Abstract
Benefits provided by urban trees are increasingly threatened by non-native pests and pathogens. Monitoring of these invasions is critical for the effective management and conservation of urban tree populations. However, a shortage of professionally collected species occurrence data is a major impediment to assessments of biological invasions in urban areas. We applied data from iNaturalist to develop a protocol for monitoring urban biological invasions using the polyphagous shot hole borer (PSHB) invasion in two urban areas of South Africa. iNaturalist records for all known PSHB reproductive host species were used together with data on localities of sites for processing plant biomass to map priority monitoring areas for detecting new and expanding PSHB infestations. Priority monitoring areas were also identified using the distribution of Acer negundo, a highly susceptible host that serves as a sentinel species for the detection of PSHB infestations. iNaturalist data provided close to 9000 observations for hosts in which PSHB is known to reproduce in our study area (349 of which were A. negundo). High-priority areas for PSHB monitoring include those with the highest density of PSHB reproductive hosts found close to the 140 plant biomass sites identified. We also identified high-priority roads for visual and baited trap surveys, providing operational guidance for practitioners. The monitoring protocol developed in this study highlights the value of citizen or community science data in informing the management of urban biological invasions. It also advocates for the use of platforms such as iNaturalist as essential tools for conservation monitoring in urban landscapes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Biological invasions are one of the five main threats to biodiversity worldwide (IPBES 2019); they can cause widespread ecological and economic damage, negatively impacting the well-being of people. With more than 57% of the global human population living in urban areas, urbanization is a major driver of global environmental change (United Nations 2018). Cities are well-connected transportation hubs that facilitate species dispersal from one region to another, allowing non-native species to spread across the globe. As a result, urban areas receive more non-native species introductions compared to rural or natural areas (Rebele 1994). With the growing urbanization and global interconnectivity of the world's human population, this rise in the introduction of non-native species will continue to increase (Perrings et al. 2010). Anthropogenic activities such as resource supplementation, increased disturbance, and transportation networks provide many opportunities for non-native species to establish and spread. Indeed, many non-native species exhibit strong adaptability to urban environments, enabling them to invade both urban ecosystems and the natural surroundings of towns and cities (Cadotte et al. 2017; Potgieter and Cadotte 2020). As urbanization expands globally, the significance of invasive species' impact on urban environments is increasing, underscoring the urgency for implementing effective strategies to mitigate their effects.
Urban forests are an important component of cities, providing a range of ecological, economic, and social benefits. They play a vital role in maintaining local biodiversity, regulating the urban microclimate, mitigating the urban heat island effect, enhancing air and water quality, and offering aesthetic and recreational opportunities for urban dwellers (Dwyer et al. 1992; Tyrväinen et al. 2005; Willis and Petrokofsky 2017). However, urban forests are also vulnerable to threats from introduced pests and pathogens, which can have significant negative impacts on biodiversity, ecosystem services, and human well-being (Paap et al. 2017). For example, the invasion of emerald ash borer (Agrilus planipennis) in the eastern United States and Canada has led to mass mortality of urban trees, negatively affecting human health and property values, while resulting in significant financial costs for treating, removing, and replacing high-value trees (Donovan et al. 2013; Schrader et al. 2021). Urban trees often face higher levels of stress than their rural counterparts, including pollution, soil compaction, and limited water and nutrient availability (Pautasso et al. 2015). These stresses can make urban trees more susceptible to pests and pathogens (Raupp et al. 2006). In addition, the proximity of urban trees to one another, coupled with planting regimes that creates monospecific stands, can facilitate the spread of pests and pathogens, allowing diseases to move quickly through tree populations and cause widespread damage. Indeed, pests and pathogens are becoming increasingly prevalent in urban areas where they are causing significant economic, environmental, and social impacts (Raum et al. 2023).
Limited resources allocated for monitoring non-native species and the increasing exposure of sites to non-native species’ introductions highlight the need for effective protocols for identifying sites most vulnerable to their establishment (Epanchin-Niell et al. 2014). The movement of plant biomass, internationally and within regions, is an important pathway for the spread of pests and pathogens (Meurisse et al. 2019). Sites where plant biomass is handled, processed, and/or disposed of (hereafter referred to as plant biomass sites) are at high risk for pest/pathogen introductions and should be prioritised for monitoring. For example, the transport of nursery stock between nurseries and their customers can facilitate the long-distance transmission of pathogens, serving as a vector for their spread (Liebhold et al. 2012; Ghelardini et al. 2017; Simamora et al. 2018; Puertolas et al. 2021). The disposal of infested plant biomass at formal (and informal) waste facilities without following appropriate handling and phytosanitary measures also presents a major dispersal pathway for pests and pathogens; waste landfills can harbour high species richness and abundance of non-native beetle species (Auclair et al. 2005; Rassati et al. 2015). The transportation of firewood by individuals for recreational activities, cooking, or residential heating purposes can also serve as a pathway for the dispersal of wood-inhabiting pests (Solano et al. 2021). These sites and activities, and consequently vectors and pathways of non-native species spread, are ubiquitous in urban landscapes (Padayachee et al. 2017) and must be well understood and incorporated in strategies for monitoring and surveillance for early detection of pests and pathogens.
Monitoring for pest and pathogen invasions is critical for the effective management and conservation of urban tree populations. Monitoring involves regular surveillance and assessment of the abundance, distribution, and impact of invasive species in urban environments. Data collected through monitoring efforts must inform management decisions and be used to prioritize actions aimed at controlling or eradicating invasive species (Potgieter et al. 2021). However, monitoring the urban forest is challenging. For example, many trees are located on private property and, in some countries, landowners can bar access to authorities attempting to gather monitoring data (Bertolino et al. 2021). Monitoring urban trees also requires time, expertise, and resources, which can be in short supply for local governments and organizations (Roman et al. 2013). However, technological advances like remote sensing and environmental DNA (eDNA) and increasingly popular citizen science initiatives (such as eBird and iNaturalist) are revolutionizing our ability to detect and respond to invasions (Allan et al. 2018). Indeed, citizen science initiatives that promote public participation in large-scale, cost-effective biodiversity identification and monitoring can overcome some of these challenges and serve as key resources for the early detection of pest/pathogen infestations.
