Abstract
The overwhelming majority of avian extinctions have occurred on islands, where introduced predators, habitat loss, disease, and human persecution have resulted in the loss of over 160 species in the last 500 years. Understanding the timing and causes of these historical extinctions can be beneficial to identifying and preventing contemporary biodiversity loss, as well as understanding the nature of island ecosystems. Tristan da Cunha (henceforth “Tristan”), the most remote inhabited island in the world, has lost three species from the main island since permanent human settlement in 1811—the Tristan Moorhen (Gallinula nesiotis), Inaccessible Finch (Nesospiza acunhae acunhae), and Tristan Albatross (Diomedea dabbenena). We used recently developed Bayesian methods, and sightings of mixed certainty compiled from historical documents, to estimate the extinction date of these three species from Tristan based on specimens. We estimate that all three species were likely extirpated from Tristan between 1869 and 1880 following a period of significant habitat alteration and human overexploitation, and only the albatross had a high probability of persistence when Black Rats (Rattus rattus) arrived in 1882, the previously assumed cause of extinction for all three species. Better estimates of extinction dates are essential for understanding the causes of historical biodiversity loss, and the combination of historical ecology with modern statistical methods has given us novel insights into the timing and therefore the causes of extinctions on one of the most isolated islands in the world.
Zusammenfassung
Aussterben von Vogelarten auf der abgelegensten bewohnten Insel der Erde
Die mit Abstand größte Anzahl ausgestorbener Vogelarten gab es auf Inseln, nachdem Räuber eingeschleppt und Lebensraum vernichtet wurde, sowie durch Krankheiten und Verfolgung durch den Menschen. All dies hat zu einem Verlust von mehr als 600 Vogelarten in den vergangenen 500 Jahren geführt. Das Verständnis von zeitlichen und ursächlichen Zusammenhängen dieses historischen Artenverlusts kann zum einen helfen, die ökologischen Besonderheiten von Inseln zu verstehen und zum anderen, akute Bedrohungen der Artenvielfalt zu erkennen und zu vermeiden. Tristan da Cunha ist die entlegenste bewohnte Insel der Erde; seit der dauerhaften Besiedlung durch Menschen (1811) sind auf der Hauptinsel drei Arten ausgestorben: die Tristan-Inselralle (Gallinula nesiotis), der Tristan-Ammerfink (Nesospiza acunhae acunhae) und der Tristan-Albatros (Diomedea dabbenena). Für die nicht immer ganz eindeutigen Sichtungen einzelner Tiere, historischen Dokumenten entnommen, wandten wir kürzlich entwickelte Bayessche Formeln an, um die Zeit des Aussterbens dieser drei Arten zu datieren; wir gehen davon aus, dass auf Tristan alle drei Arten zwischen 1869 und 1880 ausstarben. Dies geschah wohl nach einer Phase starker Besiedlungsaktivitäten und Raubbau durch Menschen, und nur der Albatros hatte noch eine gute Chance, als 1882 die Hausratte (Rattus rattus) eingeschleppt wurde, die bislang allgemein als die Ursache für das Aussterben der drei Arten angesehen wurde. Für die Erforschung der Ursachen früherer Rückgänge von Artenvielfalt sind genauere Aussterbe-Datierungen essentiell, wobei uns die Kombination von historischer Ökologie mit modernen statistischen Methoden neue Ansätze für die Datierung und damit für die Ursachenforschung vom Artensterben auf einer der abgelegensten Inseln der Erde bietet.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In the last 500 years, > 90% of avian extinctions have been of island species (Johnson and Stattersfield 1990; Loehle and Eschenbach 2012), driven by introduced predators, habitat loss, and human persecution (Steadman 2006; Duncan and Blackburn 2007; Hume and Walters 2012). Globally, rails (Gruiformes: Rallidae) and seabirds have been disproportionately affected by island extinctions and extirpations, particularly during the prehistoric settlement of Pacific islands (Steadman 1995). While the factors driving extinctions have changed over time and vary regionally, the phylogenetic and ecological predictors of historical island extinctions tend to be similar to those of modern extinctions (Boyer 2010), providing a clear rationale for historical ecology research focused on island bird extinctions.
Reconstructing even the most basic details of extinction can be difficult, however, because the final living individual is seldom known with certainty, and the statistical challenge of imperfect detection can be especially limiting on small, often remote, islands with small human populations. Despite these challenges, using statistical methods to estimate species extinction dates can provide insight into the historical ecology of ecosystems, and when combined with other data sources, such as the arrival of invasive species, or changes in habitat availability or land use, may allow us to infer the potential causes for a species’ disappearance. While researchers have focused on high-profile extinctions like that of the Dodo (Raphus cucullatus) (Roberts and Solow 2003) or for well-sampled regions like North America and Hawaii (Elphick et al. 2010), comparatively less work has been done investigating poorly documented extinctions on islands.
