Abstract
Global pollution by plastics derived from petroleum has fostered the development of carbon–neutral, biodegradable bioplastics synthesized from renewable resources such as modern biomass, yet knowledge on the impact of bioplastics on ecosystems is limited. Here we review the polylactic acid plastic with focus on synthesis, biodegradability tuning, environmental conversion to microplastics, and impact on microbes, algae, phytoplankton, zooplankton, annelids, mollusk and fish. Polylactic acid is a low weight semi-crystalline bioplastic used in agriculture, medicine, packaging and textile. Polylactic acid is one of the most widely used biopolymers, accounting for 33% of all bioplastics produced in 2021. Although biodegradable in vivo, polylactic acid is not completely degradable under natural environmental conditions, notably under aquatic conditions. Polylactic acid disintegrates into microplastics faster than petroleum-based plastics and may pose severe threats to the exposed biota.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The production of durable plastics started in 1940s (Chia et al. 2021). Plastics became an integral part of human life and as a result a huge amount is being produced every year, with around 370 million tons in 2021 (EuropeanBioplastics 2022). The non-biodegradable character of most petroleum-based plastics in addition to a low rate of recycling, for example only 9% of the total plastic produced until 2015 has been recycled (Almeshal et al. 2020), are the key factors of its accumulation in the natural environment. Plastic wastes are prone to natural weathering processes including ultraviolet radiations, oxidation, and biodegradation, which result into tiny pieces of 1 µm to 5 mm in size, termed as microplastics (Frias and Nash 2019). The presence of microplastics has been reported in almost all types of environmental media, including the ice blocks of Antarctic region (Kelly et al. 2020; Obbard et al. 2014). Marine organisms accidently take up microplastics from their ambient environment with food (Matijaković Mlinarić et al. 2022). As a result, the presence of microplastics has been reported in a wide range of organisms, ranging from zooplankton (Thery et al. 2022) to fish (Alomar et al. 2021), followed by their accumulation in the food chain via trophic transfer (Sarker et al. 2022). Consequently, a wide range of implications has been reported in the exposed individuals ranging from physical damage (Eltemsah and Bøhn 2019) to hormonal disruption (Ismail et al. 2021) and even mortality (Eom et al. 2020), suggesting that the impact of petroleum-based plastic on aquatic ecosystems is becoming severe (Nandhini et al. 2022).
Bioplastics, biodegradable and or compostable, may be considered as a relevant alternative to petroleum-based plastics. Polylactic acid is one of the most widely produced biopolymers which constituted 33% of all the bioplastics produced in 2021 (EuropeanBioplastics 2022). This aliphatic polyester is made from renewable resources (Nanda et al. 2022) and is used in agriculture, medicine and medical devices including temporary implants (Shruti and Kutralam-Muniasamy 2019), as well as for packaging, in automotive, and is now emerging for textile applications. The investigation of its degradation in natural environment is thus of great interest. Although polylactic acid is known for its in vivo biodegradable character, its biodegradation in the natural environment, notably in aquatic, is not so easy. Several review papers have summarized the applications, degradation mechanisms, modifications and ecotoxicological evaluation of polylactic acid over the last few years (Ainali et al. 2022; Chamas et al. 2020; Hubbe et al. 2021; Karamanlioglu et al. 2017; Qin et al. 2021; Ribba et al. 2022; Taib et al. 2022; Zaaba and Jaafar 2020). A comprehensive presentation on polylactic acid including the strategies available to promote biodegradability, the synthesis of microplastics and their impact on aquatic biota is still missing. This is the objective of the present contribution.
Polylactic acid as bioplastic
Polylactic acid is a low weight, in vivo biodegradable and compostable semi-crystalline bio-based polymer, synthesized from natural sources like corn starch, sugarcane or cassava roots (Grigoras 2021). For the very first time polylactic acid was synthesized in 1845 by Théophile-Jules Pelouze (Benninga 1990) and was commercialized in 1990s by Cargill and Dow Chemicals. However, polylactic acid and its copolymers were used as biomedical material during 1970s (Masutani and Kimura 2015). Due to its bio-absorbable and biocompatible nature, polylactic acid has been used in various biomedical applications ranging from instant drug delivery (Hu et al. 2003; Miao et al. 2011) to tissue engineering (Zhang and Ma 2004) (Fig. 1).