Conventional, professional survey methods often fail to adequately assess urban landscapes, resulting in a scarcity of species occurrence data (Ballard et al. 2017) which can hinder urban biological invasion assessments. However, the utilization of citizen science platforms has significantly bolstered the quantity and accessibility of species occurrence data from urban areas (Spear et al. 2017). Consequently, there are currently more data on urban species occurrences than ever before (Silvertown 2009). Indeed, some of these citizen science data have made valuable contributions to the management of biological invasions in urban areas (e.g. Crall et al. 2010). The collection of citizen science data not only addresses the challenges of accessing private land (through actively participating landowners also willing to grant access to other citizen scientists) but can also be easily expanded to encompass multiple cities (Spear et al. 2017).
Globally, iNaturalist stands as one of the most widely embraced citizen science platforms for biodiversity, with more than 3.6 million observers having contributed over 180 million observations (https://www.inaturalist.org; accessed 19 December 2023). Observers can upload geolocated records of any species, and the community aids in validating species identifications. iNaturalist has already made substantial contributions to ecological research, e.g. tracking range expansions of non-native species (Agarwal 2017), identifying new and emerging invasive species (Hiller and Haelewaters 2019), and conducting regular surveillance of invasive species (Larson et al. 2020). Nevertheless, the potential of platforms such as iNaturalist for monitoring invasive species in urban environments has not been extensively investigated.
In this study, we use iNaturalist data to develop a monitoring protocol for the early detection and surveillance of urban biological invasions using the polyphagous shot hole borer (PSHB) in two urban centres of South Africa as a case study. iNaturalist records for PSHB-reproductive host tree species were used together with data on plant biomass sites to develop a monitoring protocol for detecting new and expanding PSHB infestations.
Methods
Study taxa
Wood borers are among the most damaging pests worldwide and many species have become successful invaders, often causing substantial economic, ecological, and social costs (Raum et al. 2023). PSHB, Euwallacea fornicatus (Coleoptera: Curculionidae: Scolytinae), is an ambrosia beetle native to Southeast Asia (Stouthamer et al. 2017) that has been unintentionally introduced to many parts of the world, including Australia, California, Hawaii, Israel, South Africa, and many European countries, with varying levels of success in containing or eradicating it (van Rooyen et al. 2021). PSHB has a symbiotic relationship with at least three fungal species: Fusarium euwallaceae, Graphium euwallaceae and Paracremonium pembeum (Lynch et al. 2016), which serve as a food source for the adults and their larvae (Freeman et al. 2013). In susceptible hosts, this leads to Fusarium Dieback, a disease that, in conjunction with the boring activities of the beetle, can cause branch dieback and tree death (Freeman et al. 2016).
PSHB can establish in a wide range of native and non-native tree species in urban, agricultural, and natural landscapes (van Rooyen et al. 2021). Susceptible tree species can serve as reproductive or non-reproductive hosts for the beetle, showing different responses to infestations. Reproductive host trees are those in which both the beetles and the fungus establish, and where the beetles can reproduce. Susceptible reproductive hosts may eventually be killed by the combined impacts of the beetle and the fungus. Non-reproductive host trees are targeted and attacked by the beetle, leading to the establishment of the fungus, but the beetle does not reproduce; in such trees, the fungus may or may not cause disease but is unlikely to kill the tree (Freeman et al. 2013, 2016; Lynch et al. 2016).
Female beetles are dark brown to black and 1.8–2.5 mm long and winged. Male beetles are reddish-brown, smaller than females at 1.5 mm long, and apterous. The female beetle tunnels into the host tree forming galleries in which eggs are laid. Females lay an average of 32 eggs over their lifetime and the first adults that develop from these can appear after only 22 days under ideal conditions (Cooperband et al. 2016). Females exhibit slower development rates compared to males, although they are produced in significantly larger quantities (Cooperband et al. 2016; Umeda and Paine 2019). Generally, females are fertilized by their brothers inside the galleries after which they emerge through the original entrance tunnel and take flight to seek out new hosts. An unmated female can, however, still establish a gallery and lay eggs. These unfertilised eggs all hatch as haploid males. The female can mate with any of these, and then go on to lay diploid female eggs again (Cooperband et al. 2016). This means that a single unmated female can establish a new colony.
The beetles have special structures at the base of their mandibles called mycangia which are used to transport the spores of their fungal symbionts (Freeman et al. 2013). During the process of establishing brood galleries, adult female beetles burrow into trees, introducing fungal spores that colonize the walls of the galleries. These spores then serve as a nourishing food source for both developing larvae and adult beetles. By infiltrating the vascular tissues of trees, F. euwallaceae disrupts the transportation of water and nutrients, leading to branch dieback and tree death, known as Fusarium dieback (Freeman et al. 2013, 2016).
Host trees exhibit varying responses to beetle infestation across different species, leading to noticeable distinctions. Staining, gumming, sugary exudate, and frass (excrement and sawdust) from holes that are about 0.85 mm in diameter on living trees can all be signs of the beetle’s boring activity. The abdomen of the female beetle can often be seen protruding out of the entrance hole through the bark, guarding the developing larvae within the galleries. Advanced beetle infestations and fungal infections on highly susceptible hosts (PSHB-reproductive hosts) will eventually lead to branch dieback and tree mortality. At least 161 tree species, 77 of them native, are known to be attacked in South Africa (FABI 2023). To date, 83 (41 of which are native) tree species have been identified as reproductive hosts including maples (Acer spp.), oaks (Quercus spp.), and willows (native and alien Salix spp.), native coral trees (Erythrina spp.) and bushwillows (Combretum spp.). PSHB also poses a threat to many economically important tree crops including avocado (Persea americana), apple (Malus domestica), and various stone fruit trees (Prunus spp.) (Jones and Paine 2017; de Jager and Roets 2022, 2023)—see Table S1 for a list of known reproductive and non-reproductive host species in South Africa.
PSHB poses a substantial risk to the agricultural, forestry, and urban and natural forest sectors throughout the country (Paap et al. 2018). A study by de Wit et al. (2022) used an ex-ante assessment to predict the cost of the PSHB invasion in South Africa by modelling the potential growth in PSHB populations and forecasting the impact on the net present cost resulting from damage to natural and urban forests, commercial forestry, and the avocado industry over a 10-year period (assuming unmitigated spread). The predicted net cost is 18.45 billion international dollars (Int. $), or 0.66% of South Africa’s GDP for the baseline scenario, with the bulk of the costs coming from the removal of infested urban trees.
