Abstract
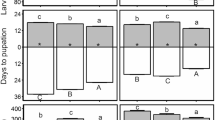
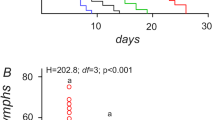
Pest management strategies relying on agrochemicals could be altered by climate change, because of the temperature-dependent toxicity of the compound involved. Many studies have explored the response of targeted pests to pesticide and temperature. Pesticides are seldom strictly selective and also affect nontarget pests. Surprisingly, the way temperature may shape these side effects of pesticides remains overlooked, limiting our understanding of the net impacts of future chemical treatments on the overall damage induced by different pests. We investigated how temperature modulates the response of a major grape insect pest (the tortricid moth Lobesia botrana) to a copper-based fungicide. We examined the lethal (larval survival) and sublethal (larval development, pupal mass, immune parameters) effects of exposure to different concentrations of copper in larval food. We found that copper concentration had negative linear effects on larval development and pupal mass. In addition, copper concentration had biphasic curvilinear effects on total phenoloxidase activity, which is indicative of hormesis (stimulation and inhibition of insect performance at low and high copper concentrations, respectively). Temperature stimulated development, while compromising immunity (total phenoloxidase activity). Significant interaction between copper concentration and temperature was detected for larval survival and phenoloxidase activity: warmer conditions improved pest tolerance to copper through temperature-driven hormesis (larval survival) or by shifting the hormesis-related peak of performance toward higher copper concentrations (phenoloxidase activity). This combination of simple and interactive effects could propagate to populations, communities and agroecosystem, with implications for future management of viticultural pests.


Similar content being viewed by others
Code availability
R scripts used to analyze the data are available upon request.
Data availability
Data involved in this study are available upon request.
References
Abbes K, Biondi A, Kurtulus A, Ricupero M, Russo A, Siscaro G, Chermiti B, Zappalà L (2015) Combined non-target effects of insecticide and high temperature on the parasitoid Bracon nigricans. PLoS ONE 10:e0138411. https://doi.org/10.1371/journal.pone.0138411
Angelova VR, Ivanov AS, Braikov DM (1999) Heavy metals (Pb, Cu, Zn and Cd) in the system soil: grapevine - grape. J Sci Food Agric 79:713–721. https://doi.org/10.1002/(SICI)1097-0010(199904)79:5%3c713::AID-JSFA229%3e3.0.CO;2-F
Baghban A, Sendi JJ, Zibaee A, Khosravi R (2014) Effect of heavy metals (Cd, Cu, and Zn) on feeding indices and energy reserves of the cotton boll worm Helicoverpa armigera Hubner (Lepidoptera: Noctuidae). J Plant Prot Res 54:367–373. https://doi.org/10.2478/jppr-2014-0055
Bengochea P, Budia F, Viñuela E, Medina P (2014) Are kaolin and copper treatments safe to the olive fruit fly parasitoid Psyttalia concolor? J Pest Sci 87:351–359. https://doi.org/10.1007/s10340-013-0543-5
Bereswill R, Golla B, Streloke M, Schulz R (2012) Entry and toxicity of organic pesticides and copper in vineyard streams: erosion rills jeopardise the efficiency of riparian buffer strips. Agric Ecosyst Environ 146:81–92. https://doi.org/10.1016/j.agee.2011.10.010
Biondi A, Mommaerts V, Smagghe G, Viñuela E, Zappalà L, Desneux N (2012) The non-target impact of spinosyns on beneficial arthropods. Pest Manag Sci 68:1523–1536. https://doi.org/10.1002/ps.3396
Brittain CA, Vighi M, Bommarco R, Settele J, Potts SG (2010) Impacts of a pesticide on pollinator species richness at different spatial scales. Basic Appl Ecol 11:106–115. https://doi.org/10.1016/j.baae.2009.11.007
Butler CD, Beckage NE, Trumble JT (2009) Effects of terrestrial pollutants on insect parasitoids. Environ Toxicol Chem 28:1111–1119. https://doi.org/10.1897/08-440.1
