Abstract
The brown marmorated stink bug Halyomorpha halys is a polyphagous insect, which has a devastating impact on agricultural production in many countries. The alteration of symbiont vertical transmission, by removing symbionts from stink bug eggs (symbiotic control), has been recently introduced in control programmes against this insect. A major advantage of this strategy is the compatibility with natural enemies, since it allows an insecticide-free approach that is not harmful to other agroecosystem components. However, the effect of anti-symbiont products on parasitism by egg parasitoids is still unexplored. Here, we investigated the impact on parasitism by native (Anastatus bifasciatus, Ooencyrtus telenomicida and Trissolcus kozlovi) and exotic (Trissolcus japonicus and Trissolcus mitsukurii) parasitoids that attack H. halys eggs, after treatment with the micronutrient biocomplex Dentamet®, used for symbiotic control. The native wasp species were tested in no-choice bioassays, showing that treatment of the egg masses did not affect emergence percentages, but the non-reproductive effects were often reduced by the biocomplex. The exotic species T. japonicus and T. mitsukurii were used in no-choice and paired choice bioassays, showing an opposite influence of Dentamet® on emergence percentage and preference in the two species. No-choice tests indicated the highest successful parasitoid emergence on biocomplex-treated egg masses for T. japonicus, while no preference in the paired comparison with eggs treated with water or untreated. In contrast, T. mitsukurii displayed the lowest parasitism after Dentamet® treatment in no-choice tests, and preferred egg masses without Dentamet® in paired choice tests. We did not record any natural symbiont acquisition by the parasitoids emerged from H. halys egg masses, indicating that the wasp fitness is very unlikely to be altered by dysbiotic effects resulting from treatments. Therefore, our results support a further implementation of symbiotic control in different crops in combination with biological control, as sustainable options for H. halys integrated pest management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The brown marmorated stink bug Halyomorpha halys (Stål) (Hemiptera: Pentatomidae) is one of the most studied pentatomid species due to its invasive potential and high polyphagy (Leskey et al. 2012a; Rice et al. 2014; Leskey and Nielsen 2018). This insect pest is native to Asia, and it was unintentionally introduced in North America in the 1990s and later in Europe in 2004 (Haye and Weber 2017; Leskey and Nielsen 2018). Halyomorpha halys attacks more than 300 species of many important agricultural crops and wild plants (Lee et al. 2013). In fruits, feeding activity induces symptoms such as deformation and discolouration, and seed abortion with early attacks (Rice et al. 2014; Bariselli et al. 2016; Bergmann et al. 2016; Bosco et al. 2018). Moreover, it is also an important household nuisance pest due to its aggregative overwintering behaviour inside buildings (Inkley 2012). Because of its invading potential, the population dynamics of H. halys have been studied under different environmental conditions, providing important information for predicting the potential spread. A survival reduction was observed in the presence of high temperature and/or low humidity, suggesting that the insect may preferentially spread in areas where heatwaves are not frequent (Scaccini et al. 2019; Fisher et al. 2020; Khadka et al. 2020).
Currently, the control of H. halys mainly relies on the use of chemical pesticides, with an important environmental impact and a huge effect on human health (Nielsen et al. 2008; Leskey et al. 2012b). Moreover, most insecticides used against this pest have broad-spectrum activity, which negatively affects natural enemies and pollinators (Leskey et al. 2012a, b; Cira et al. 2017; Kuhar and Kamminga 2017). Several methods have been proposed to reduce chemical treatments within Integrated Pest Management Crop Perimeter Restructuring programmes, e.g. border row or alternate row insecticide applications (Blaauw et al. 2015; Akotsen-Mensah et al. 2020; Ludwick et al. 2020).
The main alternative to insecticides against H. halys is biological control. Egg parasitoids are the most studied and specialized natural enemies both in the native (Lee 2015) and in the invaded areas (Abram et al. 2017; Conti et al. 2021). In the native area, the most common egg parasitoids belong to Scelionidae and Eupelmidae (Lee et al. 2013). In particular, the samurai wasp Trissolcus japonicus (Ashmead) (Hymenoptera: Scelionidae) is reported as the predominant species in China and in Japan (Yang et al. 2009, 2015; Zhang et al. 2017; Kamiyama et al. 2022). In Europe, Anastatus bifasciatus (Geoffroy) (Hymenoptera: Eupelmidae) is the main native species found emerging from field-laid and sentinel H. halys egg masses in Italy and Switzerland (Haye et al. 2015; Costi et al. 2019; Moraglio et al. 2020; Zapponi et al. 2020, 2021). Furthermore, the generalist Ooencyrtus telenomicida (Vassiliev) (Hymenoptera: Encyrtidae) was obtained from frozen sentinel H. halys eggs in Central Italy (Roversi et al. 2016), while Trissolcus kozlovi Rjachovskij (Hymenoptera: Scelionidae) was found emerging from field-laid H. halys eggs in North Italy (Moraglio et al. 2020; Scaccini et al. 2020). Both A. bifasciatus and T. kozlovi were tested in augmentative release field trials, and proved to attack H. halys freshly laid eggs, although neither was able to effectively contain the bug population in the experimental conditions (Stahl et al. 2018, 2019a, b; Moraglio et al. 2021a). Therefore, T. japonicus was considered as the most promising candidate for H. halys biological control, and host range studies were started in support of the application for authorization to the field release of the exotic parasitoid, which must be submitted and approved by regulatory agencies (Haye et al. 2020; Sabbatini-Peverieri et al. 2021). In Italy, the field release of T. japonicus has been recently authorized, and started in the summer of 2020. Meanwhile, adventive populations of T. japonicus have been detected in Switzerland and in Italy in 2017 and in 2018, respectively (Sabbatini-Peverieri et al. 2018; Stahl et al. 2019c; Moraglio et al. 2020). Furthermore, also adventive populations of Trissolcus mitsukurii (Ashmead) (Hymenoptera: Scelionidae), another Asian parasitoid of H. halys eggs in Japan (Arakawa and Namura 2002; Kamiyama et al. 2022), have been found in Europe, in Italy and in France in 2016 and 2021, respectively (Scaccini et al. 2020; Bout et al. 2021). This species has been considered as another promising candidate for biological control (Sabbatini-Peverieri et al. 2020; Caron et al.2021; Giovannini et al. 2022). However, biological control is seriously hindered by the use of insecticides (Lowenstein et al. 2019).
