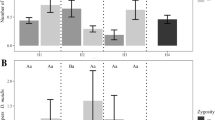
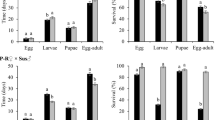
Abstract
Historically, Bt maize hybrids have been produced with the Bt alleles in a hemizygous state. Homozygous Bt transgenes increase the expression of Bt proteins in plants; however, recent studies have only briefly explored the zygosity of Bt transgenes in maize hybrids and its effects on the control of lepidopteran pests. In this study, we investigated whether an additional allelic dose of the single Bt events Bt11 and MIR162, and a pyramided version (Bt11 + MIR162 + GA21) would impact Bt protein expression in leaves and increase the control of fall armyworm, Spodoptera frugiperda, in leaves and grains of near-isogenic maize hybrids. Our results revealed that homozygous Bt hybrids had higher Cry1Ab and Vip3Aa20 protein concentrations in leaves than their hemizygous versions. However, higher concentrations of Bt proteins in homozygous versions did not increase the control of S. frugiperda. Hybrids expressing Cry1Ab, hemizygous and homozygous for the transgene, had similar mortality of S. frugiperda fed on leaves and grains as the non-Bt near-isogenic version, likely due to resistance of the pest to Cry1Ab. High susceptibility to Vip3Aa20 caused all larvae to die on hemizygous and homozygous MIR162 leaves. Also, larvae that survived on Vip3Aa20 grains did not gain weight after four days of feeding, regardless of the Bt zygosity of Vip3Aa20-expressing grains. Additional implications of Bt zygosity in maize hybrids on insect resistance management strategies are discussed.






Similar content being viewed by others
Availability of data and material
Data will be available if needed.
Code availability
R scripts will be provided if needed.
References
Amaral FSA, Guidolin AS, Salmeron E, Kanno RH, Padovez FE, Fatoretto JC, Omoto C (2020) Geographical distribution of Vip3Aa20 resistance allele frequencies in Spodoptera frugiperda (Lepidoptera: Noctuidae) populations in Brazil. Pest Manag Sci. https://doi.org/10.1002/ps.5490
Andow DA (2008) The risk of resistance evolution in insects to transgenic insecticidal crops. Colln Biosaf Rev 4:142–199
Bernardi D, Salmeron E, Horikoshi RJ, Bernardi O, Dourado PM, Carvalho RA, Martinelli S, Head GP, Omoto C (2015a) Cross-resistance between Cry1 proteins in fall armyworm (Spodoptera frugiperda) may affect the durability of current pyramided Bt maize hybrids in Brazil. PLoS ONE 10:1–15. https://doi.org/10.1371/journal.pone.0140130
Bernardi O, Bernardi D, Amado D, Sousa RS, Fatoretto J, Medeiros FC, Conville J, Burd T, Omoto C (2015b) Resistance risk assessment of Spodoptera frugiperda (Lepidoptera: Noctuidae) and Diatraea saccharalis (Lepidoptera: Crambidae) to Vip3Aa20 insecticidal protein expressed in corn. J Econ Entomol 108:2711–2719. https://doi.org/10.1093/jee/tov219
Botha AS, Erasmus A, du Plessis H, Van den Berg J (2019) Efficacy of Bt maize for control of Spodoptera frugiperda (Lepidoptera: Noctuidae) in South Africa. J Econ Entomol 112:1260–1266. https://doi.org/10.1093/jee/toz048
Burkness EC, Dively G, Patton T, Morey AC, Hutchison WD (2010) Novel Vip3A Bacillus thuringiensis (Bt) maize approaches high-dose efficacy against Helicoverpa zea (Lepidoptera: Noctuidae) under field conditions: implications for resistance management. GM Crops 1:337–343. https://doi.org/10.4161/gmcr.1.5.14765
Caligari PDS, Yapabandara YMHB, Paul EM, Perret J, Roger P, Dunwell JM (1993) Field performance of derived generations of transgenic tobacco. Theor Appl Genet 86:875–879. https://doi.org/10.1007/BF00212615
Caprio MA, Martinez JC, Porter PA, Bynum E (2015) The impact of inter-kernel movement in the evolution of resistance to dual-toxin Bt-corn varieties in Helicoverpa zea (Lepidoptera: Noctuidae). J Econ Entomol 109:307–319. https://doi.org/10.1093/jee/tov295
Carrière Y, Degain BA, Harpold VS, Unnithan GC, Tabashnik BE (2020) Gene flow between Bt and non-Bt plants in a seed mixture increases dominance of resistance to pyramided Bt corn in Helicoverpa zea (Lepidoptera: Noctuidae). J Econ Entomol 20:1–11. https://doi.org/10.1093/jee/toaa138
