Abstract
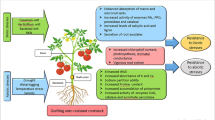
Vegetables constitute 12% of world agricultural produce. Solanaceous and cucurbitaceous crops constitute the major share of vegetable production, but their productivity is greatly impaired by plant-parasitic nematodes, among which root-knot nematodes pose the greatest threat. Due to climate change, water scarcity, shrinking of arable land, and ill effect of pesticides, a major shift in world agriculture is taking place toward sustainability, including organic and protected farming of high-value vegetable crops year-round. Grafting offers a sustainable alternative to conventional and transgenic breeding for nematode resistance. The technique can potentially reduce the phytonematode damage relying on several factors. These include R gene-mediated plant responses, efficient cross talk of defensive genes, alteration of plant metabolism and nutrient mobility, changes in plant vigor and physiological attributes, and modification of rhizosphere microbial community. Additionally, epigenetic factors also determine the success of graft union between the scion and rootstock. Grafting should also be included as a tool in integrated nematode management practices. Studies on identifying potential rootstocks and using grafted vegetable crops to manage nematode incidence are exhaustive but scattered. Here, we review the current status of vegetable grafting for nematode management using resistant rootstocks in both open-field condition and greenhouses. This is the first review, which compiles and summarizes the information on managing nematodes using grafted vegetables. We conclude the review with futuristic research perspectives of grafting that may aid in improving vegetable productivity in a sustainable manner.

Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Abd-Elgawad MMM (2022) Understanding molecular plant-nematode interactions to develop alternative approaches for nematode control. Plants 11:2141. https://doi.org/10.3390/plants11162141
Abdelmageed AHA, Gruda N (2009) Influence of grafting on growth, development and some physiological parameters of tomatoes under controlled heat stress conditions. Eur J Hortic Sci 74:16–20
Agyeman C, Okorley BA, Amissah JN, Nyaku ST (2019) Evaluation of tomato (Solanum lyicopersicum ‘Pectomech’) grafts against root-knot nematode Meloidogyne incognita. Acta Hortic 1302:177–184. https://doi.org/10.17660/ActaHortic.2021.1302.24
Ahn SJ, Im YJ, Chung GC, Cho BH, Suh SR (1999) Physiological responses of grafted-cucumber leaves and rootstock roots affected by low root temperature. Sci Hortic 81:397–408. https://doi.org/10.1016/S0304-4238(99)00042-4
Alam MM, Jairajpuri MS (1990) Nematode control strategies. In: nematode bio-control (aspects and prospects), CBS Pub Dist., Delhi, India, 5–15.
Albacete A, Martinez-Andujar C, Martinez-Pérez A, Thompson AJ, Dodd IC, Pérez-Alfocea F (2015) Unravelling rootstock × scion interactions to improve food security. J Exp Bot 66:2211–2226. https://doi.org/10.1093/jxb/erv027
Ali M, Matsuzoe N, Okubo H, Fujieda K (1992) Resistance of non-tuberous Solanum to root-knot nematode. J Jpn Soc Hortic Sci 60:921–926. https://doi.org/10.2503/jjshs.60.921
Ali MA, Azeem F, Abbas A, Joyia FA, Li H, Dababat AA (2017) Transgenic strategies for enhancement of nematode resistance in plants. Front Plant Sci 8:750. https://doi.org/10.3389/fpls.2017.00750
Ali SS (1997) Status of nematode problem and research in India. Diagnosis of key nematode pests of chickpea and pigeonpea and their management, pp 74–82.
Aloni B, Cohen R, Karni L, Aktas H, Edelstein M (2010) Hormonal signaling in rootstock–scion interactions. Sci Hortic 127:119–126. https://doi.org/10.1016/j.scienta.2010.09.003
Álvarez-Hernández JC, Castellanos-Ramos JZ, Aguirre-Mancilla CL, Huitrón-Ramírez MV, Camacho-Ferre F (2015) Influence of rootstocks on Fusarium wilt, nematode infestation, yield and fruit quality in watermelon production. Ciênc Agrotec 39:323–330. https://doi.org/10.1590/S1413-70542015000400002
Amin AW, Mona AW (2014) Protecting cucumber from Meloidogyne incognita using graft onto resistant cucurbit rootstocks and antagonistic marigold as an alternative to nematicide. Pak J Nematol 32:51–58
Anwar SA, McKenry MV, Ahmad HA (2012) Nematode and fungal communities associated with mango decline of southern Punjab. Pakistan J Zool 44:915–922
Atkinson NJ, Urwin PE (2012) The interaction of plant biotic and abiotic stresses: from genes to the field. J Exp Bot 10:3523–3544. https://doi.org/10.1093/jxb/ers100
Augustin B, Graf V, Laun N (2002) Temperature influencing efficiency of grafted tomato cultivars against root-knot nematode (Meloidogyne arenaria) and corky root (Pyrenochaeta lycopersici). J Plant Dis Prot 109:371–383
Avramidou E, Kapazoglou A, Aravanopoulos FA, Xanthopoulou A, Ganopoulos I, Tsaballa A, Madesis P, Doulis AG, Tsaftaris A (2015) Global DNA methylation changes in Cucurbitaceae inter-species grafting. Crop Breed Appl Biotechnol 15:112–116. https://doi.org/10.1590/1984-70332015v15n2n20
Bagnaresi P, Sala T, Irdani T, Scotto C, Lamontanara A, Beretta M, Rotino GL, Sestili S, Cattivalli L, Sabatini E (2013) Solanum torvum responses to the root-knot nematode Meloidogyne incognita. BMC Genom 14:540. https://doi.org/10.1186/1471-2164-14-540
