Abstract
Two new neoavian landbirds are reported from the early Miocene St Bathans Fauna from New Zealand. Aegotheles zealandivetus sp. nov. is described from several bones, among which, notably, the tarsometatarsus shows more similarity to New Guinean taxa than to Australian—New Zealand species. Zealandornis relictus gen. et sp. nov. is described from a distal end of a humerus and placed in the new family Zealandornithidae, tentatively among the ‘higher landbirds’ Telluraves, with most similarity to coliiforms. The humerus is of similar size to that of species of Colius and its gracile shaft and very shallow sulcus scapulotricipitalis suggests reduced flying ability. The new species of Aegotheles reinforces the Australasian nature of the Zealandian fauna, while in contrast, Zealandornis relictus gen. et sp. nov. appears to have no close relatives. It is as distinct as Acanthisittidae and Strigopidae among birds, or Leiopelmatidae and Sphenodontidae among the herpetofauna, and like them, represents a similar relictual taxon. Together they confer a highly evolutionarily distinctive nature to the Zealandian fauna concomitant with a minimal 60 million years of isolation.
Zusammenfassung
Zwei neue Landvögel mit verschiedenen paläobiogeografischen Implikationen aus der frühmiozänen St. Bathans-Fauna Neuseelands
Hier beschreiben wir zwei neue Landvögel aus der frühmiozänen St. Bathans-Fauna (Otago, Neuseeland). Aegotheles zealandivetus sp. nov. wird anhand mehrerer Knochen beschrieben, unter denen besonders der Tarsus eine stärkere Ähnlichkeit mit Taxa aus Neuguinea aufweist als mit australisch-neuseeländischen Arten. Zealandornis relictus gen. et sp. nov. wird nach dem distalen Ende eines Humerus beschrieben und in die neue Familie Zealandornithidae eingeordnet, vorläufig bei den „höhreren Landvögeln" Telluraves, unter denen sie den Mausvögeln am nächsten stehen. Der Humerus ist von ähnlicher Größe wie die der Colius-Arten; sein graziler Schaft sowie das Fehlen eines Sulcus scapulotricipitalis deuten auf verringerte Flugfähigkeit hin. Die neue Aegotheles-Art betont den australasiatischen Charakter der neuseeländischen Fauna, während Zealandornis relictus gen. et sp. nov. dagegen keine nahen Verwandten zu besitzen scheint. Diese Art ist so eigenständig unter den Vögeln wie die Acanthisittidae oder Strigopidae beziehungsweise die Leiopelmatidae und Sphenodontidae in der Herpetofauna und stellt ein diesen vergleichbares Relikttaxon dar. Gemeinsam verleihen sie der neuseeländischen Fauna einen evolutionär deutlich ausgeprägten eigenen Charakter, passend zu einer durch mindestens 60 Millionen Jahre währenden Isolation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
New Zealand (NZ) is well known for its historical and late Quaternary avifauna in which birds dominate among vertebrates in non-marine ecosystems (Buller 1869, 1888; Oliver 1955; Fleming 1979; Fuller 1987; Worthy and Holdaway 2002; Gibbs 2006; Norman 2018; Thomsen 2021). The evolutionary origin of this avifauna is evidenced most significantly in the early Miocene (19–16 Ma) St Bathans Fauna, deposited in lacustrine environments and exposed in sites near St Bathans in Otago, South Island (Worthy et al. 2007). This local fauna provides essentially the only window into the fossil history of terrestrial or non-marine vertebrates between the Cretaceous and the late Quaternary in New Zealand and has been the subject of extensive investigations since 2001, as reviewed by Worthy et al. (2017). Therein, more than 40 species were listed for the St Bathans Fauna, about half of which had been taxonomically described. Since then, another duck, a parrot and two rails have been described (Mather et al. 2019; Worthy et al. 2019, 2022). Notable among the remaining taxa is an owlet-nightjar (Aegothelidae: Aegotheles) and a bird identified provisionally as representing a new family. Worthy et al. (2007) referred a proximal tarsometatarsus to Aegotheles, and since then, four other fossils attributable to this genus have been identified from among more than 7900 catalogued St Bathans specimens. The single specimen, then putatively regarded to represent a new family, remained unnamed. Here we taxonomically describe these specimens to mark their place in the evolutionary history of the New Zealand avifauna. Details of the stratigraphy and sites the specimens derive from is given in Schwarzhans et al. (2012) and Worthy et al. (2022).
The Aegothelidae comprise twelve species in the monotypic genus Aegotheles (Dumbacher et al. 2003; Dickinson and Remsen 2013) and are considered to be the sister group to Trochilidae+Apodidae (e.g., Mayr 2002; Sangster 2005; Dickinson and Remsen 2013; Claramunt and Cracraft 2015; Prum et al. 2015; Kuhl et al. 2021), either in Trochiloidea within Caprimulgiformes (Dickinson and Remsen 2013) or Apodiformes (Gill et al. 2010; Worthy and Nguyen 2020), or have been distinguished at the ordinal level, see Mayr (2010). The species of Aegotheles are endemic to the Australasian region, with A. novaezealandiae Scarlett, 1968 (extinct, NZ), A. savesi (New Caledonia), A. cristatus (Australia, New Guinea), A. crinifrons (Moluccas) and the remaining species all in New Guinea (Dickinson and Remsen 2013). Dumbacher et al. (2003) used mitochondrial genes to explore the intrageneric relationships of all the currently accepted species, finding that A. savesi and A. novaezealandiae were the sister group to the remaining species and that A. cristatus was deeply nested within the New Guinea taxa. The various species group into two size classes: one of larger bodied species (A. crinifrons, A. insignis, A. tatei, A. savesi and A. novaezealandiae) and one of smaller ones (A. cristatus, A. affinis, A. bennettii, A. salvadorii, A. wallacii, A. archboldi and A. albertisi).
The New Zealand Owlet-nightjar A. novaezealandiae went extinct during the latest Holocene (Holdaway et al. 2002; Worthy and Holdaway 2002). The New Caledonian Owlet-nightjar A. savesi is exceedingly rare, not having been reported for more than 100 years since its description, except for a possible sighting in 1998 (Layard and Layard 1881; Tobias and Ekstrom, 2002). Fossil bones are, however, quite numerous (Balouet and Olson 1989; Anderson et al. 2010). The presence of a species of Aegotheles in NZ 19–16 Ma reveals a long history for the group in the region and is, therefore, of considerable interest.
The 217 indigenous breeding taxa known from the late Holocene of NZ (Worthy et al. 2017) represented 46 families (10 endemic) and 105 genera (32 endemic) prior to the catastrophic impacts of humans. Of the endemic taxa, only six families are neoavian, four of which are passeriforms. The St Bathans Fauna has to date revealed no novel endemic family-level taxa, although it has confirmed the early Miocene presence of the endemic Dinornithiformes, Apterygidae, Aptornithidae, and Acanthisittidae (Worthy et al. 2017). Therefore, recognition of a new family-level, non-passerine neoavian taxon is a significant addition to the New Zealand biota.
Materials and methods
Institutional abbreviations: AM, Australian Museum, Sydney, New South Wales, Australia; CM, Canterbury Museum, Christchurch, NZ; FUR, Palaeontology collection, Flinders University, South Australia; IANCP, collections of Institut d'Archéologie de la Nouvelle-Calédonie et du Pacifique, Nouméa, New Caledonia; NMNZ, Museum of New Zealand Te Papa Tongarewa, Wellington, NZ; SAM, South Australian Museum, Adelaide, South Australia; YPM, Yale Peabody Museum of Natural History, New Haven, Connecticut, USA; USNM, United States National Museum, Smithsonian Institution, Washington D.C., USA.
Terms for specific anatomical features follow Baumel and Witmer (1993). We follow Dickinson and Remsen (2013) for nomenclature of modern taxa, wherein taxonomic authorities can be found; we provide taxon authors only for extinct taxa at first mention.
Comparative specimens: The extensive collection of modern bird skeletons at SAM was used for wider comparisons. Below we only list specimens of key taxa that were examined for the following descriptions. Tinamidae (tinamous): Nothura darwinii CM Av26475, Av26476; Nothura maculosa, CM 2015.175.1; Tinamus solitarius, CM 2015.175.2; Crypturella obsoletus, CM 2015.175.3; Rhynchotus rufescens, CM 2015.175.4; Eudromia elegans, CM 2015.175.5. Podargidae (Frogmouths): Podargus strigoides SAM B.56413, 59039. Caprimulgidae (Nightjars), Eurostopodinae: Eurostopodus argus SAM B.55617, 56406, 56393. Aegothelidae (Owlet-nightjars): Aegotheles cristatus SAM B.11472, 32226, 32314, 32880, 40084, 47604, 48184, 48534, 48671, 49635, 55133, 55527, 56425, 58554, 58983, 59200. Aegotheles savesi Specimens from Pindai Cave A, Testpit 3 and Testpit 4, now in IANCP, Noumea, New Caledonia. Aegotheles crinifrons, Moluccan Owlet-nightjar, USNM 560816, synoptic skeleton image, male, [https://collections.nmnh.si.edu/search/birds/]. Aegotheles salvadorii, YPN ORN 104653. Aegotheles insignis, YPN ORN 104654. Aegotheles novaezealandiae CM Av16996, holotype; paratypes Av16638, 16745, 16773, 16804, 17333; 17503, 17513, 17774, 18012, 18240, 18641, 20651. Other specimens CM Av14472, 17236, 25160, 28220, 31188, 33283, 36016. NMNZ S.22454 image https://collections.tepapa.govt.nz/object/373259#open-iiifViewerCuculiformes (cuckoos), Centropodinae: Centropus phasianinus SAM B.32225, 48695. Cuculinae: Chalcites basalis SAM B.51225. Chalcites osculans SAM B.49504. Chalcites lucidus SAM B.46817. Cacomantis flabelliformis SAM B.49232, 55124, 58737. Heteroscenes pallidus SAM B.31781, 32426. Strigiformes (Owls): Tyto delicatula FUR219. Ninox novaeseelandiae, FUR 181. Coliidae (Mousebirds): Urocolius (Colius) macrourus, USNM 490679. Colius striatus, USNM 558545. Trogonidae (Trogons): Harpactes ardens, USNM 488931. Apaloderma aequatoriale USNM 291791. Pharomachrus mocinno, USNM 428247. Trogon violaceus, USNM 612331. Upupidae (Hoopoes): Upupa epops, USNM 603636; SAM B.59285. Picidae (Woodpeckers): Jynx torquilla SAM B.59286, Italy. Dryocopus pileatus SAM B.47755. Colaptes auratus SAM B.36893. Sphyrapicus varius SAM B.47764. Melanerpes erythrocephalus SAM B.47758. Coraciidae (Rollers): Eurystomus orientalis AM O.62414. Meropidae (Bee-eaters): Merops apiaster SAM B.36837, 48746. Ramphastidae (Toucans and Barbets), Megalaiminae (Asian Barbets): Psilopogon zeylanicus (=Megalaima zeylanica), USNM 343617. Ramphastinae, Ramphastini (Toucans, Toucanets): Aulacorhynchus prasinus caeruleogularis, USNM 613302. Lybiinae, Trachphonini (African Barbets), Trachyphonus vaillantii, USNM 558564. Tribe Lybiini, Tricholaema leucomelas, USNM 642369.