Carrière Y, Fabrick JA, Tabashnik BE (2016) Can pyramids and seed mixtures delay resistance to Bt crops? Trends Biotechnol 34:291–302. https://doi.org/10.1016/j.tibtech.2015.12.011
Chilcutt CF, Tabashnik BE (2004) Contamination of refuges by Bacillus thuringiensis toxin genes from transgenic maize. PNAS 101:7526–7529. https://doi.org/10.1073/pnas.0400546101
Clough RC, Pappu K, Thompson K, Beifuss K, Lane J, Delaney DE, Harkey R, Drees C, Howard JA, Hood EE (2006) Manganese peroxidase from the white-rot fungus Phanerochaete chrysosporium is enzymatically active and accumulates to high levels in transgenic maize seed. Plant Biotechnol J 4:53–62. https://doi.org/10.1111/j.1467-7652.2005.00157.x
Comissão Técnica Nacional de Biossegurança, CTNBio (2007) Parecer Técnico Nº 1100/2007. http://ctnbio.mcti.gov.br/liberacao-comercial. Accessed 17 July 2019
Comissão Técnica Nacional de Biossegurança, CTNBio (2008) Parecer Técnico Nº 1255/2008. http://ctnbio.mcti.gov.br/liberacao-comercial. Accessed 16 July 2019
Comissão Técnica Nacional de Biossegurança, CTNBio (2009) Extrato de Parecer Técnico N° 2042/2009. http://ctnbio.mcti.gov.br/liberacao-comercial. Accessed 16 July 2019
Davis FM, Ng SS, Williams WP (1992) Visual rating scales for screening whorl-stage corn resistance to fall armyworm. Mississippi State University, Mississippi State
Eghrari K, Brito AH, Baldassi A, Balbuena TS, Fernandes OA, Môro GV (2019) Homozygosis of Bt locus increases Bt protein expression and the control of Spodoptera frugiperda (Lepidoptera: Noctuidae) in maize hybrids. J Crop Prot. https://doi.org/10.1016/j.cropro.2019.104871
Farias JR, Andow DA, Horikoshi RJ, Sorgatto RJ, Fresia P, dos Santos AC, Omoto C (2014) Field-evolved resistance to Cry1F maize by Spodoptera frugiperda (Lepidoptera: Noctuidae) in Brazil. J Crop Prot 64:150–158. https://doi.org/10.1016/j.cropro.2014.06.019
Farias JR, Andow DA, Horikoshi RJ, Bernardi D, Ribeiro RDS, Nascimento ARD, Santos AC, Omoto C (2016) Frequency of Cry1F resistance alleles in Spodoptera frugiperda (Lepidoptera: Noctuidae) in Brazil. Pest Manag Sci 72:2295–2302. https://doi.org/10.1002/ps.4274
Food and Agriculture Organization of the United Nations, FAO (2018) FAOSTAT database. http://www.fao.org/faostat/en/#data/QC. Accessed 20 June 2020.
Gutha LR, Purushottam D, Veeramachaneni A, Tigulla S, Kodappully V, Enjala C, Rajput H, Anderson J, Hong B, Schmidt J, Bagga S (2018) Expression of endogenous proteins in maize hybrids in a multi-location field trial in India. Transgenic Res 27:331–342. https://doi.org/10.1007/s11248-018-0077-y
Hood EE, Devaiah SP, Fake G, Egelkrout E, Teoh KT, Requesens DV, Hayden C, Hood KR, Pappu KM, Carrol J, Howard JA (2012) Manipulating corn germplasm to increase recombinant protein accumulation. Plant Biotechnol J 10:20–30. https://doi.org/10.1111/j.1467-7652.2011.00627.x
Howard JA, Hood EE (2014) Commercial plant-produced recombinant protein products: case studies. Springer, Berlin
Huang F (2021) Resistance of the fall armyworm, Spodoptera frugiperda (J. E. Smith), to transgenic Bacillus thuringiensis Cry1F corn in the Americas: lessons and implications for Bt corn IRM in China. Ins Sci. https://doi.org/10.1111/1744-7917.12826
Huang F, Andow DA, Buschman LL (2011) Success of the high-dose/refuge resistance management strategy after 15 years of Bt crop use in North America. Entomol Exp Appl 140:1–16. https://doi.org/10.1111/j.1570-7458.2011.01138.x
Huang F, Qureshi JA, Meagher RL et al (2014) Cry1F resistance in fall armyworm Spodoptera frugiperda: Single gene versus pyramided Bt maize. PLoS ONE. https://doi.org/10.1371/journal.pone.0112958
Hutchison WD, Burkness EC, Mitchell PD, Moon RD, Leslie TW, Fleischer SJ, Abrahamson M, Hamilton KL, Steffey KL, Gray ME, Hellmich RL (2010) Areawide suppression of European corn borer with Bt maize reaps savings to non-Bt maize growers. Science 330:222–225. https://doi.org/10.1126/science.1190242
International Service for the Acquisition of Agri-Biotech Applications, ISAAA (2019) GM Approval Database. http://www.isaaa.org/gmapprovaldatabase. Accessed 22 Aug 2019.
