Abstract
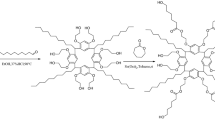
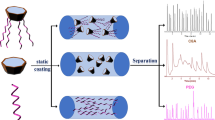
A bromine-terminated triblock copolymer (Br-PCL-PDMS-PCL-Br) was synthesized and employed as the stationary phase for capillary gas chromatography (GC). The statically coated Br-PCL-PDMS-PCL-Br column showed the efficiency of 2260 plates/m determined by 1-octanol at 120 °C and moderate polarity. For evaluating the separation performance, a mixture of 23 analytes was employed and the commercial HP-35 and PEG-20 M columns were used as the reference columns. Also, the positional and cis-/trans-isomers were utilized to investigate the separation performance on the Br-PCL-PDMS-PCL-Br column. Moreover, it showed higher resolution of chloroaniline and bromoaniline isomers on the Br-PCL-PDMS-PCL-Br column than commercial HP-35 and PEG-20 M columns, and exhibited different retention behavior with HP-35 column. All the results indicated that there were multiple molecular recognition interactions between the Br-PCL-PDMS-PCL-Br stationary phase and the analytes, including H-bonding, dipole–dipole, CH···π, dispersion and halogen-bonding interactions. This work provided a research basis for exploring the application of block copolymer materials in the field of chromatographic analysis.








Similar content being viewed by others
References
Marno AR, Thurbide KB (2022) Selective separation of polar unsaturated organics using a water stationary phase in gas chromatography. Chromatographia 85:105–113
He YR, Qi ML (2020) Separation performance of a new triptycene-based stationary phase with polyethylene glycol units and its application to analysis of the essential oil of Osmanthus fragrans Lour. J Chromatogr A 1618:460928
Mametov R, Ratiu IA, Monedeiro F, Ligor T, Buszewski B (2019) Evolution and evaluation of GC columns. Crit Rev Anal Chem 51:150–173
Liu JC, Xu L, Bai JC, Du AQ, Wu B (2019) Nitro- and fluoro-substituted tetraphenyl–phenyl grafted polysiloxanes as stationary phase for capillary gas chromatography. New J Chem 43:8290–8298
Han X, Liu JC, Wang B, Du AQ, Xu L, Wu B (2018) Synthesis and chromatographic applications of polysiloxane-based stationary phase containing nitrogen heterocyclic system. J Chromatogr A 1578:76–82
Xu L, Bai JC, Du AQ, Yang ZX, Wu B (2020) 1,4-Diphenyltriphenylene grafted polysiloxane as a stationary phase for gas chromatography. New J Chem 44:695–703
Hodjmohammadi MR, Ebrahimi P, Pourmorad F (2004) Quantitative Structure-Retention Relationships (QSRR) of Some CNS Agents Studied on DB-5 and DB-17 Phases in Gas Chromatography. QSAR Comb Sci 2004:23
Shi TT, Qi ML, Huang XB (2020) High-resolution performance of triptycene functionalized with polycaprolactones for gas chromatography. J Chromatogr A 1614:460714
Curat A, Tisse S, Andrieu A, Bar N, Villemin D, Cardinael P (2014) Physical ionic liquid/polysiloxane mixtures for tuning the polarity and the selectivity of the polysiloxane stationary phase for GC analysis. Chromatographia 77:1671–1681
Leffler VB, Mayr L, Paciok P, Du H, Dunin-Borkowski RE, Dulle M, Förster S (2019) Controlled assembly of block copolymer coated nanoparticles in 2D arrays. Angew Chem Int Ed 58:8541–8545
Dash TK, Konkimalla VB (2012) Polymeric modification and its implication in drug delivery: Poly-ε-caprolactone (PCL) as a model polymer. Mol Pharmaceutics 9:2365–2379
Rosen T, Goldberg I, Navarra W, Venditto V, Kol M (2018) Block-Stereoblock copolymers of Poly(ϵ-Caprolactone) and Poly(Lactic Acid). Angew Chem Int Ed 57:7191–7195
Sun T, Li B, Li Y, Zhao XY, Song QQ, Jiang XX, Shuai XM, Li YY, Cai ZQ, Hu SQ (2019) Amphiphilic star-shaped Calix[4]resorcinarene as stationary phase for capillary gas chromatography. Chromatographia 82:1697–1708
Salom C, Nava D, Prolongo MG, Masegosa RM (2006) Poly(e-caprolactone) + unsaturated isophthalic polyester blends: thermal properties and morphology. Eur Polym J 42:1798–1810
Sun T, Shuai XM, Ren KX, Jiang XX, Chen YJ, Zhao XY, Song QQ, Hu SQ, Cai ZQ (2019) Amphiphilic block copolymer PCL-PEG-PCL as stationary phase for capillary gas chromatographic separations. Molecules 24:3158
Sun T, Huang QC, Chen RN, Zhang W, Li QL, Wu AP, Wang GX, Hu SQ, Cai ZQ (2021) The selectivity of a polydimethylsiloxane-based triblock copolymer as the stationary phase for capillary gas chromatography. New J Chem 45:20459–20467
Mayer-Helm BX, Rauter W (2005) Determination of the minimum allowable operating temperature of stationary phases in capillary columns by inverse gas chromatography. Analyst 130:502–507
Che XY, Gong SM, Zhang H, Liu B, Wang YH (2016) The effect of junction modes between backbones and side chains of polyimides on the stability of liquid crystal vertical alignment. Phys Chem Chem Phys 18:3884–3892