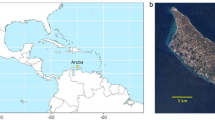
Among inhabited islands, the UK Overseas Territory of Tristan da Cunha is the most remote in the world, and comprises four main islands in the central South Atlantic Ocean [Tristan da Cunha (henceforth “Tristan”), Inaccessible, Nightingale, and Gough islands, and their associated islets; Fig. 1]. The islands were first documented in 1506 (d’Albuquerque 1875), and throughout the following centuries were visited sporadically by sealing vessels, and ships seeking supplies (Brander 1940). The main island of Tristan has been inhabited almost continuously since 1811 (Lambert 1811), and is now home to approximately 260 residents. Introduced species, mainly livestock, were brought to the island in the eighteenth century, and by the early nineteenth century feral cats (Felis catus), were present (Wace 1969; Wace and Holdgate 1976); House Mice (Mus musculus) arrived in the early nineteenth century, though the date is unknown (Wace and Holdgate 1976) and Black Rats (Rattus rattus) arrived during a shipwreck in 1882 (Barrow 1910). As the fortunes of the island residents changed through the nineteenth century, so, too, did their need to increase the number of livestock, and clear land for agriculture (Wace 1969; Wace and Holdgate 1976). This resulted in significant habitat changes, particularly the reduction in the island group’s sole woody tree Phylica arborea on the settlement plain along the north-western periphery of the island (Brander 1940; Hagen 1952; Milton et al. 1993). The island group currently has a diverse and unique avifauna, comprising > 30 breeding seabird species, and six land birds endemic to the group (Ryan 2007).
The history of introduced species and exploitation by early inhabitants, however, has resulted in the extinction of one species and the extirpation of at least two others from the main island of Tristan. The Tristan Moorhen (Gallinula nesiotis; Fig. 2), a strict Tristan endemic, is now entirely extinct (Beintema 1972; Groenenberg et al. 2008). A subspecies of the Inaccessible Finch (Nesospiza acunhae subsp. acunhae; Fig. 3) was also formerly present on the main island, from which the type specimen was collected (Cabanis 1873; Ryan 2008), but it is now entirely restricted to Inaccessible Island. Tristan Albatrosses (Diomedea dabbenena; Fig. 4) formerly bred on Tristan, but are now restricted to Gough Island, with another two to three breeding pairs on Inaccessible Island (Davies et al. 2015; McClelland et al. 2016). The Inaccessible Finch was last seen on Tristan prior to the Challenger expedition of 1873, the first major scientific expedition to the island (von Willemöes-Suhm 1876). The other two species likely disappeared from the main island long before the Norwegian scientific expedition of 1937 (Nicoll 1906; Barrow 1910; Wilkins 1923; Hagen 1952). Since the extinction of these three birds is so poorly understood, we used historical records and museum specimens to estimate the extinction dates of each species from Tristan da Cunha, to help place these losses in the context of the changing island ecosystems of the nineteenth and early twentieth centuries.

Image A from the Biodiversity Heritage Library. Digitized by the Natural History Museum. http://www.biodiversitylibrary.org. Images in b Trustees of The Natural History Museum, Tring. Used with permission
The Tristan Moorhen likely went extinct in 1874. First described by Sclater (1861) (a), now known from two specimens [b; Natural History Museum, Tring, UK (NHMUK) 1861.9.16.1 (top), NHMUK 1864.7.30.1 (bottom)].

Image A from the Biodiversity Heritage Library. Digitized by the Smithsonian Libraries. http://www.biodiversitylibrary.org. Image in b Hwa Ja-Götz, Museum für Naturkunde, Berlin. Used with permission
The Tristan Albatross likely went extinct on the main island of Tristan by 1880, in part owing to human persecution, as depicted in a (Earle 1824; used with permission), though it remains extant on Inaccessible and Gough Islands (b, c).
Methods
We compiled sighting and specimen data for the three species from historical documents, including reports of visitors to the island, ship logs, museum records, and extant museum specimens. Each record was initially assigned to one of three categories: presence, absence, or uncertain. Uncertain records were those that could either not be linked directly to the main island of Tristan (i.e., they could have been referring to Inaccessible, Nightingale or Gough islands), or if detailed ornithological notes were made on two of the three species, but not the third. We follow the template of other studies that use sighting date records with mixed uncertainty (Solow and Beet 2014; Carlson et al. 2018a), and divided presence records into three tiers: confirmed and verifiable (e.g., a museum specimen); confirmed, but unverifiable (e.g., an expert sighting); or unconfirmed, but plausible (e.g., second-hand records, or records of questionable identification).