Study site
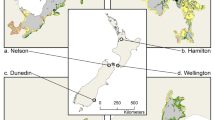
Our study area comprised two urban areas in South Africa’s Western Cape Province: Cape Town (33°55′31″S 18°25′26″E) and Stellenbosch (33°55′12″S 18°51′36″E). Both areas occur at the southwestern tip of Africa within the Cape Floristic Region (CFR), a global biodiversity hotspot (Fig. 1). Fynbos, a fire-prone and fire-adapted shrubland, is the dominant natural vegetation in the area, thriving on sandy and infertile soils (Cowling et al. 1996). The City of Cape Town alone contains 19 endemic vegetation types and over 190 endemic plant species (Holmes et al. 2012). Human population densities are high with 4.8 million and 196 000 people, respectively, in the City of Cape Town and the town of Stellenbosch (City of Cape Town 2021a; Department of Social Development 2021).
Location and land cover of the study area which comprises metropolitan Cape Town and the neighbouring town of Stellenbosch, South Africa (data: South African National Land Cover, Department of Forestry, Fisheries and the Environment 2020). The current known polyphagous shot hole borer distribution within the study area (data: City of Cape Town, updated 19 April 2023), the ambrosia beetle Euwallacea fornicatus and the entry hole and associated fungal stain are also shown
Due to strong participation by community members on platforms such as iNaturalist (https://www.inaturalist.org/) and aided by initiatives such as “The Great Southern Bioblitz” (https://www.inaturalist.org/projects/great-southern-bioblitz-2022-umbrella), the City of Cape Town and the town of Stellenbosch have comprehensive coverage of iNaturalist observations. These two adjacent areas have also recently experienced PSHB invasions (2019 and 2022 for Cape Town and Stellenbosch, respectively). There is also significant concern and interest in PSHB in these urban areas due to the importance of many highly susceptible (particularly non-native) tree species. For example, Stellenbosch is well known for the oak trees (Quercus sp.), some of which are highly susceptible PSHB-reproductive hosts, that have been extensively planted within and around the town (Donald 1978). This town is sometimes referred to as Eikestad (in Afrikaans), or Oak City (direct translation into English). Compared to Stellenbosch, the City of Cape Town has a relatively low tree canopy cover at just six percent (City of Cape Town 2021b). However, both Cape Town and Stellenbosch, the oldest and second oldest towns in South Africa, respectively, have been strongly influenced by European colonization, resulting in the introduction of various non-native species (Pooley 2018). As a result, a large proportion of the urban tree populations comprises non-native species such as Acer negundo, Quercus robur, and Platanus × hispanica. A high proportion of non-native tree species in the urban landscape can increase the probability of introduced pests and pathogens finding and establishing on suitable hosts (Colunga-Garcia et al. 2010).
To delineate the boundary of our study area, the boundary for each metropolitan area was first defined, then merged. For Cape Town’s boundary, the City of Cape Town metropolitan municipal boundary shapefile was obtained from the Municipal Demarcation Board (MDB 2022; updated 25 June 2019). To delineate the boundary for Stellenbosch, a combination of the municipal boundary and quaternary catchments was used. Spatial data for quaternary catchments of South Africa were obtained from the Department of Water and Sanitation (2011). Quaternary catchments are “hydrological units that are hierarchically nested from the primary drainage basin, through to secondary, tertiary, and quaternary level” (Nel et al. 2011). Using ArcGIS 10.7.1, those quaternary catchments which encompassed the urbanized parts of the town of Stellenbosch were selected, clipped, and merged into a single polygon. This polygon was then intersected with the Stellenbosch local municipal boundary polygon. The Cape Town and Stellenbosch boundaries were then merged into one final polygon delineating the study area (Fig. 1).
Data collection and analyses
Occurrence data of PSHB-reproductive hosts within our study area were exported from the project titled “Reproductive hosts at risk of PSHB in South Africa” in iNaturalist (https://www.inaturalist.org/projects/reproductive-hosts-at-risk-of-pshb-in-south-africa; accessed 14 February 2023). Only observations with one or more agreements on identification were included. This project includes all species from the Forestry and Agricultural Biotechnology Institute’s most up-to-date PSHB reproductive host species list (https://www.fabinet.up.ac.za/images/PSHB/8-PSHB_host_list_2022-08-02.pdf; updated on 17 April 2023).
The movement of infested wood is a crucial pathway for the spread of PSHB. For example, the informal urban firewood trade (which serves as a vital energy source and contributes to income stability for numerous urban residents) and the trade in infested nursery material poses a significant risk for long-distance dispersal of PSHB, as firewood and nursery stock are transported over extensive distances, including into natural environments (de Wit et al. 2022). The disposal of infested plant biomass at formal (and informal) waste facilities without following appropriate handling and phytosanitary measures, also presents a major dispersal pathway for PSHB (van Rooyen et al. 2021).
Spatial data for various plant biomass sites were obtained from the City of Cape Town Open Data Portal (https://odp-cctegis.opendata.arcgis.com/) and identified through consultation with local arborists and municipal workers, and our own observations. The sites identified, however, do not represent all plant biomass sites within the study area. For example, many informal dumping sites, firewood distributors and nurseries occur across the study area for which no data currently exist. However, we are confident that the sites included in our analyses provide an appropriate sample to draw reliable conclusions on where best to monitor PSHB infestations. Moreover, the approach developed in this study allows for the seamless integration of these data as they become available—the spatial analyses can be rerun to provide updated maps of priority PSHB monitoring areas.
The Box Elder (Acer negundo) has been identified as one of the most susceptible species to PSHB in South Africa and abroad (e.g. agric.wa.gov.au/borer; FR unpublished data). These “amplifier” trees significantly enhance propagule pressure thereby increasing the risk of future impacts on nearby trees. This, combined with its relative abundance in South African urban tree plantings, the rapid onset of dieback symptoms after infestation and the relative ease in which initial symptoms can be detected, make this species an ideal candidate as sentinel species for the detection of PSHB infestations. As a result, occurrence data for A. negundo trees within our study area were exported from the iNaturalist project titled “Acer negundo in South Africa” (https://www.inaturalist.org/projects/acer-negundo-in-south-africa; Accessed 14 February 2023). Given that relatively few observations of this species have been recorded within our study area, all records (including those with no supporting identifications) were retained for the analysis. This resulted in 349 records for A. negundo of which 22% had no identification agreements. As this species does not pose identification challenges, we consider this approach to be appropriate.
iNaturalist records for PSHB reproductive hosts and A. negundo were mapped separately using ArcGIS 10.7.1. These records were clipped to our study area boundary. All PSHB reproductive host records included observations of A. negundo but given the importance of A. negundo as a sentinel species for monitoring and surveillance, they were mapped separately. A grid (1 × 1 km cell size) covering the extent of the study area was then created using the Create Fishnet geoprocessing tool. A Spatial Join analysis was then used to join the grid to the PSHB reproductive hosts observation shapefile. This was also done separately for the A. negundo shapefile. This produced datasets of densities (records per square kilometre) for PSHB reproductive host trees and A. negundo for the study area. Each grid polygon was then converted to a raster dataset and reclassified.
