Abstract
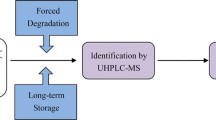
Fipronil belongs to the phenylpyrazole family of insecticides and functions by interrupting the γ-aminobutyric acid system and chloride ion channels of pests. Studies have shown Fipronil can undergo several degradation pathways in the environment to form degradants that may be toxic to non-target aquatic life. In this study, a comprehensive forced degradation study of Fipronil was carried out following ICH guidelines to generate, characterize, and identify the major degradation products of Fipronil. Stress conditions included acidic, alkaline, oxidative, thermal (solid and solution states), and photolytic (solid and solution states). Separation of six major degradation products (DPs) was achieved using reversed phase high-performance liquid chromatography (HPLC) with gradient elution. The DPs were characterized and identified using HPLC-high-resolution mass spectrometry (HPLC-HRMS) and comparison of tandem MS/MS fragmentation profiles. The Fipronil dimer required isolation through semi-preparative HPLC and analysis by nuclear magnetic resonance (NMR) to elucidate and confirm its structure. The proposed structures of DPs were based upon the given degradation pathways of Fipronil. The key results and findings of this study will provide better understanding of Fipronil stability and its major degradation products including degradation/formation pathways, which would assist designing of more stable Fipronil formulations and finished products with extended shelf-life including enhanced efficacy with decreased adverse effects.






Similar content being viewed by others
Data availability
The experimental data are available upon request.
References
Ibrahim SA et al (2003) Toxicity, repellency, and horizontal transmission of fipronil in the formosan subterranean termite (Isoptera: Rhinotermitidae). J Econ Entomol 96:461–467. https://doi.org/10.1093/jee/96.2.461
Bonmatin J-M et al (2015) Environmental fate and exposure; neonicotinoids and fipronil. Environ Sci Pollut R 22:35–67. https://doi.org/10.1007/s11356-014-3332-7
Dryden MW et al (2000) Control of fleas on naturally infested dogs and cats and in private residences with topical spot applications of fipronil or imidacloprid. Vet Parasitol 93:69–75. https://doi.org/10.1016/s0304-4017(00)00318-6
Eiden AL et al (2015) Detection of permethrin resistance and fipronil tolerance in Rhipicephalus sanguineus (acari: ixodidae) in the United States. J Med Entomol 52:429–436. https://doi.org/10.1093/jme/tjv005
Hosie AM et al (1995) Actions of the insecticide fipronil, on dieldrin-sensitive and -resistant GABA receptors of Drosophila melanogaster. Br J Pharmacol 115:909–912. https://doi.org/10.1111/j.1476-5381.1995.tb15896.x
Rosa de Oliveira P et al (2009) Action of the chemical agent fipronil on the reproductive process of semi-engorged females of the tick Rhipicephalus sanguineus (latreille, 1806) (acari: ixodidae). Ultrastructural evaluation of ovary cells. Food Chem Toxicol 47:1255–1264. https://doi.org/10.1016/j.fct.2009.02.019
Singh NS et al (2021) A comprehensive review of environmental fate and degradation of fipronil and its toxic metabolites. Environ Res 199:111316. https://doi.org/10.1016/j.envres.2021.111316
Lee S-J et al (2010) Acute illnesses associated with exposure to fipronil—surveillance data from 11 states in the United States, 2001–2007. Clin Toxicol 48:737–744. https://doi.org/10.3109/15563650.2010.507548
Nillos MG et al (2009) Enantioselectivity in fipronil aquatic toxicity and degradation. Environ Toxicol Chem 28:1825–1833. https://doi.org/10.1897/08-658.1
Jun W et al (2015) Occurrence and ecological risks from fipronil in aquatic environments located within residential landscapes. Sci Total Environ 518–519:139–147. https://doi.org/10.1016/j.scitotenv.2014.12.103
Pei Z et al (2004) Dynamics of fipronil residue in vegetable-field ecosystem. Chemosphere 57:1691–1696. https://doi.org/10.1016/j.chemosphere.2004.06.025
Jimenez JJ et al (2008) Sample preparation methods to analyze fipronil in honey by gas chromatography with electron-capture and mass spectrometric detection. J Chromatogr A 1187:40–45. https://doi.org/10.1016/j.chroma.2008.02.014
Hingmire S et al (2015) Residue analysis of fipronil and difenoconazole in okra by liquid chromatography tandem mass spectrometry and their food safety evaluation. Food Chem 176:145–151. https://doi.org/10.1016/j.foodchem.2014.12.049