Baidya S, Timila RD, Ram Bahadur KC, Manandhar HK, Manandhar C (2017) Management of root knot nematode on tomato through grafting root stock of Solanum sisymbriifolium. J Nep Agric Res 3:27–31. https://doi.org/10.3126/jnarc.v3i1.17272
Barbary A, Djian-Caporalino C, Palloix A, Castagnone-Sereno P (2015) Host genetic resistance to root-knot nematodes, Meloidogyne spp., in Solanaceae: from genes to the field. Pest Manag Sci 71:1591–1598. https://doi.org/10.1002/ps.4091
Barrett CE, Zhao X, McSorley R (2012a) Grafting for root-knot nematode control and yield improvement in organic heirloom tomato production. HortScience 47:614–620. https://doi.org/10.21273/HORTSCI.47.5.614
Barrett CE, Zhao X, Hodges AW (2012b) Cost benefit analysis of using grafted transplants for root-knot nematode management in organic heirloom tomato production. HortTechnology 22:252–257. https://doi.org/10.21273/HORTTECH.22.2.252
Bartusch K, Melnyk CW (2020) Insights into plant surgery: an overview of the multiple grafting techniques for Arabidopsis thaliana. Front Plant Sci 11:613442. https://doi.org/10.3389/fpls.2020.613442
Bie Z, Nawaz MA, Huang Y, Lee J-M, Colla G (2017) Introduction of vegetable grafting. In: Wallingford UK (ed) Vegetable grafting: principles and practices. CABI Publishing, pp 1–21
Black LL, Wu DL, Wang JF, Kalb T, Abbass D, Chen JH (2003) Grafting tomatoes for production in the hot-wet season International Cooperators’ Guide. Asian Veg Res Develop Center 3(2):551–555
Bletsos F, Thanassoulopoulos C, Roupakias D (2003) Effect of grafting on growth, yield, and Verticillium wilt of eggplant. HortScience 38:183–186. https://doi.org/10.21273/HORTSCI.38.2.183
Cabrera-Hidalgo AJ, Marban-Mendoza N, Valadez-Moctezuma E (2019) Phylogenetic relationships among Mexican populations of Nacobbus aberrans (Nematoda, Pratylenchidae) reveal the existence of cryptic (complex) species. Nematropica 49:1–11
Cáceres A, Perpina G, Ferriol M, Picó B, Gisbert C (2017) New cucumis rootstocks for melon: ‘UPV-FA’and ‘UPV-FMy.’ HortScience 52:792–797. https://doi.org/10.21273/HORTSCI11791-17
Cao ZP, Chen GK, Chen YF, Yang H, Han LF, Dawson R (2005) Comparative performance of nematode resistant rootstock and non-resistant tomato cultivars on soil biota. Allelopathy J 15:85–94
Cao L, Yu N, Li J, Qi Z, Wang D, Chen L (2016) Heritability and reversibility of DNA methylation induced by in vitro grafting between Brassica juncea and B. oleracea. Sci Rep 6:27233. https://doi.org/10.1038/srep27233
Capinera J (2020) Handbook of vegetable pests. Academic Press, London, UK
Cardarelli M, Rouphael Y, Kyriacou MC, Colla G, Pane C (2020) Augmenting the sustainability of vegetable cropping systems by configuring rootstock-dependent rhizomicrobiomes that support plant protection. Agronomy 10:1185. https://doi.org/10.3390/agronomy10081185
Cerruti E, Comino C, Catoni M, Barchi L, Valentini D, Gisbert C, Prohens J, Portis E, Lanteri S (2018) Integrated DNA methylome and transcriptome analyses highlight epigenomic changes in grafted eggplant plants. Proceedings of the LXII SIGA Annual Congress, Verona, Italy, pp 25–28
Cohen R, Burger Y, Horev C, Porat A, Edelstein M (2005) Performance of Galia-type melons grafted on to Cucurbita rootstock in Monosporascus cannonballus-infested and non-infested soils. Ann Appl Biol 146:381–387. https://doi.org/10.1111/j.1744-7348.2005.040010.x
Cohen R, Dombrovsky A, Louws FJ (2017) Grafting as agrotechnology for reducing disease damage. Veg Graft: Principles Pract CAB Int Oxfordshire UK. https://doi.org/10.1079/9781780648972.0155
Collange B, Navarrete M, Peyre G, Mateille T, Tchamitchian M (2011) Root-knot nematode (Meloidogyne) management in vegetable crop production: The challenge of an agronomic system analysis. Crop Prot 30:1251–1262. https://doi.org/10.1016/j.cropro.2011.04.016
Cook R (2004) Genetic resistance to nematodes: where is it useful? Australas Plant Pathol 33:139–150. https://doi.org/10.1071/AP04035
Cortada L, Sorribas FJ, Ornat C, Kaloshian I, Verdejo-Lucas S (2008) Variability in infection and reproduction of Meloidogyne javanica on tomato rootstocks with the Mi resistance gene. Plant Pathol 57:1125–1135. https://doi.org/10.1111/j.1365-3059.2008.01906.x
Cortada L, Sorribas FJ, Ornat C, Andrés MF, Verdejo-Lucas S (2009) Response of tomato rootstocks carrying the Mi-resistance gene to populations of Meloidogyne arenaria, M. incognita and M. javanica. Eur J Plant Pathol 124:337–343. https://doi.org/10.1007/s10658-008-9413-z
Coyne DL, Cortada L, Dalzell JJ, Claudius-Cole AO, Haukeland S, Luambano N, Talwana H (2018) Plant-parasitic nematodes and food security in Sub-Saharan Africa. Annu Rev Phytopathol 56:381–403. https://doi.org/10.1146/annurev-phyto-080417-045833
Cristóbal AJ, Mora-Aguilera G, Manzanilla-López RH, Marbán-Mendoza N, Sánchez-García P, Cid del Prado-Vera I, Evans K (2006) Epidemiology and integrated control of Nacobbus aberrans on tomato in Mexico. Nematology 8:727–737
Crow WT, Dunn RA (2005) Nematode management for nursery crops (ornamentals and planting stock of fruits and nuts). ENY-39, part of the Florida Nematode Management Guide from the Department of Entomology and Nematology, Florida Cooperative Extension Service. [available at: https://ipm.ifas.ufl.edu/pdfs/NG01100.pdf]
Crow WT (2020) Nematode management in the vegetable garden. Institute of Food and Agricultural Sciences, ENY-012. Gainesville, FL, USA, Univ Florida.