Han YM, Zhang JY, Shi L, Qi SC, Cheng J, Jin RG (2008) Improvement of thermal resistance of polydimethylsiloxanes with polymethylmethoxysiloxane as crosslinker. Polym Degrad Stab 93:242–251
Riley KE, Murray JS, Fanfrlík J, Řezáč J, Solá RJ, Concha MC, Ramos FM, Politzer P (2011) Halogen bond tunability I: the effects of aromatic fluorine substitution on the strengths of halogen-bonding interactions involving chlorine, bromine, and iodine. J Mol Model 17:3309–3318
Esrafili MD (2013) A theoretical investigation of the characteristics of hydrogen/halogen bonding interactions in dibromo-nitroaniline. J Mol Model 19:1417–1427
James EH (2020) Inorganic chemistry third edition: condensed phases. Bloomington-normal, Illinois State
Zondlo NJ (2012) Aromatic-proline interactions: electronically tunable ch/π interactions. Acc Chem Res 46:1039–1049
Schneider HJ (2015) Dispersive interactions in solution complexes. Acc Chem Res 48:1815–1822
Peluso P, Mamane V, Cossu S (2015) Liquid chromatography enantioseparations of halogenated compounds on polysaccharide-based chiral stationary phases: role of halogen substituents in molecular recognition. Chirality 27:667–684
Zhou HB, Chen Y, Plummer CM, Huang HH, Chen YM (2017) Facile and efficient bromination of hydroxyl-containing polymers to synthesize well-defined brominated polymers. Polym Chem 8:2189–2196
Mohammadkhani L, Heravi MM (2019) XtalFluor-E: a useful and versatile reagent in organic transformations. J Fluorine Chem 225:11–20
Huang AJ, Nan N, Chen M, Pu X, Tang H, Sun YL (1988) A modified sodium chloride deposition method for surface roughening of glass capillary columns. Acta Sci Nat Univ Pek 24:425–429
Ragonese C, Sciarrone D, Tranchida PQ, Dugo P, Mondello L (2012) Use of ionic liquids as stationary phases in hyphenated gas chromatography techniques. J Chromatogr A 1255:130–144
Sun T, Huang QC, Zhang W, Chen RN, Li W, Chen HP, Hu SQ, Cai ZQ (2022) Performance and selectivity of amphiphilic pillar[5]arene as stationary phase for capillary gas chromatography. J Chromatogr A 1671:463008
Berthod A, Zhou EY, Le K, Armstrong DW (1995) Determination and use of Rohrschneider-McReynolds constants for chiral stationary phases used in capillary gas chromatography. Anal Chem 67:849–857
Sun XJ, Zhu YL, Wang P, Li J, Wu CY, Xing J (2011) High temperature and highly selective stationary phases of ionic liquid bonded polysiloxanes for gas chromatography. J Chromatogr A 1218:1833–1841
Lamartine R, Perrin R, Thozet A, Perrin M (1983) Catalytic hydrogenation of organic solids-basic problems. Mol Cryst Liq Cryst 96:57–69
Hiyoshi N, Mine E, Rode CV, Sato O, Ebina T, Shirai M (2006) Control of stereoselectivity in 4-tert-butylphenol hydrogenation over a carbon-supported rhodium catalyst by carbon dioxide solvent. Chem Lett 35:1060–1061
Nie Y, Jaenicke S, van Bekkum H, Chuah G-K (2007) Stereoselective cascade hydrogenation of 4-tert-butylphenol and p-cresol over Zr-zeolite beta-supported rhodium. J Catal 246:223–231
Qi YG, Toyooka T, Nie JS, Ohta H, Koda S, Wang RS (2020) Comparative γ-H2AX analysis for assessment of the genotoxicity of six aromatic amines implicated in bladder cancer in human urothelial cell line. Toxicol In Vitro 66:104880
Duan JD, Jeffrey AM, Williams GM (2008) Assessment of the medicines lidocaine, prilocaine, and their metabolites, 2,6-dimethylaniline and 2-methylaniline, for DNA adduct formation in rat tissues. Drug Metab Dispos 36:1470
Xiong X, Qi ML (2020) Adenine-functionalized polypropylene glycol: A novel stationary phase for gas chromatography offering good inertness for acids and bases combined with a unique selectivity. J Chromatogr A 1612:460627
Funding
The work was supported by the Scientific Research Foundation of the Education Department of Liaoning Province (LJGD2020015), and the Training Project for Youth Backbone Teachers in Colleges and Universities of Luoyang Normal University.
Author information
Authors and Affiliations
Contributions
Conceptualization: ZC and TS; methodology: RC, QH, ZC, WZ, WL, KJ, YZ, and YL; formal analysis and investigation: RC, QH, ZC, TS, WZ, WL, KJ, YZ, and YL; writing—original draft preparation: RC; writing—review and editing: RC, ZC, and TS; funding acquisition: ZC and TS; resources: ZC and TS; supervision: ZC and TS.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no relevant financial or nonfinancial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, R., Cai, Z., Huang, Q. et al. A Bromine-Terminated Triblock Copolymer (Br-PCL-PDMS-PCL-Br) as the Stationary Phase for Gas Chromatography Analysis. Chromatographia 85, 883–894 (2022). https://doi.org/10.1007/s10337-022-04202-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10337-022-04202-7