Measurements: We used a graticule in a binocular microscope or TESR dial callipers, and rounded measurements to 0.1 mm.
Nomenclatural acts: To conform to the requirements of the amended International Code of Zoological Nomenclature and hence make available under that Code the new names contained herein, this article and the nomenclatural acts it contains have been registered in ZooBank, the online registration system for the ICZN. The LSID for this publication is:
urn:lsid:zoobank.org:pub:A52BB3B2-CD1D-46FE-A80A-3E64B6D97660.
Systematic palaeontology
The St Bathans aegothelid
Class Aves Linnaeus, 1758.
Subclass Strisores Baird, 1858, sensu Mayr (2010)
Order Apodiformes Peters, 1940
NMNZ S.52917 (Fig. 1) has the common avian arrangement of the trochleae metatarsorum II, III and IV directed cranially, which excludes affinity with birds having the zygodactyl condition of the trochlea metatarsi IV rotated plantarly, usually with development of an accessory trochlea (Psittaciformes, Strigiformes, Cuculiformes, Coliiformes, Piciformes (including Picidae, Ramphastidae, Bucconidae, Gabulidae, Indicatoridae), or with those having the heterodactyl condition of trochlea metatarsi II rotated plantarly (Trogoniformes). Tarsometatarsi of bucerotiforms differ markedly from the fossil in having the trochleae essentially in one plane, fossa metatarsi I deeply incising the medial margin of the shaft, trochlea metatarsi II markedly prominent medially and trochlea metatarsi III short. Tarsometatarsi of all passerines differ markedly from the fossil in that the trochleae are in one dorsoplantar plane and have near equal distal extent.
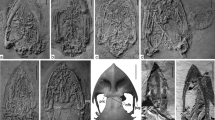
Tarsometatarsi of species of Aegotheles. Aegotheles zealandivetus sp. nov. NMNZ S.52917, distal right tarsometatarsus (A–E) in lateral (A), medial (B), distal (C), dorsal (D), plantar (E), aspects; Aegotheles cristatus SAM B.55133 (F, K); Aegotheles novaezealandiae CM Av16996 (holotype), left tarsometatarsus (reversed) (H, M); A. novaezealandiae CM Av17503 (paratype), distal left tarsometatarsus (reversed) (G). A. insignis YPM ORN 104654 (I, J); A. savesi right tarsometatarsus, (IANCP unregistered: 100–110 cm Testpit 3, Pindai, New Caledonia) (L). Abbreviations: cpl, crista plantaris lateralis; fm1, fossa metatarsi I; fvd, foramen vasculare distale; il, incisura intertrochlearis lateralis; TII, trochlea metatarsi II; TIV, trochlea metatarsi IV. Scale bars are 10 mm, and apply to images A–H. Images I–M are scaled so the distal width is the same as in (D, E)
Tarsometatarsi of procellariiforms, specifically species of Pelecanoides, which is represented by P. miokuaka Worthy et al., 2007 in the St Bathans Fauna (Worthy et al. 2007), differ greatly as follows: they have no, or a very weakly formed, fossa metatarsi 1; the foramen vasculare distale is larger and preceded by a deep sulcus; the trochlea metatarsi II is distinctly shorter than the trochlea metatarsi IV and is deeply grooved; and the trochlea metatarsi IV is grooved dorsally.
The fossil shows closest similarity to the tarsometatarsi of some caprimulgiforms, e.g., Eurostopodus argus and aegothelids, but those of species of Eurostopodus differ notably in having the proximal facet in the fossa metatarsi I prominent medially, cristae plantares medialis et lateralis more robust, distally shorter trochleae that diverge symmetrically from the shaft and are more arched so that, plantarly, the gap separating trochleae metatarsorum II and IV is less than the width of trochlea metatarsi III, and trochleae metatarsorum III and IV have shallow grooves dorsally, distally and plantarly. The tarsometatarsi of aegothelids have most similarity to the fossil.
Family Aegothelidae Bonaparte, 1853
Genus Aegotheles Vigors & Horsfield, 1827
Aegotheles zealandivetus sp. nov.
urn:lsid:zoobank.org:act:89951097-E888-4589-9FD2-8E928B40FAF0.
Holotype: NMNZ S.52917, a distal right tarsometatarsus, with part of the shaft and three trochleae exhibiting slight wear but otherwise well-preserved (Fig. 1A–E).
Etymology: From Zealandia, the continent on which New Zealand is emergent and vetus Latin adjective for old, ancient, or former, to reflect the fact that the new species was an old inhabitant of Zealandia, masculine.
Type locality: Bed HH1b, c.10 cm thick sand and coarse cobble layer, 9.5–9.58 m above base of Bannockburn Formation, Trench excavation, foot of hill 50 m across terrace from riverbank at 44.54.493°S; 169.85844°E; Manuherikia River, Home Hills Station, Otago, New Zealand. NZ Fossil Record File Number H41/f0103.
Stratigraphy and age: Bannockburn Formation, Manuherikia Group, early Miocene (19–16 Ma).
Diagnosis: The new taxon is an aegothelid having the following unique combination of characters for the distal tarsometatarsus: The fossa metatarsi I is deep, broad, not impacting on the medial profile of the shaft; the foramen vasculare distale is small, close to the incisura lateralis, and preceded by a short shallow sulcus; the trochleae diverge asymmetrically; the trochlea metatarsi II is directed somewhat medially making the medial profile concave; the trochlea metatarsi IV is slightly offset from the shaft, directed distally, resulting in convex lateral profile; the dorsal surface proximally adjacent to trochlea metatarsi III is flat, lacking a sulcus extending proximally from the incisura medialis; the distal projection of trochlea metatarsi II is slightly less than that of trochlea metatarsi IV, with 25% of the length of trochlea metatarsi III exceeding both; the trochlea metatarsi II lacks a groove dorsally; the trochlea metatarsi IV has a shallow groove distally but none dorsally; and the trochlea metatarsi III is deeply grooved dorsally, distally and plantarly, deeper than wide, with rims parallel in plantar aspect.
Differential diagnosis: Differs from Aegotheles savesi, A. novaezealandiae, A. salvadorii and A. cristatus on account of the following features: the dorsal surface proximal to trochlea metatarsi III is flat and merges with an evenly convex facies extending to the medial margin proximal to the trochlea metatarsi II without interruption by a groove extending from the intertrochlear incision (as opposed to being convex with a deep groove); the foramen vasculare distale is smaller; the trochlea metatarsi II is shorter than the trochlea metatarsi IV (as opposed to a relatively longer trochlea metatarsi II that distally exceeds trochlea metatarsi IV, or which may distally exceed trochlea metatarsi III in A. novaezealandiae).
Differs from A. novaezealandiae on account of: the trochlea metatarsi IV extends distally slightly past the trochlea metatarsi II; the trochlea metatarsi IV projects distally straight (not directed laterally); the fossa metatarsi I is larger and more excavated.
Differs from A. savesi on account of: the dorsal surface is flatter; the trochlea metatarsi II does not equal or surpass distally the trochlea metatarsi IV in medial/dorsal views; the trochlea metatarsi II is more plantarly retracted; the projection on the medial rim of the trochlea metatarsi II is smaller, even allowing for damage (A. savesi has a robust plantar flange); the trochlea metatarsi II is broader mediolaterally (dorsal view); the rims of the trochleae are more pronounced; there is a deeper furrow on trochlea metatarsi III, with rims that are parallel and not tapered in plantar aspect (A. savesi has proximally convergent rims); the trochlea metatarsi IV has a groove distally (lacking) and its outer rim lacks plantar projection (in A. savesi, the outer rim has a narrow flange extending about 30% of its depth plantar to the inner rim); the foramen vasculare distale is round, smaller and situated closer to incisura intertrochlearis; the fossa metatarsi I is wider/broader; and the cristae plantares medialis et lateralis is stronger.
Differs from Aegotheles cristatus and A. salvadorii on account of: larger size (Table 1); the trochlea metatarsi II is not as medially splayed; the trochlea metatarsi IV is directed mainly distally (more laterally) and is more laterally positioned relative to the shaft so that the profile is convex (versus straight in A. cristatus and A. salvadorii); the trochlea metatarsi III is deeply grooved; the trochlea metatarsi IV is shallowly grooved distally (in A. cristatus and A. salvadorii, all trochleae lack a groove both dorsally and distally).