A variety of methods have been developed to infer extinction dates (TE) from sighting record data, which make use of varying assumptions and data requirements (Boakes et al. 2015) and have correspondingly variable performance (Rivadeneira et al. 2009). The majority of methods use a time series of the last k sightings of a species, where sampling begins in time t0 and sightings occur at times (t0, …, tn). Most extinction dates are estimated by assuming that sightings occur as a random process driven by a given distribution over time, which ends once the species is extinct. Among these estimators, the most popular and widely used (Clements et al. 2013) is the optimal linear estimator (OLE), a non-parametric method that treats the last few sightings of a species as following a Weibull distribution. The OLE approach has proven to be an accurate and reliable estimator of extinction dates, and is commonly used to study other major extinctions, including those of other island avifauna (Roberts and Solow 2003; Collen et al. 2010).
One downside of the OLE method, however, is that it relies on a dataset of only “valid” sightings; in fact, most non-parametric extinction date estimators are sensitive to the inclusion of “invalid” sightings (Roberts et al. 2010). Consequently, a handful of more complex Bayesian methods have been developed to address variable sighting quality. The simplest approach assumes that uncertain sightings only arise after the last certain sighting (Solow et al. 2012), but a handful of models have been developed that refine and elaborate this approach (Boakes et al. 2015). Here, we use a model developed by Solow and Beet (2014), the second they propose, which assumes that the processes generating valid and invalid sightings are generated by two independent, non-homogenous Poisson processes. All certain sightings are assumed valid, while uncertain sightings might be valid or invalid (the true status of any given sighting is unknown, and the model makes no assumptions). With E being the event that a species is extinct by a given year (and \(\bar{E}\) the converse case that the species persists), and a sighting dataset t, Bayes’ theorem provides a basic expression for the probability of persistence:
The prior probability of extinction \(P\left( E \right)\) is subjective, and can potentially be used as a way to simply produce any given desired outcome. Consequently, we can set an uninformative prior where extinction and persistence are equally likely:
and the resulting posterior probability compares the likelihood of the data being generated by the two explanations:
To calculate those likelihoods, we define \(E\) as the event of extinction at a specific time \(T_{\text{E}} \le T\) and define \(\bar{E}\) as \(T_{\text{E}} > T\) for a given period of observation ending in time T. The likelihood of the data is expressed as a product of the conditional likelihoods of the certain and uncertain sighting sub-datasets:
where \(n_{\text{c}}\) and \(n_{\text{u}}\) are the number of certain and uncertain sightings, \(n_{\text{u}} ( {\hat{T}_{\text{E}} } )\) is the number of uncertain sightings before the extinction date, and \(\omega\) is a stand-in for the proportion of valid sightings. We reported the extinction dates \(\hat{T}_{\text{E}}\) with the highest posterior likelihood, based on a uniform prior on \(\hat{T}_{\text{E}}\) (which makes no assumptions about how far after the last certain sighting persistence continues to be plausible). We also report the probability of persistence of each species in 1882 under two scenarios: one where all expert-validated sightings are treated equally (all certain), and one where specimen and observation data are separated (all observations are uncertain).
However, posterior likelihoods derived from the Solow and Beet model can be misleading, due to a basic principle of sighting data: it is always more likely that extinction occurred before an interval with no valid sightings (the data have a conditional likelihood of 1) than that the species persisted but no valid sightings were generated by even the smallest rate process (conditional likelihood lower than 1). Because of this, in the absence of invalid sightings (and even with some), posteriors from the model usually peak immediately after the last certain sighting (in the absence of uncertainty, this is a given). Moreover, the posterior probability of persistence can still be high even in the year \(\hat{T}_{\text{E}}\) that maximizes \(P\left( {t|E} \right)\), as long as \(P\left( {t|\bar{E}} \right)\) is also high. Given our data limitations and this aspect of the model, we also use Bayes factors to test our certainty about our selected test year 1882:
This Bayes factor expresses our relative certainty that extinction happened by a given point, and can be comparatively low even when the posterior probability of persistence is high.
Models were implemented using the R package spatExintct (Carlson et al. 2018b), using integration increments of 0.001. Models that account for other aspects, such as detection probability and survey effort (Thompson et al. 2017) were not considered here because of our desire to include uncertain records, and the lack of survey effort data with which to parameterize the model.
Results
The Tristan Moorhen was observed from 1790 (Purdy 1816) through to the Challenger expedition in 1873 (von Willemöes-Suhm 1876), with records summarized by Beintema (1972) (Table 1), though throughout the twentieth century there was much debate as to whether it constituted a separate species from the Gough Moorhen (Gallinula comeri), which was introduced to Tristan in 1956 (Allen 1892; Wace and Holdgate 1976; Richardson 1984; Groenenberg et al. 2008). Only two museum specimens exist, both at The Natural History Museum, Tring, UK (NHMUK 1861.9.16.1, the type specimen, and NHMUK 1864.7.30.1; Fig. 2) (Sclater 1861; Groenenberg et al. 2008). The extinction likelihood for the Bayesian model peaked in 1874, and declined rapidly (Fig. 5a), with a 36–38% probability of persistence in 1882 (Bayes factors: 1.63–1.76; Fig. 6a).
