Abstract
Endosymbiosis is very common between bacteria and insects, and it has been deeply studied for over a century on model insects such as Bactrocera oleae, the key pest of the olives. It was demonstrated that “Candidatus Erwinia dacicola” is the main component of its midgut bacterial communities, acting a fundamental role in the fly’s nutrition process and thus on its fitness. In this study, Trichoderma secondary metabolites have been used to treat olive fruit fly in order to alter the “Ca. Erwinia dacicola” titer and to assess the subsequent effects on its host. The selected metabolites, 6-pentyl-α-pyrone and harzianic acid, directly affect the insect’s fitness also on the subsequent generation, but not always in a concentration-dependent manner. Aside from the direct effects, the treatments also showed a modification of the bacterial titer. Therefore, real-time qPCRs were carried out on wild individual flies highlighting natural variations of the symbiont presence and activity during the seasons. The data obtained suggest that bioactive fungal metabolites can be formulated for direct or indirect control strategies of B. oleae in integrated pest management programs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bactrocera oleae (Diptera: Tephritidae) (Rossi), the olive fruit fly (OLF), can cause significant losses in olive production and is considered a key pest of wild and cultivated olive trees worldwide (Montiel Bueno and Jones 2002; Ordano et al. 2015; Daane and Johnson 2010). The trophic activity of the larvae and the oviposition punctures of the adults interfere with the biochemical composition of the olives, altering their quality and quantity (Corrado et al. 2023; Gómez-Caravaca et al. 2008; Malheiro et al. 2015; Pereira et al. 2004). The common control strategies concern the use of traps, biological control and chemicals (Daane and Johnson 2010; Lantero et al. 2023), even if it has changed over time (Nestel et al. 2016) due to the ongoing climate change (Caselli and Petacchi 2021). The rise in insect resistance and the risk to ecosystems resulted in a reduction of accepted synthetic molecules and stricter registration requirements (Siden-Kiamos et al. 2022). Recently, researchers have focused their efforts on the modification of pest microbiomes as a novel control strategy (Qadri et al. 2020). The change of microbial pest symbionts limits the adaptability of the hosts to the seasonal and nutritional trends due to the climatic circumstances (Ferguson et al. 2018; Higuita Palacio et al. 2021). Among the whole microbiome of the OLF, the obligate bacterial endosymbiont, described for the first time by Petri (1909), "Candidatus Erwinia dacicola" is the most important and abundant species (Ben-Yosef et al. 2010; Capuzzo et al. 2005; Estes et al. 2012, 2009; Savio et al. 2012). This symbiont is present in adults in the oesophageal bulb (OB), a cephalic organ connected to the pharynx, where it can rapidly multiply (Caleca et al. 2012; Sacchetti et al. 2008). The activity of "Ca. Erwinia dacicola" is fundamental in the development of the larval stages and high mortality has been registered with the administration of antibiotics or copper oxychloride (Belcari et al. 2005; Ben-Yosef et al. 2015; Bigiotti et al. 2019a, b; Estes et al. 2012; Sinno et al. 2020; Perin et al. 2023). On this biological basis, it is possible to hypothesize control strategies of the OLF by modifying the development and action of the endosymbionts.
Microorganisms, and in particular fungi, produce secondary metabolites (SMs) through highly specific and sometimes unusual pathways (Demain and Fang 2000; Herbert 1994; Vinale et al. 2021; Zeilinger et al. 2016). Fungal strains of the genus Trichoderma are well known producers of SMs with various biological activities like biocontrol against phytopathogenic fungi (Lorito et al. 2010; Nicoletti and Vinale 2018) and biostimulants that boost crop nutrition, growth and stress response (Hermosa et al. 2013; Marra et al. 2018; Mayo-Prieto et al. 2019; Sabzi-Nojadeh et al. 2024; Vinale et al. 2009; Vinale and Sivasithamparam 2020). 6-pentyl-α-pyrone (6PP) is a Trichoderma metabolite characterized by a strong and peculiar coconut flavor and strongly correlated to the biocontrol activities of the producing strains (Mutawila et al. 2016). This compound is involved also on antibiosis, microbial competition, induction of plant resistance, and plant promotion (Marra et al. 2019; Vinale et al. 2012; Ismaiel and Ali 2017). Harzianic acid (HA) is a tetramic acid derivatives with chelating properties on different metals (De Tommaso et al. 2020; Staropoli et al. 2023; Tommaso et al. 2021; Vinale et al. 2013), able to promote plant growth and to inhibit microbial development in a concentration-dependent manner (Vinale and Sivasithamparam 2020; Vinale et al. 2009; Sawa et al. 1994; Healy et al. 2015; De Filippis et al. 2021). Recent studies were conducted to evaluate the beneficial effects of fungal metabolites on B. oleae, such as viridiol from T. viride (Sinno et al. 2020) and natural compounds extracted from Metarhizium anisopliae (Juibari et al. 2023). In this framework, we aimed to evaluate the OLF adults' fitness and microbiome responses to applications of 6PP and HA, that are known for their antimicrobial activity (Ismaiel and Ali 2017; Sawa et al. 1994). Since the symbiosis helps the host to overcome difficulties and variations of the environment (Ben-Yosef et al. 2015; Siden-Kiamos et al. 2022), a study on seasonal variations of OLF-microbiome was carried out. This will contribute to a better understanding and correlate the SMs effects to the period of administration. Laboratory bioassays and subsequent real-time qPCRs were performed on wild adults for this purpose. Moreover, an additional study was also conducted on the olfactory response of wild adults to the volatile metabolite 6PP.
Materials and methods
Production and isolation of Trichoderma spp. secondary metabolites.
The beneficial fungi Trichoderma harzianum strain M10 and Trichoderma atroviride strain P1 were maintained on potato dextrose agar (PDA; Hi Media, Mumbai, India) at room temperature and sub-cultured bimonthly. 10 Plugs of M10 and P1 from actively growing margins of PDA cultures were inoculated in 5 L flasks containing 1 L of potato dextrose broth (PDB, Hi Media, Mumbai, India). The stationary cultures were incubated for 25 days at 25 °C, then filtered and exhaustively extracted with ethyl acetate. 6PP and HA were isolated as previously described (Vinale et al. 2012, 2009).