Most similar to those of A. crinifrons and A. insignis, especially in the minimal development of a sulcus and ridge that extends proximally to the incisura medialis and the trochlea metatarsi III, respectively, and in the trochlea metatarsi II being comparatively short. Differs from A. crinifrons on account of: the shaft is more slender; the trochlea metatarsi II is shorter and level with the trochlea metatarsi IV; the trochleae metatarsorum III et IV are grooved; the trochlea metatarsi III has rims that are parallel in plantar view (convergent proximally). Differs from A. insignis on account of: the shaft is more slender; the trochlea metatarsi II is longer (IV much exceeds II in insignis); the trochlea metatarsi III is deeply grooved dorsally (lacking); the foramen vasculare distale is relatively larger.
Aegotheles tatei is the only similar-sized aegothelid that the specimen was not compared with, but it was considered conspecific with A. insignis until recently, so is probably very similar to that taxon; moreover, all these modern birds are separated from the fossil taxon by a minimum of 16 million years so are unlikely to be conspecific.
Referred specimens: All from beds on the East bank of the Manuherikia River, Home Hills Station, St Bathans, Otago, NZ. NMNZ S.42800, a proximal right tarsometatarsus, Bed HH1a, 6.88–7.0 m above base of Bannockburn Formation, at 44.907944S, 169.858222E. NZ Fossil Record File Number H41/f88. (Schwarzhans et al. 2012; Worthy et al. 2022). CM 2013.18.1127, cranial part of a right coracoid (Fig. 2A–C), Bed HH2, 21.02–21.48 m above the base of the Bannockburn Formation, at 44.907861°S 169.857389°E; NZ Fossil Record File Number H41/f87. NMNZ S.51974, proximal right ulna (Fig. 2G), Bed HH1b, details as per type locality. NMNZ S.52761, distal right tibiotarsus (Fig. 2J), Bed HH1b, details as per type locality.
Bones of species of Aegotheles: coracoids (A–D); ulnae (E–G); and tibiotarsi (H–J). Aegotheles zealandivetus sp. nov. cranial part right coracoid CM 2013.18.1127 (A–C), proximal right ulna NMNZ S.51974 (G), distal right tibiotarsus NMNZ S.52761 (J); Aegotheles cristatus SAM B.55133 (D, F, H), A. novaezealandiae CM Av28220 (paratype), proximal left ulna [reversed] (E), CM Av18240, distal left tibiotarsus reversed (K); A. insignis YPM ORN 104654, surface scan of specimen (I). Abbreviations: cs, cotyla scapularis; cv, cotyla ventralis; dsf, distal sulcus of musculus fibularis; el, tuberositas retinaculi extensoris lateralis; em, tuberositas retinaculi extensoris medialis; fah, facies articularis humeralis; fs, foramen nervi supracoracoidei; ic, incisura intercondylaris; lc, condylus lateralis; mc, condylus medialis; ol, olecranon; pcd, processus cotyla dorsalis; tlcv, tuberculum ligamentum collateralis ventralis; arrow points distal end of the processus procoracoideus. Scale bars = 10 mm
Measurements (mm): Holotype, NMNZ S.52917, distal right tarsometatarsus, preserved length 12.0; shaft width 2.1; shaft depth 1.8; distal width 5.1. NMNZ S.42800, proximal right tarsometatarsus: preserved length 11.0; proximal width 4.3; proximal depth 4.1 mm; least shaft width 1.6 mm. NMNZ S.51974, proximal right ulna: maximum proximal width 4.5. CM 2013.18.1127, cranial part right coracoid: preserved length 12.6; cotyla scapularis to proximal side facies articularis humeralis 3.5. NMNZ S.52761, distal right tibiotarsus: distal width 4.4; proximodistal length condylus lateralis 3.1; proximodistal length condylus medialis 2.9; depth condylus medialis 4.1. See Table 1.
Description and comparisons
Holotype, NMNZ S.52917: As the distal tarsometatarsus shows more morphological disparity among aegothelids than the other elements referred to the new species, the following additional features not captured in the diagnoses are detailed: 1. The fossa metatarsi I is deeply concave and extends over about 30% of shaft depth and to mid-width of the shaft. It is steep-walled medially and mainly faces plantarly. On the plantar surface and proximally-adjacent to the fossa, is a rounded elevated rugose scar. 2. The cristae plantares medialis et lateralis extend proximally along the margins of the shaft from this scar and are separated by a flat face. 3. The foramen vasculare distale is small and separated from the incisura lateralis by a distance slightly greater than its length and is preceded by a short shallow sulcus proximally. 4. The trochlea metatarsi II is retracted plantarly and overlaps the plantar half of the depth of trochlea metatarsi III. 5. The trochlea metatarsi IV is slightly retracted plantarly relative to trochlea metatarsi III. 6. The trochlea metatarsi III is deeper than wide, and its rims are near parallel plantarly. 7. The trochlea metatarsi IV is smooth dorsally but has a shallow groove plantarly that slightly impacts the distal margin. However, the outer cortical surface is worn so the groove will have been originally deeper. 8. The outer rim of trochlea metatarsi II lacks a flange projecting plantarly and is shallower than the internal rim. 9. The rims of trochlea metatarsi IV have equal depth and lack a plantar flange.
The following aegothelid specimens are referred to the new taxon, because they are of appropriate size for the holotype:
NMNZ S.42800, proximal right tarsometatarsus: This specimen was considered by Worthy et al. (2007) to be very similar to those of A. novaezealandiae and A. cristatus. Here, we note that the tuberositas musculus tibialis cranialis is more displaced from the medial shaft margin and that the sulcus defined by the impressiones retinaculi extensorii is more deeply impressed than in these taxa.
CM 2013.18.1127, cranial part right coracoid (Fig. 2A–C): This omal fragment of a coracoid has lost the greater part of the processus procoracoideus, although the foramen nervi supracoracoidei is present, and the processus acrocoracoideus is lost medial to the sulcus supracoracoideus and the impressio ligamentum acrocoracohumeralis (Fig. 2). The cotyla scapularis is a very shallow fossa. The facies articularis humeralis is more than twice as long as the length of the cotyla scapularis and so relatively longer than in A. novaezealandiae. The foramen n. supracoracoidei is relatively larger than it is in A. novaezealandiae, A. savesi, A. crinifrons and A. cristatus.
NMNZ S.51974, proximal right ulna (Fig. 2G): NMNZ S.51974 is slightly worn with loss to the tip of the processus cotylaris dorsalis, the olecranon, and the caudal margins of the cotylae linking these features. Nevertheless, it clearly exhibits the key attributes of aegothelid ulnae with a small olecranon that is not prominent proximally to the cotyla ventralis, proximal width being far wider than the craniocaudal depth between the olecranon and the cranial margin of the cotyla ventralis, and a markedly ventrally prominent tuberculum ligamentum collateralis ventralis. The incisura radialis is a flattened shallow sulcus traversed by a narrow scar passing proximoventrally over the ventral margin onto the impressio brachialis. The impressio brachialis is deep and bound caudally by a robust ridge. There are no notable differences between the fossil and compared taxa, although it is a little larger than the ulna of A. cristatus, consistent with the size differences of the referred coracoid.
NMNZ S.52761, distal right tibiotarsus (Fig. 2J): This specimen preserves the distal end from just proximal to the tuberositas retinaculi extensoris lateralis. It shows the typical aegothelid features of the condylus lateralis being wider than the condylus medialis; a narrow incisura intercondylaris; a shorter than wide pons supratendineus with its distal margin between the condyles; the area intercondylaris lacking a secondary fossa; the tuberositas retinaculi extensoris lateralis forming a prominent tuberculum lateral to the pons and extending proximally as a narrowing crista; and a small epicondylus medialis. NMNZ S.52761 is smaller than specimens of A. savesi and A. novaezealandiae but larger than those of A. cristatus (Table 1). The condyles are proximodistally shorter than distal width, being similarly proportioned to A. insignis and A. novaezealandiae, whereas in A. cristatus the length of the condyles more closely approximates distal width. In the fossil, the area proximal to the condylus lateralis and lateral to the tuberositas retinaculi extensoris lateralis is relatively broad and flattened and faces craniolaterally. This is the distal end of the sulcus of musculus fibularis and is morphologically very similar to the condition seen in A. novaezealandiae but differs from that in A. cristatus, where the sulcus is rotated onto the lateral facies at this point (Fig. 2). NMNZ S.52761 is also more similar to A. novaezealandiae in the shape of the condylus medialis. While worn distomedially, the proximal part of this condyle is relatively narrower than in both A. novaezealandiae and A. cristatus, resulting in a straighter medial profile from the shaft onto the condyle in cranial aspect (Fig. 2).
Comparisons, fossil taxa
Other than the late Holocene A. novaezealandiae compared above, the only other fossil aegothelid is the late Miocene species Quipollornis koniberi Rich & McEvey, 1977, from the early to middle Miocene Chalk Mountain Formation in New South Wales. It is, therefore, roughly of contemporary age to the St Bathans Fauna, but it is larger than both A. cristatus and A. novaezealandiae and thus larger than the new species reported here.
The holotype NMNZ S.052917 of Aegotheles zealandivetus sp. nov. shows some similarities to the distal tarsometatarsus of Procuculus minutus Harrison & Walker, 1977 (holotype, NHM A 4680) from the Eocene London Clay, which was identified as an apodiform by Mayr (2016). However, this species differs by being about half the size of NMNZ S.52917, and has a narrower more laterally directed trochlea metatarsi IV, and the trochlea metatarsi II is broader, deeply notched plantarly and more medially inflected (see Mayr 2016, fig. 2s).
A new family of higher landbirds
Order incertae sedis.
Family Zealandornithidae fam. nov.
urn:lsid:zoobank.org:act:EE0CDC0E-A6EB-4118-96FE-9F8C59D3CC09.
Included taxa: Zealandornis relictus gen. et sp. nov.