The Near analysis was used to calculate the distance from each grid cell to the nearest plant biomass sites. Priorities were then assigned based on PSHB reproductive host and A. negundo densities (per km2) and proximity to plant biomass sites. To provide more detailed operational guidance for practitioners, roads within priority grid cells were identified to determine which roads should be prioritised for visual surveys. Spatial data for roads within the study area were obtained from the City of Cape Town data portal and Humanitarian Data Exchange. These shapefiles were merged and clipped to our study area. A Spatial Join analysis was used to join the road shapefile with the PSHB reproductive host densities (with priorities). This was also done separately for the A. negundo shapefile.
Results
Our monitoring protocol, which combined citizen science data on species occurrence and spatial data on urban facilities, identified priority areas which require monitoring for the early detection and management of a highly damaging pest-pathogen complex. Using the invasion of PSHB in two urban centres in the Western Cape, South Africa as case study, priority monitoring areas were determined based on PSHB reproductive host density (using data from iNaturalist) and their proximity to plant biomass sites.
We identified and mapped 140 plant biomass sites across the study area (Fig. 2). These included arborist facilities, firewood distributors, general waste facilities, green waste facilities, landfills, mature tree nurseries, and refuse transfer stations. A total of 8654 records for 55 PSHB reproductive host taxa, and 349 for A. negundo were included in our analyses. The highest number of observations for PSHB reproductive hosts in our study area include Psoralea pinnata (1330, native), Kiggelaria africana (1101, native), Acacia longifolia (875, non-native), Acacia mearnsii (720, non-native), and Acacia melanoxylon (585, non-native). Species with the most observations in the ten highest priority areas (km2) for PSHB monitoring include Psoralea pinnata (272), Acacia longifolia (173), Quercus robur (70, non-native), Acacia melanoxylon (52), and Kiggelaria africana (46). These areas all occur within one kilometre of plant biomass sites (see Table S2).
High-priority areas for PSHB monitoring include those areas with the highest density of reproductive hosts (Fig. 3) close to plant biomass sites. These areas include Cape Town's Central Business District (CBD) and the southern suburbs which lie on the north- and south-eastern slopes of Table Mountain, respectively. Other high-priority areas include most of the urbanized parts of Stellenbosch and dozens of isolated areas in the central and south-western parts of the study area (Fig. 4a).
Densities (number of species’ observations per km2 grid cell) for a) all polyphagous shot hole borer (PSHB) reproductive hosts (n = 8654), and b) Acer negundo (n = 349), a highly susceptible reproductive host. Red = high density, green = low density. Data from iNaturalist (https://www.inaturalist.org/projects/reproductive-hosts-at-risk-of-pshb-in-south-africa” and https://www.inaturalist.org/projects/acer-negundo-in-south-africa, Accessed 14 February 2023)
Priority monitoring areas (1km2 grid cells) based on polyphagous shot hole borer (PSHB) reproductive host density and their proximity to plant biomass sites for a all reproductive hosts, and b Acer negundo. Red = high priority, green = low priority. A visual survey of a high priority Acer negundo monitoring site identified in our protocol detected a new PSHB infestation outside a nursery (photo credit: LJ Potgieter, 18 February 2023). Data from iNaturalist (https://www.inaturalist.org/projects/reproductive-hosts-at-risk-of-pshb-in-south-africa and https://www.inaturalist.org/projects/acer-negundo-in-south-africa, Accessed 14 February 2023)
High priority areas for PSHB monitoring based on the distribution of the highly susceptible, amplifier species A. negundo, include the southern suburbs on the north-eastern slopes of the Table Mountain National Park (Fig. 4b). Other high-priority areas include isolated areas in Cape Town’s CDB and in the town of Stellenbosch. A recent visual survey of a high-priority Acer negundo monitoring site identified in our protocol detected a new PSHB infestation located outside a nursery (Fig. 4b).
Lower priority areas occur in the mountainous parts along the south-western (Cape Peninsula) and south-eastern parts of the study area (Fig. 4). Protected Areas such as the Table Mountain National Park (south-west) and Steenbras Nature Reserve (south-east) make up a significant proportion of these areas. The PSHB reproductive host densities are relatively low and their proximity to plant biomass sites is high. Data deficient (few to no iNaturalist observations) areas include agricultural areas in the north-eastern parts of the study area and the Cape Flats in the central region of the study area. While these areas are characterized by low tree canopy cover and therefore low PSHB reproductive host densities, few of the trees which do occur have been captured in iNaturalist (see Discussion for further details).
Our spatial monitoring prioritisation approach also identified high-priority roads for visual surveys (see Table S2 and 3). This provides practical and operational guidance for practitioners conducting the monitoring. In addition to visual road surveys, our plant biomass site map also serves as a useful guide for site-specific monitoring for PSHB infestations (see Table S4).
Discussion
Monitoring urban biological invasions is critical for preventing the establishment and spread of invasive species, protecting natural resources, minimizing negative impacts on biodiversity and ecosystem services, and ensuring the effectiveness of management efforts (Persad and Tobin 2015). However, there are generally a lack of robust and reliable baseline monitoring data to effectively guide management action (Lindenmayer et al. 2013), especially in urban areas (Potgieter et al. 2021). The increased adoption of citizen science platforms like iNaturalist has resulted in an unprecedented abundance of data on species occurrences in urban areas (Spear et al. 2017). Recent studies have emphasised the demand for detection and management tools, and particularly the integration of citizen science into official pest and pathogen monitoring programmes (e.g. Green et al. 2023).
This study used citizen science data to develop a spatially explicit prioritisation protocol for monitoring pests/pathogens in urban areas, using PSHB invasions in two urban centres in the Western Cape, South Africa, as a case study. It shifts the focus away from ad hoc monitoring approaches and provides urban conservation practitioners with guidance on where to focus monitoring efforts to detect new or expanding infestations.