Chromatograms of T. harzianum M10 and T. atroviride P1 extracts used for harzianic acid (HA) and 6-penthyl-α-pyrone (6PP) isolation were obtained by LC–MS analysis (Fig. S3). In particular, the total crude extract and isolated metabolites (HA and 6PP) were analyzed using a quadrupole-time of flight (Q-TOF) mass spectrometer (Agilent Technologies, Santa Clara, CA, USA) coupled to a liquid chromatograph (Agilent HP 1260 Infinity Series, Agilent Technologies). Separation was achieved with a Zorbax Extend C-18 column (4.6 × 50 mm, 3.5 µm, Agilent Technologies) at 37 °C. Elution gradient: 0.1% (v/v) formic acid in water (A), and 0.1% (v/v) formic acid in acetonitrile (B); from 5 to 100% B in 6 min, isocratic at 100% B for 2 min; from 100 to 5% B in 2 min (equilibration time: 2 min; flow rate: 0.4 mL/min; injection volume: 7 µL). MassHunter Data Acquisition Software, rev. B.05.01 (Agilent Technologies) was used to set spectrometric parameters (Mayo-Prieto et al. 2019). HA and 6PP isolated form liquid cultures showed the same chromatographic and spectroscopic data of the authentic standards present in our laboratories (Vinale et al. 2008, 2009).
Effects of 6PP and HA on fitness and microbiome responses of OLF adults
New-emerged adults collected from infested olives (cv. Leccino; experimental site UTM: 445,093,878 E; 4,518,644,785 N 33 T) sampled every two weeks were used in the tests. From the same place, a constant availability of fresh green olives was assured, protecting selected branches with different layers as previously described by Jesu et al. (2022). The collected fruits, pupae and adults were placed in a controlled chamber as reported by Jesu et al. (2022). The effects of SMs on the fitness of the adult OLFs exposed were evaluated through ingestion bioassays. The diets offered ad libitum consisted of 300 mg of sucrose solubilized in 200 µl of water (negative control, CNT). Positive controls were obtained using the same sucrose and water diet, in which 100 μg/ml of the antibiotic piperacillin (Sigma) (Piperacillin); 0.5%, 0.1% and 0.02% p/p copper oxychloride (Cu) dissolved (Caleca et al. 2010). The effects of pure harzianic acid (HA) and 6-pentyl-α-pyrone (6PP) are tested at the same concentrations chosen for Cu. The same concentrations of pure HA and 6PP were used as treatments. Five biological replications with 15 males and 15 females were conducted for each test. As water source, a 20 cm diameter cotton ball, soaked in water daily, was placed on the cover net of each cage. During the bioassays, diet consumption, mortality to the exposition, mean number of sterile stings and eggs laid per drupe/female, pre-pupal mortality and sex ratio of the emerged flies were computed as previously described (Jesu et al. 2022).
Following previously established protocols (Jesu et al. 2022), “Ca. Erwinia dacicola” was quantified considering only oesophageal bulbs (OBs) dissected with surgical micro scissors (Fine Science Tools, Foster City, CA, USA) and pins from the adults’ heads. Briefly, the 16 s regions of the “Ca. Erwinia dacicola” rDNA were cloned on Escherichia coli vectors using the TOPO® TA Cloning® Kit (Life Technologies-Thermo Fisher Scientific). The qPCRs were performed using a couple of primers specific for the Ca. E. dacicola 16S rRNA gene: EdEnRev (Estes et al. 2012) and EdF1 (Estes et al. 2009. These primers were previously used by Biogiotti et al. (2019b) in a similar work, for which with a good level of sensitivity was demonstrated even if some aspecific reaction may occur. All the analysis were performed on a StepOne™ Real-Time PCR System (Applied Biosystems) with a reaction mix consisted in 10 μL of SYBR® Green PCR 2X Master Mix (Applied Biosystems, Foster City, CA, USA), 1 μL of each primer (10 ng) and water up to 20 μL. The obtained CTs were then computed to get the actual amounts of the bacterium present in the hosts after the treatments.
Seasonal variability of “Ca. Erwinia dacicola” in field
For four years, during the period of activity of the fly, adult females were monthly collected as they emerged from the pupae collected from the experimental site (UTM: 445,093,878 E; 4,518,644,785 N 33 T), pooled in groups of 5 individuals and immediately stored at − 80 °C. A survey on the seasonal variations of the symbiont in natural conditions was completed on ten pools per month following the same methodology reported in Sect. 2.2.
Olfactory response of OLF to the volatile metabolite, 6-pentyl-α-pyrone.
The 6PP olfactometry assay was carried out in a Y-glass olfactometer (Fatouros et al. 2012; Ponzio et al. 2016). The lack of volatility of HA made it less indicated to be used in such assays. 12 fresh green olives were used as the positive control, corresponding to 90% of the attractiveness of OLF adults (n. 60 ♂ and 60 ♀) in pre-tests. In the other arm, the same number of olives were used in combination with 5 μl of subsequential dilutions (10–1, 10–2, and 10–3 M) of 6PP dropped on a 9 cm2 square of filter paper (Whatman®, No.4). All the trials were carried out on newly emerged adults (60 ♂ and 60 ♀ per test). The olfactometry room was air-conditioned (24 ± 2 °C; 60 ± 10% RH) and the tests were conducted at times when the biorhythm of the species is known to be positive (Bertolini et al. 2018). As a consequence of our results, a further trial was carried out using only 6PP 10–1 M without olives, to assess whether the insect response recorded in the trial was due to the combinations of the VOCs mixture present or only to the metabolite. One fly was placed in the Y-shaped tube and its choice was recorded after 5 min. The behavior was noted as indecision when the fly did not choose any arm. Between each fly’s run, the Y-glass tube was disassembled, cleaned, and reassembled with inverted positions of the arms, then left under a clean airflow for at least 5 min.
Statistical analysis
The biological and molecular data recorded were analyzed with the PRISM GraphPad 6.01 (GraphPad Software Inc., San Diego, CA, USA) and the SPSS (IBM Corp. Released 2021, version 28.0. Armonk, NY: IBM Corp). One-way ANOVA and post hoc Student–Newman–Keulsa,b tests were performed on the recorded data of mortality, sterile stings, eggs laid, F1 and on the “Ca. Erwinia dacicola” titer obtained after the laboratory trials and on its seasonal occurrence on the field collected adults (p value < 0.05). A χ2 test was applied for the olfactometry trials using the results of the controls as the expected ones (p value = 0.05).
Results
Effects of 6-pentyl-α-pyrone and harzianic acid on Bactrocera oleae fitness
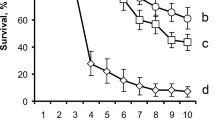
The diet consumptions were similar among the treatments (Supplementary Fig. 1). 6PP at 0.02%, Cu at 0.5% and 0.1%, and the antibiotic piperacillin caused similar direct mortality to OLF (~ 50%, Fig. 1). The lowest mean mortality was recorded with HA at 0.5% (16.7%), which appeared to have beneficial effects on the treated population (Fig. 1). Tests conducted with HA at 0.1% and 0.02% showed similar effects of the control, while the effects of all the Cu and the 0.02% 6PP treatments did not differ from the positive control Piperacillin (Fig. 1). Females appeared to be more sensitive to the compound treatments (data not shown).