Diagnosis: A small coly-like bird, with distal humeri characterised by the following unique combination of characters: a large, robust, dorsally prominent processus supracondylaris dorsalis with its tip located just proximal to the condylus dorsalis; the scar the for palmar branch of musculus extensor carpi radialis is located ventrally adjacent to the processus supracondylaris dorsalis, proximally enclosing a deep lenticular fossa adjacent to the condylus dorsalis; a deep fossa brachialis, restricted to the ventral half of the bone, extending proximal to the processus supracondylaris dorsalis, and narrowly separated from the ventral margin; an undeveloped epicondylus dorsalis, not projected dorsal to the condylus dorsalis; the condylus dorsalis is widely separated from the tuberculum supracondylare ventrale; the condylus ventralis is globose, distally prominent; a robust ventrally prominent processus flexorius–tuberculum supracondylare ventrale complex that lacks a prominent epicondylus ventralis; a distally short processus flexorius, greatly surpassed by the condylus ventralis; no distinct sulcus scapulotricipitalis (autapomorphy); the tuberculum supracondylare ventrale is robust, cranially elevated, lacking a distinct facet for the attachment of ligamentum collaterale ventrale (autapomorphy), and flattened ventrally.
The overall form, with maximum distal width about twice as wide as the proximodistal length from the condylus dorsalis to the processus supracondylaris dorsalis, the processus supracondylaris dorsalis being large and prominent, and the processus flexorius–tuberculum supracondylare ventrale complex being strongly ventrally prominent, is consistent with it deriving from a bird in one of the following neoavian ordinal lineages, Cuculiformes, Strigiformes, Caprimulgiformes, and Coraciimorphae, including Coliiformes, Leptosomatiformes, Trogoniformes, Bucerotiformes, Piciformes, Coraciiformes, and Passeriformes. It is distinguished from all these taxa by lack of a well-marked facet on the tuberculum supracondylare ventrale, and from all except podargids by a lack of a distinct sulcus scapulotricipitalis.
It is further distinguished from passeriforms by the dorsal location of the scar for the palmar branch of musculus extensor carpi radialis; from strigiforms, by the more proximal location of the processus supracondylaris dorsalis, a distally shorter processus flexorius, lack of ventral prominence of the epicondylus ventralis and smaller size; from cuculids by the processus supracondylaris dorsalis being less offset proximally from the condylus dorsalis, the scar for the palmar branch of musculus extensor carpi radialis more dorsoventrally elongate, less ventrally prominent processus flexorius–epicondylus ventralis complex, distally shorter processus flexorius, more elongate fossa musculi brachialis extending proximal to the processus supracondylaris dorsalis; from caprimulgiforms, by the more proximal location of the processus supracondylaris dorsalis, more globose condylus ventralis, and a wider tuberculum supracondylare ventrale that is more widely separated from the condylus dorsalis; further differentiated from podargids, by a larger more prominent processus supracondylaris dorsalis, a better defined lenticular sulcus proximal to condylus dorsalis; from caprimulgids, by a much larger more prominent processus supracondylaris dorsalis, where the palmar branch of musculus extensor carpi radialis is ventrally adjacent to the scar for the dorsal branch, not offset proximally from it; from aegothelids, by the lack of a dorsally prominent epicondylus dorsalis, the presence of a robust dorsally prominent processus supracondylaris dorsalis, and the lack of a ventrally projecting triangular epicondylus ventralis; from all trogoniforms and bucerotiforms, in having a robust dorsally prominent processus supracondylaris dorsalis; from piciforms, by having a distally short processus flexorius rather than one that is elongate and surpasses the condylus ventralis, and from ramphastids among piciforms, by the large dorsally prominent processus supracondylaris dorsalis; from all coraciiforms, by a globular condylus ventralis, lack of a distinct ventrally prominent epicondylus ventralis, and lack of a dorsally prominent epicondylus dorsalis; and from coliiforms, by a distally shorter processus flexorius, lack of projection for the epicondylus ventralis, and less separation of fossa musculi brachialis from the lenticular fossa proximal to the condylus dorsalis.
The lack of a distinct sulcus scapulotricipitalis confers superficial similarity to palaeognaths—lithornithids and tinamids—although this feature is also seen in many galliforms and podargids. Both lithornithids and tinamids differ by a much narrower gap between the condylus dorsalis and the tuberculum supracondylare ventrale (<tuberculum width), smaller processus supracondylaris dorsalis, a distinct and large fossa for insertion of the proximal head of musculus pronator superficialis, a stouter shaft, and much larger size. Lithornithids differ further by the fossa musculi brachialis extending to the dorsal margin and a far more distally elongate processus flexorius (Houde 1988). All compared tinamids, and see Bertelli et al. (2014), have a shallow fossa musculi brachialis of variable extent and a much smaller and not proximally hooked processus supracondylaris dorsalis. These features make it very unlikely that the fossil could be associated with Proapteryx micromeros Worthy TH, Worthy JP, Tennyson, Salisbury, Hand, Scofield, 2013, a small kiwi known from the St Bathans fauna (Worthy et al. 2013a, b). However, we explored the relative size of NMNZ S.52077 and P. micromeros, wherein a ratio of the minimum femur shaft width to distal width of the humerus of 0.66 is much higher than that for volant tinamous and very much less than that for Apteryx owenii (see SI); the distal humerus of all species of Apteryx have amorphous shapes, wherein no typical humeral morphology is present. While the proportions are consistent with a slight relative size reduction in a humerus for the given size of the femur of Proapteryx, the major differences in morphology from volant palaeognaths precludes this association.
Zealandornis relictus gen. et. sp. nov. ( Fig. 3 A – D ).
Distal right humeri. Zealandornis relictus gen. et sp. nov. NMNZ S.52077 in cranial (A), caudal (B), dorsal (C), and ventral (D) aspect. Aegotheles cristatus SAM B.55133 (E); Colius striatus USNM 558545 (F); Cacomantis flabelliformis SAM B.55124 (G). Abbreviations: cd, condylus dorsalis; cv, condylus ventralis; ed, epicondylus dorsalis; ev, epicondylus ventralis; fb, fossa musculi brachialis; flcv, facet for the attachment of ligamentum collaterale ventrale on the tuberculum supracondylare ventrale; ls, lenticular sulcus; pbmr, scar for the palmar branch of m. extensor carpi radialis; pf, processus flexorius; psd, processus supracondylaris dorsalis; sh, sulcus humerotricipitalis; tsv, tuberculum supracondylare ventrale. Scale bar = 5 mm
urn:lsid:zoobank.org:act:B36C310E-FCCB-4E7C-BE75-007911B6676B.
urn:lsid:zoobank.org:act:B733FC16-ACFA-4771-BB06-0586CDF606C3.
Holotype: NMNZ S.52077, distal right humerus (Fig. 3A–D), collected on 15 January 2008 by a University of New South Wales led (S. Hand, M. Archer, et al.) expedition, jointly involving Adelaide University of South Australia (T.H. Worthy et al.), the Museum of New Zealand (A. Tennyson) and the Canterbury Museum (R.P. Scofield).
Diagnosis: As for family.
Type locality: Bed HH1a, Manuherikia River, 6.88–7.0 m above the base of the Bannockburn Formation, East bank Manuherikia River, Home Hills Station, St Bathans, Otago, NZ at 44.907944°S, 169.858222°E. NZ Fossil Record File Number H41/f88. (Schwarzhans et al. 2012; Worthy et al. 2022).
Stratigraphy and age: Lower Bannockburn Formation, Manuherikia Group, early Miocene (19–16 Ma).
Etymology: The genus name captures the origin of this taxon from Zealandia, gender masculine, with the specific epithet to reflect one remaining or left behind.
Measurements (mm): Maximum distal width, processus supracondylaris dorsalis–epicondylus ventralis, 5.4; distal width condylus dorsalis–epicondylus ventralis, 5.0; length processus supracondylaris dorsalis–distal margin condylus dorsalis, 2.9; minimum shaft diameter as preserved, 2.0.
Description and comparisons
The distal right humerus NMNZ S.52077 has the following features. (1) A robust, dorsally prominent processus supracondylaris dorsalis (=tuberculum supracondylare dorsale) located distinctly proximal to the condylus dorsalis, on which facing proximodorsally, is a well-marked scar for the dorsal branch of musculus extensor carpi radialis (dbmr). The robustness of this process is in part due to it having a proximodistally elongate bulbous cranial expansion that distally partially overlaps the proximal extent of the condylus dorsalis. (2) The scar for the palmar branch of musculus extensor carpi radialis (pbmr) ventrally abuts the processus supracondylaris dorsalis on the cranial surface. It is dorsoventrally elongate and raised, thus contributes to defining a deep lenticular sulcus. (3) The lenticular sulcus lies proximally adjacent to the entire width of the condylus dorsalis. (4) The fossa musculi brachialis is deep, restricted to the ventral half of the bone, and while damage precludes ascertaining its original proximal extent, the preserved section extends well proximal to the processus supracondylaris dorsalis, and distally it merges with a flattened face adjacent to the ventral margin that proximally bounds the tuberculum supracondylare ventrale. Ventrally, the fossa is bound by a low and narrow ridge. (5) The tuberculum supracondylare ventrale is a robust, rounded, cranially elevated prominence that originates slightly proximal to the condylus ventralis and lacks a facet for the ligamentum collaterale ventrale. Its proximal margin is level with that of the condylus dorsalis, from which it is separated by a space much wider than the tuberculum. Although this tuberculum is a little worn, its cranially bulbous form projects cranially in the area a distinct facet occupies on this tuberculum, where such is present, so we consider that wear does not account for the lack of a facet. (6) The tuberculum supracondylare ventrale and, caudal to it the processus flexorius, together form a robust ventrally flattened prominence that is not surpassed ventrally by a distinct epicondylus ventralis. (7) The processus flexorius is very poorly developed distally, and its convex distal margin is greatly surpassed by the condylus ventralis. (8) The condylus ventralis is globose, not dorsoventrally elongate, very prominent distally, and separated from the condylus dorsalis by a distinct notch in cranial aspect. Its junction with the processus flexorius ventrally is marked by a very shallow notch in cranial aspect. (9) Caudally, the facies where the sulcus scapulotricipitalis is normally located is convex caudally and distally but wear suggests that a very shallow sulcus was the original condition. (10) The sulcus humerotricipitalis is dorsoventrally narrow and shallow. (11) The epicondylus dorsalis (ectepicondyle) is indistinct, not projecting dorsal to the condylus dorsalis. (12) There is no sign of a pit ventrally for the origin of the proximal head of musculus pronator superficialis. (13) The preserved minimum shaft diameter (2.0 mm) is relatively narrow, 40% of the distal width (5.0 mm).