Li X et al (2020) Determination of residual fipronil and its metabolites in food samples: a review. Trends Food Sci Technol 97:185–195. https://doi.org/10.1016/j.tifs.2020.01.018
Bobe A et al (1998) Kinetics and mechanisms of abiotic degradation of fipronil (hydrolysis and photolysis). J Agric Food Chem 46:2834–2839. https://doi.org/10.1021/jf970874d
Fipronil for veterinary use. Ph Eur 11(3):2869 (2018)
Hainzl D, Casida JE (1996) Fipronil insecticide: novel photochemical desulfinylation with retention of neurotoxicity. Proc Natl Acad Sci 93:12764–12767. https://doi.org/10.1073/pnas.93.23.12764
Fenoll J et al (2014) Fipronil decomposition in aqueous semiconductor suspensions using UV light and solar energy. J Taiwan Inst Chem E 45:981–988. https://doi.org/10.1016/j.jtice.2013.09.015
Qian C et al (2020) Efficient degradation of fipronil in water by microwave-induced argon plasma: mechanism and degradation pathways. Sci Total Environ 725:138487. https://doi.org/10.1016/j.scitotenv.2020.138487
Validation of analytical procedures Q2(R2). International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use (2022)
Taylor MJ et al (2008) The utility of ultra-performance liquid chromatography/electrospray ionisation time-of-flight mass spectrometry for multi-residue determination of pesticides in strawberry. Rapid Commun Mass Spectrom 22:2731–2746. https://doi.org/10.1002/rcm.3671
Lacroix MZ et al (2010) Quantification of fipronil and its metabolite fipronil sulfone in rat plasma over a wide range of concentrations by LC/UV/MS. J Chromatogr B 878:1934–1938. https://doi.org/10.1016/j.jchromb.2010.05.018
Garcia-Chao M et al (2010) Validation of an off line solid phase extraction liquid chromatography–tandem mass spectrometry method for the determination of systemic insecticide residues in honey and pollen samples collected in apiaries from NW Spain. Anal Chim Acta 672:107–113. https://doi.org/10.1016/j.aca.2010.03.011
Kumar R et al (2013) Bioefficacy and fate of fipronil and its metabolites in basmati rice under sub-tropical climatic conditions. Crop Prot 45:41–48. https://doi.org/10.1016/j.cropro.2012.11.017
Litmanovich AD, Plate NA (2000) Alkaline hydrolysis of polyacrylonitrile. On the reaction mechanism. Macromol Chem Phys 201:2176–2180. https://doi.org/10.1002/1521-3935(20001101)201:16%3c2176::AID-MACP2176%3e3.0.CO;2-5
Krieble VK, Noll CI (1939) The hydrolysis of nitriles with acids. J Am Chem Soc 61:560–563. https://doi.org/10.1021/ja01872a005
Liu D et al (2008) Enantioselective degradation of fipronil in Chinese cabbage (Brassica pekinensis). Food Chem 110:399–405. https://doi.org/10.1016/j.foodchem.2008.02.017
De Toffoli AL et al (2015) Development, validation, and application of a method for the GC-MS analysis of fipronil and three of its degradation products in samples of water, soil, and sediment. J Environ Sci Health Part B 50:753–759. https://doi.org/10.1080/03601234.2015.1058091
Acknowledgements
The authors would like to thank all the analytical scientists in the Global Pharmaceutical Technical Support (GPTS) group at BIAH North Brunswick Research Center and to the dedicated analytical team (of GPTS) in Pharmaron laboratory for their support and execution of various studies described in this paper.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
A.M.R. conceptualization, experimental design, manuscript review/editing. S.N.B. data collection, data analysis, manuscript preparation, manuscript editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent for publication
The authors’ affiliated company, Boehringer Ingelheim Animal Health (BIAH) USA Inc. approved the submission of this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This document is provided for scientific purposes only. Any reference to a brand or trademark herein is for informational purposes only and is not intended for a commercial purpose or to dilute the rights of the respective owners(s) of the brand(s) or trademark(s).
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Berger, S.N., Rustum, A.M. Comprehensive Stress Degradation Studies of Fipronil: Identification and Characterization of Major Degradation Products of Fipronil Including Their Degradation Pathways Using High Resolution-LCMS. Chromatographia 87, 249–257 (2024). https://doi.org/10.1007/s10337-024-04324-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10337-024-04324-0