Çürük S, Dasgan HY, Mansuroğlu S, Kurt Ş, Mazmanoğlu M, Antakli Ö, Tarla G (2009) Grafted eggplant yield, quality and growth in infested soil with Verticillium dahliae and Meloidogyne incognita. Pesqui Agropecu Bras 44:1673–1681. https://doi.org/10.1590/S0100-204X2009001200017
Dalakouras A, Vlachostergios D (2021) Epigenetic approaches for crop breeding: current status and perspectives. J Exp Bot 72:5356–5371. https://doi.org/10.1093/jxb/erab227
Davis AR, Perkins-Veazie P, Sakata Y, Lopez-Galarza S, Maroto JV, Lee SG, Huh YC, Sun Z, Miguel A, King SR, Cohen R (2008) Cucurbit grafting. Crit Rev. Plant Sci 27:50–74. https://doi.org/10.1080/07352680802053940
de Carvalho LM, Benda ND, Vaughan MM, Cabrera AR, Hung K, Cox T, Abdo Z, Allen LH, Teal PEA (2015) Mi-1-mediated nematode resistance in tomatoes is broken by short-term heat stress but recovers over time. J Nematol 47:133–140. PMID: 26170475
Desaeger J, Wram C, Zasada I (2020) New reduced-risk agricultural nematicides - rationale and review. J Nematol 52:e2020–e2091. https://doi.org/10.21307/jofnem-2020-091
Di Vito M, Greco N, Carella A (1983) The effect of population densities of Meloidogyne incognita on the yield of cantaloupe and tobacco. Nematologia Medit 11:169–174
Djian-Caporalino C, Fazari A, Arguel MJ, Vernie T, VandeCasteele C, Faure I, Brunoud G, Pijarowski L, Palloix A, Lefebvre V, Abad P (2007) Root-knot nematode (Meloidogyne spp.) Me resistance genes in pepper (Capsicum annuum L.) are clustered on the P9 chromosome. Theor Appl Genet 114:473–486. https://doi.org/10.1007/s00122-006-0447-3
Douda O, Manasova M, Zouhar M, Hnatek J, Stejskal V (2021) Field validation of the effect of soil fumigation of ethanedinitrile (EDN) on the mortality of Meloidogyne hapla and carrot yield parameters. Agronomy 11:208. https://doi.org/10.3390/agronomy11020208
Dutta TK, Papolu PK, Banakar P, Choudhary D, Sirohi A, Rao U (2015) Tomato transgenic plants expressing hairpin construct of a nematode protease gene conferred enhanced resistance to root-knot nematodes. Front Microbiol 6:260. https://doi.org/10.3389/fmicb.2015.00260
Dutta TK, Khan MR, Phani V (2019) Plant-parasitic nematode management via biofumigation using brassica and non-brassica plants: current status and future prospects. Curr Plant Biol 17:17–32. https://doi.org/10.1016/J.CPB.2019.02.001
Echevarría PH, Rodríguez Castro A, Vivaracho SM, Vallejo AD (2004) Influence of rootstocks and soil treatment on the yield and quality of greenhouse-grown cucumbers in Spain, In XXVI International Horticultural Congress: protected cultivation 2002: Search Struct, Syst Plant Mater, 633: 403–408. https://doi.org/10.17660/ActaHortic.2004.633.49
El-Eslamboly AASA, Deabes AAA (2014) Grafting cucumber onto some rootstocks for controlling root-knot nematodes. Minufiya J Agric Res 39:1109–1129
El-Sappah AH, Islam MM, El-Awady H, Yan S, Qi S, Liu J, Cheng G, Liang Y (2019) Tomato natural resistance genes in controlling the root-knot nematode. Genes 10:925. https://doi.org/10.3390/genes10110925
Estan MT, Martinez-Rodriguez MM, Perez-Alfocea F, Flowers TJ, Bolarin MC (2005) Grafting raises the salt tolerance of tomato through limiting the transport of sodium and chloride to the shoot. J Exp Bot 56:703–712. https://doi.org/10.1093/jxb/eri027
Expósito A, Munera M, Giné A, Lopez-Gomez M, Careres A, Pico B, Gisbert C, Medina V, Sorribas FJ (2018) Cucumis metuliferus is resistant to rootknot nematode Mi1. 2 gene (a) virulent isolates and a promising melon rootstock. Plant Pathol 67:1161–1167. https://doi.org/10.1111/ppa.12815
Expósito A, García S, Giné A, Escudero N, Sorribas FJ (2019) Cucumis metuliferus reduces Meloidogyne incognita virulence against the Mi1.2 resistance gene in a tomato–melon rotation sequence. Pest Manag Sci 75:1902–1910. https://doi.org/10.1002/ps.5297
Expósito A, Pujolà M, Achaerandio I, Giné A, Escudero N, Fullana AM, Cunquer M, Loza-Alvarez P, Sorribas FJ (2020) Tomato and melon Meloidogyne resistant rootstocks improve crop yield but melon fruit quality is influenced by the cropping season. Front Plant Sci 11:560024. https://doi.org/10.3389/fpls.2020.560024
FAOSTAT (2020) Food and Agriculture Organization of the United Nations. Statistics Division. Available online at: www.fao.org/faostat/. Accessed 17 Nov 2021).
Faske T, Hurd K (2015) Sensitivity of Meloidogyne incognita and Rotylenchulus reniformis to fluopyram. J Nematol 47:316–321. PMID: 26941460
Forghani F, Hajihassani A (2020) Recent advances in the development of environmentally benign treatments to control root-knot nematodes. Front Plant Sci 11:1125. https://doi.org/10.3389/fpls.2020.01125
Fuentes I, Stegemann S, Golczyk H, Karcher D, Bock R (2014) Horizontal genome transfer as an asexual path to the formation of new species. Nature 511:232–235. https://doi.org/10.1038/nature13291
Fuller VL, Lilley CJ, Urwin PE (2008) Nematode resistance. New Phytol 180:27–44. https://doi.org/10.1111/j.1469-8137.2008.02508.x
Galatti FDS, Franco AJ, Ito LA, Charlo HDO, Gaion LA, Braz LT (2013) Rootstocks resistant to Meloidogyne incognita and compatibility of grafting in net melon. Rev Ceres 60:432–436. https://doi.org/10.1590/S0034-737X2013000300018
Garbeva P, Elsas JD, Veen JA (2008) Rhizosphere microbial community and its response to plant species and soil history. Plant Soil 302:19–32. https://doi.org/10.1007/s11104-007-9432-0
Garcia-Lozano M, Dutta SK, Natarajan P, Tomason YR, Lopez C, Katam R, Levi A, Nimmakayala P, Reddy UK (2020) Transcriptome changes in reciprocal grafts involving watermelon and bottle gourd reveal molecular mechanisms involved in increase of the fruit size, rind toughness and soluble solids. Plant Mol Biol 102:213–223. https://doi.org/10.1007/s11103-019-00942-7
García-Mendívil HA, Sorribas FJ (2019) Fitness cost but no selection for virulence in Meloidogyne incognita after two consecutive crops of eggplant grafted onto Solanum torvum. Plant Pathol 68:1602–1606. https://doi.org/10.1111/ppa.13092
Garita SA, Bernardo VF, Guimarães MDA, Arango MC, Ruscitti MF (2019) Mycorrhization and grafting improve growth in the tomato and reduce the population of Nacobbus aberrans. Revista Ciência Agronômica 50:609–615. https://doi.org/10.5935/1806-6690.20190072