The general form of NMNZ S.52077, with a large prominent processus supracondylaris dorsalis and a strongly ventrally prominent processus flexorius–tuberculum supracondylare ventrale complex, is consistent with it deriving from a bird in one of the following neoavian ordinal lineages, Cuculiformes, Strigiformes, Caprimulgiformes, and Coraciimorphae including Coliiformes, Leptosomatiformes, Trogoniformes, Bucerotiformes, Piciformes, Coraciiformes, and Passeriformes. However, the coliiforms display the greatest similarity as follows. Detailed comparisons are made between NMNZ S.52077 and the humeri of other listed taxa in the SI.
Humeri of the colies (Urocolius macrourus USNM 490679, Colius striatus USNM 558545) have the following similarities to NMNZ S.52077: a large dorsally prominent processus dorsalis supracondylaris located proximal to the condylus dorsalis (1), with the dorsal branch of the musculus extensor carpi radialis (dbmr) on its dorsal tip, and the palmar branch (pbmr) located adjacent to it ventrally (2); a deep lenticular sulcus proximal to the condylus dorsalis, bound proximally by pbmr (3); a deep fossa musculi brachialis restricted to the ventral half of the bone, extending well proximal to the processus supracondylaris dorsalis and narrowly separated from the ventral shaft margin (4); the tuberculum supracondylare ventrale broadly separated from the condylus dorsalis (in part 5); the condylus ventralis being dorsoventrally short and proximodistally broad in cranial aspect, resulting in a globular appearance (8); and an epicondylus dorsalis not projecting dorsal to the condylus dorsalis (11).
However, coliids differ from NMNZ S.52077 in significant ways (see also Rich and Haarhoff 1985) as follows. The fossa musculi brachialis is separated by an elevated rounded ridge from the lenticular fossa proximal to the condylus dorsalis. The tuberculum supracondylare ventrale has a large triangular distally facing facet, as wide as it is elevated. The processus flexorius is robust caudally and extends distally to the same extent as the condylus ventralis. The epicondylus ventralis projects markedly ventral to the facet on the tuberculum supracondylare ventrale in species of Colius, but less so in C. (Urocolius) macrourus, wherein the processus flexorius is more projected caudally. The sulcus scapulotricipitalis is deep and confined by a robust ridge ventrally. The sulcus humerotricipitalis is deep and bound by a steep wall of the processus flexorius ventrally.
Fossil Coliiformes (Coliidae, Sandcoleidae): Humeri of fossil coliids, where comparable (Oligocolius brevitarsus Mayr, 2000, Primocolius minor Mourer-Chauviré 1988, P. sigei Mourer-Chauviré, 1988; Masillacolius brevidactylus Mayr & Peters, 1998; Necrornis palustris Milne-Edwards, 1871; Selmes absurdipes Peters, 1999), differ from NMNZ S.52077 as follows: a smaller processus supracondylaris dorsalis, more ventrally prominent epicondylus ventralis and distally projecting processus flexorius, and a distinct facet on tuberculum supracondylare ventrale located more proximal than the condylus ventralis (Mayr & Peters 1998; Mayr 2000, 2010, 2020: fig. 5).
A distinct facet on the tuberculum supracondylare ventrale located more proximal than the condylus ventralis was also present on the distal humerus of the sandcoleids Eoglaucidium pallas Fischer, 1987, Sandcoleus copiosus Houde & Olson, 1992, Anneavis anneae Houde & Olson, 1992; and Chascacocolius oscitans Houde & Olson, 1992 (Houde & Olson 1992; Mayr & Peters 1998, Fig. 2; Zelenkov & Dyke 2008; Mayr 2020, fig. 5). In all these taxa, the epicondylus ventralis is prominent ventrally and the processus flexorius is more elongate distally, although in Eoglaucidium pallas it was described as small (Mayr & Peters 1998), and the processus supracondylaris dorsalis is smaller.
Summary: Our comparisons show that extensive differences exist between the humerus described as Zealandornis relictus gen. et sp. nov. and all compared extant birds. However, given the unusual morphology of NMNZ S.52077, it is worth considering to what extent wear may have affected these comparisons. While the holotype bone is worn, the processus supracondylaris dorsalis, the condyles and their caudal facies are well preserved, as is the distal section of the fossa musculi brachialis and the adjacent shaft region. The cranial prominence of the tuberculum supracondylare ventrale and the distoventral margin of the epicondylus ventralis–processus flexorius complex are worn. However, in all taxa with a well-defined facet on the tuberculum supracondylare ventrale, the inner (dorsal) side of this facet is abruptly raised above the facies between it and the condylus dorsalis in the area proximal to the condylus ventralis; no such elevation is apparent on the fossil and no wear is evident, where such elevation would be expected. Moreover, the ventral facies next to the tuberculum supracondylare ventrale preserves unworn surface bone, precluding an extensive and prominent epicondylus ventralis having existed. Thus, while wear allows for the former presence of a low distally directed facet on the tuberculum supracondylare ventrale, all other features are apparently real.
The question then arises, to what extent could such an unusual combination of features be due to a lack of volancy in the bird this bone derives from? This possibility is signalled by the relatively gracile shaft compared to the distal width. There are few taxon-sets, where volant and flightless or weakly flying species occur in the same clade, and which have any similarity to the fossil. Perhaps the acanthisittid wrens provide the best example, as the family includes adept fliers (Acanthisitta chloris) and wholly flightless taxa, such as Dendroscansor decurvirostris Millener & Worthy, 1991, Traversia lyalli and species of Pachyplichas (see Millener 1988; Millener & Worthy 1991). Relative to A. chloris, the flightless taxa do not have a consistently narrower humeral shaft relative to the width of the distal extremity. Unexpectedly, D. decurvirostris has a relatively wider shaft than A. chloris, although the reported distal width is likely an under-estimate as the processus flexorius is missing in the measured holotype humerus. Moreover, while Traversia lyalli had a relatively reduced processus supracondylaris dorsalis, this processus was not similarly reduced in species of Pachyplichas and Dendroscansor. Similarly, there is no reduction in the size of the processus flexorius, nor does the depth of the fossa musculi brachialis vary among the volant and flightless acanthisittids. However, D. decurvirostris does have a relatively shallow and indistinct sulcus scapulotricipitalis and sulcus humerotricipitalis compared to A. chloris and species of Xenicus. Similarly, the extinct Slender-billed Greenfinch Carduelis aurelioi and the extinct Bunting Emberiza alcoveri, both from Tenerife Island in the Canary Islands, were weaker flying and flightless finches, respectively, than their relatives the Common Greenfinch C. chloris and Cabanis's Bunting E. cabanisi. The main differences between the distal humeri of these taxa and their volant congeners is that the shaft is narrower and the sulcus scapulotricipitalis is shallower distally (see Rando et al. 1999, 2010). Notably the processus supracondylaris dorsalis and processus flexorius are equally large and prominent.
These comparisons reveal that some features do not appear compromised by changes associated with flightlessness. Notably, if volant species in a lineage have a large distally elongate processus flexorius, it is not absent in their flightless relatives. Similarly, if a large dorsally prominent processus supracondylaris dorsalis is present in volant members of a lineage, it is not necessarily reduced and certainly is not lost in flightless taxa, and neither is its proximodistal location relative to the condylus dorsalis altered. Therefore, only the relatively narrow shaft and that the sulcus scapulotricipitalis and sulcus humerotricipitalis are shallow are potentially traits related to a transition to flightlessness. The remaining features that characterise this species present a unique combination that precludes affinity of this taxon to any known group of birds.
Discussion
In this contribution, we describe two new landbirds from the early Miocene, 19–16 Ma, St Bathans Fauna of NZ. The first is a member of the ‘basal landbird’ group Strisores (Prum et al. 2015; Kuhl et al. 2021), an owlet-nightjar Aegotheles zealandivetus, which is represented by five specimens collected over the last two decades. It is the second fossil species of Aegotheles known from NZ. The first is A. novaezealandiae, which is common in late Quaternary deposits of North and South islands (Scarlett 1968; Worthy and Holdaway 2002). This large and long-legged species was likely a weak flier and is the sister taxon to A. savesi of New Caledonia (Dumbacher et al. 2003), suggesting a long history on these emergent parts of Zealandia. Aegotheles zealandivetus appears to support that idea and shows that the genus may well have a continuous record on Zealandia from the early Miocene 19–16 Ma. This taxon was first noted by Worthy et al. (2007), wherein a proximal tarsometatarsus was reported, a specimen then considered little different to that of either A. cristatus of Australia or the historically extinct A. novaezealandiae from NZ.
However, here we show that while Aegotheles zealandivetus was intermediate in size between these Australasian taxa, the morphology of particularly the distal tarsometatarsus was unlike them. The Australasian taxa, including A. savesi of New Caledonia, have tarsometatarsi with an elongate trochlea metatarsi II and where trochlea metatarsi III is preceded by a distinct ridge dorsally. In contrast, the fossil has a relatively short trochlea metatarsi II and an evenly convex dorsal facies proximally adjacent to the trochleae, features it shares with the New Guinean taxa A. insignis and A. crinifrons, each of which have relatively shorter more robust tarsi than the non-New Guinea taxa. These observations suggest that A. zealandivetus is not necessarily ancestral to A. novaezealandiae, nor even part of the extant Zealandian clade, and, if so, suggests multiple dispersal events of aegothelids into Zealandia.
