Abstract
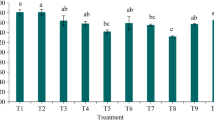
A newly introduced peach clonal rootstock, ‘Rubira’, when multiplied through mound layering, the daughter stool shoots attain excessive diameter making it difficult to perform grafting in the next season. To overcome this problem, the present study aimed to conduct budding on daughter stool shoots of ‘Rubira’ peach rootstock produced through mound layering during the summer or rainy season of the same year, further accounting for production of saleable plants within 1 year. The experiment consisted of 15 treatment combinations of budding methods (chip, shield and patch) and timing of budding (22nd May, 6th June, 21st June, 6th July, and 21st July). The results revealed that chip and shield budding performed on 22nd May and 6th June had maximum bud take success (100.00%). The chip budding performed on 22nd May resulted in significantly higher scion height (139.42 cm), scion diameter (12.68 mm), fresh weight of shoots (127.33 g), root–shoot biomass (82.41 g dry weight) and saleable plants (99.96%). Moreover, chip budding performed on May 22nd demonstrated the highest levels of leaf carbohydrate (6.65%), total sugars (1.76%) and leaf nitrogen (3.82%). The bud-take success was significantly positively correlated with saleable plant, scion height, scion diameter, number of internodes, fresh weight of root, root–shoot biomass, leaf carbohydrate and leaf nitrogen, whereas significantly negatively correlated with fresh weight of shoot. The chip budding performed during the last week of May to the first week of June was found to be the best method and time for production of quality nursery plants of peach in stool beds of ‘Rubira’ rootstock under mound layering within 1 year.





Similar content being viewed by others
Data availability statement
The data that support the findings of this study are available from the corresponding author (PV) upon reasonable request.
References
Akhtar A, Bakhsh A, Sumrah MA (2018) Standardization of optimum budding time for peach nursery in soan valley. J Agric Res 56:243–246. https://doi.org/10.13140/RG.2.2.18148.30083
Awasthi M, Negi M (2016) Effect of different time and methods of budding on bud take success of nectarine on peach (Prunus persica L.) seedling rootstocks. Impact int J Res Appl Nat Soc Sci 4:25–30
Cao KE, Zhou Z, Wang QI, Guo J, Zhao P, Tian Z et al (2016) Genome-wide association study of 12 agronomic traits in peach. Nat Commun 7:13246. https://doi.org/10.1038/ncomms13246
Chandel JS, Gautam D, Sharma NC (2005) Chip budding: an excellent method of propagation of walnut (Juglans regia L.). Acta Hortic 705:335–339. https://doi.org/10.17660/ActaHortic.2005.705.45
Chhukit K (2009) Studies on vegetative propagation of kiwifruit (Actinidia deliciosa Chev.). YSP University of Horticulture & Forestry, Solan (Dissertation)
Dimri DC, Petwal A, Kamboj P (2009) Determination of optimum time for chip budding in apple cv. ‘Red Fuji’. Indian J Horic 66:254–256. https://doi.org/10.17660/ActaHortic.2005.696.29
Dipta K (2016) Studies on vegetative propagation of walnut under protective conditions. Dissertation, Dr YSP University of Horticulture & Forestry, Solan. http://krishikosh.egranth.ac.in/handle/1/5810006458
Dubois M, Giles K, Hamilton JK, Rebers PA, Smith F (1951) A colorimetric method for determination of sugar. Nature 168:167. https://doi.org/10.1038/168167a0
FAOSTAT (2021) Area harvested and production of peach in world. http://www.fao.org/faostat/en/#data/QCL. Accessed 3 Sept 2023
Hartmann HT, Kester DE, Davies FT, Geneve RL, Wilson SB (2017) Plant propagation: principles and practices. Pearson Education, New York
Hiscox JD, Israelstam GF (1979) A method for extraction of chlorophyll from leaf tissues without masceration. Canad J Bot 57:1332–1334. https://doi.org/10.1139/b79-163
