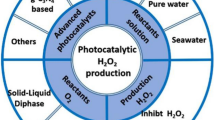
Abstract
Pollution and the rising energy demand are two major society issues, requiring urgent solutions. Here, we review the simultaneous production of hydrogen and removal of pollutants, with a focus on mechanisms, synthesis of photocatalysts, and applications. Remarkably, this method utilizes organic pollutants as sacrificial agents for photocatalytic hydrogen production, thus both reducing pollution and producing energy. Metal-doped, cocatalyst-loaded, organic and inorganic composite, heterostructure and heterojunction photocatalysts are described. We present applications to the removal of antibiotics and organic dyes. Pollutant degradation rates range from 5.3 to 100%, with hydrogen evolution of 13.7–2724.89 μmol·g–1h–1.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The world faces pressing challenges stemming from environmental degradation and energy crises (Zheng et al. 2016; Sajjadi et al. 2021). Numerous harmful substances, often highly toxic and non-biodegradable, persist in the environment, exacerbating environmental degradation. Notably, pollutants such as dyes, aromatics, hormones, and pharmaceutical compounds have garnered global attention due to the serious environmental problems they cause. For instance, the improper disposal practices of pharmaceuticals and personal care products, such as dumping them into water or landfills, contribute to environmental harm (Pandis et al. 2022; Rathi et al. 2021; Quddus et al. 2023; Kumar et al. 2023). This is exacerbated by the fact that a considerable portion of antibiotics ranging from 30 to 90% are released into water bodies as pollutants, originating from hospital waste and pharmaceutical industries, threatening ecosystems (Kumar et al. 2020a). Since the introduction of the internal combustion engine, fossil fuels have dominated the global energy supply, leading to significant energy shortages (Alkhatib et al. 2020). Transitioning to clean and renewable energy sources can alleviate this reliance on fossil fuels and mitigate greenhouse gas emissions, addressing both energy and environmental concerns (Wang et al. 2015; Farghali et al. 2023). Hydrogen emerges as a promising solution for a clean, renewable, non-toxic, highly combustible, and easily storable energy carrier (Staffell et al. 2019; Osman et al. 2023; Hantoko et al. 2024). However, realizing the full potential of hydrogen requires efficient and environmentally friendly large-scale production methods. Therefore, there is an urgent need for effective pollutant management strategies and mitigation of energy crises.
Recent attention has been directed toward leveraging photocatalysis technology for advanced water treatment and clean energy production, particularly focusing on photocatalytic hydrogen generation and the removal of organic pollutants from wastewater (Kumar et al. 2020b; Ismael 2021; Osman et al. 2020). Photocatalysis technology offers a green, efficient, affordable, and ecologically beneficial means of converting solar energy into chemical energy for the degradation of emerging contaminants and meeting energy demands (Liu et al. 2018; Lu et al., 2023a; Yu et al. 2014; Long et al. 2020). To improve the light absorption capacity and photocatalytic activity of materials, various methods such as doping, forming heterojunctions with other semiconductors, loading cocatalysts, and photosensitization are applied (Li et al. 2022a; Osman et al., 2024a). Constructing heterojunctions with various semiconductors is advantageous for enhancing specific surface area, expanding surface active sites, accelerating charge migration rate, and improving photocatalytic activity (Jia et al. 2023). Due to the photoredox characteristics of photocatalysts, photocatalysis involves both oxidation and reduction processes. For the oxidation of organic pollutants to produce carbon dioxide and water over photocatalysts, photogenerated holes or a range of free radicals (hydroxyl radicals and superoxide anions) from solar energy transformations are often utilized (Dong et al. 2017; Jiang et al. 2018). The separation of photogenerated electrons and holes, as well as the light quantum efficiency of catalysts, would be significantly increased in a system where photocatalytic organic pollutant degradation collaborates with hydrogen production over the same catalyst. Recently, there has been a surge in research interest in the idea of waste-to-energy conversion, focusing on the dual functionality of photocatalysis. In these studies, hydrogen is produced from wastewater containing organic pollutants (such as antibiotics and dyes), which is beneficial for simultaneous clean energy production and contamination of water pollutants in a single step (Jiang et al. 2021; Shen et al. 2017; Liu et al., 2020a; Xu et al. 2017). This sustainable approach promises a green future by generating clean energy from wastewater while enabling water treatment and hydrogen generation synchronously, as shown in Fig. 1.
Waste-to-energy conversion using photocatalysis. Photocatalysis technology provides an efficient and eco-friendly method for converting solar energy into chemical energy, with main applications in hydrogen production and waste pollutant degradation. Various techniques, such as doping and forming heterojunctions with semiconductors, enhance light absorption and catalytic activity. By utilizing photogenerated holes or free radicals (•O2− and •OH), photocatalysis facilitates the oxidation of organic pollutants into harmless by-products like carbon dioxide and water. The waste-to-energy conversion produces hydrogen (H2) from wastewater containing pollutants, offering simultaneous clean energy production and water treatment. This sustainable approach holds promise for a greener future. VB: valence band, CB: conduction band, e−: electron, h+: hole
The review summarizes the mechanisms involved in photocatalytic hydrogen production from wastewater and pollutant degradation. The role of sacrificial agents in photocatalytic hydrogen production, along with their importance and implications, is discussed. Additionally, the photocatalytic mechanism involved in coupled or synchronous hydrogen production and organic pollutant degradation is explained in the context of absorption, redox capability, band structure, quantum efficiency, pollutant structure, and other factors. Recent advancements in coupled photocatalytic processes for pollutant degradation and hydrogen production are summarized and analyzed.
Photocatalytic hydrogen production and pollutant degradation
Mechanism
Numerous studies have described the photocatalytic hydrogen production from water splitting using semiconductor photocatalysts (Wang et al. 2019a; Wang et al. 2022a; Ran et al. 2014). Typically, visible light-driven photocatalytic water splitting involves three key steps as illustrated in Fig. 2: (1) absorption of light photons by the photocatalyst to generate electron–hole (e− h+) pairs; (2) separation and migration of these photoexcited charge carriers (e− and h+); and (3) reduction of protons to hydrogen by e− on the conduction band (CB) and oxidation of water (Wang et al. 2019b; Kausar et al. 2022). Due to the challenging nature of four-electron water oxidation kinetics, it is often substituted by the oxidation of suitable organic substrates or sacrificial agents (e.g., methanol, ethanol, ascorbic acid, Na2S/Na2S2O3 pairs, and triethanolamine) (Yuan et al. 2018). These sacrificial agents, with higher oxidation potentials (0.08 V versus normal hydrogen electrode (NHE) for ethanol), facilitate quicker reduction of h+. It is worth noting that photogenerated e− and h+ couples may follow various pathways (Chen et al. 2010), including bulk and surface recombination, which significantly affects hydrogen evolution activity. In the context of photocatalytic hydrogen production, the top of the valence band must exceed the water oxidation potential to form oxygen (O2) (1.23 V), while the conduction band edge must be more negative than the H+ reduction potential to hydrogen (0 V) (Hisatomi et al. 2014; Yadav et al. 2022; Liu et al., 2022a).
Mechanism of photocatalytic hydrogen production. The photocatalytic hydrogen production mechanism involves the absorption of sunlight by a semiconductor photocatalyst, generating electron–hole pairs. These charges migrate to the catalyst’s surface, where electrons reduce protons to produce hydrogen (H2), and holes oxidize water molecules to form oxygen (O2). Reactive oxidative species aid in pollutant removal through surface interactions. This process utilizes solar energy to split water molecules, yielding hydrogen as a renewable energy source while contributing to environmental cleanup. H2O refers to water. VB and CB refer to the valence band and conduction band, respectively, while e− and h+ represent electron–hole pairs
Environmental pollutants encompass a range of substances, including gaseous pollutants like nitrogen oxides (NOx), ammonia (NH3), carbon monoxide (CO), and sulfur oxides (SOx), as well as aqueous pollutants such as pharmaceuticals and personal care products, heavy metal ions, dyes, phenols, and pathogenic microbes. During photocatalytic processes, photoinduced electrons and holes can facilitate pollutant reduction and oxidation, along with reacting with adsorbed O2 and water (H2O) to generate superoxide radicals (•O2−) and hydroxyl radicals (•OH). These reactive oxidative species (ROSs) participate in surface interactions with contaminants, aiding in their removal (Zhao et al. 2021a; Nie et al. 2024). The reactions involved in photocatalytic pollutant removal and hydrogen evolution processes are as follows (Eqs. 1–7):
The reaction involved in photocatalytic hydrogen evolution are:
Requirements
In recent decades, hydrogen production via photocatalytic overall water splitting has garnered significant interest (She et al. 2019; Wang et al. 2021; Zhao et al. 2022). However, the efficiency of solar-to-hydrogen conversion in pure water systems has been low, mainly due to the fast recombination of photogenerated charge carriers (Koe et al. 2020). To address this issue, sacrificial reagents are added to consume the holes and extend the life span of the electrons, reducing the Gibbs free energy of the hydrogen generation reaction and favoring hydrogen evolution (Wei et al., 2020a). This approach has gained popularity as it significantly increases the amount of hydrogen evolved. Thus, the total hydrogen evolution during photocatalysis depends largely on the presence and quality of sacrificial agents. However, traditional sacrificial agents like methanol (Chen et al. 2022a), triethanolamine (TEOA) (Lin et al. 2021), Na2S-Na2SO3 (Jang et al. 2007), ascorbic acid, and lactic acid (Liu et al., 2020b) are costly, toxic, and produce harmful substances.
Effective photocatalytic total water splitting without sacrificial agents is extremely challenging due to various influencing factors (Zou et al. 2022), highlighting the need for cheaper alternatives. One potential solution lies in utilizing contaminants as electron acceptors, suggesting a novel approach to converting wastewater containing organic contaminants into hydrogen energy before disposal, aligning with the waste-to-energy concept for sustainable solutions. Combining photocatalytic oxidation and reduction in a single system enhances photogenerated e–h separation efficiency, leading to superior organic pollutant degradation and wastewater energy utilization, as presented in Fig. 3.
Mechanism of simultaneous photocatalytic hydrogen production and pollutant degradation. For photocatalytic water splitting, a reduction reaction may occur at the conduction band (CB) position of the photocatalyst, where hydrogen ions (H+) are reduced to hydrogen (H2). An oxidation reaction may occur at the valence band (VB) position, where water (H2O) is split into oxygen (O2) and hydrogen ions. Similarly, for photocatalytic pollutant degradation, superoxide radicals (•O2−) are generated at the reduction site, and hydroxyl radicals (•OH) are generated at the oxidation site, both of which have strong oxidizing properties to degrade most pollutants
Over the past decade, significant research has been conducted on photocatalytic hydrogen production from organic pollutant waste. For instance, a study utilized a photocatalytic system to achieve simultaneous hydrogen production from antibiotic wastewater using a copper oxide/reduced graphene oxide/bismuth vanadate (Cu2O/RGO/BiVO4) photocatalyst (Shen et al. 2020). In their study, tetracycline acted as a sacrificial agent. During the aerobic decomposition of tetracycline, electrons combined with O2 to generate radical •O2− and the h+ reacted with H2O to form radical •OH, which served as reactive oxygen species for tetracycline elimination. Similarly, during the formation of hydrogen, electrons interacted with H2O to produce hydrogen. As a result, the h+ and •OH acted as scavengers, consuming tetracycline. Additionally, the incorporation of copper oxide (Cu2O) and bismuth vanadate (BiVO4) into the Z-Scheme system, facilitated by reduced graphene oxide (RGO), enhanced redox ability and photocatalytic performance.