Priority monitoring areas were determined based on PSHB reproductive host density (using species occurrence data from iNaturalist) and their proximity to plant biomass sites (Fig. 4). Roads within priority grid cells were also identified to provide urban practitioners with guidance on which roads should be prioritised for visual surveys and baited traps (Table S2 and 3). Priority monitoring areas were also identified using the distribution of A. negundo, a highly susceptible PSHB reproductive host that can serve as sentinel species for the detection of PSHB infestations. These priority monitoring areas proved crucial in detecting new infestations in our study area—a recent visual survey of a high-priority monitoring site identified in our protocol detected a new PSHB infestation on an A. negundo tree located outside a nursery (Fig. 4b). This tree has already been felled as per the City of Cape Town’s PSHB management protocol. Our monitoring protocol also allows for the assessment of the effectiveness of control measures implemented such as the removal of highly infested reproductive host trees. By monitoring susceptible reproductive host trees and plant biomass sites close to an initial infestation, it is possible to determine whether control measures are having the desired effect (slowing the rate of spread or reducing propagule pressure) and to adjust management strategies if necessary.
While visual tree surveys by trained monitors are a more accurate and precise way of detecting infestations, deploying monitoring traps at priority sites can also be used. Monitoring traps involve the use of an attractant to bait and capture pests for detection purposes. Although costly, a surveillance trapping protocol could lead to earlier detection of newly established urban tree pests and pathogens or to track the expansion of existing infestations, thereby increasing the probability of achieving successful eradication and mitigating negative impacts (Epanchin-Niell et al. 2014). The monitoring priorities identified in this study can be used to determine where to place monitoring traps. For PSHB traps, Quercivorol (a plant-based chemical lure), can be used to attract PSHB if a beetle is within the local area (Dodge et al. 2017). For example, traps with Quercivorol lures can be placed at intervals along and/or beyond the leading edge of the infestation, targeting high-priority sites. α-Copaene, another potential attractant for PSHB, shows equivalent efficacy to Quercivorol, but using both lures captures significantly more PSHB than either lures alone (Kendra et al. 2017). Traps can also be deployed at plant biomass sites such as landfills, nurseries, green waste processing facilities, and firewood storage and distribution lots. If traps are successful in capturing a pest, depending on the number and location of traps, the relevant management authority can initiate a visual survey to delimit the extent of the infestation.
Effective management of invasive pests in urban areas requires a multidisciplinary approach, involving collaboration between scientists, policymakers, and the public. Monitoring invasive species can raise awareness among the public and stakeholders about the importance of preventing the spread of invasive pests and pathogens, and the need for collaborative management efforts (Gallo and Waitt 2011). Using citizen science data in identifying monitoring priorities increases transparency and therefore public trust in management interventions (Cardoso et al. 2017). This approach can also help raise awareness and encourage action by fostering a sense of ownership and responsibility among participants.
The integration of citizen science with emerging technologies has great promise for conservation management. Many technologies are now being used by professionals for the detection and monitoring of forest pests (de Groot et al. 2023). For example, unmanned aerial vehicles (UAVs) and satellites can provide high-resolution imagery which can enhance the accuracy and granularity of community-contributed data. Equipped with various sensors, including cameras and LiDAR, these data can enable fine-scale monitoring of individual urban trees, canopy structures, and vegetation health. Integrating these data sources with citizen science efforts can enhance the robustness of urban forest monitoring.
Limitations
There are also several limitations to using community science data. iNaturalist observations are submitted voluntarily by users, so the data collected might not be representative of the entire population of a particular species. For example, certain taxa might be more popular among iNaturalist users than others, leading to sampling bias (Di Cecco et al. 2021). While iNaturalist has mechanisms in place to verify observations, such as peer review and expert identification, there is still the potential for misidentification or errors in data entry (Crall et al. 2010). This can result in inaccurate data being collected and shared on the platform. Volunteer contributors also tend have more spatially and temporally concentrated sampling effort compared to professional sampling schemes (Boakes et al. 2010). Locations with more species’ observations are also those with higher numbers of users on the platform. The greater the number of people accessing the application, the greater the amount of available information, generating a possible observation bias.
Participation in citizen science does not always reflect the demographics of the population. Individuals from historically underrepresented groups are less likely to participate resulting in a high proportion of affluent participants (NASEM 2018). For example, in our study area there are a lack of data (few to no iNaturalist observations) in the low-lying areas known as the Cape Flats which comprises mostly low-income townships and informal settlements. While these areas have low tree canopy cover and therefore have low densities of PSHB reproductive hosts, most of the residents here do not have the capacity to capture citizen science data (e.g. Anderson and O’Farrell 2012; Potgieter et al. 2019). As a result, few of the trees which do occur have been captured in iNaturalist. Despite these limitations, iNaturalist provides data in sufficient quantity and quality for research and management applications.
Additional limitations include taxonomic identification challenges based on morphological features (some taxa can only be accurately identified through DNA barcoding), and destructive sampling might be necessary to identify some taxa. Also, citizen scientists might not get access to some urban areas e.g. alongside train tracks or within military bases.
Conclusion
We have developed a spatially explicit prioritisation protocol for monitoring invasive pests/pathogens in urban areas. This monitoring approach can be a critical component of any Integrated Pest Management program, as it allows for early detection of pest infestations and provides valuable data for making informed management decisions.
Utilizing citizen science offers a cost-effective approach for gathering crucial monitoring data while fostering significant citizen engagement and can play a pivotal role in the adaptive management learning process. This study highlights the value of citizen science data in informing the management of urban biological invasions. By leveraging the power of crowdsourcing and overcoming many of the challenges of traditional survey methods, iNaturalist should be considered an essential tool for conservation monitoring in urban landscapes.
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by LJP. The first draft of the manuscript was written by LJP, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request. Various datasets used during this study are publicly accessible and made available under the City of Cape Town’s Open Data Portal, policy no. 27781 (https://odp-cctegis.opendata.arcgis.com/).
