modelling water relations of horticultural crops: a review. Sci Hortic 74:21–46. https://doi.org/10.1016/s0304-4238(98)00081-8
Jump AS, Ruiz-Benito P, Greenwood S, Allen CD, Kitzberger T, Fensham R, Martínez-Vilalta J, Lloret F (2017) Structural overshoot of tree growth with climate variability and the global spectrum of drought-induced forest dieback. Glob Change Biol 23(9):3742–3757. https://doi.org/10.1111/gcb.13636
Kaczmarek M, Fedorowicz-Strońska GK, Waśkiewicz A, Sadowski J (2017) CaCl2 treatment improves drought stress tolerance in barley (Hordeum vulgare L.). Acta Physiol Plant 39(1):41. https://doi.org/10.1007/s11738-016-2336-y
Khushboo KB, Preeti S, Meenakshi R, Vinay S, Deepak K (2018) Exogenous application of calcium chloride in wheat genotypes alleviates negative effect of drought stress by modulating antioxidant machinery and enhanced osmolyte accumulation. Vitro Cell Dev Biol Plant 54:495–507. https://doi.org/10.1007/s11627-018-9912-3
Kilpeläinen J, Barbero-López A, Vestberg M, Heiskanen J, Lehto T (2017) Does severe soil drought have after-effects on arbuscular and ectomycorrhizal root colonisation and plant nutrition? Plant Soil 418:377–386. https://doi.org/10.1111/10.1007/s11104-017-3308-8
Lee SH, Calvo-Polanco M, Chung GC, Zwiazek JJ (2010) Role of aquaporins in root water transport of ectomycorrhizal jack pine (Pinus banksiana) seedlings exposed to NaCl and fluoride. Plant Cell Environ 33(5):769–780. https://doi.org/10.1111/j.1365-3040.2009.02103.x
Lehto T, Zwiazek JJ (2011) Ectomycorrhizas and water relations of trees: a review. Mycorrhiza 21:71–90. https://doi.org/10.1007/s00572-010-0348-9
Li W, Zhou PP, Jia LP, Yu LJ, Li LX, Zhu M (2009) Limestone dissolution induced by fungal mycelia, acidic materials, and carbonic anhydrase from fungi. Mycopathologia 167:37–46. https://doi.org/10.1007/s11046-008-9143-y
Liese R, Lübbe T, Albers NW, Meier IC (2017) The mycorrhizal type governs root exudation and nitrogen uptake of temperate tree species. Tree Physiol 38:1–13. https://doi.org/10.1093/treephys/tpx131
Liu YY, Wang AY, An YN, Lian PY, Wu DD, Zhu JJ, Meinzer FC, Hao GY (2018) Hydraulics play an important role in causing low growth rate and dieback of aging Pinus sylvestris var. mongolica trees in plantations of Northeast China. Plant Cell Environ 41:7. https://doi.org/10.1111/pce.13160
Ma X, Sun M, Sa G, Zhang Y, Li L, Sun J, Shen X, Polle A, Chen S (2014) Ion fluxes in Paxillus involutus-inoculated roots of Populus × canescens, under saline stress. Environ Exp Bot 108:99–108. https://doi.org/10.1016/j.envexpbot.2013.11.016
Mahajan S, Tuteja N (2005) Cold, salinity and drought stresses: an overview. Arch Biochem Biophys 444:139–158. https://doi.org/10.1016/j.abb.2005.10.018
Mason AC (1958) The effect of soil moisture on the mineral composition of apple plants grown in pots. J Hortic Sci 33:202–211. https://doi.org/10.1080/00221589.1958.11513927
Mcdowell N, Pockman WT, Allen CD, Breshears DD, Cobb N, Kolb T, Plaut J, Sperry J, West A, Williams DG, Yepez EA (2008) Mechanisms of plant survival and mortality during drought: why do some plants survive while others succumb to drought? New Phytol 178(4):719–739. https://doi.org/10.1111/j.1469-8137.2008.02436.x
Mclaughlin SB, Wimmer R (1999) Tansley review no. 104. Calcium physiology and terrestrial ecosystem processes. New Phytol 142:373–417. https://doi.org/10.1046/j.1469-8137.1999.00420.x
Melin E, Nilsson H (1955) 45Ca used as indicator of transport of cations to pine seedlings by means of mycorrhiza mycelium. Sven Bot Tidskr 49:119–121