Sacrificial agents in hydrogen evolution
Sacrificial agents, also known as electron donors or hole scavengers, play a crucial role in photocatalytic hydrogen generation. Common sacrificial agents used in photocatalytic processes include methanol, ethanol, isopropanol, triethanolamine, formic acid, ascorbic acid, N,N-dimethylformamide (DMF), and glucose (Kumaravel et al. 2019; Huang et al. 2019; Vaiano et al. 2015). These agents, characterized by negative charge and hydroxyl groups on their surfaces (Zhu et al. 2019), are oxidized by photoexcited holes during the photocatalytic process, preventing their recombination with electrons and facilitating practical hydrogen generation, especially when biomass or abundant chemicals are employed. Most h+ oxidizing agents (•OH, •O2−) can additionally release H+ or hydrogen, resulting in a larger hydrogen evolution yield than the photocatalyst alone. The oxidation of sacrificial agents results in the production of simpler products and hydrogen. For example, methanol transforms into formaldehyde, formic acid, carbon dioxide, and hydrogen when activated by photoexcited holes or •OH radicals (Salcedo-Abraira et al. 2021). Additionally, intermediate species generated during alcohol oxidation possess reducing characteristics, enhancing hydrogen production activity indirectly. Some sacrificial substances, such as phosphoric acid and sulfide (S2−), protonate water, leading to hydrogen production when the photogenerated holes interact with protonated ions (Hao et al. 2019; Pei et al. 2019).
The performance of cobalt disulfide encapsulated with thiocyanate (CoS2@SCN) nanomaterials regarding charge transfer efficiency and hydrogen generation was significantly enhanced by employing various sacrificial agents, such as triethanolamine and organic alcohols. Among these agents, triethanolamine exhibited the highest photocatalytic activity, attributed to its negatively charged surface facilitating reactions with holes, thus improving charge transfer and hydrogen generation efficiency (Wang et al. 2022b). In another study, lactate, formic acid sodium sulfide/sodium sulfite (Na2S/Na2SO3), and triethanolamine were utilized as hole scavengers to accelerate the oxidation half-reaction for enhanced photocatalytic hydrogen evolution using molybdenum disulfide/cadmium zinc sulfide (MoS2/Cd0.6Zn0.4S). The presence of sacrificial agents simultaneously strengthened both reduction and oxidation half-reactions, thereby improving overall efficiency. Compared to other MoS2/Cd0.6Zn0.4S heterostructures, and even in the absence of sacrificial reagents, the study demonstrated that 0.8% MoS2/Cd0.6Zn0.4S with Na2S/Na2SO3 as the sacrificial agent exhibited superior hydrogen evolution reaction (HER) performance. Sacrificial electron donors, such as S2−/SO3 and S22−, facilitate hydrogen generation through direct reduction and indirect oxidation pathways, with water serving as a proton source for hydrogen evolution reactions. The optimized 0.8% MoS2/Cd0.6Zn0.4S heterostructure achieved a hydrogen evolution reaction rate of up to 13,466.50 µmol·g−1·h−1 (Wang et al. 2023).
While the addition of synthetic reagents enhances the cost of hydrogen generation and contributes to significant environmental harm, thereby limiting their suitability for photocatalysis (Bao et al. 2021), recent studies have explored synergistic catalysis involving both contaminant degradation and hydrogen generation (Li et al. 2018; Wei et al., 2020b). This approach has been applied to the treatment of dye wastewater and antibiotic wastewater, demonstrating promising potential in synergetic photocatalysis for hydrogen evolution.
In summary, sacrificial agents are integral to photocatalytic hydrogen generation. Methanol, ethanol, isopropanol, triethanolamine, formic acid, ascorbic acid, N,N-dimethylformamide, and glucose are among the agents commonly utilized for their negative charge and hydroxyl groups, facilitating oxidation by photoexcited holes, and preventing electron recombination. Their inclusion significantly enhances hydrogen production efficiency, particularly when biomass or abundant chemicals are employed. Furthermore, the oxidation of sacrificial agents produces simpler products and hydrogen, further boosting hydrogen production activity. Recent advancements exploring synergistic catalysis, combining contaminant degradation and hydrogen generation, show promise, especially in treating dye and antibiotic wastewater. This approach presents an opportunity to address challenges associated with synthetic reagents in photocatalysis, offering a more sustainable and efficient pathway for hydrogen production.
Pollutants as sacrificial agents
The role of pollutants as sacrificial agents is crucial in photocatalytic hydrogen generation due to the energetically uphill nature of water splitting (∆H0 = 286 kJ mol−1) (Kumaravel et al. 2019; Méndez et al. 2014). Conventional photocatalysts often exhibit inadequate quantum efficiency, with a significant portion of photogenerated h+ and e− prone to recombination during migration, posing challenges for smooth migration to the photocatalyst surface (Guo et al. 2023). To overcome these challenges and maximize efficiency, experiments have explored the use of organic and inorganic pollutants in the environment as electron donors or acceptors for photocatalytic reactions, aiming to develop stable and highly active catalysts. Organic pollutants like antibiotics (tetracycline, norfloxacin, ofloxacin, and others) and dyes have shown potential as sacrificial agents through photooxidation under light exposure, making them promising candidates for photocatalytic hydrogen production. Recent studies have investigated photocatalytic hydrogen production from polluted solutions, with organic contaminants serving as sacrificial agents to enhance photoreactivity (Chen et al. 2022b; Liu et al. 2023). For instance, a study examined the effect of rhodamine B, methylene orange, and levofloxacin as sacrificial agents at different pH levels (3, 5, 7, and 9) for hydrogen production using a Z-Scheme Ag/g-C3N4-Ag-Ag3PO4 photocatalyst. Levofloxacin demonstrated the highest hydrogen production (218.87 µmol) due to its positive charge at pH 5, facilitating interaction with negatively charged photocatalyst particles. Conversely, methylene orange exhibited decreased efficiency (98.24 µmol) due to electron-withdrawing groups, while rhodamine B showed low efficiency because both its molecules and photocatalyst particles were negatively charged at pH 5, hindering the breakdown process (Li et al. 2020).
Photocatalysts synthesis
Metal-doped photocatalysts
Improving photocatalytic efficiency by doping metals or nonmetals to modify the optical band gap of the photocatalyst shows promise. Doping transition and inner transition metals facilitate the conveyance of charge carriers to the photocatalyst’s surface. These metals, when used as dopants, can influence the host crystal’s lattice flaws, alter crystallinity, hinder electron–hole recombination, and adjust the light absorption region by modifying crystallinity (Sanakousar et al. 2022; Palani et al. 2022). For example, bismuth (Bi) doped graphitic carbon nitride (g-C3N4) exhibited superior optical characteristics and increased absorption (440 nm to 660 nm) compared to pure water, leading to enhanced hydrogen generation efficiency. Using amoxicillin (AMX) as a sacrificial reagent in the reaction process further improved performance, with the 2% Bi/g-C3N4 photocatalyst achieving the highest hydrogen evolution rate of 35.935.9 µmol h−1 (Wei et al. 2019). Additionally, (Hu et al. 2021) designed a novel barium (Ba2+) doped and cyano (-C≡N) comodified g-C3N4 through a salt-assisted method for hydrogen generation from antibiotic wastewater. Ba2+ ions trapped in the g-C3N4 intra-layer ring cavity through ion–dipole interaction promoted the production of cyano defects by partial heptazine ring opening, enhancing light absorption capacity and resulting in a red shift in UV spectra. The 7% BaCN-g-C3N4 photocatalyst demonstrated superior activity in tetracycline water, achieving simultaneous 63.62% tetracycline removal and hydrogen production of 10,316 mmol g−1 h−1, nearly 5.5 times greater than pure hydrogen production, which was nearly 5.5 times more than pure g-C3N4.
Cocatalyst-loaded photocatalysts
Cocatalysts are commonly employed in heterogeneous photocatalytic systems to enhance the photocatalytic hydrogen activity of the photocatalyst by improving charge separation efficiency. Additionally, cocatalysts can increase the catalyst’s resistance to photodegradation by efficiently consuming light-induced charge carriers, particularly by preventing holes from oxidizing the semiconductor surface (Yang et al. 2013). Efficient operation is facilitated by the presence of an effective cocatalyst, which promotes interfacial charge transfer at the conductor/semiconductor contact, enhancing the built-in electric field according to the fundamental concept of interface potential convergence (Chen et al. 2017). Noble metals like platinum (Pt) (Caudillo-Flores et al. 2018), palladium (Pd) (Ren et al. 2022), and gold (Au) (Reichert et al. 2015) are frequently utilized as cocatalysts due to their lower Fermi energy level compared to typical semiconductors such as titanium oxide (TiO2), making them suitable for forming a Schottky barrier at the heterojunction interface (Leung et al. 2010; Kashiwaya et al. 2018). The quantity of cocatalysts significantly influences the photocatalytic hydrogen generation capacity by providing additional active regions for hydrogen production at the photocatalyst’s surface (Li et al. 2021a). For instance, Pt-loaded nitrogen-rich g-C3N4 nanotube (CNNT) photocatalysts were employed for photocatalytic hydrogen evolution alongside bisphenol A degradation. The addition of Pt (3wt %) enhanced the photocatalytic hydrogen evolution performance of CNNTs. CNNTs containing Pt nanoparticles as a cocatalyst exhibited a substantially higher hydrogen evolution rate of 180.60 µmol h−1 compared to bulk g-C3N4 (9.267 µmol h−1) (Xu et al. 2020a). However, the high cost of precious metals limits their application as cocatalysts for energy conversion. Consequently, researchers are investigating low-cost, non-noble metal-based semiconductor cocatalysts for photocatalytic hydrogen generation, such as metal sulfides and metal phosphides (Zhang et al. 2022). For example, a novel nickel phosphide/indium oxide (Ni2P/In2O3) composite photocatalyst (NIP/INO) exhibited the best hydrogen evolution rate of 0.138 µmol h−1 with 16.12% rhodamine B degradation, representing a 10.6-fold and 1.6-fold increase over individual In2O3, respectively. Additionally, its photocatalytic activity surpassed that of the 1 wt.% Pt-decorated In2O3. Furthermore, Ni2P as a cocatalyst significantly enhanced photocatalytic performance under visible light irradiation (Yang et al. 2021a).