Recently, symbiotic control protocols were designed to prevent symbiont acquisition by nymphs by treating H. halys eggs with antibacterial substances; this approach was proposed as a suitable option for pentatomid containment (Gonella et al. 2019, 2020). In this insect family, gut symbiotic bacteria are transmitted through maternal secretions, which are smeared on egg masses during oviposition and orally acquired by the nymphs after egg hatching (Prado et al. 2006; Otero-Bravo and Sabree 2015). Since these obligate bacteria are essential for growth, development and survival of the insect, symbiont-deprived stink bugs usually display reduced survival or fitness (Taylor et al. 2014). Different active substances have been tested to eliminate the primary symbiont of H. halys, i.e. “Candidatus Pantoea carbekii” (P. carbekii), from the egg surface, showing high mortality percentages soon after nymph emergence (Mathews and Barry 2014; Taylor et al. 2017; Gonella et al. 2019). These nymphs tested negative for P. carbekii through specific qPCR analyses, indicating that the observed effects resulted from missed symbiont acquisition (Gonella et al. 2019). In particular, the application of the zinc, copper and citric acid biocomplex Dentamet® (Diachem, Italy) on egg masses caused more than 90% first instar nymph mortality. Therefore, in Italy foliar applications with Dentamet® were introduced in control programmes targeting field-laid egg masses of H. halys in several crops, as the product was provisionally authorized to control H. halys by the Italian Ministry of Agricultural, Food and Forestry Policies in 2021. Since this product does not show a direct insecticidal activity, it is expected not to have any effects on the survival of beneficial insects, including egg parasitoids; however, its effects on trophic interactions involving natural enemies—such as host preference and exploitation efficiency—have not yet been investigated.
Here, we assessed the impact of Dentamet® on parasitism by native and exotic egg parasitoids. Specifically, we measured parasitoid emergence percentages on treated and untreated egg masses under laboratory conditions. We conducted no-choice tests with three native (A. bifasciatus, O. telenomicida, T. kozlovi) and two exotic (T. japonicus and T. mitsukurii) egg parasitoids species, to assess the suitability for parasitic wasps of egg masses exposed to the biocomplex. Moreover, paired choice tests were performed using the species that showed the highest emergence percentage in no-choice tests, to evaluate their actual preference when treated egg masses were compared to untreated ones. Since several studies have shown the horizontal transmission of symbionts between parasitoids and their hosts, in some cases offering a beneficial effect to the new host (Vavre et al. 1999; Gualtieri et al. 2017; Qi et al. 2019), we also evaluated the possible acquisition of P. carbekii from H. halys egg masses in the co-evolved parasitoids, i.e. the Asian Trissolcus species.
Materials and methods
Insect rearing
Overwintered H. halys adults were collected in 2021 from wild and cultivated host plants in several sites in the Piedmont region, NW Italy. Insects were maintained in climatic chambers at 25 ± 1 °C, 65 ± 5% RH and 16:8 h L:D photoperiod, in net cages (930 × 475 × 475 mm) containing broad bean (Vicia faba L.) seedlings, apples (Malus domestica Borkh.), shelled hazelnuts (Corylus avellana L.) and green beans (Phaseolus vulgaris L.). Halyomorpha halys rearing was inspected daily to collect freshly laid egg masses (less than 24 h old) to be used in this study.
A colony of T. kozlovi had already been established in laboratory since 2017 (Moraglio et al. 2021a), while the other native (A. bifasciatus and O. telenomicida), and exotic egg parasitoids (T. japonicus and T. mitsukurii) were obtained from field-collected parasitized egg masses of H. halys, sampled from several sites in Piedmont in 2021. All wasp species were maintained separately from the stink bug rearing, in a climatic chamber at the same conditions indicated above, on fresh or frozen H. halys egg masses, in plastic containers (100 mm diameter, 50 mm height) with a mesh on the lid, wet cotton and a honey drop (applied with a needle) were provided on the lid and weekly replaced.
No-choice tests
No-choice tests were conducted by exposing three types of H. halys egg masses to the five egg parasitoid species: (a) untreated, (b) treated with ultrapure water and (c) treated with Dentamet® 1% v/v (dissolved in ultrapure water). Prior to the beginning of experiments, the egg masses were randomly assigned to different treatments; the number of eggs per mass was recorded, then the eggs were not further manipulated in order to avoid any disturbance to parasitism. The egg masses were individually placed in plastic Petri dishes (60 mm diameter) and treated with the biocomplex or water by a 200 mL hand sprayer under a fume hood, according to Gonella et al. (2019). A single spray (651 ± 7.42 µl) was applied about 20 cm away from the Petri dish. After 30 min from spray application, when the treated egg mass was dry, a single 5–10-day-old mated female of each parasitoid species was placed into a Petri dish with a single H. halys fresh egg mass of one of the three types and maintained for 48 h. The 48 h period was selected to allow the parasitization even by the less efficient egg parasitoids. Every female was used only once. All wasps were fed with a small drop of honey, placed at the top of Petri dish. Egg mass visit and oviposition were recorded according to Haye et al. (2015). In addition, an equal number of egg masses were treated as explained above, but they were not exposed to any parasitoid species, to evaluate natural host mortality. Each trial (one egg mass per treatment offered to each of the five parasitoid species, plus one egg mass per treatment unexposed to any wasp) was replicated 10 times (for a total of 180 egg masses). All trials were conducted in a climatic chamber at 26 ± 1 °C, 65 ± 5% RH. After the experiment, the egg parasitoids were removed, and all egg masses were individually reared until the emergence of stink bug nymphs and/or wasp adults. The number of unhatched H. halys eggs, and the number of emerged parasitoids were recorded for each egg mass. The two species showing the highest parasitoid efficiency in no-choice tests were used to perform a paired choice test.
Paired choice tests
After no-choice tests, paired choice tests were conducted with the two most efficient wasp species, namely T. japonicus and T. mitsukurii, to check for a possible interference with host preference caused by treatments. All wasps and egg masses were collected from the laboratory colonies as described above. A single-mated 5–10-day-old wasp female of each species was placed in an arena (h = 70 mm; diameter = 95 mm) with two egg masses; females were used only once. Each arena was composed of the following pairs of egg masses: (A) untreated vs treated with water, (B) treated with water vs treated with Dentamet® 1% v/v, and (C) untreated vs treated with Dentamet® 1% v/v. Each comparison was replicated 10 times, and a total of 120 egg masses were used for this experiment. A small drop of honey was provided in the arena as a food source for the wasp. The female was left in the arena for 3 h according to the observations of Haye et al. (2020), who suggested a limited test duration to reduce the number of wasps parasitizing both egg masses. The first egg mass visited by the parasitoid was visually checked and recorded. At the end of the experiment, wasp females were removed, and the egg masses were reared separately at 26 ± 1 °C, 65 ± 5% RH and 16:8 L:D photoperiod until the emergence of all H. halys nymphs and/or parasitoid adults. The numbers of unhatched eggs and emerged parasitoids were recorded for each egg mass.