The likelihood of Tristan Moorhen (a), Inaccessible Finch (b), and Tristan Albatross (c) extinction over time. The figure presents the posterior probability of a given extinction date \(\tau_{\text{E}}\) scaled by the area under the entire likelihood curve. The model treats specimen-based records separately and as certain observations; consequently, evaluation begins in 1873 (a), 1816 (b), and 1868 (c), the year of the last certain sighting (i.e., extinction prior to that date is not considered)
Records were the sparsest for the Inaccessible Finch on Tristan, likely owing to its small size (Table 2), and the fact that it was not used as food. Dupetit Thours (1811) first recorded the finch in 1793, and it was last definitively seen in 1852 (MacGillivray 1852), though it would not be formally described until 1873, based on a specimen collected in 1816 (Carmichael 1819; Cabanis 1873) now in the Museum für Naturkunde, Berlin (ZMB 7316; Fig. 3). The extinction likelihood peaked in 1869 (Fig. 5b), and by 1882 the probability of persistence was 28–35% (Bayes factors: 1.88–2.53; Fig. 6b).
The earliest reliable record for Tristan Albatross was from 1793 (Dupetit Thours 1811), and the last confirmed record was in 1880 (Wace and Holdgate 1976) (Table 3). Albatross were regularly recorded through the nineteenth century, as they provided a food source for residents (Earle 1832). Our analysis estimated that 1880 was the most likely \(\hat{T}_{\text{E}}\)—(Fig. 5c), but that the Tristan Albatross had a high chance of persistence when rats arrived in 1882 (p = 0.77–1.00; Bayes factors: 0.00–0.29; Fig. 6c).
Discussion
Our estimates place the likely extinction of all three extirpated species from Tristan between 1869 and 1880, with only the albatross having a high probability of persistence in 1882. In their landmark monograph, Wace and Holdgate (1976) attributed the extinction of the finch and moorhen to the presence of feral cats and Black Rats based on those species’ implication in species declines and extirpations elsewhere, though rats arrived in 1882 with the wreck of the American schooner Henry B. Paul. This date is considered fairly reliable, as shortly thereafter rats were identified as agricultural pests on the island (Wace and Holdgate 1976). Feral cats and House Mice were present on Tristan from the early nineteenth century, if not earlier (Wace 1967), and have devastating effects on island birds (van Aarde 1980; Angel et al. 2009; Medina et al. 2011).
Our results suggest the Tristan Moorhen likely went extinct in the 1870s, which is ca. 30 years prior to Beintema’s (1972) estimate of 1906, the year the Valhalla, Royal Yacht Squadron, visited Tristan but did not record the species (Nicoll 1906). Beintema (1972) assumed the arrival of rats in 1882 was the ultimate cause of the moorhen’s extinction; however, since our results suggest that they were likely gone by the time rats arrived, introduced cats and human persecution likely drove the Tristan Moorhen to extinction. Sperling (1872) reported severe predation of moorhens by cats, and given the aggressive nature of Gough Moorhen on Tristan towards domestic poultry, they may have also been perceived as a pest species and hunted (Richardson 1984).
In the decades leading up to the 1860s and 1870s, Tristan served as a trading post for visiting ships, and as a result people cleared large swaths of land and stocked the settlement plain with large amounts of livestock (Wace and Holdgate 1976). The habitat alterations likely strongly affected the Inaccessible Finch, which relied on tussock grass (Spartina arundinacea) or Phylica arborea (the only woody plant in the island group) for breeding and feeding habitat, as it does on Inaccessible Island (Fraser and Briggs 1992; Ryan and Moloney 2002). By 1842, for example, more than 60 ha of the settlement plain, comprising 10% of the its area, had been cleared for livestock grazing and agriculture (Brierly 1842). While some P. arborea habitat would have persisted (and persists today) on the cliff edges, it is much reduced and highly fragmented (Holdgate 1965). The Inaccessible Finch was most likely extinct by the late 1860s or early 1870s, and was probably the least numerous of the three extirpated species considered here. Our analyses support the conclusion that the Inaccessible Finch was extinct prior to the Challenger expedition in 1873 (Fisher et al. 1969), though the search effort during that expedition was low compared to the potential available habitat, which would have influenced the probability that the finch would have been detected if it had still been extant, and therefore its estimated extinction date (Thompson et al. 2017). Although the ultimate cause of this extinction remains unknown, it was probably driven by several concurrent pressures. The finch relied heavily on tussock grass and P. arborea, rapidly dwindling resources during that time, which suggests that habitat loss played an important role in its extinction. The decline of these plants may also have been exacerbated by trampling by livestock (Gangoso et al. 2006). Predation by cats (Medina et al. 2011, 2014) may also have played a role in the Inaccessible Finch’s extinction.