Organic and inorganic composites
The emergence of organic–inorganic composites represents a groundbreaking advancement in the realm of photocatalysis, offering highly adaptable functional photocatalysts with tailored features and enhanced energy conversion efficiency by leveraging the strengths of both organic and inorganic materials (Singh et al. 2020; Wang et al. 2017; Yang et al. 2022). Organic components, such as polymers or carbon-containing substances like metal–organic frameworks (MOFs) and carbon quantum dots (CQDs), are commonly integrated into these composites. They serve various roles, including acting as conductive elements, catalytic supports, or facilitators of electron transport (Belousov et al. 2022; Zhang et al. 2019). Inorganic materials, such as semiconductors or metal-based catalysts, play a vital role in the photocatalytic process by aiding in the decomposition of contaminants in wastewater and facilitating the water splitting reaction (Liras and Barawi 2019). In recent years, diverse organic–inorganic composites have been applied for simultaneous photocatalytic hydrogen production from organic wastewater. For instance, a Pt-loaded g-C3N4/SrTiO3 Z-Scheme heterostructure composite was developed for photocatalytic hydrogen production from acid red 1 solution, achieving an impressive 93% degradation of acid red 1 dye (50 mg/L); alongside, a simultaneous hydrogen evolution rate of 471 µmol·h−1·g−1 was achieved (Tan et al. 2020). Furthermore, the CQDs/g-C3N4 (CCN) catalyst exhibited enhanced photodegradation efficiency and an increased hydrogen evolution rate of 1291 µmol·h−1·g−1 (Zhang et al. 2020).
Heterostructures and heterojunctions
Conventional heterojunctions
A heterojunction refers to the interface between two separate semiconductors with unequal band structures, leading to band alignments. Conventionally, there are three types of heterojunction photocatalysts: type I, type II, and type III (Low et al. 2017; Kumari et al. 2022), illustrated in Fig. 4. In type I heterojunctions, the semiconductors have straddling gaps, while in type II, they have staggered gaps, and in type III, they have broken gaps. For type I heterojunctions, the conduction band and valence band of semiconductor A are, respectively, higher and lower than those of semiconductor B. In type II heterojunctions, the conduction band potential edge of semiconductor B is higher than that of semiconductor A, facilitating the transfer of photogenerated electrons from A to B and holes from B to A (Sharma et al., 2023a; Wang et al. 2013). Type III heterojunctions have a similar design to type II but with severe staggered gaps preventing band overlaps, thereby impeding electron–hole pair separation (Hyun et al. 2013). Therefore, the type III heterojunction is not appropriate for improving the separation of electron–hole pairs because the electron–hole migration and separation between the two semiconductors cannot occur. Heterojunction types are categorized based on carrier transfer channels, band positions, and arrangements, including conventional (type I, type II, and type III), p–n heterojunctions, Z-Scheme (direct and indirect), and S-Scheme heterojunctions. Despite enabling charge transfer, conventional heterojunctions may lower the redox capacity of photocarriers within the photocatalyst.
Conventional type I, type II, and type III heterojunctions. In type I heterojunctions, depicted with straddling band gaps, the conduction band (CB) and valence band (VB) of semiconductor A are, respectively, higher and lower than those of semiconductor B. Conversely, type II heterojunctions feature staggered band gaps, where the conduction band potential edge of semiconductor B is higher than that of semiconductor A, facilitating electron transfer from A to B and hole migration from B to A. Type III heterojunctions, characterized by broken band gaps, have severe staggered gaps preventing band overlaps hindering efficient electron–hole pair separation. These representations provide insights into the band alignments and carrier transfer mechanisms essential for understanding the photocatalytic properties of heterojunctions
Z-Scheme heterojunction
In response to the inherent limitations of standard type II heterojunctions in terms of redox capabilities, Z-Scheme photocatalysts have emerged as a promising alternative. While sharing a similar band energy structure to type II heterojunctions, Z-Scheme photocatalysts offer a distinct pathway for charge carrier migration (Cui et al. 2020; Stelo et al. 2020). In Z-Scheme photocatalysts, photogenerated electrons from the lower energy levels of two semiconductors with higher energy levels recombine, enabling organic molecules to react with external materials. Specifically, electrons in the conduction band of semiconductor II migrate to the valence band of semiconductor I and combine with photogenerated holes. This electron carrier route not only enhances physical charge separation within the semiconductor but also preserves the original redox potential of electrons in semiconductor I and holes in the semiconductor II valence band (Kumar et al. 2023; Rana et al. 2023). The schematic charge transfer mechanism in Z-Scheme heterojunction is depicted in Fig. 5a.
Z-Scheme (a) and S-Scheme (b) heterojunctions. Photocatalysis enables hydrogen (H2) generation by water molecule splitting and pollutant remediation through breakdown into harmless by-products under light exposure. In Z-Scheme photocatalysts, electrons from the conduction band of semiconductor II (SC II) migrate to the valence band of semiconductor I (SC I), combining with photogenerated holes. This electron transfer route enhances charge separation and maintains the redox potential of electrons in semiconductor I and holes in semiconductor II. In S-Scheme heterojunctions, the reduction photocatalyst (RP) features lower conduction band and Fermi levels, while the oxidation photocatalyst (OP) has higher valence band levels. This Fermi level difference causes electron flow from the reduction to the oxidation photocatalyst until equilibrium is achieved, inducing an internal electric field and band bending at the interface. This mechanism facilitates photoinduced carrier transfer, with electrons transitioning to the more negative conduction band and holes to the more positive valence band. CO2 refers to carbon dioxide. H2O refers to water
Yang et al. (2021b) designed a Z-Scheme ZnIn2S4-NiO/BiVO4 photocatalytic system for simultaneous hydrogen evolution and formaldehyde (HCHO) degradation under visible light irradiation. The Z-Scheme configuration facilitates the generation of reactive oxygen species (ROS) and enhances the oxidation conversion of formaldehyde (HCHO) into formic acid (HCOOH), thereby accelerating hydrogen production. Under visible light irradiation, a large number of electrons (e−) accumulate on the conduction band of ZnIn2S4 and enrich on the Pt surface (used as a cocatalyst), while holes (h+) reside on the valence band of BiVO4 and enrich on the NiO surface. Electrons on the Pt surface participate in two reactions (2H+ → H2 and O2 → •O2−), resulting in the generation of hydrogen, while holes on the NiO surface are involved in three reactions (H2O or OH− → •OH, HCHO → HCOOH + H+, and HCOO− → H+) responsible for formaldehyde degradation (Yang et al. 2021b).
Various Z-Scheme heterojunctions have been designed recently for coupled photocatalytic processes, such as CeO2/Cu–I-bpy (Bu et al. 2023) and CeO2/g-C3N4 (Zhao et al. 2021b). However, it has been reported that Z-Scheme heterojunctions lack supporting experimental evidence, and the transfer mechanism has not been comprehensively analyzed. Early Z-Scheme junctions relied on redox mediators, and the electron transfer mediated by water was prohibited (Xu et al., 2023). Subsequently, direct solid-state Z-Scheme junctions were proposed and synthesized, but they were derived from incorrect old versions and were also inaccurate. Some authors proposed a more progressive step-scheme heterojunction, known as S-Scheme, which accounts for better interfacial charge transfer between two semiconductor photocatalysts with staggered band structures, supported by experimental evidence (Fu et al. 2019; Xu et al. 2020b).
S-Scheme heterojunctions
The concept of S-Scheme heterojunctions was introduced in 2019, comprising two distinct n-type semiconductors, namely the oxidation photocatalyst (OP) and reduction photocatalyst (RP) (Li et al. 2022b; Lu et al., 2023b). The reduction photocatalyst possesses lower conduction band and Fermi levels, while the oxidation photocatalyst has higher valence band levels. Due to the disparity in Fermi levels, electrons flow from the reduction photocatalyst to the oxidation photocatalyst until equilibrium is reached (Li et al. 2024; Zhang et al. 2021). This electron transfer mechanism induces an internal electric field and band bending at the interface, facilitating photoinduced carrier transfer. As electrons and holes transition to the more negative conduction band and more positive valence band, respectively, the carrier transfer in an S-Scheme heterojunction resembles a "step" at the macroscopic level, as depicted in Fig. 5b. The resulting S-Scheme heterojunction not only enhances carrier separation through internal electric field and band bending but also preserves the maximal redox capacity of the photocatalytic system. In this context, a novel S-Scheme In2S3/Zn3In2S6 (InS/ZIS) heterojunction was developed for photocatalytic degradation of bisphenol A and hydrogen production. The S-Scheme charge transfer mechanism enhances the redox capability of Zn3In2S6 for hydrogen evolution and In2S3 for bisphenol A degradation. The internal electric field generated by the heterojunction promotes charge separation and transfer, thereby increasing the efficiency of the photocatalytic process. The S-Scheme InS/ZIS photocatalyst exhibited a high hydrogen evolution rate of 81.6 mmol·g−1·h−1, significantly surpassing the individual InS and ZIS counterparts (Yang et al. 2023).
Compared to conventional heterojunctions (types I and II), Z-Scheme heterojunction provide fast charge separation, robust redox ability, and enhanced light absorption capacity, all of which contribute to improved photocatalytic activity (Dong et al. 2023; Liu et al. 2021; Malefane et al. 2023; Li et al. 2021b; Abdel-Aziz et al. 2019). Z-Scheme heterojunctions address charge separation and recombination more effectively than conventional counterparts. However, challenges such as high recombination rates of photogenerated electron–hole pairs, limited quantum efficiency, and long-term durability issues hinder the widespread adoption of Z-Scheme photocatalyst systems.
The emergence of new and more efficient S-Scheme heterojunctions marks a significant shift in the field. Unlike Z-Scheme heterojunctions, S-Scheme counterparts have attracted considerable interest due to their well-understood charge transfer mechanism. The inherent electric field in S-Scheme heterojunctions facilitates charge separation and exhibits strong redox capabilities. The fundamental difference between S-Scheme and Z-Scheme photocatalysts lies in the transfer of electrons between semiconductor Fermi energies following heterojunction formation. In S-Scheme photocatalysts, this electron transfer leads to the formation of an internal electric field, which sustains charge separations (Jabbar et al. 2023; Su et al. 2022; Su et al. 2023; Zheng et al. 2020; Sharma et al. 2023b). In addition, S-Scheme heterojunctions achieve enhanced redox capabilities and offer protection for high-potential band edges. This feature proves highly beneficial in simultaneous hydrogen production and pollutant degradation experiments, as they involve concurrent reduction (hydrogen evolution) and oxidation (pollutant degradation) processes. The accumulation of photogenerated electrons in the high-potential conduction band of one semiconductor in S-Scheme heterojunctions leads to high-performance hydrogen generation via the e−/H+ route, while the accumulated holes in the high-potential valence band of the other semiconductor can directly oxidize pollutants or indirectly generate OH● radicals. Moreover, the charge transfer in S-Scheme heterojunctions can be confirmed through experimental techniques such as in situ X-ray photoelectron spectroscopy (XPS).
Controlling reaction parameters
The photocatalytic activity and selectivity of a photocatalyst are known to be influenced by various reaction conditions and material design characteristics. In a dual-functional system, achieving desired outcomes with high efficiency often requires the design of suitable photocatalysts and careful management of reaction parameters (Naseri et al. 2022). Figure 6 illustrates some of these crucial reaction parameters.