Ives AR, Glaum PR, Ziebarth NL, Andow DA (2011) The evolution of resistance to two-toxin pyramid transgenic crops. Ecol Appl 21:503–515. https://doi.org/10.1890/09-1869.1
James VA, Avart C, Worland B, Snape JW, Vain P (2002) The relationship between homozygous and hemizygous transgene expression levels over generations in populations of transgenic rice plants. Theor Appl Genet 104:553–561. https://doi.org/10.1007/s001220100745
Kasten P Jr, Precetti AACM, Parra JRP (1978) Dados biológicos comparativos de Spodoptera frugiperda (J.E. Smith, 1797) em duas dietas artificiais e substrato natural. Rev Agric 53:69–78
Law RD, Russel DA, Thompson LC, Schroeder SC, Middle CM, Tremaine MT, Jury TP, Delannay X, Slater S (2006) Biochemical limitations to high-level expression of humanized monoclonal antibodies in transgenic maize seed endosperm. Biochim Biophys Acta 1760:1434–1444. https://doi.org/10.1016/j.bbagen.2006.05.006
Li XJ, Wu MF, Ma J et al (2020) Prediction of migratory routes of the invasive fall armyworm in eastern China using a trajectory analytical approach. Pest Manag Sci 76:454–463. https://doi.org/10.1002/ps.5530
Lohn AF, Trtikova M, Chapela I, Van den Berg J, Plessis H, Hilbeck A (2020) Transgene behavior in Zea mays L. crosses across different genetic backgrounds: Segregation patterns, cry1Ab transgene expression, insecticidal protein concentration and bioactivity against insect pests. PloS One. https://doi.org/https://doi.org/10.1371/journal.pone.0238523
Long N, Bottoms J, Meghji M, Hart H, Que Q, Pulliam D (2012) Corn event MIR162. U.S. Patent 8,232,456, issued July 31, 2012. https://patentimages.storage.googleapis.com/93/97/af/668ff4ca3fdfab/US8232456.pdf Accessed 17 July 2019
Magalhães PC, Durães FO (2006) Fisiologia da produção de milho. Embrapa Milho e Sorgo-Circular Técnica (INFOTECA-E). https://www.embrapa.br/busca-de-publicacoes/-/publicacao/490408/fisiologia-da-producao-de-milho. Accessed 25 April 2020
Morris M, Dreher K, Ribaut JM, Khairallah M (2003) Money matters (II): costs of maize inbred line conversion schemes at CIMMYT using conventional and marker-assisted selection. Mol Breed 11:235–247. https://doi.org/10.1023/A:1022872604743
Nguyen HT, Jehle JA (2007) Quantitative analysis of the seasonal and tissue-specific expression of Cry1Ab in transgenic maize Mon810. J Plant Dis Protect 114:82–87. https://doi.org/10.1007/BF03356208
Omoto C, Bernardi O, Salmeron E, Sorgatto RJ, Dourado PM, Crivellari A, Carvalho RA, Willse A, Martinelli S, Head GP (2016) Field-evolved resistance to Cry1Ab maize by Spodoptera frugiperda in Brazil. Pest Manag Sci 72:1727–1736. https://doi.org/10.1002/ps.4201
R Core Team (2019) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org
Santos-Amaya OF, Rodrigues JV, Souza TC, Tavares CS, Campos SO, Guedes RN, Pereira EJ (2015) Resistance to dual-gene Bt maize in Spodoptera frugiperda: selection, inheritance, and cross-resistance to other transgenic events. Sci Rep. https://doi.org/10.1038/srep18243
Székács A, Lauber É, Juracsek J, Darvas B (2010) Cry1Ab toxin production of MON 810 transgenic maize. Environ Toxicol Chem 29:182–190. https://doi.org/10.1002/etc.5
Silva V, Mol HG, Zomer P, Tienstr M, Ritsema CJ, Geissen V (2019) Pesticide residues in European agricultural soils–A hidden reality unfolded. Sci Total Environ 653:1532–1545. https://doi.org/10.1016/j.scitotenv.2018.10.441
Silver A (2019) Caterpillar’s devastating march across China spurs hunt for native predator. Nature 570:286. https://doi.org/10.1038/d41586-019-01867-3
Storer NP, Babcock JM, Schlenz M, Meade T, Thompson GD, Bing JW, Huckaba RM (2010) Discovery and characterization of field resistance to Bt maize: Spodoptera frugiperda (Lepidoptera: Noctuidae) in Puerto Rico. J Econ Entomol 103:1031–1038. https://doi.org/10.1603/EC10040