Caffarra A, Rinaldi M, Eccel E, Rossi V, Pertot I (2012) Modelling the impact of climate change on the interaction between grapevine and its pests and pathogens: European grapevine moth and powdery mildew. Agric Ecosyst Environ 148:89–101. https://doi.org/10.1016/J.AGEE.2011.11.017
Calabrese EJ (2004) Hormesis: from marginalization to mainstream. A case for hormesis as the default dose-response model in risk assessment. Toxicol Appl Pharmacol 197:125–136. https://doi.org/10.1016/j.taap.2004.02.007
Calabrese EJ, Blain RB (2005) The occurrence of hormetic dose responses in the toxicological literature, the hormesis database: an overview. Toxicol Appl Pharmacol 202:289–301. https://doi.org/10.1016/j.taap.2004.06.023
Calabrese EJ, Blain RB (2011) The hormesis database: the occurrence of hormetic dose responses in the toxicological literature. Regul Toxicol Pharmacol 61:73–81. https://doi.org/10.1016/j.yrtph.2011.06.003
Castex V, Beniston M, Calanca P, Fleury D, Moreau J (2018) Pest management under climate change: the importance of understanding tritrophic relations. Sci Total Environ 616–617:397–407. https://doi.org/10.1016/J.SCITOTENV.2017.11.027
Cedergreen N, Ritz C, Streibig JC (2005) Improved empirical models describing hormesis. Environ Toxicol Chem 24:3166–3172. https://doi.org/10.1897/05-014R.1
Connell DW, Yu QJ, Verma V (2016) Influence of exposure time on toxicity: an overview. Toxicology 355–356:49–53. https://doi.org/10.1016/j.tox.2016.05.015
Cutler GC (2013) Insects, insecticides and hormesis: evidence and considerations for study. Dose-Response 11:154–177. https://doi.org/10.2203/dose-response.12-008
Cutler GC, Guedes RNC (2017) Occurrence and significance of insecticide-induced hormesis in insects. In: Duke SO, Kudsk P, Solomon K (eds) Pesticide Dose: Effects on the Environment and Target and Non-Target Organisms. American Chemical Society, Washington, pp 101–119. https://doi.org/10.1021/bk-2017-1249.ch008
Dagostin S, Schärer HJ, Pertot I, Tamm L (2011) Are there alternatives to copper for controlling grapevine downy mildew in organic viticulture? Crop Prot 30:776–788. https://doi.org/10.1016/j.cropro.2011.02.031
Debecker S, Stoks R (2019) Pace of life syndrome under warming and pollution: integrating life history, behavior, and physiology across latitudes. Ecol Monogr 89:e01332. https://doi.org/10.1002/ecm.1332
Delbac L, Thiéry D (2016) Damage to grape flowers and berries by Lobesia botrana larvae (Denis & Schiffermüller) (Lepidoptera: Tortricidae), and relation to larval age. Aust J Grape Wine Res 22:256–261. https://doi.org/10.1111/ajgw.12204
Delcour I, Spanoghe P, Uyttendaele M (2015) Literature review: impact of climate change on pesticide use. Food Res Int 68:7–15. https://doi.org/10.1016/j.foodres.2014.09.030
Deng C, Graham R, Shukla R (2001) Detecting and estimating hormesis using a model-based approach. Hum Ecol Risk Assess 7:849–866. https://doi.org/10.1080/20018091094691
Desneux N, Decourtye A, Delpuech J-M (2007) The sublethal effects of pesticides on beneficial arthropods. Annu Rev Entomol 52:81–106. https://doi.org/10.1146/annurev.ento.52.110405.091440
EFSA (2018) Review of the existing maximum residue levels for copper compounds according to Article 12 of Regulation (EC) No 396/2005. EFSA J 16:e05212. https://doi.org/10.2903/j.efsa.2018.5212
Garcia PV, Pereira N, Oliveira LM (2009) Side-effects of organic and synthetic pesticides on cold-stored diapausing prepupae of Trichogramma cordubensis. Biocontrol 54:451–458. https://doi.org/10.1007/s10526-008-9186-5
García-Esparza MA, Capri E, Pirzadeh P, Trevisan M (2006) Copper content of grape and wine from Italian farms. Food Addit Contam 23:274–280. https://doi.org/10.1080/02652030500429117
Gessler C, Pertot I, Perazzolli M (2011) Plasmopara viticola: a review of knowledge on downy mildew of grapevine and effective disease management. Phytopathol Mediterr 50:3–44
Gill HK, Garg H (2014) Pesticides: environmental impacts and management strategies. In: Larramendy ML, Soloneski S (eds) Pesticides - Toxic Aspects. InTech, Croatia, pp 187–230. https://doi.org/10.5772/57399
González-Santoyo I, Córdoba-Aguilar A (2012) Phenoloxidase: a key component of the insect immune system. Entomol Exp Appl 142:1–16. https://doi.org/10.1111/j.1570-7458.2011.01187.x