Various reaction parameters affecting photocatalytic degradation of pollutants. These reaction parameters consist mainly of irradiation time, photocatalyst dosage, dopant type and weight percentage, pollutant type and concentration, aerobic and anaerobic conditions, light source and intensity, pH of the solution, and others. Generally, longer irradiation time, higher photocatalyst dosage, lower pollutant concentration, higher light intensity and power, and aerobic conditions have positive effects on the rate of photocatalytic degradation of pollutants. However, the effects of dopant type and weight percentage, pollutant type, anaerobic conditions, light source type, and solution pH on the photocatalytic degradation of pollutants need to be judged comprehensively on a case-by-case basis and experimental data
In a coupled process, the concentration of the pollutant plays a crucial role in hydrogen generation. For instance, Wang et al. (2022b) conducted experiments on simultaneous hydrogen evolution from tetracycline (TC) wastewater solution using Bi3TaO7/ZnIn2S4 photocatalyst. They observed that the hydrogen evolution rate increased with the initial concentration of the tetracycline solution up to 40 mg·L–1. However, at higher pollutant concentrations, the hydrogen evolution decreased because the generated tetracycline intermediates in solution consumed more photoinduced holes, reducing the hydrogen evolution rate activity in the conduction band of ZnIn2S4 (Wang et al. 2022c).
Similarly, in another work, solar-driven CaF2:Yb3+, Er3+@ZnTiO3/Au/CdS Z-Scheme heterojunction was designed to investigate the coupled behavior of photocatalysts in norfloxacin solution. They found that the degradation efficiency of norfloxacin increased with decreasing concentration of norfloxacin within 180 min of light irradiation, while the photocatalytic hydrogen production rate increased with increasing concentration of norfloxacin. The photocatalysts exhibited the highest hydrogen evolution rate of 511.99 µmol g−1 in 50 mg·L–1 norfloxacin solution over 5 h. The doping of Au nanoparticles provided a conductive channel and surface plasmon (SPR) effect, while CaF2:Yb3+, and Er3+ acted as up-conversion luminescent agents which increased the light absorption capacity of the photocatalyst near-infrared region (Liu et al., 2022b).
Increasing the mass of photocatalysts offers several advantages, primarily enhancing their available surface area and porosity. This increase in mass leads to a higher surface area of the photocatalyst, providing more active sites for the adsorption and reactivity of contaminants. Consequently, degradation becomes more likely due to the increased contact between pollutant molecules and the photocatalyst. Moreover, higher loading of the catalyst results in a greater production of oxidizing radicals, further enhancing the degradation process. However, it is crucial to consider potential constraints associated with increasing the mass of the photocatalyst. One significant concern is particle aggregation, which becomes more likely as the mass of the photocatalyst rises (Ahmed and Mohamed 2023a).
The availability of oxygen (aerobic) or the absence of oxygen (anaerobic) in the reaction or process medium significantly influences photocatalytic reactions for both pollutant degradation and hydrogen evolution. A study investigated the impact of different pH levels and light sources on the degradation of cefazolin (CFZ) along with simultaneous hydrogen evolution using a TCCdSe-Ag-WO3-Ag photocatalyst. After 30 min visible light irradiation at pH levels of 2.8, 4.8, 6.8, and 8.8, the degradation efficiency for cefazolin was found to be 93.13%, 96.32%, 83.29%, and 69.14%, respectively. After 4 h of simulated sunlight exposure, the quantities of hydrogen evolution were 488.49 µmol g−1, 448.31 µmol g−1, 350.56 µmol g−1, and 258.11 µmol g−1, respectively, when the pH values were adjusted to 2.8, 4.8, 6.8, and 8.8. The photocatalyst showed the best catalytic performance under stimulated sunlight compared to ultraviolet and visible light (Li et al., 2021c).
Synchronous hydrogen production and pollutant degradation
In recent years, the extensive exploitation of natural fossil fuels to meet industrial and societal energy needs has led to the depletion of reserves, highlighting the urgent need for sustainable energy solutions. Hydrogen, touted as a clean and sustainable alternative, is projected to supplant traditional fossil fuels (Bai et al. 2018; Han et al. 2016). Research efforts have predominantly focused on two areas: photocatalytic environmental decontamination and efficient water purification (Kumar et al. 2020b; Ahmad et al. 2015; Wang and Zhuan 2020). Many studies have explored the use of sacrificial agents to enhance hydrogen production by capturing photogenerated holes, shedding light on the underlying principles (Wang et al., 2022d; Lin et al., 2022a). Untreated wastewater, rich in organic contaminants and inorganic anions, can serve as effective sacrificial agents and electron donors. By leveraging natural resources like solar energy, this approach holds promise for cost-effective energy and pollution solutions (Patsoura et al. 2006). The economic viability and effectiveness of remediation techniques are crucial factors determining their practicality. Integrating these methods enables us to address both energy and environmental challenges simultaneously.
Antibiotics removal and hydrogen production
The presence of antibiotics in wastewater has garnered increased attention due to concerns over the development of drug-resistant microbes and the potential risks to human health. Antibiotics, known for their resistance to conventional treatment methods, pose challenges as prevalent emerging pollutants (Dong et al. 2018; Gao et al. 2016; wang et al. 2024). Addressing the issue of antibiotic wastewater, researchers have turned to a novel perspective: exploring hydrogen production from such waste before its discharge into the environment. This waste-to-energy approach aligns with the principles of environmental sustainability. For instance, Kumar et al. (2021) have developed dual Z-Scheme g-C3N4/Bi4Ti3O12/Bi4O5I2 heterostructured photocatalysts for solar-coupled degradation of antibiotics like sulfadiazine and ofloxacin, alongside simultaneous hydrogen generation in anaerobic conditions. Among the photocatalysts tested, CTBT-30 (composed of 40% g-C3N4/Bi4Ti3O12/30% Bi4O5I2) demonstrated remarkable performance, producing hydrogen at a rate of 56.2 mmol·g−1·h−1 under visible light, while removing 87.1% of ofloxacin. This efficiency was attributed to the effective promotion of charge separation by I3−/I− and Bi3+/Bi5+ redox mediators, facilitating interfacial interaction with well-matched energy bands. As a result, photogenerated electrons were available for hydrogen evolution from water once most of the holes were scavenged either for direct oxidation or through the production of •OH radicals (Kumar et al. 2021).
Lin et al. (2022b) have developed Ni5P4/g-C3N4 heterojunctions (NP/CN) for the synchronous hydrogen evolution from carbamazepine wastewater under anaerobic conditions. This photocatalyst, specifically the 25% Ni5P4/g-C3N4 (25NP/CN) junction, exhibited impressive performance, with a hydrogen evolution rate of 40.1 mmol·g−1·h−1 hydrogen evolution and 94.4% carbamazepine elimination under visible light irradiation. Remarkably, under anaerobic conditions, the 25NP/CN photocatalyst demonstrated simultaneous hydrogen evolution and carbamazepine degradation, while also maintaining high performance under aerobic conditions. The primary active species responsible for carbamazepine degradation under aerobic conditions are radical •OH+, •O2−, and h+. In the absence of dissolved oxygen, photogenerated h+ and •OH degrade carbamazepine, while separated photogenerated electrons produce hydrogen through the e−/H2O route. The degradation of carbamazepine leads to the formation of two oximes, P8 and P11, characterized by the structure (R–C═N–OH). These oximes can generate cyclic intermediates through coordination with Ni. In the process of hydrogen generation, intermolecular H+ transfer occurs within these cyclic intermediates. Consequently, both intermediates can contribute to hydrogen production through this mechanism (Lin et al., 2022b).
In another work, researchers explored the dual functionality of Ag3PO4/Cs2AgBiBr6/GH photocatalysts, which facilitated both antibiotic degradation and hydrogen generation (Chen et al. 2022d). In experiments utilizing tetracycline as a sacrificial agent in a tetracycline/methanol system, the maximum production of photocatalytic hydrogen efficiency reached 7653.21 µmol·g−1, accompanied by a simultaneous photodegradation efficiency of tetracycline at 76.55%. Through electron spin resonance (ESR) and radical trapping measurements, it was determined that reactive oxygen species such as hydroxyl radicals (•OH) and superoxide radicals (•O2−) were generated via the photogenerated charge transfer pathway of the system. These reactive oxygen species played a dual role: oxidizing tetracycline and facilitating the production of hydrogen by splitting water molecules. By attacking tetracycline molecules at both nucleophilic and radical attack sites, reactive oxygen species enhance the breakdown of tetracycline. It is hypothesized that the C–C═O and C-N–C fragments on tetracycline molecules acted as sites for nucleophilic and radical attacks, respectively. The higher yield of hydrogen production was attributed to the greater contribution of electrons from tetracycline compared to methanol (Chen et al. 2022c).
The efficiency of hydrogen generation in photocatalytic water splitting is primarily restricted by the four-electron kinetic process involved in oxygen production. However, this limitation can be significantly mitigated by leveraging potent catalysts that convert the four-electron process of oxygen production into the two-electron process of generating H2O2/•OH (Liu et al. 2015; Ou et al. 2019). For instance, the Zhang group engineered Z-Scheme Ag3PO4@ MoS2 photocatalysts featuring abundant oxygen vacancies to facilitate hydrogen production from solutions containing antibiotics such as ciprofloxacin, tetracycline, ofloxacin, and norfloxacin. The electrons in the conduction band of MoS2 possess a more negative potential (− 0.49 eV) compared to the redox potential of •O2−/O2, enabling the generation of •O2− radicals. Meanwhile, the holes in the valence band of Ag3PO4, with a potential of 2.43 eV higher than that of •OH/H2O, oxidize −OH to produce •OH radicals. Antibiotics serve a dual role in this process, acting as hydrogen donors and electron donors for the catalytic reaction. The highest hydrogen yield (108 µmol·g−1) and degradation efficiency (88.36%) were measured and observed in the norfloxacin solution, while the lowest hydrogen yield (48.22 µmol·g−1) and degradation efficiency (65.81%) were recorded for the tetracycline solution. The photocatalyst facilitates the presence of electrons and holes with potent oxidation and reduction capabilities, serving as electron mediators to expedite the separation of photogenerated carriers (Zhang et al. 2023). Table 1 presents recent advancements in simultaneous photocatalytic hydrogen production and wastewater treatment.
Other pollutant removal and hydrogen production
Organic dyes, synthetic aromatic compounds, pose potential risks of carcinogenicity and mutagenicity. Categorized into cationic and anionic types, they are common pollutants in industries like textiles, plastics, and paper production (Lu and Astruc 2020). Ding et al. (2023) conducted a study wherein they synthesized ultrathin two-dimensional ZnGa-borate-LDH nanosheets aimed at facilitating photocatalytic coupled processes for hydrogen generation in Congo red (CR) dye wastewater. In their investigation, they compared the hydrogen generation rates of ZnGa-BLDH nanosheets with those ofthe other catalysts, namely carbonate anions intercalated LDH (ZnGa-CLDH-CR) and borate anions B(OH)4− intercalated LDH (ZnGa-C-BLDH-CR). Remarkably, ZnGa-BLDH-CR exhibited a hydrogen generation rate of 32 µmol·g–1·h–1, surpassing ZnGa-CLDH-CR (12.2 µmol·g–1·h–1) and ZnGa-C-BLDH-CR (12.6 µmol·g–1·h–1) by 2.6 and 2.5 times, respectively. The interlayer channel of ZnGa-BLDH, sized similarly to a water molecule, facilitates efficient transportation of reactants and products. During visible light irradiation, photogenerated electrons migrate from the CR LUMO to the conduction band of ZnGa-LDHs, enabling proton reduction and consequent hydrogen formation. Concurrently, h+ from the valence band of ZnGa-BLDH may transfer to the CR HOMO, leading to Congo red degradation. Additionally, the •OH radicals generated by h+ can further induce Congo red degradation. However, the quantity of hydrogen produced decreased by 37% after five cycles, attributed to the formation of irreversible inactive chemicals during Congo red decay (Ding et al. 2023).