References
Agarwal M (2017) First record of Dendronotus orientalis (Baba, 1932) (Nudibranchia: Dendronotidae) in the temperate eastern Pacific. BioInvasions Rec 6:135–138. https://doi.org/10.3391/bir.2017.6.2.08
Allan BM, Nimmo DG, Ierodiaconou D, VanDerWal J, Koh LP, Ritchie EG (2018) Futurecasting ecological research: the rise of technoecology. Ecosphere 9(5):e02163. https://doi.org/10.1002/ecs2.2163
Anderson PM, O’Farrell PJ (2012) An ecological view of the history of the City of Cape Town. Ecol Soc 17(3):28. https://doi.org/10.5751/ES-04970-170328
Auclair AND, Fowler G, Hennessey MK, Hogue AT, Keena M, Lance DR, McDowell RM, Oryang DO, Sawyer AJ (2005) Assessment of the risk of introduction of Anoplophora glabripennis (Coleoptera: Cerambycidae) in municipal solid waste from the quarantine area: a pathway analysis of the risk of spread and establishment. J Econ Entomol 98:47–60. https://doi.org/10.1603/0022-0493-98.1.47
Ballard HL, Robinson LD, Young AN, Pauly GB, Higgins LM, Johnson RF, Tweddle JC (2017) Contributions to conservation outcomes by natural history museum-led citizen science: examining evidence and next steps. Biol Conserv 208:87–97. https://doi.org/10.1016/j.biocon.2016.08.040
Bertolino S, Vimercati G, Paoloni D, Martinoli A, Wauters LA, Genovesi P, La Morgia V (2021) Restricted access to private properties limits management of invasive alien species: a literature review and case studies. J Environ Manage 297:113318. https://doi.org/10.1016/j.jenvman.2021.113318
Boakes EH, McGowan PJ, Fuller RA, Chang-qing D, Clark NE, O’Connor K, Mace GM (2010) Distorted views of biodiversity: spatial and temporal bias in species occurrence data. PLoS Biol 8(6):e1000385. https://doi.org/10.1371/journal.pbio.1000385
Cadotte MW, Yasui SLE, Livingstone S, MacIvor JS (2017) Are urban systems beneficial, detrimental, or indifferent for biological invasion? Biol Invasions 19(12):3489–3503. https://doi.org/10.1007/s10530-017-1586-y
Cardoso AC, Tsiamis K, Gervasini E, Schade S, Taucer F, Adriaens T, Copas K, Flevaris S, Galiay P, Jennings E, Josefsson M (2017) Citizen science and open data: a model for invasive alien species in Europe. Res Ideas Outcomes 3:e14811. https://doi.org/10.3897/rio.3.e14811
City of Cape Town (2021a) Socio-economic profile. Available at: https://www.westerncape.gov.za/provincial-treasury/files/atoms/files/SEP-LG%202021%20-%20City%20of%20Cape%20Town.pdf
City of Cape Town (2021b) Urban Forest Policy. Available at: https://www.capetown.gov.za/councilonline/_layouts/OpenDocument/OpenDocument.aspx?DocumentId=a466936a-2f99-4833-a6fa-38de3889d9ad
Colunga-Garcia M, Haack RA, Magarey RA, Margosian ML (2010) Modeling spatial establishment patterns of exotic forest insects in urban areas in relation to tree cover and propagule pressure. J Econ Entomol 103(1):108–118. https://doi.org/10.1603/ec09203
Cooperband MF, Stouthamer R, Carrillo D, Eskalen A, Thibault T, Cossé AA, Castrillo LA, Vandenberg JD, Rugman-Jones PF (2016) Biology of two members of the Euwallacea fornicatus species complex (Coleoptera: Curculionidae: Scolytinae), recently invasive in the USA, reared on an ambrosia beetle artificial diet. Agric for Entomol 18(3):223–237. https://doi.org/10.1111/afe.12155
Cowling RM, Macdonald IAW, Simmons MT (1996) The Cape Peninsula, South Africa. Biodiver Conserv 5:527–550. https://doi.org/10.1007/BF00137608
Crall AW, Newman GJ, Jarnevich CS, Stohlgren TJ, Waller DM, Graham J (2010) Improving and integrating data on invasive species collected by citizen scientists. Biol Invasions 12:3419–3428. https://doi.org/10.1007/s10530-010-9740-9
de Jager M, Roets F (2022) Pathogenicity of Fusarium euwallaceae towards apple (Malus domestica) and grapevine (Vitis vinifera). In Press, Australas Plant Dis Notes 17:8. https://doi.org/10.1007/s13314-022-00456-0
de Jager M, Roets F (2023) Pathogenicity of Fusarium euwallaceae, symbiont of the polyphagous shot hole borer beetle, to selected stone fruit trees in South Africa. J Plant Pathol 105:5–13. https://doi.org/10.1007/s42161-022-01258-0
de Groot M, Pocock MJ, Bonte J, Fernandez-Conradi P, Valdés-Correcher E (2023) Citizen science and monitoring forest pests: a beneficial alliance? Curr for Rep 9(1):15–32. https://doi.org/10.1007/s40725-022-00176-9
de Wit MP, Crookes DJ, Blignaut JN, de Beer ZW, Paap T, Roets F, van der Merwe C, van Wilgen BW, Richardson DM (2022) An assessment of the potential economic impacts of the invasive polyphagous shot hole borer (Coleoptera: Curculionidae) in South Africa. J Econ Entomol 115(4):1076–1086. https://doi.org/10.1093/jee/toac061
Di Cecco GJ, Barve V, Belitz MW, Stucky BJ, Guralnick RP, Hurlbert AH (2021) Observing the observers: How participants contribute data to iNaturalist and implications for biodiversity science. Bioscience 1:1–10. https://doi.org/10.1093/biosci/biab093
Dodge C, Coolidge J, Cooperband M, Cossé A, Carrillo D, Stouthamer R (2017) Quercivorol as a lure for the polyphagous and Kuroshio shot hole borers, Euwallacea spp. nr. fornicatus (Coleoptera: Scolytinae), vectors of Fusarium dieback. PeerJ 5:e3656. https://doi.org/10.7717/peerj.3656
Donald DGM (1978) The oak at the Cape: its care and maintenance. Veld & Flora. https://doi.org/10.10520/AJA00423203_823
Donovan GH, Butry DT, Michael YL, Prestemon JP, Liebhold AM, Gatziolis D, Mao MY (2013) The relationship between trees and human health: evidence from the spread of the emerald ash borer. Am J Prev Med 44(2):139–145. https://doi.org/10.1016/j.amepre.2012.09.066
Dwyer JF, McPherson EG, Schroeder HW, Rowntree RA (1992) Assessing the benefits and costs of the urban forest. J Arboric. https://doi.org/10.48044/jauf.1992.045