Molecular diagnosis for P. carbekii
To evaluate the occurrence of a host-to-parasitoid horizontal transmission of P. carbekii, quantitative Real Time PCR (qPCR) analysis was used to determine the presence of the H. halys symbiont in exotic wasps emerged from no-choice tests. Adults of T. japonicus and of T. mitsukurii were collected after the emergence from untreated (field-collected) egg masses and stored at -80 °C in RNAlater™ (Sigma-Aldrich, MO, USA). Three specimens emerging from the same egg mass were sampled from 10 egg masses parasitized by each species; hence a total of 30 individuals from each exotic species were used. RNA extraction was performed with the “SV Total RNA Isolation System” (Promega, WI, USA) according to the manufacturer instructions. RNA quality and concentration were assessed with a ND-1000 spectrophotometer (NanoDrop, DE, USA). First-strand cDNA was synthesized by “Reverse Transcription System” (Promega) and Random Primers. cDNA was used as a template for qPCR analyses with the P. carbekii-specific primers PcarQF/PcarQR as described by Gonella et al. (2019). Reactions were performed on a CFX ConnectTM Real-Time PCR Detection System (Bio-Rad, CA, USA) in 25 μl volume containing: 12.5 μl of SsoAdvancedTM Universal SYBR® Green Supermix (Bio-Rad), 0.1 μl of 100 μM forward and reverse primer, 11.3 μl of sterile water, and 1 μl of cDNA template. Standard curves were constructed with cloned PCR-amplified 16S rRNA gene of P. carbekii. Standard clones were obtained using the pGEM T-easy Vector Cloning Kit (Promega, WI, USA). The detection limit was calculated as the lowest concentration of cloned amplicons used for determining the standard curves that were successfully amplified, corresponding to 4.60 gene copies/sample. An additional qPCR targeting the insect’s 18S rRNA gene (MqFw/MqRv) was performed, to verify if parasitoids emerged from treated egg masses were truly devoid of P. carbekii or whether the absence of symbiont was due to sample quality. Primers were used according to Marzachì and Bosco (2005), under the conditions described by Gonella et al. (2015).
Statistical analysis
In no-choice tests, parasitoid emergence (mean number of successfully parasitized eggs per egg mass) and egg mortality (mean number of unhatched eggs per egg mass) were evaluated for each parasitoid species considering: (i) only egg masses in which at least one parasitoid had successfully emerged, or (ii) all offered egg masses (N = 10) per treatment. Separately for each parasitoid species, means were then compared among treatments using a generalized linear model (GLM) with a binomial probability distribution and a logit link function, followed by a pairwise Bonferroni post hoc test (p ≤ 0.05). For binomial probability distribution, success/fail conditions were considered as the numbers of parasitized/non-parasitized eggs and unhatched eggs/eggs with any emergence.
In paired choice tests, the percentages of wasps parasitizing only one exposed egg mass were compared with a Pearson’s Chi-square test. The number of emerged parasitoids in parasitized egg masses and the number of unhatched egg masses were then evaluated using a GLM with a binomial probability distribution followed by a sequential Bonferroni post hoc test (p ≤ 0.05). Replicates in which both egg masses were parasitized were not included in the analysis.
Statistical analyses were carried out with SPSS Statistics 27 (IBM Corp. released 2020, Armonk, NY, USA).
Results
No-choice tests
Females of all egg parasitoid species were alive at the end of the tests, with no apparent fitness perturbation; moreover, they all were able to oviposit on H. halys eggs. However, only exotic species were able to successfully parasitize eggs in all the 10 replicates, whereas native species proved to successfully parasitize eggs in 5 up to 9 of the 10 replicates (Table 1). For this reason, the results from native wasps were submitted to two separate statistical analyses, shown in Table 1 (considering only parasitized egg masses), and in Fig. 1 and Table S1 (considering all replicates). The emergence percentage from H. halys eggs was generally higher in the exotic species than in the others (Table 1). Specific responses observed for each parasitoid species are described below.
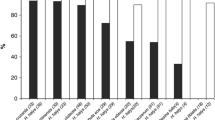
Results of no-choice tests with native parasitoid species, considering all replicates. Mean of emergence rates (a), and mean of egg mortality rates (b) recorded for H. halys egg masses exposed to different egg parasitoids and treatments. Bars indicate standard errors; asterisks indicate significant differences between treatments according to binomial GLM, with p < 0.005 (**) or p < 0.001 (***); n.s. = not significant
Anastatus bifasciatus: when only successfully parasitized egg masses were considered, emergence of A. bifasciatus was significantly different between treatments (p < 0.001), with the lowest percentage of wasps emerging from egg masses treated with Dentamet® (Table 1). Moreover, higher percentages of unhatched eggs were found in untreated and treated with water egg masses (Table 1). However, when considering all replicates, there were no significant differences among treatments in parasitoid emergence, while Dentamet® still showed the lowest percentage of unhatched eggs compared to the other two groups (Fig. 1, Table S1).
Ooencyrtus telenomicida: according to both statistical evaluations (considering only parasitized egg masses or all replicates), females of this native species did not show significant differences in emergence among treatments (Table 1, Fig. 1, Table S1). Treatment with Dentamet® produced a significant reduction of unhatched eggs when only parasitized egg masses were analysed (p < 0.001) (Table 1).
Trissolcus kozlovi: among the tested native wasps, the highest emergence percentages were observed for this species, without significant differences among the three treatments in both statistical evaluations (only parasitized or all egg masses) (Table 1, Fig. 1, Table S1). Egg masses treated with Dentamet® showed significantly lower percentages of unhatched eggs than the other two groups (Fig. 1, Table 1).
Trissolcus japonicus: emergence was significantly higher in egg masses treated with Dentamet® than in the two other groups (p < 0.001) (Table 1). Statistical analyses confirmed a significant reduction of egg mortality in the Dentamet® treatment with respect to the other groups (Table 1).
Trissolcus mitsukurii: despite the percentage of parasitoid emergence being higher than 80%, egg masses treated with Dentamet® showed significantly lower emergence (p < 0.001) than the other groups, and significantly higher percentages of unhatched eggs only in comparison to the group treated with water (Table 1).
Unexposed egg masses: the percentage of eggs that successfully hatched was very high (average 84.26%), and no significant differences were found in egg mortality in different treatments (Table 1).
Paired choice tests
Due to their higher efficiency, T. japonicus and T. mitsukurii were selected for paired choice tests. Trials revealed that some T. japonicus and T. mitsukurii females were able to parasitize both egg masses within 3 h (Fig. 2, Table 2). For these replicates, the first choice was assessed by visual observation, and selection responses were compared with a Pearson’s Chi-square test (Table 2). The first egg masses that T. japonicus females visited always corresponded to the parasitized egg masses. In cases where both egg masses were parasitized, the first visit was recorded on the egg mass that showed the higher number of emerged parasitoids. The same behaviour was observed for T. mitsukurii, except for two cases in the untreated vs treated with water pairs. In these replicates, both egg masses were parasitized but the higher number of parasitoids emerged from the second visited egg mass. The number of females that parasitized both egg masses was higher in T. mitsukurii than in T. japonicus; moreover, all the examined females parasitized at least one egg mass (Fig. 2, Table 2).