Storer NP, Kubiszak ME, King JE, Thompson GD, Santos AC (2012) Status of resistance to Bt maize in Spodoptera frugiperda: lessons from Puerto Rico. J Invertebr Pathol 110:294–300. https://doi.org/10.1016/j.jip.2012.04.007
Tabashnik BE, Brévault T, Carrière Y (2013) Insect resistance to Bt crops: lessons from the first billion acres. Nat Biotechnoly 31:510. https://doi.org/10.1038/nbt.2597
Tabashnik BE, Carrière Y (2017) Surge in insect resistance to transgenic crops and prospects for sustainability. Nat Biotechnoly 35:926–935. https://doi.org/10.1038/nbt.3974
Tabashnik BE, Van Rensburg JBJ, Carrière Y (2009) Field-evolved insect resistance to Bt crops: definition, theory, and data. J Econ Entomol 102:2011–2025. https://doi.org/10.1603/029.102.0601
Tavares WS, Costa MA, Cruz I, Silveira RD, Serrao JE, Zanuncio JC (2010) Selective effects of natural and synthetic insecticides on mortality of Spodoptera frugiperda (Lepidoptera: Noctuidae) and its predator Eriopis connexa (Coleoptera: Coccinellidae). J Environ Sci Heal B 45:557–561. https://doi.org/10.1080/03601234.2010.493493
Trtikova M, Wikmark OG, Zemp N et al (2015) Transgene expression and bt protein content in transgenic bt maize (MON810) under optimal and stressful environmental conditions. PLoS ONE 10:1–9. https://doi.org/10.1371/journal.pone.0123011
US Environmental Protection Agency, US EPA (1998) Final report of the subpanel on Bacillus thuringiensis (Bt) plant-pesticides and resistance management. https://archive.epa.gov/scipoly/sap/meetings/web/pdf/finalfeb.pdf. Accessed 31 Oct 2020
Yang F, Kerns DL, Brown S, Head GP, Huang F (2017) Pollen contamination in seed mixture increases the dominance of resistance to Bt maize in Spodoptera frugiperda (Lepidoptera: Noctuidae). Pest Manag Sci 73:2379–2385. https://doi.org/10.1002/ps.4631
Yang F, Kerns DL, Head GP, Leonard BR, Levy R, Niu Y, Huang F (2014) A challenge for the seed mixture refuge strategy in Bt maize: impact of cross-pollination on an ear-feeding pest, corn earworm. PLoS ONE. https://doi.org/10.1371/journal.pone.0112962
Yang F, Kerns DL, Head GP, Price PA, Levy R, Niu Y, Huang F (2020) Extended evaluation of Bt protein cross-pollination in seed blend plantings on survival, growth, and development of Helicoverpa zea feeding on refuge ears. Pest Manag Sci 76:1011–1019. https://doi.org/10.1002/ps.5611
Young JR (1979) Fall armyworm: control with insecticides. Fla Entomol 62:130–133
Yu SJ (1991) Insecticide resistance in the fall armyworm, Spodoptera frugiperda (JE Smith). Pestic Biochem Phys 39:84–91
Acknowledgements
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001, and the National Council for Scientific and Technological Development—Brasil (CNPq). We thank Syngenta Seeds Brazil and Gustavo Biudes for generating and providing the seeds, and Syngenta Corporation US for Bt quantification; Betiana Parody and Fabrício Francischini regarding the conduction of our partnership; Larah Martins for helping with conducting the experiments in Lavras; Naiara Zancanari, Flávia Alves, Luiz Bertasello for helping in the experiments in Jaboticabal; and, finally, Betiana Parody, Renato Carvalho, Brian Rhodes, Eddie Ross, Nadine Guyot, Douglas Sumerford, Larry Zeph, and the anonymous reviewers of this journal for the valuable contributions to this article.
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001, and the National Council for Scientific and Technological Development—Brasil (CNPq).This study was also financed in part by Syngenta Crop Protection.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by G. Smagghe.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Eghrari, K., Oliveira, S.C., Nascimento, A.M. et al. The implications of homozygous vip3Aa20- and cry1Ab-maize on Spodoptera frugiperda control. J Pest Sci 95, 115–127 (2022). https://doi.org/10.1007/s10340-021-01362-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-021-01362-7