Giannakou IO, Karpouzas DG (2003) Evaluation of chemical and integrated strategies as alternatives to methyl bromide for the control of root-knot nematodes in Greece. Pest Manag Sci 59:883–892. https://doi.org/10.1002/ps.692
Gine A, López-Gómez M, Vela MD, Ornat C, Talavera M, Verdejo-Lucas S, Sorribas FJ (2014) Thermal requirements and population dynamics of root-knot nematodes on cucumber and yield losses under protected cultivation. Plant Pathol 63:1446–1453. https://doi.org/10.1111/ppa.12217
Gisbert C, Prohens J, Raigón MD, Stommel JR, Nuez F (2011) Eggplant relatives as sources of variation for developing new rootstocks: effects of grafting on eggplant yield and fruit apparent quality and composition. Sci Hortic 128:14–22. https://doi.org/10.1016/j.scienta.2010.12.007
Goldschmidt EE (2014) Plant grafting: new mechanisms, evolutionary implications. Front Plant Sci 5:727. https://doi.org/10.3389/fpls.2014.00727
Goreta Ban S, Dumičić G, Raspudić E, Vuletin Selak G, Ban D (2014) Growth and yield of grafted cucumbers in soil infested with root-knot nematodes. Chil J Agric Res 74:29–34. https://doi.org/10.4067/S0718-58392014000100005
Grieneisen ML, Aegerter BJ, Scott Stoddard C, Zhang M (2018) Yield and fruit quality of grafted tomatoes, and their potential for soil fumigant use reduction. Meta-Anal Agron Sustain Dev 38:29. https://doi.org/10.1007/s13593-018-0507-5
Guan W, Zhao X, Hassell R, Thies J (2012) Defense mechanisms involved in disease resistance of grafted vegetables. HortScience 47:164–170. https://doi.org/10.21273/HORTSCI.47.2.164
Guan W, Zhao X, Dickson DW, Mendes ML, Thies J (2014) Root-knot nematode resistance, yield, and fruit quality of specialty melons grafted onto Cucumis metulifer. HortScience 49:1046–1051. https://doi.org/10.21273/HORTSCI.49.8.1046
Gugino BK, Abawi GS, Ludwig JW (2006) Damage and management of Meloidogyne hapla using oxamyl on carrot in New York. J Nematol 38:483–490. PMID: 19259467
Hammond-Kosack KE, Jones JD (1997) Plant disease resistance genes. Annu Rev Plant Biol 48:575–607. https://doi.org/10.1146/annurev.arplant.48.1.575
Huang W, Liao S, Lv H, Khaldun ABM, Wang Y (2015) Characterization of the growth and fruit quality of tomato grafted on a woody medicinal plant, Lycium chinense. Sci Hortic 197:447–453. https://doi.org/10.1016/j.scienta.2015.10.005
Huitrón-Ramírez MV, Ricárdez-Salinas M, Camacho-Ferre F (2009) Influence of grafted watermelon plant density on yield and quality in soil infested with melon necrotic spot virus. HortScience 44:1838–1841. https://doi.org/10.21273/HORTSCI.44.7.1838
Igarashi I, Kanno T, Kawaide T (1987) Disease and pest resistance of wild Cucumis species and their compatibility as rootstock for muskmelon, cucumber and watermelon. Bull Natl Res Inst Veg Ornam Plants Tea Series A 1:173–185
Ioannou N (2001) Integrating soil solarization with grafting on resistant rootstocks for management of soil-borne pathogens of eggplant. J Hortic Sci Biotechnol 76:396–401. https://doi.org/10.1080/14620316.2001.11511383
Iseri ÖD, Körpe DA, Sahin FI, Haberal M (2015) High salt induced oxidative damage and antioxidant response in tomato grafted on tobacco. Chil J Agric Res 75:192–201. https://doi.org/10.4067/S0718-58392015000200008
Ishibashi N, Shimizu K (1970) Gall formation by root-knot nematode, Meloidogyne incognita (Kofoid and White, 1919) Chitwood, 1949, in the grafted tomato plants, and accumulation of phosphates on the gall tissues (Nematoda: Tylenchida). Appl Ent Zool 5:105–111. https://doi.org/10.1303/aez.5.105
Jamison J (2020) Grafting Tamarillo and other solanaceous crops for nematode resistance. ECHO Development Notes, Issue No.149. https://www.echocommunity.org/en/resources/5d428a30-14cc-4e43-94f0-3bfb1cd1fff3
Jones JT, Haegeman A, Danchin EG, Gaur HS, Helder J, Jones MG, Kikuchi T, Manzanilla-López R, Palomares-Rius JE, Wesemael WM, Perry RN (2013) Top 10 plant-parasitic nematodes in molecular plant pathology. Mol Plant Pathol 14:946–961. https://doi.org/10.1111/mpp.12057
Kaskavalci G, Tuzel Y, Dura O, Oztekyn GO (2009) Effects of alternative control methods against Meloidogyne incognita in organic tomato production. Ekoloji 18:23–31
Katan J, Gamliel A (2014). Plant health management: soil solarization encyclopedia of agriculture and food systems. In: Encyclopedia of Agriculture and Food Systems, https://doi.org/10.1016/B978-0-444-52512-3.00256-4
Kearn J, Ludlow E, Dillon J, O’Connor V, Holden-Dye L (2014) Fluensulfone is a nematicide with a mode of action distinct from anticholinesterases and macrocyclic lactones. Pest Biochem Physiol 109:44–57. https://doi.org/10.1016/j.pestbp.2014.01.004
Keatinge JDH, Lin LJ, Ebert AW, Chen WY, Hughes JA, Luther GC, Wang JF, Ravishankar M (2014) Overcoming biotic and abiotic stresses in the Solanaceae through grafting: current status and future perspectives. Biol Agric Hortic 30:272–287. https://doi.org/10.1080/01448765.2014.964317
Keinath AP, Wechter WP, Rutter WB, Agudelo PA (2019) Cucurbit rootstocks resistant to Fusarium oxysporum f. sp. niveum remain resistant when coinfected by Meloidogyne incognita in the field. Plant Dis 103:1383–1390. https://doi.org/10.1094/PDIS-10-18-1869-RE
Kemble JM, Meadows IM, Jennings KM, Walgenbach JF (2019) Southeastern U.S. vegetable crop handbook. Auburn University, Auburn
Khah EM (2005) Effect of grafting on growth, performance and yield of aubergine (Solanum melongena L.) in the field and greenhouse. J Food Agric Environ 3:92–94. https://doi.org/10.22069/IJPP.2012.746
Khan MR, Jain RK, Ghule TM, Pal S (2014) Root knot nematodes in India. Compr Monogr All India Co-Ord Res Proj Plant Parasit Nematodes Integr Approach Control, Indian Agri Res Inst, New Delhi 29:1–78
Kim DG, Ferris H (2002) Relationship between crop losses and initial population densities of Meloidogyne arenaria in winter-grown oriental melon in Korea. J Nematol 34:43–49. PMID: 19265907
King SR, Davis AR, Liu W, Levi A (2008) Grafting for disease resistance. HortScience 43:1673–1676. https://doi.org/10.21273/HORTSCI.43.6.1673
King SR, Davis AR, Zhang X, Crosby K (2010) Genetics, breeding and selection of rootstocks for Solanaceae and Cucurbitaceae. Sci Hortic 127:106–111. https://doi.org/10.1016/j.scienta.2010.08.001
Ko KD (1999) Response of cucurbitaceous rootstock species to biological and environmental stresses (in Korean with English summary). PhD Dissertation, Seoul National University, Suwon, Korea.