Trissolcus japonicus females did not show any preference among treatments, choosing indifferently between all paired egg masses (Fig. 2, Table 2). Percentages of emerging parasitoids and egg mortality were not significantly different within each pair examined (Table 2, Table S2).
Trissolcus mitsukurii females chose untreated egg masses significantly more times when compared with egg masses treated with water or treated with Dentamet®. Egg masses treated with Dentamet® were chosen significantly less times when compared with egg masses untreated or treated with water (Fig. 3, Table 2). However, the percentages of parasitized eggs were always above 75% (Table 2).
Results of paired choice tests performed with T. mitsukurii females exposed to egg masses of the following groups: untreated (white boxes), treated with water (grey boxes), treated with Dentamet® (black boxes). Asterisks indicate significant differences between the number of egg parasitoids that chose one or the other egg mass according to the Pearson’s Chi-square test, with p < 0.050 (*) or p < 0.005 (**); n.s.: not significant
Evaluation of P. carbekii horizontal transmission
Molecular analysis was performed on the two exotic parasitoids emerging from untreated egg masses, to verify if the primary symbiont of H. halys may be horizontally transferred from the egg mass surface to the wasp. However, the presence of live P. carbekii cells was not found in either analysed species through Real time PCR, as only standard samples showed successful amplification. All wasp samples tested positive when submitted to insect-targeted qPCR reactions, confirming their suitable DNA quality.
Discussion
This work aimed at assessing the potential interference between the application of Dentamet® on H. halys egg masses and parasitism by both indigenous and exotic egg parasitoids. Our experimental observations firstly confirmed the absence of direct harmful effects on parasitoid females resulting from their contact with egg masses treated with biocomplex Dentamet®, as expected since this product does not contain any insecticidal molecule. The need for pest management strategies that involve a substantial reduction of insecticide use has been widely demanded to reduce the impact on beneficial insects (Leskey et al. 2020; Ludwick et al. 2020); therefore, finding a novel technique that is conservative for parasitoid wasps can considerably support the implementation of IPM programmes.
In no-choice tests using native parasitoids, the recorded parasitoid emergence percentages were consistent with previous observations (Roversi et al. 2016; Stahl et al. 2018; Andreadis et al. 2021; Moraglio et al. 2021a, b; Rot et al. 2021), with only A. bifasciatus being significantly affected by treatment with Dentamet®. However, a reduction of unhatched eggs was reported in replicates subjected to Dentamet® application for all native species. Since the percentages of unhatched eggs were higher in egg masses exposed to native parasitoids than in unexposed ones, a considerable contribution of non-reproductive effects is suggested for these species. These effects include host feeding and oviposition damage (i.e., oviposition followed by host killing and failure of wasp emergence) (Abram et al. 2016; Stahl et al. 2019b; Moraglio et al. 2021b). Treatment with Dentamet® may deter from host feeding more than from oviposition; establishing the actual contribution of the mechanisms causing non-reproductive effects under different conditions may deserve further investigations. Overall, no evident negative impact on the fitness of these wasp species was found in short-term, since the progeny number was not affected (considering all performed replicates). However, our data suggest that a long-term evaluation may be required to estimate if the total longevity and fecundity of native wasp females exposed to treated eggs may be hampered due to limitation of host feeding. On a food web perspective, the outcome of field treatments with Dentamet® will fit into the already complex dynamics that are being created by the introduction of the exotic stink bug host and parasitoids; this intricate web deserves further study.
As expected, no-choice tests with exotic wasps generally showed higher parasitization levels, as T. japonicus and T. mitsukurii were the sole species able to parasitize all the exposed egg masses. Emergence percentages were similar to those previously reported (Sabbatini-Peverieri et al. 2020), and they were always above 74%. On the other hand, the percentage of unhatched eggs after exposure to these wasp species was generally lower than for native parasitoids. This may be caused by (i) a higher efficiency of adult emergence after oviposition, and (ii) a lower host feeding behaviour (Abram et al. 2014, 2016, 2019; Kaser et al. 2018). Trissolcus japonicus most efficiently parasitized egg masses treated with Dentamet® in comparison to the other two types. Several substances (e.g. host cuticular hydrocarbons and plant volatiles) have been shown to attract T. japonicus (Akotsen-Mensah et al. 2021; Arif et al. 2021). We cannot exclude that some of them may share components with those included in the odour blend resulting after our treatment, even though the volatile compounds associated with egg masses treated with Dentamet® are currently unknown. The higher wasp emergence percentage in the group treated with Dentamet® was also the major cause for the reduced percentage of unhatched eggs, whereas a little contribution of non-reproductive effects is expected for this species.
In contrast, T. mitsukurii showed significantly lower emergence percentages in egg masses treated with Dentamet®, despite the high proportion of parasitized eggs (> 81%) for all treatments. Since the exposure to H. halys egg masses in the absence of the host plant was reported to be inefficient in attracting this wasp (Rondoni et al. 2022), the odour blend emitted after treatment with Dentamet® may further impair the wasp recognition of the host eggs, at least in our experimental conditions, where no plant was offered to the wasp. The elimination of P. carbekii may interfere with the attraction exerted by some symbiont-derived volatiles; the interference with attraction for volatiles may be exacerbated by the general non-preference of T. mitsukurii for wet eggs, as evidenced by the untreated vs treated with water paired choice test. Strikingly, we observed a possible interaction of treatment with non-reproductive effects, as egg masses treated with Dentamet® showed the highest percentages of unhatched eggs, with a statistical significance when compared with the water-treated ones. Future work is required to clarify whether the recorded lower attraction by T. mitsukurii is associated or not with unsuitability of Dentamet®-treated eggs; however, as these eggs are less attractive, the possible unsuitability does not seem to be of concern. Furthemore, all percentages of unhatched eggs after exposure to T. mitsukurii were similar to the natural proportion observed for egg masses that were not exposed to any parasitoid, indicating that the alteration of non-reproductive effects after treatment with Dentamet® is still modest for this wasp.