Tristan Albatross, though still extant on Gough and with a small relict population on Inaccessible Island, were likely extirpated from Tristan in the 1880s, which is broadly similar to some previous estimates (Wace and Holdgate 1976), though much earlier than others (Barrow 1910). The high probability of persistence in 1882 could also be the result of the low number of verifiable early sightings or specimens. There are also some unconfirmed records of birds in the 1950s and 1960s, though no photographs, or specimens (Ryan et al. 2001; De Roy et al. 2008). While they can also be affected negatively by introduced rodents, including mice (Cuthbert and Hilton 2004; Wanless et al. 2009; Davies et al. 2015), there is no evidence for mouse predation of albatrosses on Tristan, and the rat population that arrived in 1882 would have likely been too small to affect the population significantly, but equally may have been the final blow for a population already destined for extinction. On Gough Island, albatrosses were apparently unaffected by mice for many years (Swales 1965; Wace and Holdgate 1976). Cats, however, would have likely had a significant impact, as might have dogs (Canis familiaris), through both predation and disturbance (Wace and Holdgate 1976).
On Inaccessible Island, Tristan Albatross numbers declined markedly from around 200 breeding pairs in the 1870s to only two or three pairs by 1937, but have remained stable since (Hagen 1952; Stoltenhoff 1952; McClelland et al. 2016). This pattern is likely a result of predation and disturbance by introduced pigs (Sus scrofa), which were eradicated in the 1930s (Fraser et al. 1988). Large numbers of pigs and cattle (Bos taurus) were brought to Tristan, particularly in the 1850s–1880s (Wace and Holdgate 1976), but by then, Tristan Albatross were likely restricted to the base, an area of the island’s volcano starting at 500–700 m above sea level, where livestock were largely absent at the time (Earle 1832).
A likely significant contributor to the albatross’s extirpation from Tristan was overexploitation. Albatross adults, chicks, and eggs were regularly consumed by the island’s human population, and provided as provisions to passing ships (MacGillivray 1852; Barrow 1910; Hagen 1952; Fraser et al. 1988). Indeed, Earle (1832) depicted such a harvest in his account of time spent on the island (Fig. 4). That Tristan Albatross eggs were still regularly harvested when MacGillivray visited the island in 1852, a mere 30 years before their estimated extirpation, suggests a fairly high hunting pressure, as juvenile birds would have survived at sea, and continued to return to the island to attempt breeding for > 10 years after the last chick fledged, owing to the species’ lengthy juvenile pre-breeding stage (Ryan et al. 2001; Cuthbert et al. 2004; Wanless et al. 2009), as was the case with Short-tailed Albatross (Phoebastria albatrus) on Torishima, Japan, which were absent for several years until juveniles returned to re-establish the breeding colony (Deguchi et al. 2017). The decline in visiting ships in the 1870s and 1880s may have increased the importance of albatross as a subsistence food for the island population as trade decreased (Wace and Holdgate 1976).
That the three species were extirpated in close succession is notable, and is likely due in part to population growth and the fortunes of island residents. The period from 1870 to 1880 saw the peak human population (just over 100 people) until 1920, and was coincident with the great abundance and rapid rise in livestock, driven by Tristan’s diminishing role as a resupply station for ships sailing the South Atlantic Ocean (Wace and Holdgate 1976). While the arrival of Black Rats was previously suggested as the ultimate cause of extinction of the Inaccessible Finch and Tristan Moorhen in particular (e.g., Holdgate 1965), two of the three local extinctions had the greatest likelihood of occurring before the arrival of Black Rats in 1882, with the albatross likely still extant in that year. By the 1870s, the populations of all three species would have been very small, and after a long history of exploitation, habitat loss, and the impacts of other introduced mammals, it seems that they may have already disappeared from Tristan by the time rats arrived. Even if populations persisted beyond 1882, the cumulative impact of other pressures had likely reduced them to small relict populations prone to rapid extinction from the newly arrived threat of invasive rodents.
Combining historical ecology with modern statistical analysis has given us novel insights into the likely factors resulting in the extinction or extirpation of island species in one of the most remote corners of the world. Understanding the likely causes of extinctions relies on robust estimation of extinction dates and an understanding of the pressures on a population in the period leading up to extinction. The statistical advances in extinction date estimation now allow us to move beyond simple conjecture, especially for data-sparse island species, meaning we can now more accurately infer the causes of extinction and better understand the historical threats to island species. A deeper understanding of the causes of these extinctions may then lead to better and more informed conservation strategies for currently threatened island species.