Koenning SR, Overstreet C, Noling JW, Donald PA, Becker JO, Fortnum BA (1999) Survey of crop losses in response to phytoparasitic nematodes in the United States for 1994. J Nematol 31:587–618. PMID: 19270925
Kokalis-Burelle N, Rosskopf EN (2011) Microplot evaluation of rootstocks for control of Meloidogyne incognita on grafted tomato, muskmelon, and watermelon. J Nematol 43:166–171. PMID: 23431109
Kokalis-Burelle N, Bausher MG, Rosskopf EN (2009) Greenhouse evaluation of Capsicum rootstocks for management of Meloidogyne incognita on grafted bell pepper. Nematropica 39:121–132
Kokalis-Burelle N, Iriarte FB, Butler DM, Hong JC, Rosskopf EN (2014) Nematode management in Florida vegetable and ornamental production. Outlooks Pest Manag 25:287–293. https://doi.org/10.1564/v25_aug_10
Kokalis-Burelle N, Butler DM, Hong JC, Bausher MG, McCollum G, Rosskopf EN (2016) Grafting and Paladin Pic-21 for nematode and weed management in vegetable production. J Nematol 48:231–240. PMID: 28154429
Kumari A, Kumar J, Kumar A, Chaudhury A, Singh SP (2015) Grafting triggers differential responses between scion and rootstock. PLoS ONE 10:e0124438. https://doi.org/10.1371/journal.pone.0124438
Kunwar S, Paret ML, Freeman JH, Ritchie L, Olson SM, Colee J, Jones JB (2017) Foliar applications of acibenzolar-S-methyl negatively affect the yield of grafted tomatoes in fields infested with Ralstonia solanacearum. Plant Dis 101:890–894. https://doi.org/10.1094/PDIS-03-16-0331-RE
Lahm GP, Desaeger J, Smith BK, Pahutski TF, Rivera MA, Meloro T, Kucharczyk R, Lett RM, Daly A, Smith BT (2017) The discovery of fluazaindolizine: a new product for the control of plant parasitic nematodes. Bioorg Med Chem Lett 27:1572–1575. https://doi.org/10.1016/j.bmcl.2017.02.029
Lamberti F (1979) Economic importance of Meloidogyne spp. in sub-tropical and Mediterranean climate. In: Root knot nematode (Meloidogyne species). Systemics, Biology and Control, Academic press, London, UK, 341–357.
Landi S, d’Errico G, Roversi PF, d’Errico FP (2018) Management of the root-knot nematode Meloidogyne incognita on tomato with different combinations of nematicides and a resistant rootstock: preliminary data, Redia, 101:47–52, https://doi.org/10.19263/REDIA-101.18.07
Lee M, Oda JL (2003) Grafting of herbaceous vegetable and ornamental crops. Hortic Rev 28:61–124. https://doi.org/10.1002/9780470650851.ch2
Lewsey MG, Hardcastle TJ, Melnyk CW, Molnar A, Valli A, Urich MA, Nery JR, Baulcombe DC, Ecker JR (2016) Mobile small RNAs regulate genome-wide DNA methylation. Proc Natl Acad Sci USA 113:E801–E810. https://doi.org/10.1073/pnas.1515072113
Li Y, Li C, Bai L, He C, Yu X (2016) MicroRNA and target gene responses to salt stress in grafted cucumber seedlings. Acta Physiol Plant 38:42. https://doi.org/10.1007/s11738-016-2070-5
Liu B, Ren J, Zhang Y, An J, Chen M, Chen H, Xu C, Ren H (2015) A new grafted rootstock against root-knot nematode for cucumber, melon, and watermelon. Agron Sustain Dev 35:251–259. https://doi.org/10.1007/s13593-014-0234-5
Lopes-Caitar VS, Pinheiro JB, Marcelino-Guimaraes FC (2019) Nematodes in horticulture: an overview. J Hort Sci Crop Res 1:106
Lopez-Gomez M, Verdejo-Lucas S (2014) Penetration and reproduction of root-knot nematodes on cucurbit species. Eur J Plant Pathol 138:863–871. https://doi.org/10.1007/s10658-013-0359-4
Lopez-Perez JA, Le Strange M, Kaloshian I, Ploeg AT (2006) Differential response of Mi gene-resistant tomato rootstocks to root-knot nematodes (Meloidogyne incognita). Crop Prot 25:382–388. https://doi.org/10.1016/J.CROPRO.2005.07.001
Louws FJ, Rivard CL, Kubota C (2010) Grafting fruiting vegetables to manage soil borne pathogens, foliar pathogens, arthropods and weeds. Sci Hortic 127:127–146. https://doi.org/10.1016/j.scienta.2010.09.023
Mándoki Z, Pénzes B (2012) Effects of using chemical-free root-knot nematode (Meloidogyne incognita) control methods on the occurrence of blossom-end rot in pepper. J Plant Prot Res 52:337–341. https://doi.org/10.2478/v10045-012-0055-y
Martínez-Ballesta MC, Alcaraz-López C, Muries B, Mota-Cadenas C, Carvajal M (2010) Physiological aspects of rootstock–scion interactions. Sci Hortic 127:112–118. https://doi.org/10.1016/j.scienta.2010.08.002
Matsuzoe N, Okubo H, Fuiieda K (1993) Resistance of tomato plants grafted on Solanum rootstocks to bacterial wilt and root-knot nematode. J Jpn Soc Hortic Sci 61:865–872. https://doi.org/10.2503/jjshs.61.865
Melnyk CW, Molnar A, Bassett A, Baulcombe DC (2011) Mobile 24 nt small RNAs direct transcriptional gene silencing in the root meristems of Arabidopsis thaliana. Curr Biol 21:1678–1683. https://doi.org/10.1016/j.cub.2011.08.065
Milligan SB, Bodeau J, Yaghoobi J, Kaloshian I, Zabel P, Williamson VM (1998) The root knot nematode resistance gene Mi from tomato is a member of the leucine zipper, nucleotide binding, leucine-rich repeat family of plant genes. Plant Cell 10:1307–1319. https://doi.org/10.1105/tpc.10.8.1307
Molnar A, Melnyk CW, Bassett A, Hardcastle TJ, Dunn R, Baulcombe DC (2010) Small silencing RNAs in plants are mobile and direct epigenetic modification in recipient cells. Science 328:872–875. https://doi.org/10.1126/science.1187959
Moncada A, Miceli A, Vetrano F, Mineo V, Planeta D, D’Anna F (2013) Effect of grafting on yield and quality of eggplant (Solanum melongena L.). Sci Hort 149:108–114. https://doi.org/10.1016/j.scienta.2012.06.015
Moosavi MR (2015) Damage of the root-knot nematode Meloidogyne javanica to bell pepper, Capsicum annuum. J Plant Dis Prot 122:244–249. https://doi.org/10.1007/BF03356559
Morra L, Bilotto M (2006) Evaluation of new rootstocks for resistance to soil-borne pathogens and productive behaviour of pepper (Capsicum annuum L.). J Hortic Sci Biotechnol 81:518–524. https://doi.org/10.1080/14620316.2006.11512097
Moura GS, Franzener G (2017) Biodiversity of nematodes biological indicators of soil quality in the agroecosystems. Arq Inst Biol 84:1–8. https://doi.org/10.1590/1808-1657000142015
Mudge K, Janick J, Scofield S, Goldschmidt EE (2009) A history of grafting. Horticultural Reviews, vol 35. John Wiley and Sons Inc, New Jersey, pp 437–493