The results recorded in no-choice experiments using the exotic parasitoids T. japonicus and T. mitsukurii were confirmed also in paired choice tests. Haye et al. (2020) suggested that a period of less than 12 h is preferable for paired choice tests involving egg parasitoids of Pentatomidae, to limit the number of females using both egg masses for oviposition, which results in hampered interpretation of host preference. Therefore, we provided the females with egg masses for as little as 3 h. Even in this short period, all females of both species were able to parasitize at least one egg mass, and some of them could still parasitize both egg masses. A diverging behaviour was observed for the two wasp species: both species tended to attack only one of the two masses (more than 50% of parasitism events), but only T. mitsukurii showed a significant preference, mostly discarding egg mass treated with Dentamet®. After the egg mass was chosen, also the number of parasitized eggs was different for the two species: emergence percentage for T. japonicus was not affected by treatments, whereas we recorded a larger offspring on untreated or water treated egg masses for T. mitsukurii, consistently with the results of no-choice tests. The mechanisms (e.g. altered volatile composition) causing such differential behaviour in the host selection process in T. japonicus and T. mitsukurii after egg mass exposure to Dentamet® requires further investigations.
Besides the diverging outcome of the short-term preference exhibited by the two Trissolcus species, we assessed the occurrence of possible long-term effects on the coevolved host-parasitoid interaction occurring between these exotic wasps and their host H. halys. Specifically, we assessed the possible occurrence of a natural horizontal transmission of P. carbekii from stink bug egg masses (in untreated specimens) to the adult parasitoids, to figure out if a beneficial relation may have established between the symbiont and wasps over time. The symbiont is vertically transmitted through the egg mass surface (Prado et al. 2006), and this route may have exposed egg parasitoids to acquire the bacterium during emergence from the egg. Horizontal transmission of bacterial symbionts from host to parasitoids have been documented in a number of insects (Dicke et al. 2020), and it may be required for wasp long-term performance, resulting in impaired competition of symbiont-deprived parasitoids. However, qPCR screening of cDNA from untreated samples of both exotic egg parasitoids suggested a lack of P. carbekii acquisition from the host, allowing us to exclude any detrimental effects due to possible changes in the microbial community of wasps emerged from treated egg masses.
Taken together, our results support the combination of biological control, especially when performed by exotic egg parasitoids, and symbiotic control (at least using Dentamet® or other products with a similar mode of action). Such an integrated approach offers a potential control effectiveness close to 100% with total avoidance of insecticidal molecules. In fact, the efficiency recorded for T. japonicus and T. mitsukurii was always above 75%; furthermore, as reported by Gonella et al. (2019), more than 90% of H. halys nymphs emerging from egg masses treated with Dentamet® are deprived of P. carbekii and die before reaching the second instar, highlighting the powerful potential of a combined effect. Nonetheless, further work will be needed to clarify the containment of H. halys using symbiotic control under field conditions. Indeed, possible failure in reaching the egg masses through field application of the anti-symbiont formulate may reduce the final pest mortality; similarly, parasitoid populations may be unable to reach all the egg masses. On the other hand, the combined effect offered by a combined approach may become even synergistic in the field, with special regard to the diverging response observed for T. japonicus and T. mitsukurii in terms of egg mass selection. In the field, the unavoidable concurrent presence of egg masses reached by foliar treatments and egg masses that escape the sprays, may offer a choice for T. japonicus and T. mitsukurii. Egg mass selection may be differentially driven by the treatment for the two wasp species, resulting in a broader range of attacked masses. While T. japonicus can parasitize egg masses regardless of the presence of Dentamet® on their surface (providing an increment of mortality from treated egg masses), T. mitsukurii may seek for egg masses that were not reached by the treatment. It is worth dedicating future work to analyse such a scenario, since it would increase the final suppression effect, also limiting the competition between the two wasp species.
Author contributions
BO, STM, EG, AA and LT conceived the ideas and designed the methodology; BO, STM and FT collected the data; BO and STM analysed the data; BO, STM and EG wrote the manuscript; FT, LT and AA critically reviewed the manuscripts; AA and LT acquired funds. All authors contributed critically to the drafts and gave final approval for publication.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Abram PK, Gariepy TD, Boivin G, Brodeur J (2014) An invasive stink bug as an evolutionary trap for an indigenous egg parasitoid. Biol Invasions 16:1387–1395. https://doi.org/10.1007/s10530-013-0576-y
Abram PK, Brodeur J, Burte V, Boivin G (2016) Parasitoid-induced host egg abortion: an underappreciated component of biological control services provided by egg parasitoids. Biol Control 98:52–60. https://doi.org/10.1016/j.biocontrol.2016.04.002
Abram PK, Brodeur J, Urbaneja A, Tena A (2019) Nonreproductive effects of insect parasitoids on their hosts. Annu Rev Entomol 64:259–276. https://doi.org/10.1146/annurev-ento-011118-111753