Guedes RNC, Cutler GC (2014) Insecticide-induced hormesis and arthropod pest management. Pest Manag Sci 70:690–697. https://doi.org/10.1002/ps.3669
Guedes RNC, Smagghe G, Stark JD, Desneux N (2016) Pesticide-induced stress in arthropod pests for optimized integrated pest management programs. Annu Rev Entomol 61:43–62. https://doi.org/10.1146/annurev-ento-010715-023646
Gutierrez AP, Ponti L, Gilioli G, Baumgärtner J (2017) Climate warming effects on grape and grapevine moth (Lobesia botrana) in the Palearctic region. Agric for Entomol 20:255–271. https://doi.org/10.1111/afe.12256
Hardin MR, Benrey B, Coll M, Lamp WO, Roderick GK, Barbosa P (1995) Arthropod pest resurgence: an overview of potential mechanisms. Crop Prot 14:3–18. https://doi.org/10.1016/0261-2194(95)91106-P
Hill MP, Macfadyen S, Nash MA (2017) Broad spectrum pesticide application alters natural enemy communities and may facilitate secondary pest outbreaks. PeerJ 5:e4179. https://doi.org/10.7717/peerj.4179
Holmstrup M, Bindesbøl A-M, Oostingh GJ, Duschl A, Scheil V, Köhler H-R, Loureiro S, Soares AMVM, Ferreira ALG, Kienle C, Gerhardt A, Laskowski R, Kramarz PE, Bayley M, Svendsen C, Spurgeon DJ (2010) Interactions between effects of environmental chemicals and natural stressors: a review. Sci Tot Environ 408:3746–3762. https://doi.org/10.1016/j.scitotenv.2009.10.067
Hooper MJ, Ankley GT, Cristol DA, Duschl A, Scheil V, Köhler HR, Loureiro S, Soares AMVM, Ferreira ALG, Kienle C, Gerhardt A, Laskowski R, Kramarz PE, Bayley M, Svendsen C, Spurgeon DJ (2013) Interactions between chemical and climate stressors: a role for mechanistic toxicology in assessing climate change risks. Environ Toxicol Chem 32:32–48. https://doi.org/10.1002/etc.2043
Iltis C, Martel G, Thiéry D, Moreau J, Louâpre P (2018) When warmer means weaker: high temperatures reduce behavioural and immune defences of the larvae of a major grapevine pest. J Pest Sci 91:1315–1326. https://doi.org/10.1007/s10340-018-0992-y
Iltis C, Louâpre P, Pecharová K, Thiéry D, Zito S, Bois B, Moreau J (2019) Are life-history traits equally affected by global warming? A case study combining a multi-trait approach with fine-grain climate modeling. J Insect Physiol 117:103916. https://doi.org/10.1016/J.JINSPHYS.2019.103916
Iltis C, Moreau J, Pecharová K, Thiéry D, Louâpre P (2020) Reproductive performance of the European grapevine moth Lobesia botrana (Tortricidae) is adversely affected by warming scenario. J Pest Sci 93:679–689. https://doi.org/10.1007/s10340-020-01201-1
Iltis C, Louâpre P, Vogelweith F, Thiéry D, Moreau J (2021) How to stand the heat? Post-stress nutrition and developmental stage determine insect response to a heat wave. J Insect Physiol 131:104214. https://doi.org/10.1016/j.jinsphys.2021.104214
Kaunisto S, Ferguson LV, Sinclair BJ (2016) Can we predict the effects of multiple stressors on insects in a changing climate? Curr Opin Insect Sci 17:55–61. https://doi.org/10.1016/j.cois.2016.07.001
Kleynhans E, Conlong DE, Terblanche JS (2014) Host plant-related variation in thermal tolerance of Eldana saccharina. Entomol Exp Appl 150:113–122. https://doi.org/10.1111/eea.12144
Lai H-Y, Juang K-W, Chen B-C (2010) Copper concentrations in grapevines and vineyard soils in central Taiwan. Soil Sci Plant Nutr 56:601–606. https://doi.org/10.1111/j.1747-0765.2010.00494.x
Lamichhane JR, Osdaghi E, Behlau F, Köhl J, Jones JB, Aubertot JN (2018) Thirteen decades of antimicrobial copper compounds applied in agriculture. A review. Agron Sustain Dev 38:1–18. https://doi.org/10.1007/s13593-018-0503-9
Laskowski R, Bednarska AJ, Kramarz PE, Loureiro S, Scheil V, Kudłek J, Holmstrup M (2010) Interactions between toxic chemicals and natural environemental factors: a meta analysis and case studies. Sci Tot Environ 408:3763–3774. https://doi.org/10.1016/j.scitotenv.2010.01.043
Li HB, Zheng YT, Sun DD, Wang JJ, Du YZ (2014) Combined effects of temperature and avermectins on life history and stress response of the western flower thrips, Frankliniella occidentalis. Pestic Biochem Physiol 108:42–48. https://doi.org/10.1016/j.pestbp.2013.12.002