References
Allen JA (1892) Description of a new gallinule, from Gough Island. Bull Am Mus Nat Hist 4:57–58
Angel A, Wanless RM, Cooper J (2009) Review of impacts of the introduced House Mouse on islands in the Southern Ocean: are mice equivalent to rats? Biol Invasions 11:1743–1754
Barrow KM (1910) Three years in Tristan da Cunha. Skeffington & Son, London
Beintema AJ (1972) The history of the Island Hen (Gallinula nesiotis), the extinct flightless gallinule of Tristan da Cunha. Bull Br Ornithol Club 92:106–113
Boakes EH, Rout TM, Collen B (2015) Inferring species extinction: the use of sighting records. Methods Ecol Evol 6:678–687
Bourne WRP, David ACF (1981) Nineteenth century bird records from Tristan da Cunha. Bull Br Ornithol Club 101:247–256
Boyer AG (2010) Consistent ecological selectivity through time in Pacific island avian extinctions. Conserv Biol 24:511–519
Brander J (1940) Tristan da Cunha, 1506–1902. Allen and Unwin, London
Brierly OW (1842) Diary kept while aboard R. Y. S. WANDERER (Capt. Bushby, R. N.), voyaging from Plymouth to Australia. (Entries in fair and rough logs from March 18–21st, 1842). MS in Mitchell Library, Sydney (ML A528)
Broekhuysen GJ, Macnae W (1949) Observations on the birds of Tristan da Cunha Islands and Gough Island in February and early March, 1948. Ardea 37:97–113
Brooke RK (1975) Layard’s extralimital records in his Birds of South Africa and in the South African Museum. Bull Br Ornithol Club 96:75–80
Cabanis JL (1873) Ueber zwei neue Finkenarten des Berliner Museums: Crithagra insularis and Nesospiza acunhae von Tristan d’Acunha. J Ornithol 21:152–155
Carlson CJ, Bond AL, Burgio KR (2018a) Estimating the extinction date of the Thylacine with mixed certainty data. Conserv Biol 32:477–483
Carlson CJ, Burgio KR, Dallas TA, Bond AL (2018b) Spatial extinction date estimation: a novel method for reconstructing spatiotemporal patterns of extinction and identifying potential zones of rediscovery. bioRxiv:279679
Carmichael D (1819) Some accounts of the island of Tristan da Cunha and of its natural production. Trans Linn Soc Lond 12:483–513
Clements CF, Worsfold NT, Warren PH, Collen B, Clark N, Blackburn TM, Petchey OL (2013) Experimentally testing the accuracy of an extinction estimator: Solow’s optimal linear estimation model. J Anim Ecol 82:345–354
Collen B, Purvis A, Mace GM (2010) When is a species really extinct? Testing extinction inference from a sighting record to inform conservation assessment. Divers Distrib 16:755–764
Cuthbert RJ, Hilton GM (2004) Introduced House Mice Mus musculus: a significant predator of threatened and endemic birds on Gough Island, South Atlantic Ocean? Biol Conserv 117:483–489
Cuthbert RJ, Sommer E, Ryan P, Cooper J, Hilton G (2004) Demography and conservation of the Tristan Albatross Diomedea [exulans] dabbenena. Biol Conserv 117:471–481
d’Albuquerque A (1875) The commentaries of Alfonso d’Alberquerque, second Viceroy of India. Translated from the Portuguese edition of 1774, with notes and an introduction by Walter de Gray Birch. Hakluyt Society, London
Davies D, Dilley BJ, Bond AL, Cuthbert RJ, Ryan PG (2015) Trends and tactics of mouse predation on Tristan Albatross Diomedea dabbenena chicks at Gough Island, South Atlantic Ocean. Avian Conserv Ecol 10:5
De Roy T, Fitter J, Jones M (2008) Albatross: their world, their ways. Helm, London
Deguchi T, Sato F, Eda M, Izumi H, Suzuki H, Suryan RM, Lance EW, Hasegawa H, Ozaki K (2017) Translocation and hand-rearing result in Short-tailed Albatrosses returning to breed in the Ogasawara Islands 80 years after extirpation. Anim Conserv 20:341–349
Duncan RP, Blackburn TM (2007) Causes of extinction in island birds. Anim Conserv 10:149–150
Dupetit Thours A (1811) Melanges de botaniques et des voyages. 5. Description de l’ile de Tristan d’Acugna, et esquisse de sa flore. Arthus Bertrand, Paris
Earle A (1824) Summit of Tristan de Acunha: a man killing albatross [watercolour]. PIC Solander Box A30 #T54 NK12/15. National Library of Australia, Canberra
Earle A (1832) A narrative of a nine months’ residence in New Zealand in 1827; together with a journal of residence in Tristan d’Acunha, an island situated between South America and the Cape of Good Hope. Longman, Rees, Orme, Brown, Green, and Longman, London
Elliott HFI (1953) The fauna of Tristan da Cunha. Oryx 2:41–56
Elliott HFI (1957) A contribution to the ornithology of the Tristan da Cunha group. Ibis 99:545–586