Abram PK, Hoelmer KA, Acebes-Doria A, Andrews H, Beers EH, Bergh JC, Bessin R, Biddinger D, Botch P, Buffington ML, Cornelius ML, Costi E, Delfosse ES, Dieckhoff C, Dobson R, Donais Z, Grieshop M, Hamilton G, Haye T, Hedstrom C, Herlihy MV, Hoddle MS, Hooks CRR, Jentsch P, Joshi NK, Kuhar TP, Lara J, Lee JC, Legrand A, Leskey TC, Lowenstein D, Maistrello L, Mathews CR, Milnes JM, Morrison WR III, Nielsen AL, Ogburn EC, Pickett CH, Poley K, Pote J, Radl J, Shrewsbury PM, Talamas E, Tavella L, Walgenbach JF, Waterworth R, Weber DC, Welty C, Wiman NG (2017) Indigenous arthropod natural enemies of the invasive brown marmorated stink bug in North America and Europe. J Pest Sci 90:1009–1020. https://doi.org/10.1007/S10340-017-0891-7
Akotsen-Mensah C, Blaauw BR, Short B, Leskey TC, Bergh JC, Polk D, Nielsen AL (2020) Using IPM-CPR as a management program for apple orchards. J Econ Entomol 113:1894–1902. https://doi.org/10.1093/jee/toaa087
Akotsen-Mensah C, Blaauw BR, Rivera MJ, Rodriguez-Saona C, Nielsen AL (2021) Behavioral response of Halyomorpha halys (Hemiptera: Pentatomidae) and its egg parasitoid Trissolcus japonicus (Hymenoptera: Scelionidae) to host plant odors. Front Ecol Evol 9:696814. https://doi.org/10.3389/fevo.2021.696814
Andreadis SS, Gogolashvili NE, Fifis GT, Navrozidis EI, Thomidis T (2021) First report of native parasitoids of Halyomorpha halys (Hemiptera: Pentatomidae) in Greece. Insects 12:984. https://doi.org/10.3390/insects12110984
Arakawa R, Namura Y (2002) Effects of temperature on development of three Trissolcus spp. (Hymenoptera: Scelionidae), egg parasitoids of the brown marmorated stink bug, Halyomorpha halys (Hemiptera: Pentatomidae). Entomol Sci 5:215–218
Arif MA, Alınç T, Guarino S, Colazza S, Cusumano A, Peri E (2021) Only females oviposit: chemical discrimination of adult stink bug sex by the egg parasitoid Trissolcus japonicus. Front Ecol Evol 9:652561. https://doi.org/10.3389/fevo.2021.652561
Bariselli M, Bugiani R, Maistrello L (2016) Distribution and damage caused by Halyomorpha halys in Italy. Bull OEPP 46:332–334. https://doi.org/10.1111/epp.12289
Bergmann EJ, Venugopal PD, Martinson HM, Raupp MJ, Shrewsbury PM (2016) Host plant use by the invasive Halyomorpha halys (Stål) on woody ornamental trees and shrubs. PLoS ONE 11:e0149975. https://doi.org/10.1371/journal.pone.0149975
Blaauw BR, Polk D, Nielsen AL (2015) IPM-CPR for peaches: incorporating behaviorally-based methods to manage Halyomorpha halys and key pests in peach. Pest Manag Sci 71:1513–1522. https://doi.org/10.1002/ps.3955
Bosco L, Moraglio ST, Tavella L (2018) Halyomorpha halys, a serious threat for hazelnut in newly invaded areas. J Pest Sci 91:661–670. https://doi.org/10.1007/s10340-017-0937-x
Bout A, Tortorici F, Hamidi R, Warot S, Tavella L, Thomas M (2021) First detection of the adventive egg parasitoid of Halyomorpha halys (Stål) (Hemiptera: Pentatomidae) Trissolcus mitsukurii (Ashmead) (Hymenoptera: Scelionidae) in France. Insects 12:761. https://doi.org/10.3390/insects12090761
Caron V, Yonow T, Paull C, Talamas EJ, Avila GA, Hoelmer KA (2021) Preempting the arrival of the Brown Marmorated Stink Bug, Halyomorpha halys: biological control options for Australia. Insects 12:581. https://doi.org/10.3390/insects12070581
Cira TM, Burkness EC, Koch RL, Hutchison WD (2017) Halyomorpha halys mortality and sublethal feeding effects following insecticide exposure. J Pest Sci 90:1257–1268. https://doi.org/10.1007/s10340-017-0871-y
Conti E, Avila G, Barratt B, Cingolani F, Colazza S, Guarino S, Hoelmer K, Laumann RA, Maistrello L, Martel G, Peri E, Rodriguez-Saona C, Rondoni G, Rostás M, Roversi PF, Sforza RFH, Tavella L, Wajnberg E (2021) Biological control of invasive stink bugs: review of global state and future prospects. Entomol Exp Appl 169:28–51. https://doi.org/10.1111/EEA.12967
Costi E, Haye T, Maistrello L (2019) Surveying native egg parasitoids and predators of the invasive Halyomorpha halys in Northern Italy. J Appl Entomol 143:299–307. https://doi.org/10.1111/JEN.12590
Dicke M, Cusumano M, Poelman EH (2020) Microbial symbionts of parasitoids. Annu Rev Entomol 65: 171–190. https://doi.org/10.1146/annurev-ento-011019-024939
Fisher JJ, Rijal JP, Zalom FG (2020) Temperature and humidity interact to influence brown marmorated stink bug (Hemiptera: Pentatomidae), survival. Environ Entomol 20:1–9. https://doi.org/10.1093/ee/nvaa146
Giovannini L, Sabbatini-Peverieri G, Marianelli L, Rondoni G, Conti E, Roversi PF (2022) Physiological host range of Trissolcus mitsukurii, a candidate biological control agent of Halyomorpha halys in Europe. J Pest Sci 95: 605–618. https://doi.org/10.1007/s10340-021-01415-x
Gonella E, Pajoro M, Marzorati M, Crotti E, Mandrioli M, Pontini M, Bulgari D, Negri I, sacchi L, Chouaia B, daffonchio D, Alma, A. (2015) Plant-mediated interspecific horizontal transmission of an intracellular symbiont in insects. Sci Rep 5:15811. https://doi.org/10.1038/srep15811
Gonella E, Orrù B, Alma A (2019) Egg masses treatment with micronutrient fertilizers has a suppressive effect on newly-emerged nymphs of the brown marmorated stink bug Halyomorpha halys. Entomol Gen 39:231–238. https://doi.org/10.1127/entomologia2019/0819
Gonella E, Orrù B, Marasco R, Daffonchio D, Alma A (2020) Disruption of host-symbiont associations for the symbiotic control and management of pentatomid agricultural pest-a review. Front Microbiol 11:547031. https://doi.org/10.3389/fmicb.2020.547031
Gualtieri L, Nugnes F, Nappo AG, Gebiola M, Bernardo U (2017) Life inside a gall: closeness does not favour horizontal transmission of Rickettsia between a gall wasp and its parasitoid. FEMS Microbiol Ecol. https://doi.org/10.1093/femsec/fix087
Haye T, Fischer S, Zhang J, Gariepy T (2015) Can native egg parasitoids adopt the invasive brown marmorated stink bug, Halyomorpha halys (Hemiptera: Pentatomidae), in Europe? J Pest Sci 88:693–705. https://doi.org/10.1007/s10340-015-0671-1
Haye T, Weber DC (2017) Special issue on the brown marmorated stink bug, Halyomorpha halys: an emerging pest of global concern. J Pest Sci 90:987–988. https://doi.org/10.1007/s10340-017-0897-1
Haye T, Moraglio ST, Stahl J, Visentin S, Gregorio T, Tavella L (2020) Fundamental host range of Trissolcus japonicus in Europe. J Pest Sci 93:171–182. https://doi.org/10.1007/s10340-019-01127-3