Polylactic acid has been used as a potential alternative to petroleum-based plastics in many sectors. In agriculture, polylactic acid is used in greenhouses, drip irrigation and as a protective layer against soil erosion. Medical applications of polylactic acid range from surgery to dentistry. It is also used in automotive industry to produce different parts of vehicles. It is also used in increasing amount in packaging to produce cartoons, cups and plastic lined paper bags
Due to its mechanical properties, for example, Young modulus of 1–3 GPa and elongation at break around 6–7% for poly(l-lactide), and biodegradable nature, polylactic acid has also been utilized for short-term use ranging from packing to coating (Garlotta 2001) (Fig. 1), since the mid of 1990s, with the aim to replace petroleum-based plastics. Polylactic acid has a glass transition temperature (Tg) around 60 °C and is relatively brittle. Improvements are being achieved by means of copolymerization (Meimoun et al. 2021), blending, compounding and additives (Kfoury et al. 2013), which reflect the hopes and interests of both the researchers and public for replacing petroleum-based plastics with the bioplastic, polylactic acid.
Synthesis of polylactic acid
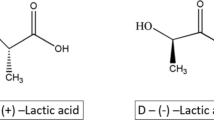
The precursor of polylactic acid is lactic acid, 2-hydroxy propionic acid, which exists in two stereo isomeric forms (Fig. 2). They are mainly produced by microbial fermentation and/or chemical synthesis. During chemical synthesis, an equal amount of l-lactic acid and d-lactic acid is produced (Juodeikiene et al. 2015), while during fermentation either type of lactic acid is produced depending on the type of bacteria used. It is then subjected to purification process in which a number of techniques are used, ranging from nanofiltration and electrodialysis to reactive distillation (Msuya et al. 2017). Once purified, an oligo(lactic acid) is synthesized by the process of polycondensation, which gives rise to a cyclic dimer, lactide, by depolymerization reaction (Filachione and Costello 1952; Masutani and Kimura 2015) (Fig. 2). l-lactide, d-lactide and meso lactide are obtained, as shown in Fig. 2. Polylactic acid can be synthesized from these monomers either by ring-opening polymerization or polycondensation, catalyzed by metallic and organic catalysts mainly. However, ring-opening polymerization was the first method of polylactic acid synthesis (Taib et al. 2022) and still being used for the manufacturing of polylactic acid on industrial scale. The resultant stereo forms of polylactic acid, poly(l-lactide), poly(d-lactide), and poly(dl-lactide) (Nair et al. 2013), and their properties depend on the stereo isomeric form of lactide and the catalyst used for their synthesis. For example, the resultant polylactic acid can be either semi-crystalline, poly(l-lactide) or poly(l-lactide), or amorphous, poly(dl lactic acid) (Naser et al. 2021).
Synthetic route of stereo forms of polylactic acid from biomass. Stereo forms of lactic acid are synthesized from biomass. Oligo(lactic acid) is formed from lactic acid by polycondensation. Oligo(lactic acid) gives rise to different stereo forms of lactide through depolymerization reaction. Polylactide is mainly synthesized by the ring opening polymerization of lactide in the presence of catalyst
Biobased versus biodegradable/degradable nature of polylactic acid
According to the International Union of Pure and Applied Chemistry, biodegradable plastics are macromolecular substances which can be degraded as a result of biological activity, leading to reduction in molecular weight. Not all bioplastics are degradable. For example, biobased polyamides are not degradable (Vardar et al. 2022). The misconception that all bioplastics are biodegradable became the base of their popularity (Reddy et al. 2013).
The biodegradation of polylactic acid depends on several factors including its material properties, first and higher order structure, environmental conditions, ultraviolet radiations, temperature, pH and humidity (Karamanlioglu et al. 2017), catalytic species, microorganisms and/or enzyme (Nampoothiri et al. 2010; Qi et al. 2017). The biodegradation of polylactic acid occurs in two main steps: fragmentation and mineralization. Fragmentation of polylactic acid is achieved by means of hydrolysis which can be biotic or abiotic. For instance, biotic hydrolysis involves microorganisms and/or enzymes, whereas abiotic hydrolysis involves mechanical weathering. The second step, mineralization, is achieved by microbes resulting into CO2, water and methane (Fig. 3) depending on the presence and/or absence of oxygen in the medium.