Elphick CS, Roberts DL, Michael Reed J (2010) Estimated dates of recent extinctions for North American and Hawaiian birds. Biol Conserv 143:617–624
Faustini A (n.d.) The annals of Tristan da Cunha
Fisher J, Simon N, Vincent J (1969) The red book: wildlife in danger. Collins, London
Fraser MW, Briggs DJ (1992) New information on the Nesospiza buntings at Inaccessible Island, Tristan da Cunha, and notes on their conservation. Bull Br Ornithol Club 112:191–205
Fraser MW, Ryan PG, Watkins BP (1988) The seabirds of Inaccessible Island, South Atlantic Ocean. Cormorant 16:7–33
Gangoso L, Donázar JA, Scholz S, Palacios CJ, Hiraldo F (2006) Contradiction in conservation of island ecosystems: plants, introduced herbivores and avian scavengers in the Canary Islands. Biodivers Conserv 15:2231
Groenenberg DSJ, Beintema AJ, Dekker RWRJ, Gittenberger E (2008) Ancient DNA elucidates the controversy about the flightless island hens (Gallinula sp.) of Tristan da Cunha. PLoS ONE 3:e1835
Hagen Y (1952) Birds of Tristan da Cunha. Results Nor Sci Exped Tristan da Cunha 20:1–248
Holdgate MW (1965) The fauna of the Tristan da Cunha islands. Philos Trans R Soc Lond B Biol Sci 249:361–424
Hume JP, Walters M (2012) Extinct birds. Poyser, London
Johnson TH, Stattersfield AJ (1990) A global review of island endemic birds. Ibis 132:167–180
Lambert J (1811) Letter to William Lockerby. In: Thurn EI, Wharton LC (eds) The journal of William Lockerby, sandalwood trader in the Fijian Islands during the years 1808–1809, with an introduction and other papers connected with the earliest European visitors in the islands. Hakluyt Society, London, pp 219–224
Layard EL (1869a) Further notes on South-African ornithology. Ibis 14:361–378
Layard EL (1869b) The South African Museum. S Afr Mag 3:467–468
Loehle C, Eschenbach W (2012) Historical bird and terrestrial mammal extinction rates and causes. Divers Distrib 18:84–91
MacGillivray J (1852) Journal of observations aboard H. M. S. Herald. Manuscripts ADM 7-851 and 7-852. Public Records Office, London
Mathews GM, Gordon JG (1932) The birds of Tristan da Cunha. Nov Zool 38:13–48
McClelland GTW, Bond AL, Sardana A, Glass T (2016) Rapid population estimate of a surface-nesting seabird on a remote island using a low-cost unmanned aerial vehicle. Mar Ornithol 44:215–220
Medina FM, Bonnaud E, Vidal E, Tershy BR, Zavaleta ES, Donlan CJ, Keitt BS, Le Corre M, Horwath SV, Nogales M (2011) A global review of the impacts of invasive cats on island endangered vertebrates. Glob Change Biol 17:3503–3510
Medina F, Bonnaud E, Vidal E, Nogales M (2014) Underlying impacts of invasive cats on islands: not only a question of predation. Biodivers Conserv 23:327–342
Milner J, Brierly OW (1869) The cruise of the H. M. S. Galatea, Captain H. R. H. the Duke of Edinburgh, K.G. in 1867–1868. Allen, London
Milton SJ, Ryan PG, Moloney CL, Cooper J, Dean WRJ, Medeiros JAC (1993) Disturbance and demography of Phylica arborea (Rhamnaceae) on the Tristan–Gough group of islands. Bot J Linn Soc 111:55–70
Moseley HN (1879) Chapter V. Tristan da Cunha, Inaccessible Island, Nightingale Island. In: Notes by a naturalist an account of observations made during the voyage of H. M. S. “Challenger” round the world in the years 1872–1876, under the command of Capt. Sir G. S. Nares, R. N., K. C. B., F. R. S., and Capt. F. T. Thomson. Macmillan, London, pp 108–137
Nicoll MJ (1906) On the birds collected and observed during the voyage of the ‘Valhalla’, R. Y. S., from November 1905 to May 1906. Ibis 48:666–712
Nolloth MS (1856) Visit of H. M. S. Frolic to Tristan da Cunha. Naut Mag 25:401–415
Purdy J (1816) The oriental navigator: or, directions for sailing to, from, and upon the coasts of, the East-Indies, China, Australia, &c. Whittle and Holmes Laurie, London
Richardson ME (1984) Aspects of the ornithology of the Tristan da Cunha group and Gough Island, 1972–1974. Cormorant 12:123–201
Rivadeneira MM, Hunt G, Roy K (2009) The use of sighting records to infer species extinctions: an evaluation of different methods. Ecology 90:1291–1300
Roberts DL, Solow AR (2003) Flightless birds: when did the Dodo become extinct? Nature 426:245
Roberts DL, Elphick CS, Reed JM (2010) Identifying anomalous reports of putatively extinct species and why it matters. Conserv Biol 24:189–196
Rogers RA (1927) The lonely isle. Allen and Unwin, London