Epanchin-Niell RS, Brockerhoff EG, Kean JM, Turner JA (2014) Designing cost-efficient surveillance for early detection and control of multiple biological invaders. Ecol Appl 24(6):1258–1274. https://doi.org/10.1890/13-1331.1
Forestry and Agricultural Biotechnology Institute (FABI) (2023) The Polyphagous Shothole Borer host list. Available at: https://www.fabinet.up.ac.za/index.php/research-groups/pshb-new/background-info/host-trees
Freeman S, Sharon M, Maymon M, Mendel Z, Protasov A, Aoki T, Eskalen A, O’Donnell K (2013) Fusarium euwallaceae sp. nov. - a symbiotic fungus of Euwallacea sp., an invasive ambrosia beetle in Israel and California. Mycol 105(6):1595–1606. https://doi.org/10.3852/13-066
Freeman S, Sharon M, Dori-Bachash M, Maymon M, Belausov E, Maoz Y, Margalit O, Protasov A, Mendel Z (2016) Symbiotic association of three fungal species throughout the life cycle of the ambrosia beetle Euwallacea nr. fornicatus. Symbiosis 68(1–3):115–128. https://doi.org/10.1007/s13199-015-0356-9
Gallo T, Waitt D (2011) Creating a successful citizen science model to detect and report invasive species. Bioscience 61(6):459–465. https://doi.org/10.1525/bio.2011.61.6.8
Ghelardini L, Luchi N, Pecori F, Pepori AL, Danti R, Della Rocca G, Capretti P, Tsopelas P, Santini A (2017) Ecology of invasive forest pathogens. Biol Invasions 19:3183–3200. https://doi.org/10.1007/s10530-017-1487-0
Green S, Dehnen-Schmutz K, Drakulic J, Eschen R, Orazio C, Douma JC, Lundén K, Colombari F, Jactel H (2023) Awareness, detection and management of new and emerging tree pests and pathogens in Europe: stakeholders’ perspectives. NeoBiota 84:9–40. https://doi.org/10.3897/NEOBIOTA.84.95761
Hiller T, Haelewaters D (2019) A case of silent invasion: citizen science confirms the presence of Harmonia axyridis (Coleoptera, Coccinellidae) in Central America. PLoS ONE. https://doi.org/10.1371/journal.pone.0220082
Holmes PM, Rebelo AG, Dorse C, Wood J (2012) Can Cape Town’s unique biodiversity be saved? Balancing conservation imperatives and development needs. Ecol Soc. https://doi.org/10.5751/ES-04552-170228
Jones ME, Paine TD (2017) Differences among avocado cultivars in susceptibility to polyphagous shot hole borer (Euwallacea sp.). Entomol Exp Appl 163(3):296–304. https://doi.org/10.1111/eea.12577
Kendra PE, Owens D, Montgomery WS, Narvaez TI, Bauchan GR, Schnell EQ, Tabanca N, Carrillo D (2017) α-Copaene is an attractant, synergistic with quercivorol, for improved detection of Euwallacea nr. fornicatus (Coleoptera: Curculionidae: Scolytinae). PLoS ONE 12(6):e0179416. https://doi.org/10.1371/journal.pone.0179416
Larson ER, Graham BM, Achury R, Coon JJ, Daniels MK, Gambrell DK, Jonasen KL, King GD, LaRacuente N, Perrin-Stowe TI, Reed EM (2020) From DNA to citizen science: emerging tools for the early detection of invasive species. Front Ecol Environ 18:194–202. https://doi.org/10.1002/fee.2162
Liebhold AM, Brockerhoff EG, Garrett LJ, Parke JL, Britton KO (2012) Live plant imports: the major pathway for forest insect and pathogen invasions of the US. Front Ecol Environ 10(3):135–143. https://doi.org/10.1890/110198
Lindenmayer DB, Piggott MP, Wintle BA (2013) Counting the books while the library burns: Why conservation monitoring programs need a plan for action. Front Ecol Environ 11(10):549–555. https://doi.org/10.1890/120220
Lynch SC, Twizeyimana M, Mayorquin JS, Wang DH, Na F, Kayim M, Kasson MT, Thu PQ, Bateman C, Rugman-Jones P, Hulcr J (2016) Identification, pathogenicity and abundance of Paracremonium pembeum sp. nov. and Graphium euwallaceae sp. nov.—two newly discovered mycangial associates of the polyphagous shot hole borer (Euwallacea sp.). Mycol 108(2):313–329. https://doi.org/10.3852/15-063
Meurisse N, Rassati D, Hurley BP, Brockerhoff EG, Haack RA (2019) Common pathways by which non-native forest insects move internationally and domestically. J Pest Sci 92:13–27
National Academies of Sciences Engineering and Medicine (NASEM) (2018) Learning through citizen science: enhancing opportunities by design. The National Academies Press, Washington, DC. https://doi.org/10.17226/25183
Nel JL, Driver A, Strydom WF, Maherry A, Petersen C, Hill L, Roux DJ, Nienaber S, Van Deventer H, Swartz S, Smith-Adao LB (2011) Atlas of freshwater ecosystem priority areas in South Africa: Maps to support sustainable development of water resources. WRC Report No. TT 500/11
Paap T, Burgess TI, Wingfield MJ (2017) Urban trees: bridge-heads for forest pest invasions and sentinels for early detection. Biol Invasions 19:3515–3526. https://doi.org/10.1007/s10530-017-1595-x
Paap T, De Beer ZW, Migliorini D, Nel WJ, Wingfield MJ (2018) The polyphagous shot hole borer (PSHB) and its fungal symbiont Fusarium euwallaceae: a new invasion in South Africa. Australas Plant Pathol 47:231–237. https://doi.org/10.1007/s13313-018-0545-0
Padayachee AL, Irlich UM, Faulkner KT, Gaertner M, Procheş Ş, Wilson JR, Rouget M (2017) How do invasive species travel to and through urban environments? Biol Invasions 19:3557–3570. https://doi.org/10.1007/s10530-017-1596-9
Pautasso M, Schlegel M, Holdenrieder O (2015) Forest health in a changing world. Microb Ecol 69:826–842. https://doi.org/10.1007/s00248-014-0545-8
Perrings C, Fenichel E, Kinzig A (2010) Globalization and invasive alien species: trade, pests, and pathogens. In: Perrings C, Mooney HA, Williamson M (eds) Bioinvasions and globalization: ecology, economics, management and policy. Oxford University Press, New York, pp 42–55
Persad AB, Tobin PC (2015) Evaluation of ash tree symptoms associated with emerald ash borer infestation in urban forests. Arboric Urban for 41(2):103–109. https://doi.org/10.48044/jauf.2015.011