Effects of harzianic acid, 6-pentyl-α-pyrone and copper oxychloride on Bactrocera oleae expressed as mean mortality (%). The data were recorded after 14 days of continuous exposition ad libitum of the insects (ingestion bioassays). CNT ( ) = negative control; Piperacillin (
) = negative control; Piperacillin ( = positive control; Cu (
= positive control; Cu ( ) = Copper oxychloride, positive control; HA (
) = Copper oxychloride, positive control; HA ( = Harzianic a
= Harzianic a cid; 6PP () = 6-pentyl-α-pyrone. On the X axis are shown the concentrations used: 0.5% (p/p), 0.1% (p/p) and 0.02% (p/p). Piperacillin is reported for the only concentration used: 100 µg/ml. Each bar represents the mean ± standard deviations mean of five independent experiments. Different letters (a, b, c, d) mean significative differences (ANOVA and Student–Newman–Keuls tests, p value < 0.05)
cid; 6PP () = 6-pentyl-α-pyrone. On the X axis are shown the concentrations used: 0.5% (p/p), 0.1% (p/p) and 0.02% (p/p). Piperacillin is reported for the only concentration used: 100 µg/ml. Each bar represents the mean ± standard deviations mean of five independent experiments. Different letters (a, b, c, d) mean significative differences (ANOVA and Student–Newman–Keuls tests, p value < 0.05)
All the treatments altered the OLF’s reproductive activity, measured through the number of sterile stings and oviposition rates. Interestingly, all the treatments strongly increased the number of sterile stings (Supplementary Fig. 2) while reducing the number of laid eggs. The strongest effect was obtained with Cu at 0.5% (Fig. 2).
Effects of harzianic acid, 6-pentyl-α-pyrone and copper oxychloride on Bactrocera oleae expressed as number of laid eggs per female. CNT ( ) = negative control; Piperacillin (
) = negative control; Piperacillin ( = positive control; Cu (
= positive control; Cu ( ) = Copper oxychloride positive control; HA (
) = Copper oxychloride positive control; HA ( )= Harzianic acid; 6PP (
)= Harzianic acid; 6PP ( ) = 6-pentyl-α-pyrone. On the X axis are shown the concentrations used: 0.5% (p/p), 0.1% (p/p) and 0.02% (p/p). Piperacillin is reported for the only concentration used: 100 µg/ml. Each bar represents the mean ± standard deviations mean of five independent experiments. Different letters (a, b, c, d) mean significative differences (ANOVA and Student–Newman–Keuls tests, p value < 0.05)
) = 6-pentyl-α-pyrone. On the X axis are shown the concentrations used: 0.5% (p/p), 0.1% (p/p) and 0.02% (p/p). Piperacillin is reported for the only concentration used: 100 µg/ml. Each bar represents the mean ± standard deviations mean of five independent experiments. Different letters (a, b, c, d) mean significative differences (ANOVA and Student–Newman–Keuls tests, p value < 0.05)
The effects of all SMs on the preimaginal development of the fly were evident (Fig. 3). The eggs laid after Cu at 0.5% and piperacillin administration did not hatch. The Cu-expositions strongly impacted the F1, where half of the observed individuals stopped their development as pupae (Fig. 3).
Number of living emerged F1 individuals of Bactrocera oleae per olive, after treatments with harzianic acid, 6-pentyl-α-pyrone and copper oxychloride. CNT ( ) = negative control; piperacillin (
) = negative control; piperacillin ( ) = positive control; Cu (
) = positive control; Cu ( ) = copper oxychloride, positive control; HA (
) = copper oxychloride, positive control; HA ( ) = harzianic acid; 6PP (
) = harzianic acid; 6PP ( ) = 6-pentyl-α-pyrone. On the X axis are shown the concentrations used: 0.5% (p/p), 0.1% (p/p) and 0.02% (p/p). Piperacillin is reported for the only concentration used: 100 µg/ml. Each bar represents the mean ± standard deviations mean of five independent experiments. Different letters (a, b, c, d) mean significative differences (ANOVA and Student–Newman–Keuls tests, p value < 0.05)
) = 6-pentyl-α-pyrone. On the X axis are shown the concentrations used: 0.5% (p/p), 0.1% (p/p) and 0.02% (p/p). Piperacillin is reported for the only concentration used: 100 µg/ml. Each bar represents the mean ± standard deviations mean of five independent experiments. Different letters (a, b, c, d) mean significative differences (ANOVA and Student–Newman–Keuls tests, p value < 0.05)
The number of puparia (data not shown) and new adults (Fig. 3) were significantly higher for the control than for all the other treatments. The sex ratio estimated on the weighted averages of the exposed olives showed a majority of emerged females in most of the tests, particularly after the 6PP 0.1% application (3 ♀ per 1 ♂) (Supplementary Table 1).
Treatments with 6-pentyl-α-pyrone and harzianic acid affect the Bactrocera oleae symbiont “Candidatus Erwinia dacicola”
The analysis of the symbiont titer did not highlight significant differences (p > 0.05), even if there is a dose-dependent reduction trend, which is pointed up with the administration of HA, 6PP, and Cu. An exception is the administration of 0.1% Cu (Fig. 4), where there is an increase in the presence of “Ca. Erwinia dacicola”.
Amount of “Ca. Erwinia dacicola” in oesophageal bulbs of B. oleae after treatments with harzianic acid, 6-pentyl-α-pyrone and copper oxychloride, expressed in number of copies. The bacterium was detected by Real-Time qPCRs. CNT ( ) = negative control; piperacillin (
) = negative control; piperacillin ( ) = positive control; Cu (
) = positive control; Cu ( ) = copper oxychloride positive control; HA (
) = copper oxychloride positive control; HA ( ) = harzianic acid; 6PP (
) = harzianic acid; 6PP ( ) = 6-pentyl-α-pyrone. On the X axis are shown the concentrations used: 0.5% (p/p), 0.1% (p/p) and 0.02% (p/p). Piperacillin is reported for the only concentration used: 100 µg/ml. Each bar represents the mean ± standard deviations mean of five independent experiments. No significative differences were recorded (ANOVA and Student–Newman–Keuls tests, p value > 0.05). Solid (HA), dashed (6PP) and dotted (Cu) lines represent the linear trend of the treatments
) = 6-pentyl-α-pyrone. On the X axis are shown the concentrations used: 0.5% (p/p), 0.1% (p/p) and 0.02% (p/p). Piperacillin is reported for the only concentration used: 100 µg/ml. Each bar represents the mean ± standard deviations mean of five independent experiments. No significative differences were recorded (ANOVA and Student–Newman–Keuls tests, p value > 0.05). Solid (HA), dashed (6PP) and dotted (Cu) lines represent the linear trend of the treatments
Seasonal variability of “Ca. Erwinia dacicola” in field
The amount of “Ca. Erwinia dacicola” reached similar values during the same periods of the four years of the trial (Fig. 5). Each year, the highest peak of presence was recorded in August, the lowest in September and in December (Fig. 5). The overall recorded climatic conditions are similar considering the month and year of data collection. When differences between humidity and temperatures were recorded during the year, a subsequent and coherent difference in the symbiont titer was recorded (Supplementary Fig. 3).