Ryan PG (ed) (2007) Field guide to the animals and plants of Tristan da Cunha and Gough Island. Pisces, Newbury
Ryan PG (2008) Taxonomic and conservation implications of ecological speciation in Nesospiza buntings on Tristan da Cunha. Bird Conserv Int 18:20–29
Ryan PG, Moloney CL (2002) Breeding behaviour, clutch size and egg dimensions of Neospiza buntings at Inaccessible Island, Tristan da Cunha. Ostrich 73:52–58
Ryan PG, Cooper J, Glass JP (2001) Population status, breeding biology and conservation of the Tristan Albatross Diomedea [exulans] dabbenena. Bird Conserv Int 11:35–48
Sclater PL (1861) On the island-hen of Tristan d’Acunha. Proc Zool Soc Lond 1861:260–263
Solow AR, Beet AR (2014) On uncertain sightings and inference about extinction. Conserv Biol 28:1119–1123
Solow A, Smith W, Burgman M, Rout T, Wintle B, Roberts D (2012) Uncertain sightings and the extinction of the Ivory-billed Woodpecker. Conserv Biol 26:180–184
Sperling RM (1872) Letter. Ibis 4:74–79
Steadman DW (1995) Prehistoric extinctions of Pacific island birds: biodiversity meets zooarchaeology. Science 267:1123–1131
Steadman DW (2006) Extinction and biogeography of tropical Pacific birds. University of Chicago Press, Chicago
Stirling W (1843) Narrative of the wreck of the ship Tiger, of Liverpool. Roberts, Exeter
Stoltenhoff F (1952) Notes on birds on Inaccessible Island. In: Rosenthal E (ed) Shelter from the spray. Cape Times, Parrow
Stresemann E (1952) Birds collected by Capt. Dugald Carmichael on Tristan da Cunha 1816–1817. Ibis 95:146–147
Swales MK (1965) The sea-birds of Gough Island. Ibis 107(17–42):215–229
Thompson CJ, Koshkina V, Burgman MA, Butchart SHM, Stone L (2017) Inferring extinctions. II. A practical, iterative model based on records and surveys. Biol Conserv 214:328–335
van Aarde RJ (1980) The diet and feeding behaviour of feral cats, Felis catus at Marion Island. S Afr J Wildl Res 10:123–128
von Willemöes-Suhm R (1876) Preliminary report to Professor Wyville Thomson F. R. S., director of civilian scientific staff, on observations made during the earlier part of the voyage of H. M. S. ‘Challenger’. Proc R Soc Lond 24:569–585
Wace NM (1967) Alien plants in the Tristan da Cunha islands. Toward a new relationship of man and nature in temperate lands. Part III. Changes due to introduced species. IUCN publications new series no. 9. IUCN, Morges, pp 46–60
Wace NM (1969) The discovery, exploitation and settlement of the Tristan da Cunha Islands. Proc R Geogr Soc Aust (S Aust Branch) 70:11–40
Wace NM, Holdgate MW (1976) Man and nature in the Tristan da Cunha islands. IUCN Monogr 6:1–114
Wanless RM, Ryan PG, Altwegg R, Angel A, Cooper J, Cuthbert RJ, Hilton GM (2009) From both sides: dire demographic consequences of carnivorous mice and longlining for the critically endangered Tristan Albatrosses on Gough Island. Biol Conserv 142:1710–1718
Wilkins GH (1923) Report on the birds collected during the voyage of the ‘Quest’ (Shackleton-Rowett expedition) to the Southem Atlantic. Ibis 5:474–511
Winterbottom JM (1976) Keytel’s birds from Tristan da Cunha. Ostrich 47:69–70
Acknowledgements
We are indebted to the library staff at the Royal Society for the Protection of Birds for their invaluable assistance in locating historical documents, and without whom this work would not have been possible. Numerous sources were also available on the Biodiversity Heritage Library website (http://www.biodiversitylibrary.org/). We also thank S. Mvungi of the Niven Library (Percy FitzPatrick Institute of African Ornithology, University of Cape Town), S. Frahnert (ZMB), M. Adams, and H. van Grouw (NHMUK) for providing references, information relating to specimens, and for access to the collections. A. Butler (BioStatistics Scotland) translated the models from Matlab into R. Comments from C. Stringer and two anonymous reviewers improved this manuscript.
Data availability
All data are contained in the supplemental material. R code is available at figshare at: https://dx.doi.org/10.6084/m9.figshare.5852961.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by C. Barbraud.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Bond, A.L., Carlson, C.J. & Burgio, K.R. Local extinctions of insular avifauna on the most remote inhabited island in the world. J Ornithol 160, 49–60 (2019). https://doi.org/10.1007/s10336-018-1590-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10336-018-1590-8