Hodge JE, Hofreiter BT (1962) Determination of reducing sugars and carbohydrates. In: Whistler RL, Wolfrom ML (eds) Methods in carbohydrate chemistry. Academic Press, NewYork, pp 380–394
Howard BH, Skene DS, Coles JS (1974) The effects of different grafting methods upon the development of one-year-old nursery apple trees. J Hortic Sci 49:287–295. https://doi.org/10.1080/00221589.1974.11514581
Iqbal M, Singh KK (2020) Propagation of temperate fruit crops. In: Innovative agriculture and botany. Victorious Publishers, pp 119–135
Jackson ML (1973) Soil chemical analysis. Prentice Hall, New Delhi
Jaipal (2019) Standardization of grafting techniques on different rootstocks in low chill peach cv. ‘Shan-e-Punjab’. Dissertation, Chaudhary Charan Singh Haryana Agricultural University, Haryana. http://krishikosh.egranth.ac.in/handle/1/5810142491
Karna AK, Varu DK, Panda PA, Hota D (2017) Standardization of grafting time and height on success of softwood grafting in mango (Mangifera indica L.). J Pharmacogn Phytochem 6:2803–2807
Kasmi M, Rama P, Bari H, Edlira K, Rabeta A (2013) Budding of walnut (Juglans regia L.). Albanian J Agric Sci 12:465–469
Kumari (2019) Standardization of budding techniques for propagation of Sweet Cherry (Prunus avium L.). Dissertation, Dr YSP University of Horticulture & Forestry, Solan. http://krishikosh.egranth.ac.in/handle/1/5810135839
Layne DR, Bassi D (2008) The peach: botany, production and uses. Cabi Wallingford
Layne REC (1987) Peach rootstocks. In: Rom RC, Carson RF (eds) Rootstocks for fruit crops. Wiley, New York, pp 185–216
Layne REC, Jui PY (1994) Genetically diverse peach seedling rootstocks affect long-term performance of ‘Redhaven’ peach on fox sand. J Am Soc Hortic Sci 119:1303–1311
Ljung K (2013) Auxin metabolism and homeostasis during plant development. Development 140:943–950. https://doi.org/10.1242/dev.086363
Mayer NA, Bianchi VJ, Feldberg NP, Morini S (2017) Advances in peach nectarine and plum propagation. Rev Bras Frutic. https://doi.org/10.1590/0100-29452017355
Muniyappan C, Rajangam J, Kumar CSR, Venkatesan K (2019) The Standardization of method and time of propagation in jamun (Syzygium cuminii Skeels) var. ‘Konkan Bahadoli. J Pharmacogn Phytochem 8:467–471
NHB (2022) National horticulture board, ministry of agriculture and farmers welfare 2022–2023. https://nhb.gov.in/. Accessed 21 Mar 2024
Pinghai D, Rongting X (1993) Effect of phenols on the survival of walnut grafting. Acta Hortic 311:134–140. https://doi.org/10.17660/ActaHortic.1993.311.18
Powell AE, Lenhard M (2012) Control of organ size in plants. Curr Biol 22:R360–R367. https://doi.org/10.1016/j.cub.2012.02.010
Pramanick KK (2018) Rootstock for stone fruits—a new prospective. JOJ Hortic Arboric. https://doi.org/10.19080/JOJHA.2018.01.555574
Rawat L (2006) Studies on seasonal influence of chip budding on bud success and growth of apple (Malus×domestica Borkh) cv. ‘Royal Delicious’. Dissertation, GBP University of Agriculture & Technology. http://krishikosh.egranth.ac.in/handle/1/5810137482
Rom RC, Carlson RF (1987) Rootstocks for fruit crops. Wiley, New York
Sandhya (2021) Effects of budding methods and time on bud take and growth of apricot (Prunus armeniaca L.) nursery plants. Dissertation, Dr YSP University of Horticulture & Forestry, Solan. https://krishikosh.egranth.ac.in/handle/1/5810177191
Sauge MH, Kervella J, Pascal T (1998) Settling behaviour and reproductive potential of the green peach aphid Myzus persicae on peach varieties and a related wild Prunus. Entomol Exp Appl 86:233–242
Sauge MH, Lacroze JP, Poëssel JL, Pascal T, Kervella J (2002) Induced resistance by Myzus persicae in the peach cultivar ‘Rubira’. Entomol Exp Appl 102:29–37. https://doi.org/10.1023/A:1015282702657