Economic viability, waste management, and environmental impact
The scalable production of hydrogen from wastewater emerges as a promising technique, poised to tackle several pressing concerns spanning economic feasibility, waste management, and environmental challenges (Osman et al., 2024b). Leveraging wastewater as a readily available source holds immense potential for unlimited photocatalytic hydrogen production. Crucial to the economic viability of this process is the energy source employed for hydrogen manufacturing. The adoption of renewable energy sources, particularly solar energy, stands to significantly slash energy costs, thereby enhancing the economic attractiveness of hydrogen generation.
Moreover, the prevailing market dynamics surrounding hydrogen and its various applications play a pivotal role in determining the economic feasibility of wastewater-derived hydrogen production. The demand for hydrogen across sectors such as transportation and industry greatly influences the viability of this technology. Integrating wastewater hydrogen generation into existing wastewater treatment plants (WWTPs) presents a dual advantage: boosting overall wastewater treatment plant’s efficiency while yielding a valuable hydrogen fuel resource. This integration not only mitigates the environmental impact of wastewater disposal but also curtails the release of hazardous compounds into the environment.
Furthermore, exploring renewable energy sources for hydrogen production fosters the generation of "clean energy" with a minimal carbon footprint, thereby advancing environmental sustainability (Farghali et al. 2022). By effectively utilizing both renewable energy and water resources, hydrogen generation from wastewater embodies a holistic approach to resource conservation. It concurrently addresses pressing economic, environmental, and resource management challenges, underscoring its potential as a transformative technology in the quest for sustainable development.
It is worth noting that the economic feasibility of utilizing hydrogen from wastewater is subject to various factors, including geographical location, energy pricing structures, and governmental support mechanisms. Similarly, the environmental impact hinges on the specific methodologies and technologies deployed in the process. While the potential benefits of producing sustainable hydrogen from wastewater are significant in terms of addressing economic, waste treatment, and environmental challenges, its success is contingent upon several key factors.
Continuous advancements in technology will be essential to optimize the efficiency and cost-effectiveness of hydrogen production from wastewater. Additionally, favorable economic conditions, such as incentives or subsidies for renewable energy initiatives, will play a crucial role in fostering the widespread adoption of this technology. Moreover, supportive legislation and regulatory frameworks can provide the necessary impetus for investment and innovation in the field of wastewater hydrogen generation.
In summary, while the production of sustainable hydrogen from wastewater holds tremendous promise for addressing multifaceted challenges, its long-term viability and beneficial impact necessitate ongoing improvements in technology, conducive economic conditions, and supportive policy measures. These combined efforts are essential to ensure the continued advancement and widespread implementation of this transformative technology.
Conclusion
We have reviewed photocatalytic processes aimed at simultaneously producing clean energy and removing pollutants. However, it is essential to address substantial obstacles to ensure continued advancements in this field as follows: Enhancing practical applications of photocatalysis involves improving photocatalyst characteristics, synthesis methods, and stability. This encompasses refining preparation procedures, and developing materials with enhanced light spectrum responsiveness, robust charge transfer capabilities, improved catalytic activity, and prolonged usefulness. To overcome constraints, researchers need to explore active components, multiphase photocatalytic processes, and semiconductor energy-level designs. Understanding photogenerated carrier migration, interface recombination, and the impact of catalysts or contaminants on reaction activity is crucial for surmounting challenges and boosting overall efficiency.
To thoroughly investigate the decomposition processes of organic contaminants in photocatalytic degradation, it is essential to analyze their oxidation potential, which is intricately linked to their chemical structure. Moreover, semiconductors based on S-Scheme heterojunctions play a pivotal role in this research due to their robust redox capacity and enhanced charge carrier separation. They are crucial for examining the concurrent evolution of hydrogen and the degradation of pollutants. Moreover, practical energy production from wastewater necessitates extensive research on reaction parameters and ambient conditions, particularly in areas that have received less attention, such as the impact of pH, electrolytes, temperature, irradiation time, and catalyst dosage on simultaneous hydrogen production and pollutant degradation. The existing uncertainty surrounding responses in aerobic and anaerobic environments underscores the importance of gathering more experimental data to validate the underlying processes.
Addressing obstacles related to wastewater deterioration and photocatalytic hydrogen evolution is crucial. A significant challenge lies in determining the source of hydrogen, often achieved using antibiotics as sacrificial reagents, to gain a comprehensive understanding of the waste-to-energy conversion process. Additionally, optimizing the process by initiating anaerobic photocatalytic hydrogen generation enhances wastewater treatment efficacy. To achieve thorough and efficient pollutant degradation, it is essential to analyze the by-products produced under anaerobic conditions. Also, before being employed extensively for practical wastewater treatment, the photocatalytic hydrogen generation method for pollutant degradation primarily remains within the realm of laboratory research. The fundamental challenge lies in the complexity of the pollutants found in real wastewater, often comprising a diverse array of unidentified contaminants, and necessitating significant investment expenditures.
Data availability
No data were used for the research described in the article.
References
Abdel-Aziz HM et al (2019) Carbamazepine removal from aqueous solution by green synthesis zero-valent Iron/Cu nanoparticles with ficus benjamina leaves’ extract. Int J Environ Res 13:843–852. https://doi.org/10.1007/s41742-019-00220-w
Ahmad H et al (2015) Hydrogen from photo-catalytic water splitting process: a review. Renew Sustain Energy Rev 43:599–610. https://doi.org/10.1016/j.rser.2014.10.101
Ahmed MA, Mohamed AA (2023a) Recent progress in semiconductor/graphene photocatalysts: synthesis, photocatalytic applications, and challenges. RSC Adv 13:421–439. https://doi.org/10.1039/D2RA07225D
Ahmed MA, Mohamed AA (2023b) Advances in ultrasound-assisted synthesis of photocatalysts and sonophotocatalytic processes: a review. iScience 27:108583. https://doi.org/10.1016/j.isci.2023.108583
Alkhatib II et al (2020) Metal-organic frameworks for photocatalytic CO2 reduction under visible radiation: a review of strategies and applications. Catal Today 340:209–224. https://doi.org/10.1016/j.cattod.2018.09.032
Bai J et al (2018) Integrating MoS2 on sulfur-doped porous g-C3N4 iostype heterojunction hybrids enhances visible-light photocatalytic performance. J Alloy Compound 768:766–774. https://doi.org/10.1016/j.jallcom.2018.07.286
Bao S et al (2021) Amino-assisted AHMT anchored on graphene oxide as high performance adsorbent for efficient removal of Cr (VI) and Hg (II) from aqueous solutions under wide pH range. J Hazard Mater 416:125825. https://doi.org/10.1016/j.jhazmat.2021.125825
Belousov AS et al (2022) Metal–organic framework-based heterojunction photocatalysts for organic pollutant degradation: design, construction, and performances. J Chem Technol Biotechnol 97:2675–2693. https://doi.org/10.1002/jctb.7091
Bu D et al (2023) Co-catalyst free direct Z–scheme photocatalytic system with simultaneous hydrogen evolution and degradation of organic pollutants. Int J Hydro Energy 48:576–585. https://doi.org/10.1016/j.ijhydene.2022.09.196
Caudillo-Flores U et al (2018) Bimetallic Pt-Pd co-catalyst Nb-doped TiO2 materials for H2 photo-production under UV and Visible light illumination. Appl Catal B 238:533–545. https://doi.org/10.1016/j.apcatb.2018.07.047
Chen X et al (2010) Semiconductor-based photocatalytic hydrogen generation. Chem Rev 110:6503–6570. https://doi.org/10.1021/cr1001645
Chen S et al (2017) Particulate photocatalysts for overall water splitting. Nat Rev Mater 2:1–17. https://doi.org/10.1038/natrevmats.2017.50
Chen Z et al (2022c) Achieving simultaneous hydrogen evolution and organic pollutants degradation through the modification of Ag3PO4 using Cs2AgBiBr6 quantum dots and graphene hydrogel. Sep Purif Technol 302:122079. https://doi.org/10.1016/j.seppur.2022.122079
Chen J-L et al (2022a) Cu2O-loaded TiO2 heterojunction composites for enhanced photocatalytic H2 production. J Mol Struct 1247:131294. https://doi.org/10.1016/j.molstruc.2021.131294
Chen R et al (2022bd) Rational designing of dual-functional photocatalysts for simultaneous hydrogen generation and organic pollutants degradation over Cd0.5Mn0.5S/CoP. Int J Hydro Energy 47:32921–32927. https://doi.org/10.1016/j.ijhydene.2022.07.174
Chen Z et al (2022b) S-scheme Cs2AgBiBr6/Ag3PO4 heterojunction with efficient photocatalysis performance for H2 production and organic pollutant degradation under visible light. Sep Purif Technol 295:121250. https://doi.org/10.1016/j.seppur.2022.121250
Chen R et al (2022e) Highly efficient simultaneous hydrogen evolution and organic pollutants removal using CdS decorated with CoP nanosheet under visible light. Chem Eng J Adv 11:100339. https://doi.org/10.1016/j.ceja.2022.100339
Cui K et al (2020) Facile synthesis of intercalated Z-scheme Bi2O4/g-C3N4 composite photocatalysts for effective removal of 2-Mercaptobenzothiazole: degradation pathways and mechanism. J Taiwan Inst Chem Eng 111:212–221. https://doi.org/10.1016/j.jtice.2020.03.007