Titer (number of copies) of “Ca. Erwinia dacicola” in oesophageal bulbs of newly emerged wild adults of B. oleae (10 pools of 5 females) from August (AUG) to December (DEC), in 2018, 2019, 2020 and 2021. Each bar represents the mean ± standard deviations of ten independent pools of five females each. Different letters (a, b, c, d) mean significative differences (ANOVA and Student–Newman–Keuls tests, p value < 0.05)
Application of the volatile metabolite 6-pentyl-α-pyrone on Bactrocera oleae: olfactometry tests
The application of 6PP at 10–3 M to the olives did not alter the behavior of the OLFs, and the response was equivalent to the control carried out with only olives (Fig. 6). 6PP at 10–2 and 10–1 M cause a high number of cases of indecision: around 30% of the tested flies, while the remaining are approximately equally distributed between repellency and attractiveness. During the assays, the doubtful flies always showed a peculiar behavior: they remained stationary at the beginning of the tube and frantically cleaned their antennae, mouthparts and legs. Due to this behavior, additional olfactometry tests have been performed using 6PP 10–1 M alone. The data registered with this experiment confirmed this effect on flies (Fig. 6).
Distribution (%) of the choices of the Bactrocera oleae adults (60♀ + 60♂) in terms of attractiveness (black), repulsiveness (white) and indecision (gray) in two-way olfactometry tests with different concentrations of 6-pentyl-α-pyrone. Below the dashed line: AIR versus AIR = control with both the lateral chambers left empty; AIR versus OLIVES = control with 12 olives, attractive, in contraposition with clean air. Over the dashed line: AIR versus 6PP 10–3 + OLIVES = 150 μl of 6PP 10–3 M in combination with 12 fresh green olives and in contraposition to clean air; AIR versus 6PP−2 + OLIVES = 150 μl of 6PP 10–2 M in combination with 12 fresh green olives and in contraposition to clean air; AIR versus 6PP−1 + OLIVES = 150 μl of 6PP 10–1 M in combination with 12 fresh green olives and in contraposition to clean air. AIR versus 6PP−1 = 150 μl of 6PP−1 M were offered in contraposition to clean air. Each bar represents the percentage of choice. For each row, the sum of the white, black and gray bars is equal to 100%. The χ2 tests were carried out using the results recorded in the control tests as expected ones and are displayed separately: versus AIR versus OLVES control (external brackets); versus AIR versus AIR control (internal brackets). * = significance (p value < 0.05)
Discussions
Insect pests are related to microbial consortia (mainly bacteria) that impact the physiology and all the possible multicomponent interactions (Gurung et al. 2019). The information on the composition of the microbiome is considered a crucial point to control specific pest (Gurung et al. 2019).
Insects and their symbionts coevolved for long time and have established dynamic relationships, offering a stable habitat, disposability of nutrients and effective defensive strategies (Moran and Telang 1998; Parker et al. 1997). Among the several known relationships between microorganisms and tephritids the relationship of OLF with “Ca. Erwinia dacicola” is characterized by the survival of the symbiont even during the destructive holometabolic process of the host (Degani and Gordani 2022; Estes et al. 2009). This characteristic makes the bacterium an interesting and persistent target for Integrated Pest Management (IPM) strategies. In this context, it could be useful to explore the use of this symbiont and its characteristics for managing tephritid pests more sustainably and effectively. The biosynthesis of 6PP by selected Trichoderma species is involved in the biocontrol activities of the producing strain against phytopathogenic fungi (Mutawila et al. 2016). This metabolite exhibits various actions in this way, including direct inhibition of spores germination, inhibition of mycelium pigmentation and suppression of hyphal growth (Degani and Gordani 2022; Parker et al. 1997). Noticeably, 6-PP is considered a food-grade volatile compound, which means it meets safety standards for use in food products. As a result, it could potentially be applied in crop protection with a large possibility of applications.
Harzianic acid (HA) is recognized for its involvement in both beneficial activities for the radical uptake of minerals (e.g., iron) and the promotion of growth in biological and ecological contexts. It serves as a key factor that influences the competitive interactions between microorganisms and can promote the growth of certain species while inhibiting the growth of others (Vinale et al. 2009).
The treatments with 6PP and HA influence the phenology of the OLF and similar results have been recorded with the application of essential oils (Rizzo et al. 2020), viridiol (Sinno et al. 2020) and smoke waters (Jesu et al. 2022). The mortality of treated OLF, similar to the control, and similar consumption among the treatments suggest that the tephritid does not detect the presence of the tested compounds in the diet. Further tests will be needed to assess whether the fly chooses diets supplemented with SMs even when it has the opportunity for different choices, to check the better possible formulation for the development of novel IPM technologies. The larger number of sterile punctures after the applications leads to a consequent better preservation of the fruits. The high mortality of the adults obtained with 6PP at 0.02% and Cu at 0.1%, comparable with that determined by the administration of the antibiotic piperacillin, confirmed a direct link between the survival of the fly and the presence of “Ca. Erwinia dacicola” described in the literature (Ben-Yosef et al. 2014, 2015; Capuzzo et al. 2005; Jurkevitch 2011). The antimicrobial activity of 6PP already reported (Ismaiel and Ali 2017) is confirmed in this study and explains the adults’ mortality and the difficulty in completing the larval development of B. oleae (Belcari et al. 2005; Caleca et al. 2010; Sacchetti et al. 2004; Tsolakis et al. 2011; Tzanakakis 1985). Ingestion of Cu at 0.5% reduced the adult emergences, while HA, with a dose-dependent effect, and 6PP determined a large number of sterile punctures. This effect has also been registered with Cu at 0.1% suggesting a direct effect on the microbiota of the host. The unexpected outlier in the molecular outcomes revealing an increase in bacterial titer with treatment with Cu 0.1% could be related to the non-high specificity of the premiers (Bigiotti et al. 2019b) or by an interference resulting from this Cu concentration. The dose-dependent bacteriostatic effect recorded for both HA and 6PP suggests that the use of fungal secondary metabolites acting on the pest symbionts is a promising control tool (Matloub et al. 2021; Pietri and Liang 2020). 6PP has been used also for olfactometer assays since this Trichoderma volatile metabolite in preliminary experiments revealed a good attitude to modify the fly behavior (unpublished data). The OLF responses at different concentrations of 6PP are consistent with what is already reported (Rizzo et al. 2020; Sinno et al. 2020; Jesu et al. 2022). The indecision is usually defined as the insect moving from one arm to the other and is related to the perception of a similar offer. This behavior for the OLF is usually shown when only air was injected into both arms of the olfactometer (Fig. 6, AIR-to-AIR control). The exposition to 6PP at 10–2 and 10–1 M induced the OLFs to continuously cleaning of the antennae, legs and mouth appendages, related to difficulties in the reception of odors or to a paroxysmal repellency. The analysis of the “Ca. Erwinia dacicola” titer after the administrations showed high variability within the replications but with an overall reduction trend. This bacterium is essential for insect development, in particular for its sustenance, both during the larval (Ben-Yosef et al. 2015) and at the adult stage (Ben-Yosef et al. 2010, 2014, 2015). A cyclical variability among the replicates correlated to the starting dates of the bioassays, has been demonstrated during the seasonal recording of the symbiont titer on adults. The higher titers were observed with higher temperatures and lower relative humidity corresponding to the natural conditions during which the OLF is known to slow down the number of generations related to a scarcity of nutrients and drupes to sting (Siden-Kiamos et al. 2022). The ability of the fly to adapt its reproduction rate when bad climatic conditions occur is already reported (Tzanakakis 2003). Moreover, the highlighted titer cyclical fluctuations can be related to several factors, following climatic and spatiotemporal patterns (Kounatidis et al. 2008) and other endogenous and exogenous variations (Ordano et al. 2015). The high variability among the replicates after the administration of SMs could be due to the seasonal variability of the titer in the OB of wild newly emerged adults we found during all the periods of activity of the fly. These fluctuations highlight how the starting conditions can modify the effectiveness of the treatments.