Pooley S (2018) The long and entangled history of humans and invasive introduced plants on South Africa’s Cape Peninsula. In: Queiroz AI, Pooley S (eds) Histories of bioinvasions in the Mediterranean. Springer, Cham, pp 219–251. https://doi.org/10.1007/978-3-319-74986-0_10
Potgieter LJ, Cadotte MW (2020) The application of selected invasion frameworks to urban ecosystems. NeoBiota 62:365–386. https://doi.org/10.3897/neobiota.62.50661
Potgieter LJ, Gaertner M, O’Farrell PJ, Richardson DM (2019) Perceptions of impact: invasive alien plants in the urban environment. J Environ Manage 229:76–87. https://doi.org/10.1016/j.jenvman.2018.05.080
Potgieter LJ, Aronson MFJ, Brandt AJ, Cook CN, Gaertner M, Mandrak NE, Richardson DM, Shrestha N, Cadotte MW (2021) Prioritization and thresholds for managing biological invasions in urban ecosystems. Urban Ecosyst 25(1):253–271. https://doi.org/10.1007/s11252-021-01144-0
Puertolas A, Bonants PJM, Boa E, Woodward S (2021) Application of real-time PCR for the detection and quantification of oomycetes in ornamental nursery stock. J Fungi 7:87. https://doi.org/10.3390/jof7020087
Rassati D, Faccoli M, Marini L et al (2015) Exploring the role of wood waste landfills in early detection of non-native wood-boring beetles. J Pest Sci 88:563–572. https://doi.org/10.1007/s10340-014-0639-6
Raum S, Collins CM, Urquhart J, Potter C, Pauleit S, Egerer M (2023) Tree insect pests and pathogens: a global systematic review of their impacts in urban areas. Urban Ecosyst. https://doi.org/10.1007/s11252-022-01317-5
Raupp MJ, Cumming AB, Raupp EC (2006) Street tree diversity in eastern North America and its potential for tree loss to exotic borers. Arboric Urban for 32:297. https://doi.org/10.4236/ojf.2023.131005
Rebele F (1994) Urban ecology and special features of urban ecosystems. Glob Ecol Biogeogr 4:173–187. https://doi.org/10.2307/2997649
Roman LA, McPherson EG, Scharenbroch BC, Bartens J (2013) Identifying common practices and challenges for local urban tree monitoring programs across the United States. Arboric Urban for 39(6):292–299. https://doi.org/10.48044/jauf.2013.038
van Rooyen E, Paap T, DeBeer W, Townsend G, Fell S, Nel WJ, Morgan S, Hill M, Gonzalez A, Roets F (2021) The polyphagous shot hole borer beetle: current status of a perfect invader in South Africa. S Afr J Sci; 117(11–12):1–10. https://doi.org/10.17159/sajs.2021/9736
Schrader G, Baker R, Baranchikov Y, Dumouchel L, Knight KS, McCullough DG, Orlova-Bienkowskaja MJ, Pasquali S, Gilioli G (2021) How does the Emerald Ash Borer (Agrilus planipennis) affect ecosystem services and biodiversity components in invaded areas? EPPO Bulletin 51(1):216–228. https://doi.org/10.1111/epp.12734
Silvertown J (2009) A new dawn for citizen science. Trends Ecol Evol 24:467–471. https://doi.org/10.1016/j.tree.2009.03.017
Simamora AV, Paap T, Howard K, Stukely MJ, Hardy GESJ, Burgess TI (2018) Phytophthora contamination in a nursery and its potential dispersal into the natural environment. Plant Dis 102(1):132–139. https://doi.org/10.1094/PDIS-05-17-0689-RE
Solano A, Rodriguez SL, Greenwood L, Dodds KJ, Coyle DR (2021) Firewood transport as a vector of forest pest dispersal in North America: a scoping review. J Econ Entomol 114(1):14–23. https://doi.org.ez.sun.ac.za/10.1093/jee/toaa278
Spear DM, Pauly GB, Kaiser K (2017) Citizen science as a tool for augmenting museum collection data from urban areas. Front Ecol Evol 5:195. https://doi.org/10.3389/fevo.2017.00086
Stouthamer R, Rugman-Jones P, Thu PQ, Eskalen A, Thibault T, Hulcr J, et al. (2017) Tracing the origin of a cryptic invader: Phylogeography of the Euwallacea fornicatus (Coleoptera: Curculionidae: Scolytinae) species complex. Agric For Entomol 19(4):366–375. https://doi.org/10.1111/afe.12215
Tyrväinen L, Pauleit S, Seeland K, De Vries S (2005) Benefits and uses of urban forests and trees. Urban forests and trees: a reference book. pp 81–114. https://doi.org/10.1007/3-540-27684-X_5
Umeda C, Paine T (2019) Temperature can limit the invasion range of the ambrosia beetle Euwallacea nr. fornicatus. Agric for Entomol 21:1–7. https://doi.org/10.1111/afe.12297
United Nations (2018) World urbanization prospects: the 2018 revision. Department of Economic and Social Affairs, Population Division. https://doi.org/10.18356/b9e995fe-en
Willis KJ, Petrokofsky G (2017) The natural capital of city trees. Science 356(6336):374–376. https://doi.org/10.1126/science.aam9724
Acknowledgements
The authors would like to thank Paul Barker and Phumudzo Ramabulana for their valuable contributions to this work.
Funding
Open access funding provided by Stellenbosch University. LJP and DMR acknowledge support from the DST-NRF Centre of Excellence for Invasion Biology. DMR acknowledges support from Mobility 2020 project no. CZ.02.2.69/0.0/0.0/18_053/0017850 (Ministry of Education, Youth and Sports of the Czech Republic) and long-term research development project RVO 67985939 (Czech Academy of Sciences). FR acknowledge financial support from Vergelegen and Lourensford estates, Hortgro (V-19-USE-PM06) and Winetech (US FR 20–01). MWC acknowledge financial support from Natural Sciences and Engineering Research Council of Canada (#386,151).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Communicated by Salvatore Arpaia.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Potgieter, L.J., Cadotte, M.W., Roets, F. et al. Monitoring urban biological invasions using citizen science: the polyphagous shot hole borer (Euwallacea fornicatus). J Pest Sci (2024). https://doi.org/10.1007/s10340-024-01744-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10340-024-01744-7