Ding W et al (2023) Ultrathin 2D ZnGa-borate-LDH nanosheets for boosting dye-sensitized photocatalytic coupled reaction of H2 production with pollutant degradation. Colloid Surf a: Physicochem Eng Asp 657:130575. https://doi.org/10.1016/j.colsurfa.2022.130575
Dong W-H et al (2017) Coupling of photodegradation of RhB with photoreduction of CO2 over rGO/SrTi0. 95Fe0. 05O3− δ catalyst: a strategy for one-pot conversion of organic pollutants to methanol and ethanol. J Catal 349:218–225. https://doi.org/10.1016/j.jcat.2017.02.004
Dong H et al (2018) Synthesis of g-C3N4 by different precursors under burning explosion effect and its photocatalytic degradation for tylosin. Appl Catal B 230:65–76. https://doi.org/10.1016/j.apcatb.2018.02.044
Dong J et al (2023) Construction of Z-scheme MnO2/BiOBr heterojunction for photocatalytic ciprofloxacin removal and CO2 reduction. Acta Phys-Chim Sin 39:2212011
Farghali M et al (2022) Integration of biogas systems into a carbon zero and hydrogen economy: a review. Environ Chem Lett 20:2853–2927. https://doi.org/10.1007/s10311-022-01468-z
Farghali M et al (2023) Social, environmental, and economic consequences of integrating renewable energies in the electricity sector: a review. Environ Chem Lett 21:1381–1418. https://doi.org/10.1007/s10311-023-01587-1
Fu J et al (2019) Ultrathin 2D/2D WO3/g-C3N4 step-scheme H2-production photocatalyst. Appl Catal B: Environ 243:556–565. https://doi.org/10.1016/j.apcatb.2018.11.011
Gao B et al (2016) Identification of intermediates and transformation pathways derived from photocatalytic degradation of five antibiotics on ZnIn2S4. Chem Eng J 304:826–840. https://doi.org/10.1016/j.cej.2016.07.029
Guo W et al (2023) Progress on simultaneous photocatalytic degradation of pollutants and production of clean energy: a review. Chemosphere 339:139486. https://doi.org/10.1016/j.chemosphere.2023.139486
Han W et al (2016) Techno-economic evaluation of a combined bioprocess for fermentative hydrogen production from food waste. Biores Technol 202:107–112. https://doi.org/10.1016/j.biortech.2015.11.072
Hantoko D et al (2024) Carbon–neutral hydrogen production by catalytic methane decomposition: a review. Environ Chem Lett. https://doi.org/10.1007/s10311-024-01732-4
Hao X et al (2019) Self-constructed facet junctions on hexagonal CdS single crystals with high photoactivity and photostability for water splitting. Appl Catal B 244:694–703. https://doi.org/10.1016/j.apcatb.2018.12.006
Hisatomi T et al (2014) Recent advances in semiconductors for photocatalytic and photoelectrochemical water splitting. Chem Soc Rev 43:7520–7535. https://doi.org/10.1039/C3CS60378D
Hu X et al (2021) Metal-ion-assisted construction of cyano group defects in g-C3N4 to simultaneously degrade wastewater and produce hydrogen. Chem Eng J 423:130278. https://doi.org/10.1016/j.cej.2021.130278
Huang W-Y et al (2019) C-H activation derived CPPs for photocatalytic hydrogen production excellently accelerated by a DMF cosolvent. J Mater Chem A 7:24222–24230. https://doi.org/10.1039/C9TA06444C
Hyun JK et al (2013) Nanowire heterostructures. Annu Rev Mater Res 43:451–479. https://doi.org/10.1146/annurev-matsci-071312-121659
Ismael M (2021) Ferrites as solar photocatalytic materials and their activities in solar energy conversion and environmental protection: a review. Solar Energy Mater Solar Cell 219:110786
Jabbar ZH et al (2023) Design and construction of a robust ternary Bi5O7I/Cd0 5Zn0 5S/CuO photocatalytic system for boosted photodegradation of antibiotics via dual-S-scheme mechanisms: environmental factors and degradation intermediates. Environ Res 234:116554. https://doi.org/10.1016/j.envres.2023.116554
Jang JS et al (2007) Optimization of CdS/TiO2 nano-bulk composite photocatalysts for hydrogen production from Na2S/Na2SO3 aqueous electrolyte solution under visible light (λ≥ 420 nm). J Photochem Photobiol, A 188:112–119. https://doi.org/10.1016/j.jphotochem.2006.11.027
Jia J et al (2023) Recent advances on g–C3N4–based Z-scheme photocatalysts: Structural design and photocatalytic applications. Int J Hydrogen Energy 48:196–231. https://doi.org/10.1016/j.ijhydene.2022.09.272
Jiang X-H et al (2018) Photodegradation of organic pollutants coupled with simultaneous photocatalytic evolution of hydrogen using quantum-dot-modified g-C3N4 catalysts under visible-light irradiation. ACS Sustain Chem Eng 6:12695–12705. https://doi.org/10.1021/acssuschemeng.8b01695
Jiang X-H et al (2021) Isotypic heterojunction based on Fe-doped and terephthalaldehyde-modified carbon nitride for improving photocatalytic degradation with simultaneous hydrogen production. Chin Chem Lett 32:2782–2786. https://doi.org/10.1016/j.cclet.2021.01.011
Kashiwaya S et al (2018) Fermi level positions and induced band bending at single crystalline anatase (101) and (001) surfaces: origin of the enhanced photocatalytic activity of facet engineered crystals. Adv Energy Mater 8:1802195. https://doi.org/10.1002/aenm.201802195
Kausar F et al (2022) Recent trends in photocatalytic water splitting using titania based ternary photocatalysts-a review. Int J Hydro Energy 47:22371–22402. https://doi.org/10.1016/j.ijhydene.2022.05.058
Koe WS et al (2020) An overview of photocatalytic degradation: photocatalysts, mechanisms, and development of photocatalytic membrane. Environ Sci Pollut Res 27:2522–2565. https://doi.org/10.1007/s11356-019-07193-5
Kumar R et al (2020a) Photocatalytic degradation of cefoxitin sodium antibiotic using novel BN/CdAl2O4 composite. J Clean Prod 246:119076. https://doi.org/10.1016/j.jclepro.2019.119076
Kumar A et al (2020b) Bio-inspired and biomaterials-based hybrid photocatalysts for environmental detoxification: a review. Chem Eng J 382:122937. https://doi.org/10.1016/j.cej.2019.122937
Kumar A et al (2021) Construction of dual Z-scheme g-C3N4/Bi4Ti3O12/Bi4O5I2 heterojunction for visible and solar powered coupled photocatalytic antibiotic degradation and hydrogen production: Boosting via I−/I3− and Bi3+/Bi5+ redox mediators. Appl Catal B: Environ 284:119808. https://doi.org/10.1016/j.apcatb.2020.119808
Kumar A et al (2023) A review on S-scheme and dual S-scheme heterojunctions for photocatalytic hydrogen evolution, water detoxification and CO2 reduction. Fuel 333:126267. https://doi.org/10.1016/j.fuel.2022.126267
Kumaravel V et al (2019) Photocatalytic hydrogen production: role of sacrificial reagents on the activity of oxide, carbon, and sulfide catalysts. Catalysts 9:276. https://doi.org/10.3390/catal9030276
Kumari P et al (2022) Engineering Schottky-like and heterojunction materials for enhanced photocatalysis performance–a review. Mater Adv 3:2309–2323. https://doi.org/10.1039/D1MA01062J
Leung DY et al (2010) Hydrogen production over titania-based photocatalysts. Chemsuschem 3:681–694. https://doi.org/10.1002/cssc.201000014
Li D et al (2018) A dual-function photocatalytic system for simultaneous separating hydrogen from water splitting and photocatalytic degradation of phenol in a twin-reactor. Appl Catal B 239:268–279. https://doi.org/10.1016/j.apcatb.2018.08.010
Li S et al (2020) Fabrication of highly active Z-scheme Ag/g-C3N4-Ag-Ag3PO4 (1 1 0) photocatalyst photocatalyst for visible light photocatalytic degradation of levofloxacin with simultaneous hydrogen production. Chem Eng J 382:122394. https://doi.org/10.1016/j.cej.2019.122394
Li B et al (2021b) All-solid-state direct Z-scheme NiTiO3/Cd 0.5Zn0.5 S heterostructures for photocatalytic hydrogen evolution with visible light. J Mater Chem A 9:10270–10276. https://doi.org/10.1039/D1TA01220G
Li K et al (2021a) MXenes as noble-metal-alternative co-catalysts in photocatalysis. Chin J Catal 42:3–14. https://doi.org/10.1016/S1872-2067(20)63630-0
Li S et al (2021b) Boosted electron-transfer by coupling Ag and Z-scheme heterostructures in CdSe-Ag-WO3-Ag for excellent photocatalytic H2 evolution with simultaneous degradation. Chem Eng J 417:129298. https://doi.org/10.1016/j.cej.2021.129298
Li X et al (2022a) Principles, synthesis and applications of dual Z-scheme photocatalysts. Coord Chem Rev 467:214596. https://doi.org/10.1016/j.ccr.2022.214596
Li Y et al (2022b) Review on g-C3N4-based S-scheme heterojunction photocatalysts. J Mater Sci Technol 125:128–144. https://doi.org/10.1016/j.jmst.2022.02.035
Li Y et al (2022c) A 3D C@TiO2 multishell nanoframe for simultaneous photothermal catalytic hydrogen generation and organic pollutant degradation. J Colloid Interfac Sci 609:535–546. https://doi.org/10.1016/j.jcis.2021.11.052
Li T et al (2024) S-scheme heterojunction in photocatalytic hydrogen production. J Mater Sci Technol 169:82–104. https://doi.org/10.1016/j.jmst.2023.04.049
Lin W-C et al (2021) Effect of energy bandgap and sacrificial agents of cyclopentadithiophene-based polymers for enhanced photocatalytic hydrogen evolution. Appl Catal B 298:120577. https://doi.org/10.1016/j.apcatb.2021.120577
Lin Y et al (2022a) Construction and preparation of coated Z-scheme NaGdF4: Nd3+, Yb3+, Tm3+@ CdS-Pd@ NiO photocatalyst with core-shell structure for organic pollutant degradation with synchronous hydrogen evolution. Appl Catal A 643:118758. https://doi.org/10.1016/j.apcata.2022.118758
Lin X et al (2022b) A dual-functional integrated Ni5P4/g-C3N4 S-scheme heterojunction for high performance synchronous photocatalytic hydrogen evolution and multi-contaminant removal with a waste-to-energy conversion. J Molecul Liq 366:120147. https://doi.org/10.1016/j.molliq.2022.120147
Liras M, Barawi M (2019) Hybrid materials based on conjugated polymers and inorganic semiconductors as photocatalysts: from environmental to energy applications. Chem Soc Rev 48:5454–5487. https://doi.org/10.1039/C9CS00377K
Liu J et al (2015) Metal-free efficient photocatalyst for stable visible water splitting via a two-electron pathway. Science 347:970–974. https://doi.org/10.1126/science.aaa3145
Liu J et al (2018) Efficient photocatalytic hydrogen evolution on N-deficient g-C3N4 achieved by a molten salt post-treatment approach. Appl Catal B 238:465–470. https://doi.org/10.1016/j.apcatb.2018.07.021
Liu M et al (2020a) Adsorption enhanced photocatalytic degradation sulfadiazine antibiotic using porous carbon nitride nanosheets with carbon vacancies. Chem Eng J 382:123017