In conclusion, the data obtained suggest that B. oleae microbiota alteration using microbial metabolites is a promising tool for IPM and the application of Trichoderma-based products, using selected strains and metabolites (i.e., 6PP and HA), can affect the insect biology and improve olive production and quality (Dini et al. 2020; Lombardi et al. 2020).
Authors contributions
We want to thank the partners of the MIUR project LINFA (grant number Linfa 03PE_00026_1) as part of the Ph.D. doctoral scholarship of the Programma Operativo Nazionale Ricerca e Innovazione 2014-2020 (CCI 2014IT16M2OP005), Fondo Sociale Europeo, Azione I.1 “Dottorati Innovativi con caratterizzazione Industriale”, Codice Borsa: DOT178289 – Borsa 3. This study was carried out within the Agritech National Research Center and received funding from the European Union Next-Generation EU (PIANO NAZIONALE DI RIPRESA E RESILIENZA (PNRR)—MISSIONE 4 COMPONENTE 2, INVESTIMENTO 1.4–D.D. 1032 17/06/2022, CN00000022). This manuscript reflects only the authors’ views and opinions, neither the European Union nor the European Commission can be considered responsible for them.
Availability of data and materials
The supporting data for the findings of this paper are available on request from the corresponding author.
References
Belcari A, Sacchetti P, Rosi MC, Del Pianta R (2005) The use of copper products to control the olive fly (Bactrocera oleae) in Central Italy. IOBC/WPRS Bull 28:45–48
Ben-Yosef M, Aharon Y, Jurkevitch E, Yuval B (2010) Give us the tools and we will do the job: symbiotic bacteria affect olive fly fitness in a diet-dependent fashion. Proc r Soc B Biol Sci 277:1545–1552. https://doi.org/10.1098/rspb.2009.2102
Ben-Yosef M, Pasternak Z, Jurkevitch E, Yuval B (2014) Symbiotic bacteria enable olive flies (Bactrocera oleae) to exploit intractable sources of nitrogen. J Evol Biol 27:2695–2705. https://doi.org/10.1111/jeb.12527
Ben-Yosef M, Pasternak Z, Jurkevitch E, Yuval B (2015) Symbiotic bacteria enable olive fly larvae to overcome host defences. R Soc Open Sci. https://doi.org/10.1098/rsos.150170
Bertolini E, Kistenpfennig C, Menegazzi P, Keller A, Koukidou M, Helfrich-Förster C (2018) The characterization of the circadian clock in the olive fly Bactrocera oleae (Diptera: Tephritidae) reveals a Drosophila-like organization. Sci Rep 8:1–12. https://doi.org/10.1038/s41598-018-19255-8
Bigiotti G, Pastorelli R, Belcari A, Sacchetti P (2019a) Symbiosis interruption in the olive fly: effect of copper and propolis on Candidatus Erwinia dacicola. J Appl Entomol 143(4):357–364. https://doi.org/10.1099/ijs.0.63653-0
Bigiotti G, Pastorelli R, Guidi R, Belcari A, Sacchetti P (2019b) Horizontal transfer and finalization of a reliable detection method for the olive fruit fly endosymbiont Candidatus Erwinia Dacicola. BMC Biotechnol 19(2):1–12. https://doi.org/10.1186/s12896-019-0583-x. (PMID: 31847845)
Caleca V, Lo Verde G, Lo Verde V, Palumbo Piccionello M, Rizzo R (2010) Control of Bactrocera oleae and Ceratitis capitata in organic orchards: use of clays and copper products. Acta Hortic 873:227–234. https://doi.org/10.17660/ActaHortic.2010.873.24
Caleca V, Belcari A, Sacchetti P (2012) Lotta alla mosca delle olive in olivicoltura integrata e biologica, pp 27–33
Capuzzo C, Firrao G, Mazzon L, Squartini A, Girolami V (2005) “Candidatus Erwinia dacicola”, a coevolved symbiotic bacterium of the olive fly Bactrocera oleae (Gmelin). Int J Syst Evol Microbiol 55:1641–1647. https://doi.org/10.1099/ijs.0.63653-0
Caselli A, Petacchi R (2021) Climate change and major pests of mediterranean olive orchards: are we ready to face the global heating? InSects 12(9):802. https://doi.org/10.3390/insects12090802
Corrado G, Mataffo A, Garonna AP, Rao R, Basile B (2023) Investigation of fruit growth patterns, olive fly Bactrocera oleae (Rossi) infestation, and genetic diversity in Italian olive cultivars. Appl Sci 13(17):9929. https://doi.org/10.3390/app13179929
Daane KM, Johnson MW (2010) Olive fruit fly: Managing an ancient pest in modern times. Annu Rev Entomol 55:151–169. https://doi.org/10.1146/annurev.ento.54.110807.090553
De Filippis A, Nocera FP, Tafuri S, Ciani F, Staropoli A, Comite E, Bottiglieri A, Gioia L, Lorito M, Woo SL, Vinale F, De Martino L (2021) Antimicrobial activity of harzianic acid against Staphylococcus pseudintermedius. Nat Prod Res 35(23):5440–5445. https://doi.org/10.1080/14786419.2020.1779714
De Tommaso G, Salvatore MM, Nicoletti R, DellaGreca M, Vinale F, Bottiglieri A, Staropoli A, Salvatore F, Lorito M, Iuliano M, Andolfi A (2020) Bivalent metal-chelating properties of harzianic acid produced by Trichoderma pleuroticola associated to the gastropod Melarhaphe neritoides. Molecules. https://doi.org/10.3390/molecules25092147
Degani O, Gordani A (2022) New antifungal compound, 6-pentyl-α-pyrone, against the maize late wilt pathogen, Magnaporthiopsis maydis. Agronomy. https://doi.org/10.3390/agronomy12102339
Demain AL, Fang A (2000) The natural functions of secondary metabolites. In: Fletcher A (ed) History of modern biotechnology I. Advances in biochemical engineering/biotechnology. Springer, Berlin, pp 1–39
Dini I, Graziani G, Fedele FL, Sicari A, Vinale F, Castaldo L, Ritieni A (2020) Effects of trichoderma biostimulation on the phenolic profile of extra-virgin olive oil and olive oil by-products. Antioxidants 9:1–12. https://doi.org/10.3390/antiox9040284