The second new taxon described herein is likely to be a member of the Telluraves or ‘higher landbirds’ (Yuri et al. 2013; Kuhl et al. 2021), given its morphology is most similar to that of Coliidae. We place Zealandornis relictus in a new family to reflect our contention that the fossil humerus NMNZ S.52077 represents a novel and highly evolutionarily distinctive taxon (May 1990). Discovering a taxon this distinctive among NZ’s fossil terrestrial vertebrate biota is not unexpected. New Zealand, the emergent part of the continent Zealandia (Mortimer et al. 2017), has a long history of producing highly distinctive taxa from long-isolated lineages that had vicariant connections to Gondwana and/or a record of very old (pre-Miocene) dispersal events (Worthy and Holdaway 2002; Gibbs 2006; Trewick et al. 2007; Goldberg et al. 2008; Boyer and Giribet 2009; Wallis and Trewick 2009; Giribet and Boyer, 2010; Worthy et al. 2017). Iconic, or evolutionarily distinctive, extant taxa such as the rhynocephalian tuatara Sphenodon punctatus and the several species of leiopelmatid frogs attest to ancient vicariant origins (Roelants et al. 2007; Jones et al. 2009; Worthy et al. 2013a, b; Carr et al. 2015), as do a host of invertebrate taxa especially from freshwater and soil/litter environments (Apte et al. 2007; Wesenera and VandenSpiegel 2009; Allwood et al. 2010; Toon et al. 2010; Buckley et al. 2011; Marshall 2011; Sun et al. 2014). Some birds attest to minimally a pre-Oligocene dispersal event, if they are not actually vicariant in origin, such as the acanthisittid wrens (Acanthisittidae) and strigopoid (Strigopidae, Nestoridae) parrots (Ericson et al. 2002; Schweizer et al. 2010; Joseph et al. 2012; Jarvis et al. 2014; Mitchell et al. 2016). The moa (Dinornithiformes; 6 genera, 9 species) and kiwi (Apterygidae) derive from ancient dispersal events by volant ancestors, having sister groups in the South American tinamou and Madagascan aepyornithids, respectively (Bunce et al. 2009; Phillips et al. 2010; Mitchell et al. 2014; Yonezawa et al. 2017). Other recently extinct groups, such as the adzebills (Aptornis) are now understood to be the sister taxon to African and Madagascan sarothrurids and their relatives in New Guinea and the Caribbean (Boast et al. 2019; Oswald et al. 2021). NZ clearly has many relictual taxa.
New Zealand is not alone as an island continent preserving relictual taxa. Madagascar is another Gondwanan continental fragment that has been long isolated and that also preserves many disparate and unique faunal components, such as the Cuckoo-roller Leptosomus discolor, a monotypic endemic family with fossil relatives widespread in the Northern Hemisphere (Mayr 2008), and the endemic mesites (Mesitornithidae) with two genera and three species (Dickinson and Remsen 2013). Other examples include the well-known elephant birds Aepyornithidae (Hansford and Turvey 2018) that went extinct during the late Holocene and a host of endemic mammals, most notably the lemurs (Suborder Strepsirrhini, 8 families [Groves 2005]).
It is noteworthy that NZ and Madagascar have recently identified faunal links among such old endemic taxa. First, and unheralded by morphology, is the kiwi and its aepyornithid sister taxon (Mitchell et al. 2014; Yonezawa et al. 2017), and more recently the Aptornis–sarothrurid pairing (Boast et al. 2019; Oswald et al. 2021). However, Zealandornis relictus may be a member of the wider Coliiformes group, or one related to it. Presently, coliiforms have low extant diversity (6 species) and are confined to Africa, yet in the Eocene to Oligocene, were widespread in the Northern Hemisphere across Europe and America (Houde and Olson 1992; Mayr and Peters 1998; Mayr 2000; Zelenkov and Dyke 2008; Ksepka et al. 2017). Regardless of its true affinities that must await further material, Z. relictus appears to be another distinctive relictual taxon, e.g., like the acanthisittids, leptosomids, and the colies, but one whose lineage did not survive to the present.
The rarity of Zealandornis relictus in the St Bathans Fauna suggests it was a bird that inhabited the terrestrial rather than aquatic habitats surrounding palaeolake Manuherikia. Several other such terrestrial taxa are known from 1 to 3 specimens despite more than 7900 fossil bones having been catalogued (authors’ data). Examples of rare taxa in this biota include a large terrestrial turtle, a sphenodontid, the frog Leiopelma miocaenale, and, among birds, a species of Collocalia, an undescribed small accipitrid, and the large parrot Heracles inexpectatus (see in Worthy et al. 2017, 2019). Rarity of taxa that lived outside the zone of deposition underpins the need for large sample sizes in assessing faunal composition.
In summary, our description of Aegotheles zealandivetus adds a quintessential Australasian taxon to the early Miocene St Bathans Fauna, confirming a long history for this genus in the region. In contrast, Zealandornis relictus represents a new bird family, one as evolutionarily distinct as acanthisittid wrens or leiopelmatid frogs. It should be listed along with other New Zealand birds long separated from their nearest relatives, such as dinornithiforms, apterygids, aptornithids, strigopids and acanthisittids. Collectively these have all contributed to the evolutionarily highly distinctive nature of the Zealandian avifauna.
References
Allwood J, Gleeson D, Mayer G, Daniels S, Beggs JR, Buckley TR (2010) Support for vicariant origins of the New Zealand Onychophora. J Biogeog 37:669–681
Anderson A, Sand C, Petchey F, Worthy TH (2010) Faunal extinction and human habitation in New Caledonia: initial results and implications of new research at the Pindai Caves. J Pac Archaeol 1:89–109
Apte S, Smith PJ, Wallis GP (2007) Mitochondrial phylogeography of the New Zealand freshwater crayfishes, Paranephrops spp. Mol Ecol 16:1897–1908
Baird SF (1858) Birds [pp. 18, 128-592]: Reports of explorations and surveys to ascertain the most practicable and economical route for a railroad from the Mississippi River to the Pacific Ocean. U.S. Congress, Washington
Balouet J-C, Olson SL (1989) Fossil birds from late Quaternary deposits in New Caledonia. Smith Contr Zool 469:1–38
Baumel JJ, Witmer LM (1993) Osteologia. In: Baumel JJ, King AS, Breazile JE, Evans HE, Vanden Berge JC (eds) Handbook of avian anatomy: Nomina anatomica avium, vol 23, 2nd edn. Publications of the Nuttall Ornithological Club, Cambridge, pp 45–132
Bertelli S, Chiappe LM, Mayr G (2014) Phylogenetic interrelationships of living and extinct Tinamidae, volant palaeognathous birds from the New World. Zool J Linn Soc 172:145–184
Boast AP, Chapman B, Herrera MB, Worthy TH, Scofield RP, Tennyson AJD, Houde P, Bunce M, Cooper A, Mitchell KJ (2019) Mitochondrial genomes from New Zealand’s extinct adzebills (Aves: Aptornithidae: Aptornis) support a sister-taxon relationship with the Afro-Madagascan Sarothruridae. Diversity 11:24. https://doi.org/10.3390/d11020024
Bonaparte CL (1853) Classification ornithologique pars series. C R Hebdomadaires Séances L’academy Sci 37(18):641–647
Boyer SL, Giribet G (2009) Welcome back New Zealand: regional biogeography and Gondwanan origin of three endemic genera of mite harvestmen (Arachnida, Opiliones, Cyphophthalmi. J Biogeog 36:1084–1099
Buckley TR, James S, Allwood J, Bartlam S, Howitt R, Prada D (2011) Phylogenetic analysis of New Zealand earthworms (Oligochaeta: Megascolecidae) reveals ancient clades and cryptic taxonomic diversity. Mol Phylo Evol 58:85–96
Buller WL (1869) Essay on the ornithology of New Zealand. Trans Proc NZ Inst 1:213–231
Buller WL (1888) A history of the birds of New Zealand, 2nd edition, 2 vol. Buller, London
Bunce M, Worthy TH, Phillips MJ, Holdaway RN, Willerslev E, Haile J, Shapiro B, Scofield RP, Drummond A, Kamp PJJ, Cooper A (2009) The evolutionary history of the extinct ratite moa and New Zealand Neogene paleogeography. PNAS 106:20646–20651. https://doi.org/10.1073/pnas.0906660106
Carr LM, McLenachan PA, Waddell PJ, Gemmell NJ, Penny D, Ebach M (2015) Analyses of the mitochondrial genome of Leiopelma hochstetteri argues against the full drowning of New Zealand. J Biogeog 42:1066–1076
Claramunt S, Cracraft J (2015) A new time tree reveals Earth history’s imprint on the evolution of modern birds. Sci Adv 1:e1501005. https://doi.org/10.1126/sciadv.1501005
Dickinson EC, Remsen JV (2013) The Howard and Moore complete checklist of the birds of the world, vol 1. Aves Press Limited, Eastbourne, p 461
Dumbacher JP, Pratt TK, Fleischer RC (2003) Phylogeny of the owlet-nightjars (Aves: Aegothelidae) based on mitochondrial DNA sequence. Mol Phylo Evol 29:540–549
Ericson PG, Christidis L, Cooper A, Irestedt M, Jackson J, Johansson US, Norman JA (2002) A Gondwanan origin of passerine birds supported by DNA sequences of the endemic New Zealand wrens. Proc R Soc B 269:235–241
Fischer K (1987) Eulenreste (Eoglaucidium pallas nov. gen., nov. sp., Strigiformes, Aves) aus der mitteleozänen Braunkohle des Geiseltals bei Halle (DDR). Mitteilungen aus dem Zoologischen Museum, Berlin, 63, Supplement: Annalen für Ornithologie, 11:137–142, 6 text-figs.; Berlin
Fleming CA (1979) The geological history of New Zealand and its life. Auckland University Press/Oxford University Press, Auckland
Fuller E (1987) Extinct Birds. Viking Rainbird, London
Gibbs G (2006) Ghosts of Gondwana. The history of life in New Zealand. Craig Potton Publ, Nelson
Gill BJ, Bell BD, Chambers GK, Medway DG, Palma RL, Scofield RP, Tennyson AJD, Worthy TH (2010) Checklist of the birds of New Zealand, Norfolk and Macquarie Islands, and the Ross Dependency, Antarctica, 4th edn. Wellington, Ornithological Society of NZ
Giribet G, Boyer SL (2010) ‘Moa’s Ark’ or ‘Goodbye Gondwana’, is the origin of New Zealand’s terrestrial invertebrate fauna ancient, recent, or both? Invert Syst 24:1–8
Goldberg J, Trewick SA, Paterson AM (2008) Evolution of New Zealand’s terrestrial fauna, a review of molecular evidence. Phil Trans R Soc B 363:3319–3334
Groves CP (2005) Order Primates. In: Wilson DE, Reeder DM (eds) Mammal species of the World: a taxonomic and geographic reference, 3rd edn. The Johns Hopkins University Press, Baltimore, pp 111–184
Hansford JP, Turvey ST (2018) Unexpected diversity within the extinct elephant birds (Aves: Aepyornithidae) and a new identity for the world’s largest bird. R Soc Open Sci 5:181295. https://doi.org/10.1098/rsos.181295
Harrison CJO, Walker CA (1977) Birds of the British Lower Eocene. Tertiary Res Special Paper 3:1–52
Holdaway RN, Jones MD, Beavan Athfield NR (2002) Late Holocene extinction of the New Zealand owlet-nightjar Aegotheles novaezealandiae. J Roy Soc NZ 32(4):653–667
Houde PW (1988) Paleognathous birds from the early Tertiary of the Northern Hemisphere. Publ Nuttall Ornithology Club, Cambridge, Massachusetts, p 22
Houde P, Olson S (1992) A radiation of coly-like birds from the early Eocene of North America (Aves: Sandcoleiformes new order). In: Campbell KE (ed.) Papers in Avian Paleontology honoring Pierce Brodkorb. Sci Ser Nat Hist Mus Los Angeles City 36:137–160, 21 text-figs., 2 pls.