Murata G, Uehara T, Lee HJ, Mizutani M, Kadota Y, Shinmura Y, Saito T, Uesugi K (2022) Solanum palinacanthum Dunal as a potential eggplant rootstock resistant to root-knot nematodes. J Phytopathol. https://doi.org/10.1111/jph.13067
Nakamura S, Hondo K, Kawara T, Okazaki Y, Saito K, Kobayashi K, Yaeno T, Yamaoka N, Nishiguchi M (2015) Conferring high-temperature tolerance to nontransgenic tomato scions using graft transmission of RNA silencing of the fatty acid desaturase gene. Plant Biotechnol J 14:783–790. https://doi.org/10.1111/pbi.12429
Noling JW (2019) Nematode management in tomatoes, peppers, and eggplant. University of Florida publication Series no ENY-032, USA
Nyaku ST, Amissah N (2018) Grafting: an effective strategy for nematode management in tomato genotypes. In: Recent advances in tomato breeding and production, Intech Open. https://doi.org/10.5772/intechopen.82774
Oka Y, Offenbach R, Pivonia S (2004) Pepper rootstock graft compatibility and response to Meloidogyne javanica and M. incognita. J Nematol 36:137–141. PMID: 19262798
Oka Y, Shuker S, Tkachi N (2012) Systemic nematicidal activity of fluensulfone against the root-knot nematode Meloidogyne incognita on pepper. Pest Manag Sci 68:268–275. https://doi.org/10.1002/ps.2256
Olthof THA, Potter JW (1973) The relationship between population densities of Pratylenchus penetrans and crop losses in summer-maturing vegetables in Ontario. Phytopathology 63:577–582
Olthof THA, Potter JW, Peterson EA (1974) Relationship between population densities of Heterodera schachtii and losses in vegetable crops in Ontario. Phytopathology 64:549–554
Ornat C, Sorribas FJ (2008) Integrated management of root-knot nematodes in Mediterranean horticultural crops. Integrated management and biocontrol of vegetable and grain crops nematodes. Springer, Dordrecht, pp 295–319
Owusu SB, Kwoseh CK, Starr JL, Davies FT (2016) Grafting for management of root-knot nematodes, Meloidogyne incognita, in tomato (Solanum lycopersicum L.). Nematropica 46:14–21
Pardo-Alonso JL, Carreño-Ortega A, Martínez-Gaitán CC, Callejón-Ferre ÁJ (2019) Combined influence of cutting angle and diameter differences between seedlings on the grafting success of tomato using the splicing technique. Agronomy 9:5. https://doi.org/10.3390/agronomy9010005
Pham TT, Giang BL, Nguyen NH, Yen P, Hoang V, Ha B, Le N (2020) Combination of mycorrhizal symbiosis and root grafting effectively controls nematode in replanted coffee soil. Plants 9:555. https://doi.org/10.3390/plants9050555
Phani V, Khan MR, Dutta TK (2021) Plant-parasitic nematodes as a potential threat to protected agriculture: current status and management options. Crop Prot 144:105573. https://doi.org/10.1016/j.cropro.2021.105573
Ploeg A, Phillips M (2001) Damage to melon (Cucumis melo L.) cv. Durango by Meloidogyne incognita in Southern California. Nematology 3:151–157. https://doi.org/10.1163/156854101750236277
Pocurull M, Fullana AM, Ferro M, Valero P, Escudero N, Saus E, Gabaldón T, Sorribas FJ (2020) Commercial formulates of Trichoderma induce systemic plant resistance to Meloidogyne incognita in tomato and the effect is additive to that of the Mi-1.2 resistance gene, Front Microbiol, 10:3042. https://doi.org/10.3389/fmicb.2019.03042
Pofu KM, Mashela PW, Mphosi MS (2011) Management of Meloidogyne incognita in nematode susceptible watermelon cultivars using nematode resistant Cucumis africanus and Cucumis myriocarpus rootstocks. Afr J Biotech 10:8790–8793. https://doi.org/10.5897/AJB10.1252
Pofu KM, Mashela PW, Shimelis H (2012) Inter-generic grafting in watermelon for managing Meloidogyne species: a review. Sci Res Essays 7:107–113
Pokharel R (2011) Importance of plant parasitic nematodes in Colorado crops. Fact Sheet No. 2.952, Crop series - Disease, Western Colorado Research Center, Colorado State University.
Poudel R, Jumpponen A, Kennelly MM, Rivard CL, Gomez-Montano L, Garrett KA (2019) Rootstocks shape the rhizobiome: rhizosphere and endosphere bacterial communities in the grafted tomato system. Appl Environ Microbiol 85:e01765-e1818. https://doi.org/10.1128/AEM.01765-18
Pulavarty A, Egan A, Karpinska A, Horgan K, Kakouli-Duarte T (2021) Plant parasitic nematodes: a review on their behaviour, host interaction, management approaches and their occurrence in two sites in the republic of Ireland. Plants 10:2352. https://doi.org/10.3390/plants10112352
Rahman MA, Rashid MA, Salam MA, Masud MA, Masum AS, Hossain MM (2002) Performance of some grafted eggplant genotypes on wild Solanum root stocks against root-knot nematode. Online J Biol Sci 2:446–448
Rao MS, Umamaheswari R, Chakravarty AK (2015) Plant parasitic nematodes: a major stumbling block for successful crop cultivation under protected conditions in India. Current Sci 108:13–14
Rich JR, Brito JA, Kaur R, Ferrell JA (2009) Weed species as hosts of Meloidogyne: a review. Nematropica 39:157–185
Rivard CL, O’Connell S, Peet MM, Louws FJ (2010) Grafting tomato with interspecific rootstock to manage diseases caused by Sclerotium rolfsii and southern root-knot nematode. Plant Dis 94:1015–1021. https://doi.org/10.1094/PDIS-94-8-1015
Rivero RM, Ruiz JM, Romero L (2003a) Role of grafting in horticultural plants under stress conditions. J Food Agric Environ 1:70–74
Rivero RM, Ruiz JM, Romero L (2003b) Can grafting in tomato plants strengthen resistance to thermal stress? J Sci Food Agric 83:1315–1319
Robinson AF, Inserra RN, Caswell-Chen EP, Vovlas N, Troccoli A (1997) Rotylenchulus species: identification, distribution, host ranges, and crop plant resistance. Nematropica 27:127–180
Ros C, Guerrero MM, Martiinez MA, Lacasa A, Bello A (2006) Integrated management of Meloidogyne resistance in sweet pepper in greenhouses. IOBC WPRS Bulletin 29:319
Ros C, Martínez V, Sánchez-Solana F, López-Marín J, Lacasa CM, del Mar Guerrero M, Lacasa A (2018) Combination of biosolarization and grafting to control Meloidogyne incognita in greenhouse pepper crops. Crop Prot 113:33–39. https://doi.org/10.1016/j.cropro.2018.07.007
Rosskopf E, Pisani C, Di Gioia F (2017) Crop specific grafting methods, rootstocks and scheduling: tomato. Grafting manual: how to produce grafted vegetable plants (www. vegetablegrafting.org).