Shah R, Sharma A, Wali V, Bakshi P, Kumar R, Gupta R (2017) Response of budding methods and time on bud success and budding growth of peach (Prunus persica) on different rootstocks. Indian J Agric Sci 87:669–676. https://doi.org/10.56093/ijas.v87i5.70196
Sharma JB, Chauhan N, Rana K, Bakshi M (2020a) Evaluation of rootstocks for temperate fruit crops—a review. Int J Curr Microbiol App Sci 9:3533–3539. https://doi.org/10.20546/ijcmas.2020.911.422
Sharma N, Sharma SD, Kumar D (2009) Selection of stone fruit rootstock for drought tolerance and amenability to clonal multiplication. Biol Forum 1:36–46
Sharma R, Kumar A (2019) Influence of indole butyric acid on propagability of clonal rootstock of Prunus species through cuttings and stooling. J Pharmacogn Phytochem 8:2483–2487
Sharma R, Singh PN, Dimri DC, Uniyal S, Nirgude V, Singh M (2020b) Optimization of different propagating technique and time period to enhance higher success rate in propagation of low chill peach cv. ‘Shan-e-Punjab. J Plant Dev Sci 12:99–103
Soodan AS, Wafri BA, Kaul AK (1994) Peach diversity in Kashmir. Indian Hortic 32:1–44
Thakur P (2021) Effect of time of planting and growing media on stem cuttings of kiwifruit (Actinidia deliciosa chev.) under polyhouse. Dissertation, Dr YSP University of Horticulture & Forestry, Solan. https://krishikosh.egranth.ac.in/handle/1/5810176649
Trinchera A, Pandozy G, Rinaldi S, Crinò P, Temperini O et al (2013) Graft union formation in artichoke grafting onto wild and cultivated cardoon: An anatomical study. J Plant Physiol 170:1569–1578. https://doi.org/10.1016/j.jplph.2013.06.018
Tsipouridis C, Thomidis T (2005) Effect of 14 peach rootstocks on the yield fruit quality mortality girth expansion and resistance to frost damages of ‘May Crest’ peach variety and their susceptibility on Phytophthora citrophthora. Sci Hortic 103:421–428. https://doi.org/10.1016/j.scienta.2004.08.004
Usman MG, Rafii MY, Martini MY, Oladosu Y, Kashiani P (2017) Genotypic character relationship and phenotypic path coefficient analysis in chilli pepper genotypes grown under tropical condition. J Sci Food Agric 97:1164–1171. https://doi.org/10.1002/jsfa.7843
Vatankhah M, Jafarpour M, Shams D (2015) Effect of time method of budding and type of scion on bud take of sour cherry scions on to ‘Mahaleb’ rootstocks. Int J Agron Agric Res 6:233–239
Wareing PF, Hanney CEA, Digby J (1964) The role of endogenous hormones in cambial activity and xylem differentiation. In: Zimmerman MH (ed) The formation of wood in forest trees. Academic Press, New York, pp 323–344 https://doi.org/10.1016/B978-1-4832-2931-7.50023-9
Xu F, Chu C, Xu Z (2020) Effects of different fertilizer formulas on the growth of loquat rootstocks and stem lignifications. Sci Rep 10:1033. https://doi.org/10.1038/s41598-019-57270-5
Zheng Y, Crawford GW, Chen X (2014) Archaeological evidence for peach (Prunus persica) cultivation and domestication in China. Plos One 9:106595. https://doi.org/10.1371/journal.pone.0106595
Acknowledgements
The authors are thankful to the Professor and Head, Department of Fruit Science and Director of Research, Dr. YS Parmar University of Horticulture and Forestry, Solan (India), for providing the necessary facilities to carry out this experiment.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
P. Verma, N.C. Sharma, P. Verma, U. Sharma and S. Thakur declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Verma, P., Sharma, N.C., Verma, P. et al. Studies on the Production of Budded Nursery Plants in Stool Beds of ‘Rubira’ Peach (Prunus persica [Linnaeus] Batsch). Applied Fruit Science (2024). https://doi.org/10.1007/s10341-024-01094-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10341-024-01094-9