Jarvis ED, Mirarab S, Aberer AJ, Li B, Houde P, Li C, Ho SYW, Faircloth BC, Nabholz B, Howard JT, Suh A, Weber CC, da Fonseca RR, Li J, Zhang F, Li H, Zhou L, Narula N, Liu L, Ganapathy G, Boussau B, Bayzid MS, Zavidovych V, Subramanian S, Gabaldón T, Capella-Gutiérrez S, Huerta-Cepas J, Rekepalli B, Munch K, Schierup M, Lindow B, Warren WC, Ray D, Green RE, Bruford MW, Zhan X, Dixon A, Li S, Li N, Huang Y, Derryberry EP, Bertelsen MF, Sheldon FH, Brumfield RT, Mello CV, Lovell PV, Wirthlin M, Schneider MPC, Prosdocimi F, Samaniego JA, Velazquez AMV, Alfaro-Núñez A, Campos PF, Petersen B, Sicheritz-Ponten T, Pas A, Bailey T, Scofield P, Bunce M, Lambert DM, Zhou Q, Perelman P, Driskell AC, Shapiro B, Xiong Z, Zeng Y, Liu S, Li Z, Liu B, Wu K, Xiao J, Yinqi X, Zheng Q, Zhang Y, Yang H, Wang J, Smeds L, Rheindt FE, Braun M, Fjeldsa J, Orlando L, Barker FK, Jønsson KA, Johnson W, Koepfli K-P, O’Brien S, Haussler D, Ryder OA, Rahbek C, Willerslev E, Graves GR, Glenn TC, McCormack J, Burt D, Ellegren H, Alström P, Edwards SV, Stamatakis A, Mindell DP, Cracraft J, Braun EL, Warnow T, Jun W, Gilbert MTP, Zhang G (2014) Whole-genome analyses resolve early branches in the tree of life of modern birds. Science 346:1320–1331
Jones MEH, Tennyson AJD, Worthy JP, Evans SE, Worthy TH (2009) A sphenodontine (Rhynchocephalia) from the Miocene of New Zealand and palaeobiogeography of the tuatara (Sphenodon). Proc R Soc B 276:1385–1390
Joseph L, Toon A, Schirtzinger EE, Wright TF, Schodde R (2012) A revised nomenclature and classification for family-group taxa of parrots (Psittaciformes). Zootaxa 3205:26–40. https://doi.org/10.11646/zootaxa.3205.1.2
Ksepka DT, Stidham TA, Williamson TE (2017) Early Paleocene landbird supports rapid phylogenetic and morphological diversification of crown birds after the K-Pg mass extinction. Proc Natl Acad Sci USA 114(30):8047–8052. https://doi.org/10.1073/pnas.1700188114
Kuhl H, Frankl-Vilches C, Bakker A, Mayr G, Nikolaus G, Boerno ST, Klages S, Timmermann B, Gahr M (2021) An unbiased molecular approach using 3′-UTRs resolves the avian family-level tree of life. Mol Biol Evol 38(1):108–127. https://doi.org/10.1093/molbev/msaa191
Layard EL, Layard ELC (1881) Notes on the avifauna of New Caledonia and the New Hebrides. Ibis ser. IV, 5(17):132–139, pl. 5.
Linnaeus C (1758) Systema Naturae pe regni tria naturae, secundum classes, ordines, genera, species, cum characteribus, differentiss, synonymis, lacis. 10th edition. Laurentius Salvius, Stockholm, 824 pp.
Marshall BA (2011) A new species of Latia Gray, 1850 (Gastropoda: Pulmonata: Hygrophila: Chilinoidea: Latiidae) from Miocene Palaeo-lake Manuherikia, southern New Zealand, and biogeographic implications. Molluscan Res 31:47–52
Mather EK, Tennyson AJD, Scofield RP, De Pietri VL, Hand SJ, Archer M, Handley WD, Worthy TH (2019) Flightless rails (Aves; Rallidae) from the early Miocene St Bathans Fauna, Otago. New Zealand J Syst Palaeontol 17(5):423–449
May RM (1990) Taxonomy as destiny. Nature 347:129–130
Mayr G (2000) A new mousebird (Coliiformes: Coliidae) from the Oligocene of Germany. J Ornithol 141:85–92
Mayr G (2002) Osteological evidence for paraphyly of the avian order Caprimulgiformes (nightjars and allies). J Für Ornithol 143:82–97
Mayr G (2008) The Madagascan “Cuckoo-roller” (Aves: Leptosomidae) is not a roller — notes on the phylogenetic affinities and evolutionary history of a “living fossil.” Acta Ornithol 43:226–230
Mayr G (2010) Phylogenetic relationships of the paraphyletic ‘caprimulgiform’ birds (nightjars and allies). J Zool Syst Evol Res 48(2):126–137. https://doi.org/10.1111/j.1439-0469.2009.00552.x
Mayr G (2016) The world’s smallest owl, the earliest unambiguous charadriiform bird, and other avian remains from the early Eocene Nanjemoy Formation of Virginia (USA). Paläontol Z 90:747–763
Mayr G (2020) An updated review of the middle Eocene avifauna from the Geiseltal (Germany), with comments on the unusual taphonomy of some bird remains. Geobios 62:45–59
Mayr G, Peters DS (1998) The mousebirds (Aves: Coliiformes) from the Middle Eocene of Grube Messel (Hessen, Germany). Senckenb Lethaea 78(1/2):179–197
Millener PR (1988) Contributions to New Zealand’s Late Quaternary avifauna. I: Pachyplichas, a new genus of wren (Aves: Acanthisittidae), with two new species. J R Soc NZ 18(4):383–406
Millener PR, Worthy TH (1991) Contributions to New Zealand’s Late Quaternary avifauna II: Dendroscansor decurvirostris, a new genus and species of wren (Aves: Acanthisittidae). J R Soc NZ 21(2):179–200
Milne-Edwards A (1871) [1869–1871]) Recherches anatomiques et paléontologiques pour servir à l’historie des oiseaux fossiles de la France, vol 2. Victor Masson et fils, Paris
Mitchell KJ, Llamas B, Soubrier J, Rawlence NJ, Worthy TH, Wood J, Lee MSY, Cooper A (2014) Ancient DNA reveals elephant birds and kiwi are sister taxa and clarifies ratite bird evolution. Science 344:898–900. https://doi.org/10.1126/science.1251981
Mitchell KJ, Wood JR, Llamas B, McLenachan PA, Kardailsky O, Scofield RP, Worthy TH, Cooper A (2016) Ancient mitochondrial genomes clarify the evolutionary history of New Zealand’s enigmatic acanthisittid wrens. Mol Phylo Evol 102:295–304
Mortimer N, Campbell HJ, Tulloch AJ, King PR, Stagpoole VM, Wood RA, Rattenbury MS, Sutherland R, Adams CJ, Collot J, Seton M (2017) Zealandia: earth’s hidden continent. GSA Today 27:27–35. https://doi.org/10.1130/GSATG321A.1
Mourer-Chauviré C (1988) Le gisement du Bretou (Phosphorites du Quercy, Tarn-et-Garonne, France) et sa faune de vertébrés de l'Eocène supérieur. II Oiseaux. - Palaeontographica (A) 205:29–50, 5 text-figs, 8 tabs, 4 pls.