Rouphael Y, Kyriacou MC, Colla G (2018) Vegetable grafting: a toolbox for securing yield stability under multiple stress conditions. Front Plant Sci 8:2255. https://doi.org/10.3389/fpls.2017.02255
Sakata Y, Ohara T, Sugiyama M (2007) The history and present state of grafting of Cucurbitaceous vegetables in Japan, In: Proceedings of the Third International Symposium on Cucurbits. Acta Hortic, 731:159–170. https://doi.org/10.17660/ActaHortic.2007.731.22
Sargın S, Devran Z (2021) Degree of resistance of Solanum torvum cultivars to Mi-1.2-virulent and avirulent isolates of Meloidogyne incognita, Meloidogyne javanica, and Meloidogyne luci. J Nematol 53:1–7. https://doi.org/10.21307/jofnem-2021-068
Sasser JN, Freckman DW (1987) A world perspective on nematology: The role of the society. In: Veech JA, Dickson NW (eds) Vistas on nematology. Society of Nematologists, Hyattsville, Maryland, US, pp 7–14
Sato K, Uehara T, Holbein J, Sasaki-Sekimoto Y, Gan P, Bino T, Yamaguchi K, Ichihashi Y, Maki N, Shigenobu S, Ohta H, Franke RB, Siddique S, Grundler FMW, Suzuki T, Kadota Y, Shirasu K (2021) Transcriptomic analysis of resistant and susceptible responses in a new model root-knot nematode infection system using Solanum torvum and Meloidogyne arenaria. Front Plant Sci 12:680151. https://doi.org/10.3389/fpls.2021.680151
Savvas D, Colla G, Rouphael Y, Schwarz D (2010) Amelioration of heavy metal and nutrient stress in fruit vegetables by grafting. Sci Hortic 127:156–161. https://doi.org/10.1016/j.scienta.2010.09.011
Seinhorst JW (1970) Dynamics of population of plant parasitic nematodes. Annu Rev Phytopathol 8:131–156. https://doi.org/10.1146/annurev.py.08.090170.001023
Sidorova T, Miroshnichenko D, Kirov I, Pushin A, Dolgov S (2021) Effect of grafting on viral resistance of non-transgenic plum scion combined with transgenic PPV-resistant rootstock. Front Plant Sci 12:621954. https://doi.org/10.3389/fpls.2021.621954
Sigüenza C, Schochow M, Turini T, Ploeg A (2005) Use of Cucumis metuliferus as a rootstock for melon to manage Meloidogyne incognita. J Nematol 37:276–280. PMID: 19262873
Sikora RA, Fernandez E (2005) Nematode parasites of vegetables. In: Sikora R, Coyne D, Hallmann J, Timper P (eds) Plant parasitic nematodes in subtropical and tropical agriculture. CAB International, Wallingford, UK, pp 319–392
Slomczynska U, South MS, Bunkers G, Edgecomb D, Wyse-Pester D, Selness S, Ding Y, Christiansen J, Ediger K, Miller W (2014) Tioxazafen: a new broadspectrum seed treatment nematicide. ACS Symposium Series, vol 1204. Discovery and synthesis of crop protection products. Monsanto Corporation, Chesterfield, Missouri, pp 129–147
Song Y, Ling N, Ma J, Wang J, Zhu C, Raza W, Shen Y, Huang Q, Shen Q (2016) Grafting resulted in a distinct proteomic profile of watermelon root exudates relative to the un-grafted watermelon and the rootstock plant. J Plant Growth Regul 35:778–791. https://doi.org/10.1007/s00344-016-9582-5
Sorribas FJ, Ornat C, Verdejo-Lucas S, Galeano M, Valero J (2005) Effectiveness and profitability of the Mi-resistant tomatoes to control root-knot nematodes. Eur J Plant Pathol 111:29–38. https://doi.org/10.1007/s10658-004-1982-x
Sorribas FJ, Djian-Caporalino C, Mateille T (2020) Nematodes. In: Gullino M, Albajes R, Nicot P (eds) Integrated pest and disease management in greenhouse crops, plant pathology in the 21st century. Springer, Cham. https://doi.org/10.1007/978-3-030-22304-5_5
Spanò R, Mascia T, Kormelink R, Gallitelli D (2015) Grafting on a non-transgenic tolerant tomato variety confers resistance to the infection of a Sw5-breaking strain of tomato spotted wilt virus via RNA silencing. PLoS ONE 10:e0141319. https://doi.org/10.1371/journal.pone.0141319.t001
Spanò R, Ferrara M, Gallitelli D, Mascia T (2020) The role of grafting in the resistance of tomato to viruses. Plants 9:1042. https://doi.org/10.3390/plants9081042
Stegemann S, Keuthe M, Greiner S, Bock R (2012) Horizontal transfer of chloroplast genomes between plant species. Proc Natl Acad Sci USA 109:2434–2438. https://doi.org/10.1073/pnas.1114076109
Stetina SR, Smith JR, Ray JD (2014) Identification of Rotylenchulus reniformis resistant Glycine lines. J Nematol 46:1–7. PMID: 24643425
Sturhan D, Brzeski MW (1991) Stem and bulb nematodes, Ditylenchus spp. In: Nickle WR (ed) Manual of agricultural nematology. Marcel Dekker, New York, USA, pp 423–464
Subbotin SA, Rius JEP, Castillo P (2021) Systematics of root-knot nematodes (Nematoda: Meloidogynidae). Brill. https://doi.org/10.1163/9789004387584_008
Talavera M, Verdejo-Lucas S, Ornat C, Torres J, Vela MD, Macias FJ, Cortada L, Arias DJ, Valero J, Sorribas FJ (2009) Crop rotations with Mi gene resistant and susceptible tomato cultivars for management of root-knot nematodes in plastic-houses. Crop Prot 28:662–667. https://doi.org/10.1016/j.cropro.2009.03.015
Taylor AL, Sasser JN (1978) Biology, identification and control of root-knot nematodes. North Carolina State University Graphics, Raleigh
Thies JA (2021) Grafting for managing vegetable crop pests. Pest Manag Sci 77:4825–4835. https://doi.org/10.1002/ps.6512
Thies JA, Ariss JJ, Hassell RL, Olson S, Kousik CS, Levi A (2010) Grafting for management of southern root-knot nematode, Meloidogyne incognita, in watermelon. Plant Dis 94:1195–1199. https://doi.org/10.1094/PDIS-09-09-0640
Thies JA, Ariss JJ, Kousik CS, Hassell RL, Levi A (2016) Resistance to southern root-knot nematode (Meloidogyne incognita) in wild watermelon (Citrullus lanatus var. citroides). J Nematol 48:14–19. PMID: 27168648