Estes AM, Hearn DJ, Bronstein JL, Pierson EA (2009) The olive fly endosymbiont, “Candidatus Erwinia dacicola”, switches from an intracellular existence to an extracellular existence during host insect development. Appl Environ Microbiol 75:7097–7106. https://doi.org/10.1128/AEM.00778-09
Estes AM, Hearn DJ, Burrack HJ, Rempoulakis P, Pierson EA (2012) Prevalence of “Candidatus Erwinia dacicola” in wild and laboratory olive fruit fly populations and across developmental stages. Environ Entomol 41:265–274. https://doi.org/10.1603/EN11245
Fatouros NE, Lucas-Barbosa D, Weldegergis BT, Pashalidou FG, van Loon JJA, Dicke M, Harvey JA, Gols R, Huigens ME (2012) Plant volatiles induced by herbivore egg deposition affect insects of different trophic levels. PLoS ONE. https://doi.org/10.1371/journal.pone.0043607
Ferguson LV, Dhakal P, Lebenzon JE, Heinrichs DE, Bucking C, Sinclair BJ (2018) Seasonal shifts in the insect gut microbiome are concurrent with changes in cold tolerance and immunity. Funct Ecol 32:2357–2368. https://doi.org/10.1111/1365-2435.13153
Gómez-Caravaca AM, Cerretani L, Bendini A, Segura-Carretero A, Fernández-Gutiérrez A, Del Carlo M, Compagnone D, Cichelli A (2008) Effects of fly attack (Bactrocera oleae) on the phenolic profile and selected chemical parameters of olive oil. J Agric Food Chem 56:4577–4583. https://doi.org/10.1021/jf800118t
Gurung K, Wertheim B, Falcao Salles J (2019) The microbiome of pest insects: it is not just bacteria. Entomol Exp Appl 167:156–170. https://doi.org/10.1111/eea.12768
Healy AR, Vinale F, Lorito M, Westwood NJ (2015) Total synthesis and biological evaluation of the tetramic acid based natural product harzianic acid and its stereoisomers. Org Lett 17(3):692–695. https://doi.org/10.1021/ol503717r
Herbert BR (1994) The biosynthesis of secondary metabolites, 2nd edn. Springer, Amsterdam. https://doi.org/10.1007/978-94-009-5833-3
Hermosa R, Rubio MB, Cardoza RE, Nicolás C, Monte E, Gutiérrez S (2013) The contribution of Trichoderma to balancing the costs of plant growth and defense. Int Microbiol 16:69–80. https://doi.org/10.2436/20.1501.01.181
Higuita Palacio MF, Montoya OI, Saldamando CI, García-Bonilla E, Junca H, Cadavid-Restrepo GE, Moreno-Herrera CX (2021) Dry and rainy seasons significantly alter the gut microbiome composition and reveal a key Enterococcus sp. (Lactobacillales: Enterococcaceae) core component in Spodoptera frugiperda (Lepidoptera: Noctuidae) corn strain from Northwestern Colombia. J Insect Sci. https://doi.org/10.1093/jisesa/ieab076
Ismaiel AA, Ali DMI (2017) Antimicrobial properties of 6-pentyl-α-pyrone produced by endophytic strains of Trichoderma koningii and its effect on aflatoxin B1 production. Biologia 72(12):1403–1415. https://doi.org/10.1515/biolog-2017-0173
Jesu G, Laudonia S, Bonanomi G, Flematti G, Germinara SG, Pistillo M, Giron D, Bézier A, Vinale F (2022) Biochar-derived smoke waters affect Bactrocera oleae Behavior and control the olive fruit fly under field conditions. Agronomy 12:2834. https://doi.org/10.3390/agronomy12112834
Juibari MM, Zibaee A, Abbasi Mozhdehi MR (2023) Toxicity and physiological interruptions of a proteinaceous toxin from Metarhizium anisopliae against the olive fruit pest, Bacterocera oleae (Diptera: Tephritidae). Comp Biochem Physiol c: Toxicol Pharmacol 271:109681. https://doi.org/10.1016/j.cbpc.2023.109681
Jurkevitch E (2011) Riding the Trojan horse: combating pest insects with their own symbionts. Microb Biotechnol 4:620–627. https://doi.org/10.1111/j.1751-7915.2011.00249.x
Kounatidis I, Papadopoulos NT, Mavragani-Tsipidou P, Cohen Y, Tertivanidis K, Nomikou M, Nestel D (2008) Effect of elevation on spatio-temporal patterns of olive fly (Bactrocera oleae) populations in northern Greece. J Appl Entomol 132:722–733. https://doi.org/10.1111/j.1439-0418.2008.01349.x
Lantero E, Matallanas B, Callejas C (2023) Current status of the main olive pests: useful integrated pest management strategies and genetic tools. Appl Sci 13:12078. https://doi.org/10.3390/app132112078
Lombardi N, Caira S, Troise AD, Scaloni A, Vitaglione P, Vinale F, Marra R, Salzano AM, Lorito M, Woo SL (2020) Trichoderma applications on strawberry plants modulate the physiological processes positively affecting fruit production and quality. Front Microbiol 11:1–17. https://doi.org/10.3389/fmicb.2020.01364
Lorito M, Woo SL, Harman GE, Monte E (2010) Translation research on Trichoderma: from ‘omics to the field. Annu Rev Phytopathol 48:395–417. https://doi.org/10.1146/annurev-phyto-073009-114314
Malheiro R, Casal S, Baptista P, Pereira JA (2015) Physico-chemical characteristics of olive leaves and fruits and their relation with Bactrocera oleae (Rossi) cultivar oviposition preference. Sci Hortic 194:208–214. https://doi.org/10.1016/j.scienta.2015.08.017
Marra R, Vinale F, Cesarano G, Lombardi N, D’Errico G, Crasto A, Mazzei P, Piccolo A, Incerti G, Woo SL, Scala F, Bonanomi G (2018) Biochars from olive mill waste have contrasting effects on plants, fungi and phytoparasitic nematodes. PLoS ONE. https://doi.org/10.1371/journal.pone.0198728
Marra R, Lombardi N, D’Errico G, Troisi J, Scala G, Vinale F, Woo SL, Bonanomi G, Lorito M (2019) Application of trichoderma strains and metabolites enhances soybean productivity and nutrient content. J Agric Food Chem 67:1814–1822. https://doi.org/10.1021/acs.jafc.8b06503