Muhsin TM, Zwiazek JJ (2002) Ectomycorrhizas increase apoplastic water transport and root hydraulic conductivity in Ulmus americana seedlings. New Phytol 153:153–158. https://doi.org/10.1046/j.0028-646X.2001.00297.x
Nehls U, Plassard C (2018) Nitrogen and phosphate metabolism in ectomycorrhizas. New Phytol 220:1047–1058. https://doi.org/10.1111/nph.15257
Pei ZM, Ghassemian M, Kwak CM, McCourt P, Schroeder JI (1998) Role of farnesyl transferase in ABA regulation of guard cell anion channels and plant water loss. Science 282(5387):287–290. https://doi.org/10.1126/science.282.5387.287
Pei ZM, Murata Y, Benning G, Thomine S, Klüsener B, Allen GJ, Grill E, Schroeder JI (2000) Calcium channels activated by hydrogen peroxide mediate abscisic acid signalling in guard cells. Nature 406:731–734. https://doi.org/10.1038/35021067
Perakis SS, Maguire DA, Bullen TD, Cromack K, Waring RH, Boyle JR (2006) Coupled nitrogen and calcium cycles in forests of the Oregon Coast Range. Ecosystems 9:63–74. https://doi.org/10.1007/s10021-004-0039-5
Plan L (2005) Factors controlling carbonate dissolution rates quantified in a field test in the Austrian alps. Geomorphology 68:201–212. https://doi.org/10.1016/j.geomorph.2004.11.014
Qaderi M, Martel AB, Dixon S (2019) Environmental factors influence plant vascular system and water regulation. Plants (Basel) 8(3):65. https://doi.org/10.3390/plants8030065
Quirk J, Beerling DJ, Banwart SA, Kakonyi G, Romero-Gonzalez ME, Leake JR (2012) Evolution of trees and mycorrhizal fungi intensifies silicate mineral weathering. Biol Lett 8:1006–1011. https://doi.org/10.1098/rsbl.2012.0503
Reddy AS, Ali GS, Celesnik H, Day IS (2011) Coping with stresses: roles of calcium and calcium/calmodulin-regulated gene expression. Plant Cell 23:2010–2032. https://doi.org/10.1105/tpc.111.084988
Rusakov AV, Vlasov AD, Zelenskaya MS, Frank-Kamenetskaya OV, Vlasov DY (2016) The crystallization of calcium oxalate hydrates formed by interaction between microorganisms and minerals biogenic—abiogenic interactions in natural and anthropogenic systems. Springer, Cham, pp 357–377. https://doi.org/10.1007/978-3-319-24987-2_28
Schoonmaker AL, Teste FP, Simard SW, Guy RD (2007) Tree proximity, soil pathways and common mycorrhizal networks: their influence on the utilization of redistributed water by understory seedlings. Oecologia 154:455–466. https://doi.org/10.1007/s00442-007-0852-6
Seifikalhor M, Aliniaeifard S, Shomali A, Azad N, Hassani B, Lastochkina O, Li T (2019) Calcium signaling and salt tolerance are diversely entwined in plants. Plant Signal Behav 14:1665455. https://doi.org/10.1080/15592324.2019.1665455
Siemion J, Burns DA, Murdoch PS, Germain RH (2011) The relation of harvesting intensity to changes in soil, soil water, and stream chemistry in a northern hardwood forest, Catskill Mountains, USA. For Ecol Manag 261:1510–1519. https://doi.org/10.1016/j.foreco.2011.01.036
St Clair SB, Sharpe WE, Lynch JP (2007) Key interactions between nutrient limitation and climatic factors in temperate forests: a synthesis of the sugar maple literature. Can J For Res 38(3):401–414. https://doi.org/10.1139/X07-161
Swaty RL, Gehring CA, Ert MV, Theimer TC, Keim P, Whitham TG (1998) Temporal variation in temperature and rainfall differentially affects ectomycorrhizal colonization at two contrasting sites. New Phytol 139(4):733–739. https://doi.org/10.1046/j.1469-8137.1998.00234.x
Tedersoo L, Bahram M (2019) Mycorrhizal types differ in ecophysiology and alter plant nutrition and soil processes. Biol Rev 94:1857–1880. https://doi.org/10.1111/brv.12538