Mackie KA, Müller T, Kandeler E (2012) Remediation of copper in vineyards: a mini review. Environ Pollut 167:16–26. https://doi.org/10.1016/j.envpol.2012.03.023
Martín-Vertedor D, Ferrero-García JJ, Torres-Vila LM (2010) Global warming affects phenology and voltinism of Lobesia botrana in Spain. Agric for Entomol 12:169–176. https://doi.org/10.1111/j.1461-9563.2009.00465.x
Matzrafi M (2019) Climate change exacerbates pest damage through reduced pesticide efficacy. Pest Manag Sci 75:9–13. https://doi.org/10.1002/ps.5121
Miotto A, Ceretta CA, Brunetto G, Nicoloso FT, Girotto E, Farias JG, Tiecher TL, De Conti L, Trentin G (2014) Copper uptake, accumulation and physiological changes in adult grapevines in response to excess copper in soil. Plant Soil 374:593–610. https://doi.org/10.1007/s11104-013-1886-7
Moreau J, Villemant C, Benrey B, Thiéry D (2010) Species diversity of larval parasitoids of the European grapevine moth (Lobesia botrana, Lepidoptera: Tortricidae): the influence of region and cultivar. Biol Control 54:300–306. https://doi.org/10.1016/j.biocontrol.2010.05.019
Moschos T (2006) Yield loss quantification and economic injury level estimation for the carpophagous generations of the European grapevine moth Lobesia botrana Den. et Schiff. (Lepidoptera: Tortricidae). Int J Pest Manag 52:141–147. https://doi.org/10.1080/09670870600639179
Muller K, Thiéry D, Delbac L, Moreau J (2016) Mating patterns of the European grapevine moth, Lobesia botrana (Lepidoptera: Tortricidae) in sympatric and allopatric populations. Biol J Linn Soc 120:685–699. https://doi.org/10.1111/bij.12901
Musser FR, Shelton AM (2005) The influence of post-exposure temperature on the toxicity of insecticides to Ostrinia nubilalis (Lepidoptera: Crambidae). Pest Manag Sci 61:508–510. https://doi.org/10.1002/ps.998
Noyes PD, McElwee MK, Miller HD, Clark BW, Van Tiem LA, Walcott KC, Erwin KN, Levin ED (2009) The toxicology of climate change: environmental contaminants in a warming world. Environ Int 35:971–986. https://doi.org/10.1016/j.envint.2009.02.006
Op de Beeck L, Verheyen J, Olsen K, Stoks R (2017) Negative effects of pesticides under global warming can be counteracted by a higher degradation rate and thermal adaptation. J Appl Ecol 54:1847–1855. https://doi.org/10.1111/1365-2664.12919
Pavan F, Bigot G, Cargnus E, Zandigiacomo P (2014) Influence of the carpophagous generations of the European grapevine moth Lobesia botrana on grape bunch rots. Phytoparasitica 42:61–69. https://doi.org/10.1007/s12600-013-0338-5
Pennington T, Reiff JM, Theiss K, Entling MH, Hoffmann C (2018) Reduced fungicide applications improve insect pest control in grapevine. Biocontrol 63:687–695. https://doi.org/10.1007/s10526-018-9896-2
Pertot I, Caffi T, Rossi V, Mugnai L, Hoffmann C, Grando MS, Gary C, Lafond D, Duso C, Thiéry D, Mazzoni V, Anfora G (2017) A critical review of plant protection tools for reducing pesticide use on grapevine and new perspectives for the implementation of IPM in viticulture. Crop Prot 97:70–84. https://doi.org/10.1016/j.cropro.2016.11.025
Pölkki M, Kangassalo K, Rantala MJ (2014) Effects of interaction between temperature conditions and copper exposure on immune defense and other life-history traits of the blow fly Protophormia terraenovae. Environ Sci Technol 48:8793–8799. https://doi.org/10.1021/es501880b
Reineke A, Thiéry D (2016) Grapevine insect pests and their natural enemies in the age of global warming. J Pest Sci 89:313–328. https://doi.org/10.1007/s10340-016-0761-8
Ricupero M, Abbes K, Haddi K, Kurtulus A, Desneux N, Russo A, Siscaro G, Biondi A, Zappalà L (2020) Combined thermal and insecticidal stresses on the generalist predator Macrolophus pygmaeus. Sci Total Environ 729:138922. https://doi.org/10.1016/j.scitotenv.2020.138922
Savopoulou-Soultani Μ, Stavridis DG, Tzanakakis ΜΕ (1990) Development and reproduction of Lobesia botrana on vine and olive inflorescences. Entomol Hell 8:29–35. https://doi.org/10.12681/eh.13978