Matloub AA, Maamoun AA, Abdel-Aziz NF, Samour EA, El-Rafie HM (2021) Eco-friendly secondary metabolites from conyza dioscoridis against Spodoptera littoralis. Egypt J Chem 64:341–357. https://doi.org/10.21608/EJCHEM.2020.38753.2798
Mayo-Prieto S, Marra R, Vinale F, Rodríguez-González Á, Woo SL, Lorito M, Gutiérrez S, Casquero PA (2019) Effect of Trichoderma velutinum and Rhizoctonia solani on the metabolome of bean plants (Phaseolus vulgaris L). Int J Mol Sci. https://doi.org/10.3390/ijms20030549
Montiel Bueno A, Jones OT (2002) Alternative methods for controlling the olive fly, Bactrocera oleae, involving semiochemicals. In: Use pheromones other semiochemicals in Integrated Protection IOBC wprs Bull, vol. 25, p 25
Moran NA, Telang A (1998) Bacteriocyte-associated symbiotic of insects: a variety of insect groups harbor ancient prokaryotic endosymbionts. Bioscience 48:295–304. https://doi.org/10.2307/1313356
Mutawila C, Vinale F, Halleen F, Lorito M, Mostert L (2016) Isolation, production and in vitro effects of the major secondary metabolite produced by Trichoderma species used for the control of grapevine trunk diseases. Plant Pathol 65:104–113. https://doi.org/10.1111/ppa.12385
Nestel D, Rempoulakis P, Yanovski L, Miranda MA, Papadopoulos NT (2016) The evolution of alternative control strategies in a traditional crop: Economy and policy as drivers of olive fly control. Adv Insect Control Resist Manag. https://doi.org/10.1007/978-3-319-31800-4_4
Nicoletti R, Vinale F (2018) Bioactive compounds from marine-derived Aspergillus, Penicillium Talaromyces and Trichoderma Species. Mar Drugs 16:408. https://doi.org/10.3390/md16110408
Ordano M, Engelhard I, Rempoulakis P, Nemny-Lavy E, Blum M, Yasin S, Lensky IM, Papadopoulos NT, Nestel D (2015) Olive fruit fly (Bactrocera oleae) population dynamics in the eastern mediterranean: influence of exogenous uncertainty on a monophagous frugivorous insect. PLoS ONE 10:e0127798. https://doi.org/10.1371/journal.pone.0127798
Parker SR, Cutler HG, Jacyno JM, Hill RA (1997) Biological activity of 6-pentyl-2h-pyran-2-one and its analogs. J Agric Food Chem 45:2774–2776. https://doi.org/10.1021/jf960681a
Pereira JA, Rui Alves M, Casal S, Oliveira M (2004) Effect of olive fruit fly infestation on the quality of olive oil from Chemlali cultivar during ripening. Ital J Food Sci 3:355–365. https://doi.org/10.1016/j.fct.2010.08.031
Perin C, Martinez-Sañudo I, Carofano I, Mori N, Santoiemma G, Squartini A, Tondello A, Mazzon L (2023) Impairing the development of an olive fly pest by targeting its symbiotic bacteria in egg-infested fruits. Entomol Gen. https://doi.org/10.1127/entomologia/2023/1916
Petri L (1909) Ricerche sopra i batteri Intestinali della Mosca Olearia. Memorie della Regia Stazione di Patologia Vegetale di Roma (in Italian), Roma, pp 1–29
Pietri JE, Liang D (2020) Insecticidal activity of doxycycline against the common bedbug. Antimicrob Agents Chemother 64:1–6. https://doi.org/10.1128/AAC.00005-20
Ponzio C, Cascone P, Cusumano A, Weldegergis BT, Fatouros NE, Guerrieri E, Dicke M, Gols R (2016) Volatile-mediated foraging behaviour of three parasitoid species under conditions of dual insect herbivore attack. Anim Behav 111:197–206. https://doi.org/10.1016/j.anbehav.2015.10.024
Qadri M, Short S, Gast K, Hernandez J, Wong ACN (2020) Microbiome innovation in agriculture: development of microbial based tools for insect pest management. Front Sustain Food Syst 4:1–20. https://doi.org/10.3389/fsufs.2020.547751
Rizzo R, Lo Verde G, Sinacori M, Maggi F, Cappellacci L, Petrelli R, Vittori S, Reza Morshedloo M, Yvette Fofie NGB, Benelli G (2020) Developing green insecticides to manage olive fruit flies? Ingestion toxicity of four essential oils in protein baits on Bactrocera oleae. Ind Crops Prod 143:111884. https://doi.org/10.1016/j.indcrop.2019.111884
Sabzi-Nojadeh M, Pouresmaeil M, Amani M, Younessi-Hamzekhanlu M, Maggi F (2024) Colonization of Satureja hortensis L. (Summer savory) with Trichoderma harzianum alleviates salinity stress via improving physio-biochemical traits and biosynthesis of secondary metabolites. Ind Crops Prod 208:117831. https://doi.org/10.1016/j.indcrop.2023.117831
Sacchetti P, Granchietti A, Landini S, Viti C, Giovannetti L, Belcari A (2008) Relationships between the olive fly and bacteria. J Appl Entomol 132:682–689. https://doi.org/10.1111/j.1439-0418.2008.01334.x
Sacchetti P, Belcari A, Del Pianta R (2004) Utilizzo di prodotti ad azione antibatterica per il controllo della mosca delle olive, in: La Difesa Dai Fitofagi in Condizioni Di Olivicoltura Biologica, Atti Accademia Nazionale Dell’Olivo e Dell’Olio, Spoleto, 29–30 October 2002, pp 23–33
Savio C, Mazzon L, Martinez-Sañudo I, Simonato M, Squartini A, Girolami V (2012) Evidence of two lineages of the symbiont “Candidatus Erwinia dacicola” in Italian populations of Bactrocera oleae (Rossi) based on 16S rRNA gene sequences. Int J Syst Evol Microbiol 62:179–187. https://doi.org/10.1099/ijs.0.030668-0
Sawa R, Mori Y, Iinuma H, Naganawa H, Hamada M, Yoshida S, Furutani H, Kajimura Y, Fuwa T, Takeuchi T (1994) Harzianic acid, a new antimicrobial antibiotic from a fungus. J Antibiot 47(6):731–732. https://doi.org/10.7164/antibiotics.47.731
Siden-Kiamos I, Koidou V, Livadaras I, Skoufa E, Papadogiorgaki S, Papadakis S, Chalepakis G, Ioannidis P, Vontas J (2022) Dynamic interactions between the symbiont Candidatus Erwinia dacicola and its olive fruit fly host Bactrocera oleae. Insect Biochem Mol Biol 146:103793. https://doi.org/10.1016/j.ibmb.2022.103793