Upadhyaya H, Panda SK, Dutta BK (2011) CaCl2 improves postdrought recovery potential in Camellia sinensis (L) O. Kuntze. Plant Cell Rep 30:495–503. https://doi.org/10.1007/s00299-010-0958-x
van Mantgem PJ, Stephenson NL, Byrne JC, Daniels LD, Franklin JF, Fulé PZ, Harmon ME, Larson AJ, Smith JM, Taylor AH, Veblen TT (2009) Widespread increase of tree mortality rates in the western United States. Science 323:521–524. https://doi.org/10.1126/science.1165000
Wang WH, Chen J, Liu TW, Chen J, Han AD, Simon M, Dong XJ, He JX, Zheng HL (2014) Regulation of the calcium-sensing receptor in both stomatal movement and photosynthetic electron transport is crucial for water use efficiency and drought tolerance in Arabidopsis. J Exp Bot 65:223–234. https://doi.org/10.1093/jxb/ert362
Warren JM, Brooks JR, Meinzer FC, Eberhart JL (2008) Hydraulic redistribution of water from pinus ponderosa trees to seedlings: evidence for an ectomycorrhizal pathway. New Phytol 178:382–394. https://doi.org/10.1111/j.1469-8137.2008.02377.x
Watmough SA, Dillon PJ (2003) Calcium losses from a forested catchment in south-central Ontario, Canada. Environ Sci Technol 37:3085–3089. https://doi.org/10.1021/es034034t
White PJ (2001) The pathways of calcium movement to the xylem. J Exp Bot 52:891. https://doi.org/10.1093/jexbot/52.358.891
White PJ, Broadley MR (2003) Calcium in plants. Ann Bot 92:487–511. https://doi.org/10.1093/aob/mcg164
Xu C, Li X, Zhang L (2013) The effect of calcium chloride on growth, photosynthesis, and antioxidant responces of Zoysia japonica under drought conditions. PLoS ONE 8:e68214. https://doi.org/10.1371/journal.pone.0068214
Yin D, Wang H, Qi J (2020) The enhancement effect of calcium ions on ectomycorrhizal fungi-mediated drought resistance in Pinus sylvestris var. mongolica. J Plant Growth Regul. https://doi.org/10.1007/s00344-020-10197-y
Zhang T, Yang J, Sun Y, Kang Y, Yang J, Qi Z (2018) Calcium deprivation enhances non-selective fluid-phase endocytosis and modifies membrane lipid profiles in Arabidopsis roots. J Plant Physiol 226:22–30. https://doi.org/10.1016/j.jplph.2018.04.002
Zhao M, Running SW (2010) Drought-induced reduction in global terrestrial net primary production from 2000 through. Science 329(5994):940–943. https://doi.org/10.1126/science.1192666
Zhou YB, Zou XM (2017) From matching site with trees towards matching calcium with trees. J Nanjing For Univ 41:1–8 (in Chinese)
Zhu JJ, Kang HZ, Xu ML (2007) Natural regeneration barriers of Pinus sylvestris var. mongolica plantations in southern Keerqin sandy land, China. Acta Ecol Sin 27(10):4086–4095 (in Chinese)
Acknowledgements
This research was supported jointly by the National Natural Science Foundation of China (Grant Nos. 31800364, 31700552, 31400611); The Special Fund for Forest Scientific Research in the Public Welfare (Grant No. 201304216); and Fundamental Research Funds for Universities (LSNQN201708); We thank Jin Y. Qi for technical support.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
No conflict of interest exits in the submission of this manuscript, and manuscript is approved by all authors for publication. We would like to declare on behalf of our co-authors that the work described was original research.
Additional information
Communicated by Peter Annighoefer.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, Y., Zhang, T., Zhou, Y. et al. Ectomycorrhizal symbioses increase soil calcium availability and water use efficiency of Quercus acutissima seedlings under drought stress. Eur J Forest Res 140, 1039–1048 (2021). https://doi.org/10.1007/s10342-021-01383-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-021-01383-y