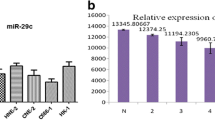
Abstract
Objective
The aim of the study was to construct miRNA-451 expression vector pLMP-miRNA-451 which could help identify the functions of miRNA-451 in SGC-7901 cell.
Methods
Total RNA was extracted from SGC-7901 cells to synthesized cDNA. The synthesized cDNA encoding pre-miRNA-451 was amplified by polymerase chain reaction (PCR). The PCR product was separated by electrophoresis on 1% agarose gel and then recovered and purified. The purified cDNA fragments of miRNA-451 precursor sequence was then ligated with vector pLMP for 1 h by using DNA ligase to form pLMPmiRNA-451 plasmid. After that, the pLMP-miRNA-451 plasmid was transformed into E. coli DH5α strain expression system to clone and amplificate. The purified pLMP-miRNA-451 extracted from E. coli DH5α via transformation and clone screening was identificatied with restriction enzyme digestion and DNA sequencing. At last, pLMP-miRNA-451 was transfected into SGC-7901 cells with lip2000. Real-time PCR was used for detection of the miRNA-451, the transfection efficiency was observed under fluorescence microscopy and cell counting kit-8 assay was conduced to evaluate the effect of miRNA-451 on SGC-7901 cell proliferation.
Results
Our results showed that pLMP-miRNA-451 expression vector was not only constructed successfully and effectively infected SGC-7901 cells, but also could repress the SGC-7901 cell proliferation.
Conclusion
The constructed plasmid pLMP-miRNA-451 could used for further studies of miRNA-451 in SGC-7901 cell lines.
Similar content being viewed by others
References
Greenlee RT, Hill-Harmon MB, Murray T, et al. Cancer statistics, 2001. CA Cancer J Clin, 2001, 51: 15–36.
Hundahl SA, Phillips JL, Menck HR. The National Cancer Data Base Report on poor survival of U.S. gastric carcinoma patients treated with gastrectomy: Fifth Edition American Joint Committee on Cancer staging, proximal disease, and the “different disease” hypothesis. Cancer, 2000, 88: 921–932.
Edwards JK, Pasqualini R, Arap W, et al. MicroRNAs and ultraconserved genes as diagnostic markers and therapeutic targets in cancer and cardiovascular diseases. J Cardiovasc Transl Res, 2010, 3: 271–279.
Fabbri M. miRNAs as molecular biomarkers of cancer. Expert Rev Mol Diagn, 2010, 10: 435–444.
Jackson A, Linsley PS. The therapeutic potential of microRNA modulation. Discov Med, 2010, 9: 311–318.
Bian HB, Pan X, Yang JS, et al. Upregulation of microRNA-451 increases cisplatin sensitivity of non-small cell lung cancer cell line (A549). J Exp Clin Cancer Res, 2011, 30: 20.
Kim Y, Roh S, Lawler S, et al. miR451 and AMPK mutual antagonism in glioma cell migration and proliferation: a mathematical model. PLoS One, 2011, 6: e28293.
Godlewski J, Nowicki MO, Bronisz A, et al. MicroRNA-451 regulates LKB1/AMPK signaling and allows adaptation to metabolic stress in glioma cells. Mol Cell, 2010, 37: 620–632.
Esquela-Kerscher A, Slack FJ. Oncomirs — microRNAs with a role in cancer. Nat Rev Cancer, 2006, 6: 259–269.
Small EM, Frost RJ, Olson EN. MicroRNAs add a new dimension to cardiovascular disease. Circulation, 2010, 121: 1022–1032.
Kolfschoten IG, Roggli E, Nesca V, et al. Role and therapeutic potential of microRNAs in diabetes. Diabetes Obes Metab, 2009, 11 Suppl 4: 118–129.
Hui AB, Lenarduzzi M, Krushel T, et al. Comprehensive microRNA profiling for head and neck squamous cell carcinomas. Clin Cancer Res, 2010, 16: 1129–1139.
Gal H, Pandi G, Kanner AA, et al. MIR-451 and Imatinib mesylate inhibit tumor growth of Glioblastoma stem cells. Biochem Biophys Res Commun, 2008, 376: 86–90.
Nan Y, Han L, Zhang A, et al. MiRNA-451 plays a role as tumor suppressor in human glioma cells. Brain Res, 2010, 1359: 14–21.
Bitarte N, Bandres E, Boni V, et al. MicroRNA-451 is involved in the self-renewal, tumorigenicity, and chemoresistance of colorectal cancer stem cells. Stem Cells, 2011, 29: 1661–1671.
Scholl V, Hassan R, Zalcberg IR. miRNA-451: A putative predictor marker of Imatinib therapy response in chronic myeloid leukemia. Leuk Res, 2012, 36: 119–121.
Bandres E, Bitarte N, Arias F, et al. microRNA-451 regulates macrophage migration inhibitory factor production and proliferation of gastrointestinal cancer cells. Clin Cancer Res, 2009, 15: 2281–2290.
Brenner B, Hoshen MB, Purim O, et al. MicroRNAs as a potential prognostic factor in gastric cancer. World J Gastroenterol, 2011, 17: 3976–3985.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by a grant from the Natural Science Foundation of China (No. 8100098).
Rights and permissions
About this article
Cite this article
Chen, B., Xu, X. Construction of the human miRNA-451 expression vector and its expression in gastric carcinoma cell line SGC-7901. Chin. -Ger. J. Clin. Oncol. 12, 379–384 (2013). https://doi.org/10.1007/s10330-013-1176-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10330-013-1176-0