Liu H et al (2020b) CoP nanoparticles as cocatalyst modified the CdS/NiWO 4 p–n heterojunction to produce hydrogen efficiently. New J Chem 44:1426–1438. https://doi.org/10.1039/C9NJ05977F
Liu X et al (2021) Advances in 2D/2D Z-scheme heterojunctions for photocatalytic applications. Solar RRL 5:2000397. https://doi.org/10.1002/solr.202000397
Liu Z et al (2022a) Semiconductor heterojunctions for photocatalytic hydrogen production and Cr (VI) reduction: A review. Mater Res Bull 147:111636. https://doi.org/10.1016/j.materresbull.2021.111636
Liu Z et al (2022b) Solar-light-driven Z-scheme CaF2:Yb3+, Er3+@ZnTiO3/Au/CdS photocatalyst with efficient charge transfer and light utilization for enhanced norfloxacin degradation with simultaneous hydrogen evolution. J Phys Chem Solids 161:110493. https://doi.org/10.1016/j.jpcs.2021.110493
Liu S et al (2023) Cu atoms on UiO-66-NH2/ZnIn2S4 nanosheets enhance photocatalytic performance for recovering hydrogen energy from organic wastewater treatment. Appl Catal B 330:122572. https://doi.org/10.1016/j.apcatb.2023.122572
Long Z et al (2020) Historical development and prospects of photocatalysts for pollutant removal in water. J Hazard Mater 395:122599. https://doi.org/10.1016/j.jhazmat.2020.122599
Low J et al (2017) Heterojunction Photocatalysts. Adv Mater 29:1601694. https://doi.org/10.1002/adma.201601694
Lu F, Astruc D (2020) Nanocatalysts and other nanomaterials for water remediation from organic pollutants. Coord Chem Rev 408:213180. https://doi.org/10.1016/j.ccr.2020.213180
Lu J et al (2023b) Review on multi-dimensional assembled S-scheme heterojunction photocatalysts. J Mater Sci Technol 160:214–239. https://doi.org/10.1016/j.jmst.2023.03.027
Lu N et al (2023a) Electrospun semiconductor-based nano-heterostructures for photocatalytic energy conversion and environmental remediation: opportunities and challenges. Energy Environ Mater 6:e12338. https://doi.org/10.1002/eem2.12338
Ma G et al (2020) Amorphous Ti (IV)-modified flower-like ZnIn2S4 microspheres with enhanced hydrogen evolution photocatalytic activity and simultaneous wastewater purification. J Mater Chem c 8:2693–2699. https://doi.org/10.1039/C9TC05456A
Malefane ME et al (2023) Understanding the principles and applications of dual Z-scheme heterojunctions: how far can we go? J Phys Chem Lett 14:1029–1045. https://doi.org/10.1021/acs.jpclett.2c03387
Méndez JO et al (2014) Production of hydrogen by water photo-splitting over commercial and synthesised Au/TiO2 catalysts. Appl Catal B 147:439–452. https://doi.org/10.1016/j.apcatb.2013.09.029
Naseri A et al (2022) Recent advances on dual-functional photocatalytic systems for combined removal of hazardous water pollutants and energy generation. Res Chem Intermed 48:911–933. https://doi.org/10.1007/s11164-022-04677-x
Nie C et al (2024) Advancements in S-scheme heterojunction materials for photocatalytic environmental remediation. J Mater Sci Techno 169:182–198
Osman AI et al (2020) Exploring the photocatalytic hydrogen production potential of titania doped with alumina derived from foil waste. Int J Hydro Energy 45:34494–34502. https://doi.org/10.1016/j.ijhydene.2020.02.065
Osman AI et al (2023) Biofuel production, hydrogen production and water remediation by photocatalysis, biocatalysis and electrocatalysis. Environ Chem Lett 21:1315–1379. https://doi.org/10.1007/s10311-023-01581-7
Osman AI et al (2024a) Coordination-driven innovations in low-energy catalytic processes: advancing sustainability in chemical production. Coord Chem Rev 514:215900
Osman AI et al (2024b) Synergy between vertical farming and the hydrogen economy. Environ Chem Lett 22:1–6. https://doi.org/10.1007/s10311-023-01648-5
Ou H et al (2019) Solvated electrons for photochemistry syntheses using conjugated carbon nitride polymers. ACS Catal 9:2949–2955. https://doi.org/10.1021/acscatal.9b00314
Palani G et al (2022) Metal-doped graphitic carbon nitride nanomaterials for photocatalytic environmental applications—a review. Nanomaterials 12:1754. https://doi.org/10.3390/nano12101754
Pandis PK et al (2022) Key points of advanced oxidation processes (AOPs) for wastewater, organic pollutants and pharmaceutical waste treatment: a mini review. Chem Eng 6:8. https://doi.org/10.3390/chemengineering6010008
Patsoura A et al (2006) Enhancement of photoinduced hydrogen production from irradiated Pt/TiO2 suspensions with simultaneous degradation of azo-dyes. Appl Catal B 64:171–179. https://doi.org/10.1016/j.apcatb.2005.11.015
Pei L et al (2019) Hierarchical Zn1-xCdxS microclusters with superior visible-light-driven photocatalytic hydrogen generation performance. J Alloy Compd 809:151869. https://doi.org/10.1016/j.jallcom.2019.151869
Qu Z et al (2017) A new visible-light-induced Z-scheme photocatalytic system: Er3+:Y3Al5O12/(MoS2/NiGa2O4)-(BiVO4/PdS) for refractory pollutant degradation with simultaneous hydrogen evolution. Molecul Catal 441:10–20. https://doi.org/10.1016/j.mcat.2017.08.001
Qu Z et al (2018) Carbon quantum dots/KNbO3 hybrid composites with enhanced visible-light driven photocatalytic activity toward dye waste-water degradation and hydrogen production. Molecul Catal 445:1–11. https://doi.org/10.1016/j.mcat.2017.11.002
Quddus F et al (2023) Environmentally benign nanoparticles for the photocatalytic degradation of pharmaceutical drugs. Catalysts 13:511. https://doi.org/10.3390/catal13030511
Ran J et al (2014) Earth-abundant cocatalysts for semiconductor-based photocatalytic water splitting. Chem Soc Rev 43:7787–7812. https://doi.org/10.1039/C3CS60425J
Rana A et al (2021) Pharmaceutical pollutant as sacrificial agent for sustainable synergistic water treatment and hydrogen production via novel Z- scheme Bi7O9I3/B4C heterojunction photocatalysts. J Molecul Liq 343:117652. https://doi.org/10.1016/j.molliq.2021.117652
Rana S et al (2023) Recent advances in perovskite-based Z-scheme and S-scheme heterojunctions for photocatalytic CO2 reduction. Chemosphere 339:139765. https://doi.org/10.1016/j.chemosphere.2023.139765
Rathi BS et al (2021) A review on effective removal of emerging contaminants from aquatic systems: current trends and scope for further research. J Hazard Mater 409:124413. https://doi.org/10.1016/j.jhazmat.2020.124413
Reichert R et al (2015) Au/TiO2 photo (electro) catalysis: the role of the Au cocatalyst in photoelectrochemical water splitting and photocatalytic H2 evolution. J Phys Chem C 119:24750–24759. https://doi.org/10.1021/acs.jpcc.5b08428
Ren X et al (2022) The fabrication of Pd single atoms/clusters on COF layers as co-catalysts for photocatalytic H2 evolution. ACS Appl Mater Interfac 14:6885–6893. https://doi.org/10.1021/acsami.1c23465
Sajjadi M et al (2021) Lignin-derived (nano) materials for environmental pollution remediation: current challenges and future perspectives. Int J Biol Macromol 178:394–423. https://doi.org/10.1016/j.ijbiomac.2021.02.165
Salcedo-Abraira P et al (2021) Nickel phosphonate MOF as efficient water splitting photocatalyst. Nano Res 14:450–457. https://doi.org/10.1007/s12274-020-3056-6
Sanakousar F et al (2022) Recent progress on visible-light-driven metal and non-metal doped ZnO nanostructures for photocatalytic degradation of organic pollutants. Mater Sci Semicond Process 140:106390. https://doi.org/10.1016/j.mssp.2021.106390
Shao X et al (2022) In-situ irradiated XPS investigation on 2D/1D Cd0.5Zn0.5S/Nb2O5 S-scheme heterojunction photocatalysts for simultaneous promotion of antibiotics removal and hydrogen evolution. Colloid Surfac a: Physicochem Eng Asp 652:129846. https://doi.org/10.1016/j.colsurfa.2022.129846
Sharma P et al (2023a) Current scenario in ternary metal indium sulfides-based heterojunctions for photocatalytic energy and environmental applications: a review. Mater Today Commun. https://doi.org/10.1016/j.mtcomm.2023.106741
Sharma P et al (2023b) Recent progress in photocatalytic applications of metal tungstates based Z-scheme and S-scheme heterojunctions. J Ind Eng Chem 132:1–12. https://doi.org/10.1016/j.jiec.2023.11.004
She H et al (2019) Synthesis of non-noble metal nickel doped sulfide solid solution for improved photocatalytic performance. Appl Catal B 245:439–447. https://doi.org/10.1016/j.apcatb.2019.01.002
Shen H et al (2017) All-solid-state Z-scheme system of RGO-Cu2O/Fe2O3 for simultaneous hydrogen production and tetracycline degradation. Mater Today Energy 5:312–319. https://doi.org/10.1016/j.mtener.2017.07.008
Shen H et al (2020) 2D/2D/3D architecture Z-scheme system for simultaneous H2 generation and antibiotic degradation. Fuel 280:118618. https://doi.org/10.1016/j.fuel.2020.118618
Singh S et al (2020) Hybrid organic–inorganic materials and composites for photoelectrochemical water splitting. ACS Energy Lett 5:1487–1497. https://doi.org/10.1021/acsenergylett.0c00327
Staffell I et al (2019) The role of hydrogen and fuel cells in the global energy system. Energy Environ Sci 12:463–491. https://doi.org/10.1039/C8EE01157E
Stelo F et al (2020) Recent advances in Bi2MoO6 based Z-scheme heterojunctions for photocatalytic degradation of pollutants. J Alloy Compd 829:154591. https://doi.org/10.1016/j.jallcom.2020.154591
Su B et al (2022) S-scheme CoTiO3/Cd9. 51Zn0. 49S10 heterostructures for visible-light driven photocatalytic CO2 reduction. J Mater Sci Technol 124:164–170. https://doi.org/10.1016/j.jmst.2022.01.030
Su B et al (2023) S-Scheme Co9S8@ Cd0 8Zn0 2S-DETA hierarchical nanocages bearing organic CO2 activators for photocatalytic syngas production. Adv Energy Mater 13:2203290. https://doi.org/10.1002/aenm.202203290
Sun C et al (2021) Photocatalytic Hd production with simultaneous wastewater purification over flower-like 1T/2H-MoS2-decorated CNT/CNU isotype heterojunction photocatalyst. Appl Surfac Sci 569:151072. https://doi.org/10.1016/j.apsusc.2021.151072
Tan C-E et al (2020) Facile approach for Z-scheme type Pt/g-C3N4/SrTiO3 heterojunction semiconductor synthesis via low-temperature process for simultaneous dyes degradation and hydrogen production. Int J Hydro Energy. 45:13330–13339. https://doi.org/10.1016/j.ijhydene.2020.03.034
Tang L et al (2019) Fabrication of Z-scheme photocatalyst, Er3+:Y3Al5O12@NiGa2O4-MWCNTs-WO3, and visible-light photocatalytic activity for degradation of organic pollutant with simultaneous hydrogen evolution. Renew Energy 138:474–488. https://doi.org/10.1016/j.renene.2019.01.113