Servia MJ, Péry ARR, Heydorff M, Garric J, Lagadic L (2006) Effects of copper on energy metabolism and larval development in the midge Chironomus riparius. Ecotoxicology 15:229–240. https://doi.org/10.1007/s10646-005-0054-0
Thiéry D, Moreau J (2005) Relative performance of European grapevine moth (Lobesia botrana) on grapes and other hosts. Oecologia 143:548–557. https://doi.org/10.1007/s00442-005-0022-7
Thiéry D, Louâpre P, Muneret L, Rusch A, Sentenac G, Vogelweith F, Iltis C, Moreau J (2018) Biological protection against grape berry moths. A review. Agron Sustain Dev 38:15. https://doi.org/10.1007/s13593-018-0493-7
van Ooik T, Rantala MJ, Saloniemi I (2007) Diet-mediated effects of heavy metal pollution on growth and immune response in the geometrid moth Epirrita autumnata. Environ Pollut 145:348–354. https://doi.org/10.1016/j.envpol.2006.03.008
van Ooik T, Pausio S, Rantala MJ (2008) Direct effects of heavy metal pollution on the immune function of a geometrid moth, Epirrita autumnata. Chemosphere 71:1840–1844. https://doi.org/10.1016/j.chemosphere.2008.02.014
Vieira A, Oliveira L, Garcia P (2001) Effects of conventional pesticides on the preimaginal developmental stages and on adults of Trichogramma cordubensis (Hymenoptera: Trichogrammatidae). Biocontrol Sci Technol 11:527–534. https://doi.org/10.1080/09583150120067553
Vogelweith F, Thiéry D (2018) An assessment of the non-target effects of copper on the leaf arthropod community in a vineyard. Biol Control 127:94–100. https://doi.org/10.1016/j.biocontrol.2018.08.011
Vogelweith F, Thiéry D, Moret Y, Moreau J (2013) Immunocompetence increases with larval body size in a phytophagous moth. Physiol Entomol 38:219–225. https://doi.org/10.1111/phen.12025
Vogelweith F, Thiéry D, Moret Y, Colin E, Motreuil S, Moreau J (2014) Defense strategies used by two sympatric vineyard moth pests. J Insect Physiol 64:54–61. https://doi.org/10.1016/j.jinsphys.2014.03.009
Xie W, Wang S, Wu Q, Feng Y, Pan H, Jiao X, Zhou L, Yang X, Fu W, Teng H, Xu B, Zhang Y (2011) Induction effects of host plants on insecticide susceptibility and detoxification enzymes of Bemisia tabaci (Hemiptera: Aleyrodidae). Pest Manag Sci 67:87–93. https://doi.org/10.1002/ps.2037
Yu YL, Huang LJ, Wang LP, Wu JC (2012) The combined effects of temperature and insecticide on the fecundity of adult males and adult females of the brown planthopper Nilaparvata lugens Stål (Hemiptera: Delphacidae). Crop Prot 34:59–64. https://doi.org/10.1016/j.cropro.2011.08.026
Zhao JH, Ho P, Azadi H (2011) Benefits of Bt cotton counterbalanced by secondary pests? Perceptions of ecological change in China. Environ Monit Assess 173:985–994. https://doi.org/10.1007/s10661-010-1439-y
Acknowledgements
We are extremely grateful to Lionel Delbac for his help in maintaining the insect stock. We also want to thank Sébastien Zito and Benjamin Bois for providing climatic data. We acknowledge the valuable technical contribution of Léa Gastal et Louis Sébert during our experiments. We would like to express our gratitude to Emma Parfitt for English revision. We thank the Editor Chris Cutler and the two anonymous reviewers for their relevant suggestions on the draft.
Funding
This work was supported by the Conseil Régional de Bourgogne Franche-Comté through the Plan d’Actions Régional pour l’Innovation (PARI) and two other funding sources (FABER LOUAPRE AGREE-BGS, VALEACLIM-BOIS 20184-07116), and the European Union through the PO FEDER-FSE Bourgogne 2014/2020 programs.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no conflict of interest.
Ethical approval
All applicable institutional and/or national guidelines for the care and use of animals were followed.
Additional information
Communicated by Chris Cutler.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Iltis, C., Moreau, J., Hübner, P. et al. Warming increases tolerance of an insect pest to fungicide exposure through temperature-mediated hormesis. J Pest Sci 95, 827–839 (2022). https://doi.org/10.1007/s10340-021-01398-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-021-01398-9