Sinno M, Bézier A, Vinale F, Giron D, Laudonia S, Garonna AP, Pennacchio F (2020) Symbiosis disruption in the olive fruit fly, Bactrocera oleae (Rossi), as a potential tool for sustainable control. Pest Manag Sci. https://doi.org/10.1002/ps.5875
Staropoli A, Cuomo P, Salvatore MM, De Tommaso G, Iuliano M, Andolfi A, Tenore GC, Capparelli R, Vinale F (2023) Harzianic acid activity against Staphylococcus aureus and its role in calcium regulation. Toxins (basel). https://doi.org/10.3390/toxins15040237
Tommaso GD, Salvatore MM, Nicoletti R, Dellagreca M, Vinale F, Staropoli A, Salvatore F, Lorito M, Iuliano M, Andolfi A (2021) Coordination properties of the fungal metabolite harzianic acid toward toxic heavy metals. Toxics. https://doi.org/10.3390/toxics9020019
Tsolakis H, Ragusa E, Tarantino P (2011) Control of Bactrocera oleae by low environmental impact methods: NPC methodology to evaluate the efficacy of lure-and-kill method and copper hydroxide treatments. Bull Insectol 64:1–8
Tzanakakis ME (2003) Seasonal development and dormancy of insects and mites feeding on olive: a review. J Zool 52:87–224
Tzanakakis ME (1985) Considerations on the possible usefulness of olive fruit fly symbionticides in integrated control in olive groves. In: Cavalloro R, Crovetti A (eds) Proceedings of integrated control in olive groves CEC7FAO/IOBC International Joint Meeting Pisa, 3–6 April 1984, pp 386–393
Vinale F, Sivasithamparam K (2020) Beneficial effects of Trichoderma secondary metabolites on crops. Phyther Res 34:2835–2842. https://doi.org/10.1002/ptr.6728
Vinale F, Sivasithamparam K, Ghisalberti EL, Marra R, Barbetti MJ, Li H, Woo SL, Lorito M (2008) A novel role for Trichoderma secondary metabolites in the interactions with plants. Physiol Mol Plant Pathol 72:80–86. https://doi.org/10.1016/j.pmpp.2008.05.005
Vinale F, Flematti G, Sivasithamparam K, Lorito M, Marra R, Skelton BW, Ghisalberti EL (2009) Harzianic acid, an antifungal and plant growth promoting metabolite from Trichoderma harzianum. J Nat Prod 72:2032–2035. https://doi.org/10.1021/np900548p
Vinale F, Sivasithamparam K, Ghisalberti EL, Ruocco M, Woo SL, Lorito M (2012) Trichoderma secondary metabolites that affect plant metabolism. Nat Prod Commun. https://doi.org/10.1177/1934578x1200701133
Vinale F, Nigro M, Sivasithamparam K, Flematti G, Ghisalberti EL, Ruocco M, Varlese R, Marra R, Lanzuise S, Eid A, Woo SL, Lorito M (2013) Harzianic acid: a novel siderophore from Trichoderma harzianum. FEMS Microbiol Lett (Oxford Academic). https://doi.org/10.1111/1574-6968.12231
Vinale F, Sivasithamparam K, Zeilinger S, Gutiérrez S (2021) Fungal secondary metabolism. In: Zaragoza Ó, Casadevall A (eds) Encyclopedia of mycology. Elsevier Inc., New York, pp 54–63. https://doi.org/10.1016/B978-0-12-819990-9.00031-7
Zeilinger S, Gruber S, Bansal R, Mukherjee PK (2016) Secondary metabolism in Trichoderma—chemistry meets genomics. Fungal Biol Rev. https://doi.org/10.1016/j.fbr.2016.05.001
Acknowledgements
We want to thank the partners of the MIUR project LINFA (grant number Linfa 03PE_00026_1) as part of the Ph.D. doctoral scholarship of the Programma Operativo Nazionale Ricerca e Innovazione 2014-2020 (CCI 2014IT16M2OP005), Fondo Sociale Europeo, Azione I.1 “Dottorati Innovativi con caratterizzazione Industriale”, Codice Borsa: DOT178289 – Borsa 3. This study was carried out within the Agritech National Research Center and received funding from the European Union Next-Generation EU (PIANO NAZIONALE DI RIPRESA E RESILIENZA (PNRR)—MISSIONE 4 COMPONENTE 2, INVESTIMENTO 1.4–D.D. 1032 17/06/2022, CN00000022). This manuscript reflects only the authors’ views and opinions, neither the European Union nor the European Commission can be considered responsible for them.
Funding
Open access funding provided by Università degli Studi di Napoli Federico II within the CRUI-CARE Agreement. This study was carried out within the Agritech National Research Center and received funding from the European Union Next-Generation EU (PIANO NAZIONALE DI RIPRESA E RESILIENZA (PNRR)—MISSIONE 4 COMPONENTE 2, INVESTIMENTO 1.4—D.D. 1032 17/06/2022, CN00000022).
Author information
Authors and Affiliations
Contributions
GJ: Conceptualization, Methodology, Validation, Investigation, Formal analysis, Resources, Data Curation, Writing-Original Draft, Visualization & Writing-Review; FV: Conceptualization, Methodology, Validation, Resources, Writing- Review & Editing, Supervision; SL: Conceptualization, Methodology, Validation, Resources, Writing-Review & Editing, Supervision; ML: Conceptualization, Methodology, Resources, Fouding, Supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they do not have personal relationships nor known competing interests that could have influenced the work reported in this paper. Indicated commercial names in this publication are just to provide specific information and are not recommendation or endorsement by the authors.
Ethical approval
Not applicable.
Additional information
Communicated by Bin Tang.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jesu, G., Vinale, F., Lorito, M. et al. Trichoderma metabolites 6-pentyl-α-pyrone and harzianic acid affect the reproduction and microbiome of Bactrocera oleae. J Pest Sci (2024). https://doi.org/10.1007/s10340-024-01796-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10340-024-01796-9