Norman G (2018) Bird stories. a history of the birds of New Zealand. Potton & Burton, Nelson, NZ
Oliver WRB (1955) New Zealand birds. AH & AW Reed, Wellington
Oswald JA, Terrill RS, Stucky BJ, LeFebvre MJ, Steadman DW, Guralnick RP, Allen JM (2021) Ancient DNA from the extinct Haitian cave-rail (Nesotrochis steganinos) suggests a biogeographic connection between the Caribbean and Old World. Biol Lett 17:20200760. https://doi.org/10.1098/rsbl.2020.0760
Peters JL (1940) Check-list of birds of the world, vol 4. Harvard University Press, Cambridge
Peters S (1999) New genus, new species, a sandicoleiform bird from the Oil Shale of Messel (Germany, Middle Eocene). Proceedings of the 4th International meeting of the Society of Avian Paleontology and Evolution. Washington, USA, June 1996. Smithsonian Contrib Paleobiol 89:217–222.
Phillips MJ, Gibb GC, Crimp EA, Penny D (2010) Tinamous and moa flock together: mitochondrial genome sequence analysis reveals independent losses of flight among ratites. Syst Biol 59:90–107. https://doi.org/10.1093/sysbio/syp079
Prum RO, Berv JS, Dornburg A, Field DJ, Townsend JP, Lemmon EM, Lemmon AR (2015) A comprehensive phylogeny of birds (Aves) using targeted next-generation DNA sequencing. Nature 526:569–573
Rando JC, López M, Seguí B (1999) A new species of extinct flightless passerine (Emberizidae: Emberiza) from the Canary Islands. Condor 101(1):1–13
Rando JC, Alcover JA, Illera JC (2010) Disentangling ancient interactions: a new extinct passerine provides insights on character displacement among extinct and extant island finches. PLoS One 5(9):e12956. https://doi.org/10.1371/journal.pone.0012956
Rich PV, McEvey AR (1977) A new owlet-nightjar from the early to mid-Miocene of Eastern New South Wales. Mem Nat Mus Victoria 38:247–252, pl. 8.
Rich PV, Haarhoff PJ (1985) Early Pliocene Coliidae (Aves, Coliiformes) from Langebaanweg, South Africa. Ostrich 56:20–41
Roelants K, Gower DJ, Wilkinson M, Loader SP, Biju SD, Guillaume K, Moriau L, Bossuyt F (2007) Global patterns of diversification in the history of modern amphibians. Proc Natl Acad Sci USA 104:887–892
Sangster G (2005) A name for the clade formed by owlet-nightjars, swifts and hummingbirds (Aves). Zootaxa 799:1–6
Scarlett RJ (1968) An owlet-nightjar from New Zealand. Notornis 15(4):254–266
Schwarzhans W, Scofield RP, Tennyson AJD, Worthy JP, Worthy TH (2012) Fish remains, mostly otoliths, from the non-marine Early Miocene of Otago. New Zealand Acta Palaeontol Pol 57(2):319–350
Schweizer M, Seehausen O, Güntert M, Hertwig ST (2010) The evolutionary diversification of parrots supports a taxon pulse model with multiple trans-oceanic dispersal events and local radiations. Mol Phylo Evol 54:984–994
Sun Y, He X, Glenny D (2014) Transantarctic disjunctions in Schistochilaceae (Marchantiophyta) explained by early extinction events, post-Gondwanan radiations and palaeoclimatic changes. Mol Phylo Evol 76:89–201
Thomsen T (2021) The lonely islands. The evolutionary phenomenon that is New Zealand. New Holland Publishers, Auckland
Tobias JA, Ekstrom MM (2002) The New Caledonian Owlet-nightjar Aegotheles savesi rediscovered? Bull Br Ornithol Club 122(4):282–285
Toon A, Pérez-Losada M, Schweitzer CE, Feldmann RM, Carlson M, Crandall KA (2010) Gondwanan radiation of the Southern Hemisphere crayfishes Decapoda: Parastacidae): evidence from fossils and molecules. J Biogeog 37:2275–2290
Trewick SA, Paterson AM, Campbell HJ (2007) Hello New Zealand. J Biogeog 34:1–6. https://doi.org/10.1111/j.1365-2699.2006.01643.x
Vigors NA, Horsfield T (1827) A description of the Australian birds in the collection of the Linnean Society; with an attempt at arranging them according to their natural affinities. Trans Linn Soc Lond 15:170–331
Wallis GP, Trewick SA (2009) New Zealand phylogeography, evolution on a small continent. Mol Ecol 18:3548–3580. https://doi.org/10.1111/j.1365-294X.2009.04294.x
Wesenera T, VandenSpiegel D (2009) A first phylogenetic analysis of giant pill-millipedes (Diplopoda, Sphaerotheriida), a new model Gondwanan taxon, with special emphasis on island gigantism. Cladistics 25:545–573
Worthy TH, Holdaway RN (2002) The lost world of the moa: Prehistoric life of New Zealand. Indiana University Press, Indiana
Worthy TH, Nguyen JMT (2020) An annotated checklist of the fossil birds of Australia. Trans R Soc South Aust 144(1):66–108. https://doi.org/10.1080/03721426.2020.1756560
Worthy TH, Tennyson AJD, Jones C, McNamara JA, Douglas BJ (2007) Miocene waterfowl and other birds from Central Otago, New Zealand. J Syst Palaeontol 5:1–39
Worthy TH, Tennyson AJD, Scofield RP, Hand SJ (2013a) Early Miocene fossil frogs (Anura: Leiopelmatidae) from New Zealand. J Roy Soc NZ 43(4):211–230. https://doi.org/10.1080/03036758.2013.825300
Worthy TH, De Pietri VL, Scofield RP (2017) Recent advances in avian palaeobiology in New Zealand with implications for understanding New Zealand’s geological, climatic and evolutionary histories. NZ J Zool 43(3):177–211
Worthy TH, Hand SJ, Archer M, Scofield RP, De Pietri VL (2019) Evidence for a giant parrot from the Early Miocene of New Zealand. Biol Lett 15(8):20190467
Worthy TH, Scofield RP, Salisbury SW, Hand SJ, De Pietri VL, Blokland JC, Archer M (2022) A new species of Manuherikia (Aves: Anatidae) provides evidence of faunal turnover in the St Bathans Fauna New Zealand. Geobios 70:87–107. https://doi.org/10.1016/j.geobios.2021.08.002
Worthy TH, Worthy JP, Tennyson AJD, Salisbury SW, Hand SJ, Scofield RP (2013b) Miocene fossils show that kiwi (Apteryx, Apterygidae) are probably not phyletic dwarves. In: Göhlich UB & Kroh A (eds) Paleornithological Research 2013b – Proceedings of the 8th International Meeting of the Society of Avian Paleontology and Evolution; Vienna, Natural History Museum Vienna; pp 63–80.
Yonezawa T, Segawa T, Mori H, Campos PF, Hongoh Y, Endo H, Akiyoshi A, Kohno N, Nishida S, Wu J (2017) Phylogenomics and morphology of extinct palaeognaths reveal the origin and evolution of the ratites. Curr Biol 27:68–77. https://doi.org/10.1016/j.cub.2016.10.029)
Yuri T, Kimball RT, Harshman J, Bowie RC, Braun MJ, Chojnowski JL, Han K-L, Hackett SJ, Huddleston CJ, Moore WS, Reddy S, Sheldon FH, Steadman DW, Witt CC, Braun EL (2013) Parsimony and model-based analyses of indels in avian nuclear genes reveal congruent and incongruent phylogenetic signals. Biology 2:419–444
Zelenkov NV, Dyke GJ (2008) The fossil record and evolution of mousebirds (Aves: Coliiformes). Palaeontology 51(6):1403–1418. https://doi.org/10.1111/j.1475-4983.2008.00814.x
Acknowledgements
We thank Euan and Ann Johnstone of Home Hills Station and Tony and Jack Enright of Dunstanburn Station, Southern Lakes Holdings Ltd for access to their land to investigate these fossil deposits. We are grateful to many people who helped with the excavations of these fossils over the years, particularly thank Alan Tennyson (NMNZ). We specifically thank the several members of The Waterhouse Club, South Australia, who gave their time in 2008. For providing access to collections and loaning comparative material, we thank Collections Managers, Maya Penck (SAM), Leah Tsang (AM), and Christopher Milensky, (USNM). We thank Mark Robbins and Kristof Zyskowski (YPM) for images of A. insignis and A. albertisi.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. This work was largely supported by the Australian Research Council Discovery Projects DP0770660 (to Suzanne Hand, Michael Archer) and DP120100486 (to Trevor Worthy, Suzanne Hand, Steven Salisbury, Richard Paul Scofield, and Alan Tennyson) and by the New Zealand Marsden Fund Council from Government funding, managed by the Royal Society Te Apārangi (CTM1601, Vanesa De Pietri, Richard Paul Scofield). In addition, Canterbury Museum, Flinders University, The Museum of New Zealand Te Papa Tongarewa, the University of New South Wales, The University of Queensland, and multiple individual volunteers contributed financial and in-kind support to the fieldwork leading to this research spread over several field seasons.
Author information
Authors and Affiliations
Contributions
Trevor Worthy conceived the project, made initial identifications and wrote the manuscript. Richard Paul Scofield and Vanesa De Pietri, jointly with Trevor Worthy, made detailed comparisons and collected comparative data. Trevor Worthy and Richard Paul Scofield made photographic images and Richard Paul Scofield created images from digital surface scan data. All authors partook in fieldwork, edited and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Communicated by F. Bairlein.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Worthy, T.H., Scofield, R.P., Salisbury, S.W. et al. Two new neoavian taxa with contrasting palaeobiogeographical implications from the early Miocene St Bathans Fauna, New Zealand. J Ornithol 163, 643–658 (2022). https://doi.org/10.1007/s10336-022-01981-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10336-022-01981-6