Inkley DB (2012) Characteristics of home invasion by the brown marmorated stink bug (Hemiptera: Pentatomidae). J Entomol Sci 47:125–130. https://doi.org/10.18474/0749-8004-47.2.125
Kamiyama MT, Matsuura K, Hata T, Yoshimura T, Yang CCS (2022) Seasonal parasitism of native egg parasitoids of brown marmorated stink bug (Halyomorpha halys) in Japan. J Pest Sci 95:1067–1079. https://doi.org/10.1007/s10340-021-01455-3
Kaser JM, Nielsen AL, Abram PK (2018) Biological control effects of non-reproductive host mortality caused by insect parasitoids. Ecol Appl 28:1081–1092. https://doi.org/10.1002/eap.1712
Khadka A, Hodges AC, Leppla NC, Tillman PG (2020) The effects of relative humidity on Halyomorpha halys (Stål) (Hemiptera: Pentatomidae) egg hatch, nymph survival, and adult reproduction. Fla Entomol 103:136–138. https://doi.org/10.1653/024.103.0424
Kuhar TP, Kamminga K (2017) Review of the chemical control research on Halyomorpha halys in the USA. J Pest Sci 90:1021–1031. https://doi.org/10.1007/s10340-017-0859-7
Lee DH, Short BD, Joseph SV, Bergh JC, Leskey TC (2013) Review of the biology, ecology, and management of Halyomorpha halys (Hemiptera: Pentatomidae) in China, Japan, and the Republic of Korea. Environ Entomol 42:627–641. https://doi.org/10.1603/EN13006
Lee DH (2015) Current status of research progress on the biology and management of Halyomorpha halys (Hemiptera: Pentatomidae) as an invasive species. Appl Entomol Zool 50:277–290. https://doi.org/10.1007/S13355-015-0350-Y
Leskey TC, Hamilton GC, Nielsen AL, Polk DF, Rodriguez-Saona C, Bergh JC, Herbert DA, Kuhar TP, Pfeiffer D, Dively GP, Hooks CRR, Raupp MJ, Shrewsbury PM, Krawczyk G, Shearer PW, Whalen J, Koplinka-Loehr C, Myers E, Inkley D, Hoelmer KA, Lee DH, Wright SE (2012a) Pest status of the brown marmorated stink bug, Halyomorpha halys in the USA. Outlooks Pest Manag 23:218–226. https://doi.org/10.1564/23oct07
Leskey TC, Lee DH, Short BD, Wright SE (2012b) Impact of insecticides on the invasive Halyomorpha halys (Hemiptera: Pentatomidae): analysis of insecticide lethality. J Econ Entomol 105:1726–1735. https://doi.org/10.1603/EC12096
Leskey TC, Nielsen AL (2018) Impact of the invasive brown marmorated stink bug in North America and Europe: history, biology, ecology, and management. Annu Rev Entomol 63:599–618. https://doi.org/10.1146/annurev-ento-020117-043226
Leskey TC, Short BD, Ludwick DC (2020) Comparison and refinement of integrated pest management tactics for Halyomorpha halys (Hemiptera: Pentatomidae) management in apple orchards. J Econ Entomol 113:1725–1734. https://doi.org/10.1093/jee/toaa067
Lowenstein DM, Andrews H, Mugica A, Wiman NG (2019) Sensitivity of the egg parasitoid Trissolcus japonicus (Hymenoptera: Scelionidae) to field and laboratory-applied insecticide residue. J Econ Entomol 112:2077–2084. https://doi.org/10.1093/jee/toz127
Ludwick DC, Patterson J, Leake LB, Carper L, Leskey TC (2020) Integrating Trissolcus japonicus (Ashmead, 1904) (Hymenoptera: Scelionidae) into management programs for Halyomorpha halys (Stål, 1855) (Hemiptera: Pentatomidae) in apple orchards: impact of insecticide applications and spray patterns. Insects 11:833. https://doi.org/10.3390/insects11120833
Marzachì C, Bosco D (2005) Relative quantification of chrysanthemum yellows (16Sr I) phytoplasma in its plant and insect host using real-time polymerase chain reaction. Mol Biotech 30:117–127. https://doi.org/10.1385/MB:30:2:11
Mathews CR, Barry S (2014) Compost tea reduces egg hatch and early-stage nymphal development of Halyomorpha halys (Hemiptera: Pentatomidae). Fla Entomol 97:1726–1732. https://doi.org/10.1653/024.097.0448
Moraglio ST, Tortorici F, Pansa MG, Castelli G, Pontini M, Scovero S, Visentin S, Tavella L (2020) A 3-year survey on parasitism of Halyomorpha halys by egg parasitoids in northern Italy. J Pest Sci 93:183–194. https://doi.org/10.1007/S10340-019-01136-2
Moraglio ST, Tortorici F, Visentin S, Pansa MG, Tavella L (2021a) Trissolcus kozlovi in north Italy: host specificity and augmentative releases against Halyomorpha halys in hazelnut orchards. Insects 12:464. https://doi.org/10.3390/insects12050464
Moraglio ST, Tortorici F, Giromini D, Pansa MG, Visentin S, Tavella L (2021b) Field collection of egg parasitoids of Pentatomidae and Scutelleridae in Northwest Italy and their efficacy in parasitizing Halyomorpha halys under laboratory conditions. Entomol Exp Appl 169:52–63. https://doi.org/10.1111/eea.12966
Nielsen AL, Shearer PW, Hamilton GC (2008) Toxicity of insecticides to Halyomorpha halys (Hemiptera: Pentatomidae) using glass-vial bioassays. J Econ Entomol 101:1439–1442. https://doi.org/10.1093/jee/101.4.1439
Otero-Bravo A, Sabree ZL (2015) Inside or out? Possible genomic consequences of extracellular transmission of crypt dwelling stinkbug mutualists. Front Ecol Evol 3:64. https://doi.org/10.3389/fevo.2015.00064
Prado SS, Rubinoff D, Almeida RPP (2006) Vertical transmission of a pentatomid caeca-associated symbiont. Ann Entomol Soc Am 99:577–585. https://doi.org/10.1603/0013-8746
Qi LD, Sun JT, Hong XY, Li YX (2019) Diversity and phylogenetic analyses reveal horizontal transmission of endosymbionts between whiteflies and their parasitoids. J Econ Entomol 112:894–905. https://doi.org/10.1093/jee/toy367
Rice KB, Bergh CJ, Bergmann EJ, Biddinger DJ, Dieckhoff C, Dively G, Fraser H, Gariepy T, Hamilton G, Haye T, Herbert A, Hoelmer K, Hooks CR, Jones A, Krawczyk G, Kuhar T, Martinson H, Mitchell W, Nielsen AL, Pfeiffer DG, Raupp MJ, Rodriguez-Saona C, Shearer P, Shrewsbury P, Venugopal PD, Whalen J, Wiman NG, Leskey TC, Tooker JF (2014) Biology, ecology, and management of brown marmorated stink bug (Hemiptera: Pentatomidae). J Integr Pest Manag 5:1–13. https://doi.org/10.1603/IPM14002
Rondoni G, Chierici E, Giovannini L, Sabbatini-Peverieri G, Roversi PF, Conti E (2022) Olfactory responses of Trissolcus mitsukurii to plants attacked by target and non-target stink bugs suggest low risk for biological control. Sci Rep 12:1880. https://doi.org/10.1038/s41598-022-05873-w