Tileubayeva Z, Avdeenko A, Avdeenko S, Stroiteleva N, Kondrashev S (2021) Plant-parasitic nematodes affecting vegetable crops in greenhouses. Saudi J Biol Sci 28:548–5433. https://doi.org/10.1016/j.sjbs.2021.05.075
Tsaballa A, Xanthopoulou A, Madesis P, Tsaftaris A, Nianiou-Obeidat I (2021) Vegetable grafting from a molecular point of view: the involvement of epigenetics in rootstock-scion interactions. Front Plant Sci 11:621999. https://doi.org/10.3389/fpls.2020.621999
Tzortzakakis EA, Bletsos FA, Avgelis AD (2006) Evaluation of Solanum rootstock accessions for control of root-knot nematodes and tobamoviruses. J Plant Dis Prot 113:188–189. https://doi.org/10.1007/BF03356178
Venema JH, Dijk BE, Bax JM, van Hasselt PR, Elzenga JTM (2008) Grafting tomato (Solanum lycopersicum) onto the rootstock of a high-altitude accession of Solanum habrochaites improves suboptimal-temperature tolerance. Environ Exp Bot 63:359–367. https://doi.org/10.1016/j.envexpbot.2007.12.015
Verdejo-Lucas S, Sorribas FJ (2008) Resistance response of the tomato rootstock SC 6301 to Meloidogyne javanica in a plastic house. Eur J Plant Pathol 121:103–107. https://doi.org/10.1007/s10658-007-9243-4
Verdejo-Lucas S, Talavera M (2019) Pathogenic potential, parasitic success and host efficiency of Meloidogyne incognita and M. javanica on cucurbitaceous plant genotypes. Eur J Plant Pathol 153:1287–1297. https://doi.org/10.1007/s10658-018-01642-6
Verdejo-Lucas S, Cortada L, Sorribas FJ, Ornat C (2009) Selection of virulent populations of Meloidogyne javanica by repeated cultivation of Mi resistance gene tomato rootstocks under field conditions. Plant Pathol 58:990–998. https://doi.org/10.1111/j.1365-3059.2009.02089.x
Viaene NM, Abawi GS (1996) Damage threshold of Meloidogyne hapla to lettuce in organic soil. J Nematol 28:537–545. PMID: 19277172
Vinícius-Marin M, Santos LS, Gaion LA, Rabelo HO, Franco CA, Diniz GM, Silva EH, Braz LT (2017) Selection of resistant rootstocks to Meloidogyne enterolobii and M. incognita for okra (Abelmoschus esculentus L. Moench). Chil J Agric Res 77:58–64. https://doi.org/10.4067/S0718-58392017000100007
Walters SA, Wehner TC, Daykin ME, Barker KR (2006) Penetration rates ofroot-knot nematodes into Cucumis sativus and C. metuliferus roots andsubsequent histological changes. Nematropica 36:231–242
Wang KH, McSorley R (2008) Exposure time to lethal temperatures for Meloidogyne incognita suppression and its implication for soil solarisation. J Nematol 40:7–12. PMID: 19259512
Wang J, Jiang L, Wu R (2017) Plant grafting: how genetic exchange promotes vascular reconnection. New Phytol 214:56–65. https://doi.org/10.1111/nph.14383
Wei C, Li M, Qin J, Xu Y, Zhang Y, Wang H (2019) Transcriptome analysis reveals the effects of grafting on sweetpotato scions during the full blooming stages. Genes Genom 41:895–907. https://doi.org/10.1007/s13258-019-00823-9
Williamson VM, Roberts PA (2009) Mechanisms and genetics of resistance. In: Perry RN, Moens M, Starr JL (eds) Root-knot nematodes. CABI international, Wallingford. https://doi.org/10.1079/9781845934927.0301
Wu R, Wang X, Lin Y, Ma Y, Liu G, Yu X, Zhong S, Liu B (2013) Inter-species grafting caused extensive and heritable alterations of DNA methylation in solanaceae plants. PLoS ONE 8:e61995. https://doi.org/10.1371/journal.pone.0061995
Xanthopoulou A, Tsaballa A, Ganopoulos I, Kapazoglou A, Avramidou E, Aravanopoulos FA, Moysiadis T, Osathanunkul M, Tsaftaris A, Doulis AG, Kalivas A, Sarrou E, Martens S, Nianiou-Obeidat I, Madesis P (2019) Ιntra-species grafting induces epigenetic and metabolic changes accompanied by alterations in fruit size and shape of Cucurbita pepo L. Plant Growth Regul 87:93–108. https://doi.org/10.1007/s10725-018-0456-7
Ye DY, Qi YH, Cao SF, Wei BQ, Zhang HS (2017) Histopathology combined with transcriptome analyses reveals the mechanism of resistance to Meloidogyne incognita in Cucumis metuliferus. J Plant Physiol 212:115–124. https://doi.org/10.1016/j.jplph.2017.02.002
Yilmaz S, Celik I, Zengin S (2011) Combining effects of soil solarization and grafting on plant yield and soil-borne pathogens in cucumber. Int J Plant Prod 5(1):95–104. https://doi.org/10.22069/IJPP.2012.723
Yin H, Yan B, Sun J, Jia P, Zhang Z, Yan X, Chai J, Ren Z, Zheng G, Liu H (2012) Graft-union development: a delicate process that involves cell–cell communication between scion and stock for local auxin accumulation. J Exp Bot 63:4219–4232. https://doi.org/10.1093/jxb/ers109
Zhang G, Mao Z, Wang Q, Song J, Nie X, Wang T, Zhang H, Guo H (2019) Comprehensive transcriptome profiling and phenotyping of rootstock and scion in a tomato/potato heterografting system. Physiol Plant 166:833–847. https://doi.org/10.1111/ppl.12858
Acknowledgements
MTG acknowledges Director, ICAR-IIVR, Varanasi, India, for providing infrastructural assistance to carry out the vegetable grafting experiments. We also thank Dr. C Sellaperumal, Scientist, ICAR-IIVR, Varanasi, India, for helping in our grafting experiments.
Funding
Not applicable.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Consent to participate
Not applicable.
Consent for publication
All authors agreed with the content and gave explicit consent to submit the manuscript in the present form.
Ethics approval
Not applicable.
Additional information
Communicated by Antonio Biondi
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Phani, V., Gowda, M.T. & Dutta, T.K. Grafting vegetable crops to manage plant-parasitic nematodes: a review. J Pest Sci 97, 539–560 (2024). https://doi.org/10.1007/s10340-023-01658-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-023-01658-w