Vaiano V et al (2015) Simultaneous production of CH4 and H2 from photocatalytic reforming of glucose aqueous solution on sulfated Pd-TiO2 catalysts. Oil Gas Sci Technol-Revue d’IFP Energies Nouvelles 70:891–902. https://doi.org/10.2516/ogst/2014062
Wang J, Zhuan R (2020) Degradation of antibiotics by advanced oxidation processes: an overview. Sci Total Environ 701:135023. https://doi.org/10.1016/j.scitotenv.2019.135023
Wang Y et al (2013) Visible light driven type II heterostructures and their enhanced photocatalysis properties: a review. Nanoscale 5:8326–8339. https://doi.org/10.1039/C3NR01577G
Wang M et al (2015) Integration of organometallic complexes with semiconductors and other nanomaterials for photocatalytic H2 production. Coord Chem Rev 287:1–14. https://doi.org/10.1016/j.ccr.2014.12.005
Wang W et al (2017) Recent progress in metal-organic frameworks for applications in electrocatalytic and photocatalytic water splitting. Adv Sci 4:1600371. https://doi.org/10.1002/advs.201600371
Wang Z et al (2019a) Recent developments in heterogeneous photocatalysts for solar-driven overall water splitting. Chem Soc Rev 48:2109–2125. https://doi.org/10.1039/C8CS00542G
Wang S et al (2019b) Crystal facet engineering of photoelectrodes for photoelectrochemical water splitting. Chem Rev 119:5192–5247. https://doi.org/10.1021/acs.chemrev.8b00584
Wang J et al (2019c) An anti-symmetric dual (ASD) Z-scheme photocatalytic system: (ZnIn2S4/Er3+:Y3Al5O12@ZnTiO3/CaIn2S4) for organic pollutants degradation with simultaneous hydrogen evolution. Int J Hydro Energy 44:6592–6607. https://doi.org/10.1016/j.ijhydene.2019.01.214
Wang Z et al (2021) Advances in designing heterojunction photocatalytic materials. Chin J Catal 42:710–730
Wang G et al (2022a) 2D materials and heterostructures for photocatalytic water-splitting: a theoretical perspective. J Phys D Appl Phys 55:293002. https://doi.org/10.1088/1361-6463/ac5771
Wang L et al (2022a) Effects of various alcohol sacrificial agents on hydrogen evolution based on CoS2@SCN nanomaterials and its mechanism. Chemosphere 286:131558. https://doi.org/10.1016/j.chemosphere.2021.131558
Wang K et al (2022c) 0D/3D Bi3TaO7/ZnIn2S4 heterojunction photocatalyst towards degradation of antibiotics coupled with simultaneous H2 evolution: In situ irradiated XPS investigation and S-scheme mechanism insight. Appl Surfac Sci 596:153444. https://doi.org/10.1016/j.apsusc.2022.153444
Wang L et al (2022d) Effects of various alcohol sacrificial agents on hydrogen evolution based on CoS2@ SCN nanomaterials and its mechanism. Chemosphere 286:131558. https://doi.org/10.1016/j.chemosphere.2021.131558
Wang Y et al (2022e) Hydrogen generation from photocatalytic treatment of wastewater containing pharmaceuticals and personal care products by Oxygen-doped crystalline carbon nitride. Separ Purif Technol 296:121425. https://doi.org/10.1016/j.seppur.2022.121425
Wang Y et al (2023) Plate-on-plate structured MoS2/Cd0.6Zn0.4S Z-scheme heterostructure with enhanced photocatalytic hydrogen production activity via hole sacrificial agent synchronously strengthen half-reactions. J Colloid Interfac Sci 630:341–351. https://doi.org/10.1016/j.jcis.2022.10.053
Wang T et al (2024) Efficient visible-light photocatalysis of chloramphenicol using novel engineered biochar-based Ti-doped Bi2WO6 composite: Mechanisms, degradation pathways, and applications. Separation and Purification Technology, 332: 125780. https://doi.org/10.1016/j.seppur.2023.125780
Wei Z et al (2019) Photocatalytic hydrogen evolution with simultaneous antibiotic wastewater degradation via the visible-light-responsive bismuth spheres-g-C3N4 nanohybrid: Waste to energy insight. Chem Eng J 358:944–954. https://doi.org/10.1016/j.cej.2018.10.096
Wei Z et al (2020b) Simultaneous visible-light-induced hydrogen production enhancement and antibiotic wastewater degradation using MoS2@ ZnxCd1-xS: Solid-solution-assisted photocatalysis. Chin J Catal 41:103–113. https://doi.org/10.1016/S1872-2067(19)63479-0
Wei Z et al (2020a) A review on photocatalysis in antibiotic wastewater: pollutant degradation and hydrogen production. Chin J Catal 41:1440–1450
Xu Z et al (2017) Waste-to-energy conversion on graphitic carbon nitride: utilizing the transformation of macrolide antibiotics to enhance photoinduced hydrogen production. ACS Sustain Chem Eng 5:9667–9672. https://doi.org/10.1021/acssuschemeng.7b03088
Xu F et al (2020a) Nitrogen-rich graphitic carbon nitride nanotubes for photocatalytic hydrogen evolution with simultaneous contaminant degradation. J Colloid Interfac Sci 560:555–564. https://doi.org/10.1016/j.jcis.2019.10.089
Xu Q et al (2020b) S-Scheme heterojunction photocatalyst. Chem 6:1543–1559. https://doi.org/10.1016/j.chempr.2020.06.010
Xu Q et al (2023) Problems and mistakes for electron transfer mechanism in Z-scheme photocatalytic system. Acta Physico-Chim Sinica. https://doi.org/10.3866/PKU.WHXB202211009
Yadav AA et al (2022) Visible light-responsive CeO2/MoS2 composite for photocatalytic hydrogen production. Catalysts 12:1185. https://doi.org/10.3390/catal12101185
Yang J et al (2013) Roles of cocatalysts in photocatalysis and photoelectrocatalysis. Acc Chem Res 46:1900–1909. https://doi.org/10.1021/ar300227e
Yang L et al (2021a) Significant enhancement of photocatalytic H2 production simultaneous with dye degradation over Ni2P modified In2O3 nanocomposites. Sep Purif Technol 263:118366. https://doi.org/10.1016/j.seppur.2021.118366
Yang R et al (2021b) Highly efficient photocatalytic hydrogen evolution and simultaneous formaldehyde degradation over Z-scheme ZnIn2S4-NiO/BiVO4 hierarchical heterojunction under visible light irradiation. Chem Eng J 423:130164. https://doi.org/10.1016/j.cej.2021.130164
Yang H et al (2022) Inorganic-organic hybrid photocatalysts: syntheses, mechanisms, and applications. Chin J Catal 43:2111–2140. https://doi.org/10.1016/S1872-2067(22)64096-8
Yang L et al (2023) S-scheme In2S3/Zn3In2S6 microsphere for efficient photocatalytic H2 evolution with simultaneous photodegradation of bisphenol A. Appl Surfac Sci. 612:155848. https://doi.org/10.1016/j.apsusc.2022.155848
Yu C et al (2014) Design and fabrication of heterojunction photocatalysts for energy conversion and pollutant degradation. Chin J Catal 35:1609–1618. https://doi.org/10.1016/S1872-2067(14)60170-4
Yuan Y-J et al (2018) Cadmium sulfide-based nanomaterials for photocatalytic hydrogen production. J Mater Chem A 6:11606–11630. https://doi.org/10.1039/C8TA00671G
Zhang S et al (2017) Photocatalytic wastewater purification with simultaneous hydrogen production using MoS2 QD-decorated hierarchical assembly of ZnIn2S4 on reduced graphene oxide photocatalyst. Water Res 121:11–19. https://doi.org/10.1016/j.watres.2017.05.013
Zhang M et al (2019) Rational design 2D/2D BiOBr/CDs/g-C3N4 Z-scheme heterojunction photocatalyst with carbon dots as solid-state electron mediators for enhanced visible and NIR photocatalytic activity: kinetics, intermediates, and mechanism insight. J Catal 369:469–481. https://doi.org/10.1016/j.jcat.2018.11.029
Zhang L et al (2020) Metal-free carbon quantum dots implant graphitic carbon nitride: enhanced photocatalytic dye wastewater purification with simultaneous hydrogen production. Int J Mol Sci 21:1052. https://doi.org/10.3390/ijms21031052
Zhang X et al (2021) In situ fabrication of a novel S-scheme heterojunction photocatalyts Bi2O3/P-C3N4 to enhance levofloxacin removal from water. Sep Purif Technol 268:118691. https://doi.org/10.1016/j.seppur.2021.118691
Zhang G et al (2022) Metal sulfides for photocatalytic hydrogen production: current development and future challenges. Solar Rrl 6:2200587. https://doi.org/10.1002/solr.202200587
Zhang S et al (2023) Interfacial oxygen vacancy modulated Ag3PO4 @MoS2 Z-scheme system for efficient photocatalytic hydrogen recovery from antibiotic wastewater. Appl Catal B: Environ 330:122584. https://doi.org/10.1016/j.apcatb.2023.122584
Zhao Y et al (2021a) Recent advances in photocatalytic decomposition of water and pollutants for sustainable application. Chemosphere 276:130201
Zhao W et al (2021b) A novel Z-scheme CeO2/g-C3N4 heterojunction photocatalyst for degradation of Bisphenol A and hydrogen evolution and insight of the photocatalysis mechanism. J Mater Sci Technol 85:18–29. https://doi.org/10.1016/j.jmst.2020.12.064
Zhao H et al (2022) Transition-metal-based cocatalysts for photocatalytic water splitting. Small Struct 3:2100229. https://doi.org/10.1002/sstr.202100229
Zheng Q et al (2016) Visible-light-responsive graphitic carbon nitride: rational design and photocatalytic applications for water treatment. Environ Sci Technol 50:12938–12948. https://doi.org/10.1021/acs.est.6b02579
Zheng J et al (2020) Design of porous double-shell Cu2O@CuCo2O4 Z-Scheme hollow microspheres with superior redox property for synergistic photocatalytic degradation of multi-pollutants. Chem Eng J 389:124339. https://doi.org/10.1016/j.cej.2020.124339
Zhu R et al (2019) Z scheme system ZnIn2S4/RGO/BiVO4 for hydrogen generation from water splitting and simultaneous degradation of organic pollutants under visible light. Renew Energy 139:22–27. https://doi.org/10.1016/j.renene.2019.02.049
Zou Y et al (2022) Photocatalytic performance and mechanism of hydrogen evolution from water over ZnCdS/Co@CoO in sacrificial agent-free system. Int J Hydro Energy 47:25289–25299
Acknowledgements
This research was funded by the following grants, including the Key Research and Development Program of Shaanxi Province (No. 2023-LL-QY-42), the Xi’an University of Architecture and Technology Research Initiation Grant Program (No. 1960323102), and the Xi’an University of Architecture and Technology Special Program for Cultivation of Frontier Interdisciplinary Fields (No. 1960523142).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Declarations
AIO declares that he is the Editor of Environmental Chemistry Letters.
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kumar, A., Sharma, P., Sharma, G. et al. Simultaneous hydrogen production and photocatalytic pollutant removal: a review. Environ Chem Lett 22, 2405–2424 (2024). https://doi.org/10.1007/s10311-024-01756-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-024-01756-w