Rot M, Maistrello L, Costi E, Bernardinelli I, Malossini G, Benvenuto L, Trdan S (2021) Native and non-native egg parasitoids associated with brown marmorated stink bug (Halyomorpha halys [Stål, 1855]; Hemiptera: Pentatomidae) in Western Slovenia. Insects 12:505. https://doi.org/10.3390/insects12060505
Roversi PF, Binazzi F, Marianelli L, Costi E, Maistrello L, Sabbatini-Peverieri G (2016) Searching for native egg-parasitoids of the invasive alien species Halyomorpha halys Stal (Heteroptera: Pentatomidae) in Southern Europe. Redia 99:63–70. https://doi.org/10.19263/REDIA-99.16.01
Sabbatini-Peverieri G, Talamas S, Bon MC, Marianelli L, Bernadirelli I, Malossini G, Benvenuto L, Roversi PF, Hoelmer K (2018) Two Asian egg parasitoids of Halyomorpha halys (Stål) (Hemiptera, Pentatomidae) emerge in northern Italy: Trissolcus mitsukurii (Ashmead) and Trissolcus japonicus (Ashmead) (Hymenoptera, Scelionidae). J Hymenopt Res 67:37–53. https://doi.org/10.3897/JHR.67.30883
Sabbatini-Peverieri G, Dieckhoff C, Giovannini L, Marianelli L, Roversi PF, Hoelmer K (2020) Rearing Trissolcus japonicus and Trissolcus mitsukurii for biological control of Halyomorpha halys. Insects 11:787. https://doi.org/10.3390/insects11110787
Sabbatini-Peverieri G, Boncompagni L, Mazza G, Paoli F, Dapporto L, Giovannini L, Marianelli L, Hoelmer K, Roversi PF (2021) Combining physiological host range, behavior and host characteristics for predictive risk analysis of Trissolcus japonicus. J Pest Sci 94:1003–1016. https://doi.org/10.1007/S10340-020-01311-W
Scaccini D, Duso C, Pozzebon A (2019) Lethal effects of high temperatures on brown marmorated stink bug adults before and after overwintering. Insects 10:355. https://doi.org/10.3390/insects10100355
Scaccini D, Falagiarda M, Tortorici F, Martinez-Sanudo I, Tirello P, Reyes-Domínguez Y, Gallmetzer A, Tavella L, Zandigiacomo P, Duso C, Pozzebon A (2020) An insight into the role of Trissolcus mitsukurii as biological control agent of Halyomorpha halys in northeastern Italy. Insects 11:306. https://doi.org/10.3390/insects11050306
Stahl JM, Babendreier D, Haye T (2018) Using the egg parasitoid Anastatus bifasciatus against the invasive brown marmorated stink bug in Europe: can non-target effects be ruled out? J Pest Sci 91:1005–1017. https://doi.org/10.1007/S10340-018-0969-X
Stahl JM, Babendreier D, Marazzi C, Caruso S, Costi E, Maistrello L, Haye T (2019a) Can Anastatus bifasciatus be used for augmentative biological control of the brown marmorated stink bug in fruit orchards? Insects 10:108–121. https://doi.org/10.3390/insects10040108
Stahl JM, Babendreier D, Haye T (2019b) Life history of Anastatus bifasciatus, a potential biological control agent of the brown marmorated stink bug in Europe. Biocontrol 129:178–186. https://doi.org/10.1016/j.biocontrol.2018.10.016
Stahl JM, Tortorici F, Pontini M, Bon MC, Hoelmer K, Marazzi C, Tavella L, Haye T (2019c) First discovery of adventive populations of Trissolcus japonicus in Europe. J Pest Sci 92:371–379. https://doi.org/10.1007/S10340-018-1061-2
Taylor CM, Coffey PL, DeLay BD, Dively GP (2014) The importance of gut symbionts in the development of the brown marmorated stink bug, Halyomorpha halys (Stål). PLoS ONE 9:e90312. https://doi.org/10.1371/journal.pone.009031
Taylor CM, Johnson V, Dively G (2017) Assessing the use of antimicrobials to sterilize brown marmorated stink bug egg masses and prevent symbiont acquisition. J Pest Sci 90:1287–1294. https://doi.org/10.1007/s10340-016-0814-z
Vavre F, Fleury F, Lepetit D, Fouillet P, Boulétreau M (1999) Phylogenetic evidence for horizontal transmission of Wolbachia in host-parasitoid associations. Mol Biol Evol 16:1711–1723. https://doi.org/10.1093/oxfordjournals.molbev.a026084
Yang ZQ, Yao YX, Qiu LF, Li ZX (2009) A new species of Trissolcus (Hymenoptera: Scelionidae) parasitizing eggs of Halyomorpha halys (Heteroptera: Pentatomidae) in China with comments on its biology. Ann Entomol Soc Am 02:39–47. https://doi.org/10.1603/008.102.0104
Yang YL, Zhong YZ, Zhang F, Zhou CQ, Yang SY, Zhang JP (2015) Parasitic capacity of Trissolcus halyomorphae and T. flavipes (Hymenoptera: Scelionidae) on eggs of Halyomorpha halys. Environ Entomol 7:1257–1262
Zapponi L, Bon MC, Fouani JM, Anfora G, Schmidt S, Falagiarda M (2020) Assemblage of the egg parasitoids of the invasive stink bug Halyomorpha halys: insights on plant host associations. Insects 11:1–17. https://doi.org/10.3390/insects11090588
Zapponi L, Tortorici F, Anfora G, Bardella S, Bariselli M, Benvenuto L, Bernardinelli I, Butturini A, Caruso S, Colla R, Costi E, Culatti P, Di Bella E, Falagiarda M, Giovannini L, Haye T, Maistrello L, Malossini G, Marazzi C, Marianelli L, Mele A, Michelon L, Moraglio ST, Pozzebon A, Preti M, Salvetti M, Scaccini D, Schmidt A, Szalatnay D, Roversi PF, Tavella L, Tommasini MG, Vaccari G, Zandigiacomo P, Sabbatini-Peverieri G (2021) Assessing the distribution of exotic egg parasitoids of Halyomorpha halys in Europe with a large-scale monitoring program. Insects 12:316. https://doi.org/10.3390/insects12040316
Zhang J, Zhang F, Gariepy T, Mason P, Gillespie D, Talamas E, Haye T (2017) Seasonal parasitism and host specificity of Trissolcus japonicus in northern China. J Pest Sci 90:1127–1141. https://doi.org/10.1007/S10340-017-0863-Y
Acknowledgements
The authors thank Sara Scovero and Sara Morello for their essential help in insect rearing and support during experimental work.
Funding
Open access funding provided by Università degli Studi di Torino within the CRUI-CARE Agreement. Research was partially funded by Regione Piemonte.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed while conducting this research.
Additional information
Communicated by Chris Cutler.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Orrù, B., Moraglio, S.T., Tortorici, F. et al. No adverse effects of symbiotic control on the parasitism of Halyomorpha halys by egg parasitoids. J Pest Sci 96, 1091–1102 (2023). https://doi.org/10.1007/s10340-022-01576-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-022-01576-3