A huge amount of polylactic acid is produced each year. After its use, a small portion is either recycled or incinerated. The rest of polylactic acid goes to dumping sites where it is prone to environmental degradation to form low molecular weight polymer. The low molecular weight polymer is either degraded by microbes, to form carbon dioxide, water, methane and biomass, or fragmented, to form pieces of less than 5 mm in size termed as microplastics. Microplastics accumulate in phytoplankton and then in zooplankton via ingestion, hence make their way to the aquatic food chain. The prey–predator relationship in aquatic ecosystems makes the microplastics to reach higher trophic levels, including humans
Many studies have assessed the biodegradation of polylactic acid and its blends with other polymers. Narancic et al. (2018) studied the biodegradation of polylactic acid and other polymers along with some of their blends using standards of biodegradation. The authors reported that polylactic acid alone or other polylactic acid-based plastics were not degraded in 56 days experiment in a simulated aquatic environment, anaerobic sludge, derived from wastewater treatment plant, under 35 ± 2 °C. The authors concluded that polylactic acid and its blends are similar to non-biodegradable plastics in terms of biodegradation in aquatic environment. Bagheri et al. (2017) studied the degradation of polylactic acid, along with other biopolymers and synthetic polymers, in artificial freshwater and sea water, in a thermostatic chamber at 25 °C and under fluorescence light of 16 h light and 8 h dark cycle. The authors found that polylactic acid did not show any significant degradation as compared to others during a 400 days experiment. Other studies have also reported very low or negligible degradation rate of polylactic acid in the aquatic environment (Martin et al. 2014; Pinto et al. 2015; Tsuji and Suzuyoshi 2002a).
Similarly, Weinstein et al. (2020) conducted a 32-week experiment in intertidal salt marshes, where samples were submerged at high tide for 6 h and exposed to low tide for approximately 6 h too, and compared the degradation of polylactic acid, as a commercial biobased case study polymer, to conventional petro-based plastics. The conventional petro-based plastics included poly(ethylene terephthalate), high density polyethylene and polystyrene, some of which contain additives to enhance the degradation. The authors reported the formation of biofilms and microplastics, and polylactic acid showed the slowest rate of degradation of all polymers tested. Kliem et al. (2020) proposed that low temperatures along with low bacterial density make the sea water unsuitable for the biodegradation of polylactic acid.
It can be concluded from these results that polylactic acid is having no or very low degradability in the natural environment at room temperature. Consequently, like majority of other petroleum-based plastics, polylactic acid will be fragmented by mechanical weathering and due to uncommon microorganisms for polylactic acid degradation and assimilation (Ribba et al. 2022), these fragments will turn to microplastics by the actions of several environmental factors including interaction with biota (Wang et al. 2021), which will have impact on aquatic biota. It should be noted in turn that polylactic acid is a compostable polymer which can be degraded under controlled conditions. In fact, the degradation of polylactic acid requires a high temperature (55–175 °C), which is beyond the limits of the natural environment (Garlotta 2001; Haider et al. 2019).
Modifications strategies for promoting the biodegradability of polylactic acid
Efforts are being made to modify polylactic acid for long term utilities. Literature survey reveals that blending with other polymers in the presence of eventual compatibilizers and the use of additives, such as plasticizers, have a significant effect on the degradability of polylactic acid as shown in Table 1.
Plasticizers
Plasticizers are used as additives which increase elongation at break, the stretching of plastic before it breaks, along with reduction in glass transition temperature and elastic modulus. Many studies have reported the use of different additives as plasticizers which promote degradation of polylactic acid. For example, Arrieta et al. (2014b) reported that the use of polyethylene glycol and acetyl-tri-n-butyl citrate, as plasticizer, can enhance polylactic acid degradation, where weight loss was higher than 90% after 28 days of incubation under composting conditions. In another example, Gardella et al. (2017) investigated the effects of a dendrimer like structure, a hyperbranched polyglycerol core poly(d-lactide) arms, as a plasticizer on the degradation rate of polylactic acid under composting conditions. The authors reported a faster degradation and increase in hydrophilicity of the polymer, up to about 30%. The authors attributed the faster degradation to increased hydrophilicity of the modified polylactic acid. Similar results were also reported by many other authors including Xie et al. (2014) and Sharma et al. (2021) while investigating the degradation of plasticized polylactic acid.
The degradation of modified polylactic acid depends on the nature of the additive used. In some cases, the use of plasticizer is shown to have very little or no effects on the degradation of polylactic acid. For example, Balart et al. (2018) reported that the use of epoxidized linseed oil, as plasticizer and or compatibilizer, can delay the degradation of polylactic acid. The authors reported that plasticized polylactic acid showed a weight loss of 61% only, while this value for unplasticized polylactic acid was 86%. Likewise, literature review reveals that other additives, when used as plasticizer, including d-limonene, glucose pentaacetate, sucrose octaacetate and glucose hexanoate esters, are reported to have slight or no effects on polylactic acid in terms of degradation (Fortunati et al. 2014; Nair et al. 2012; Yang and Hakkarainen 2015).
Blends with hydrophilic polymers
Blending with hydrophilic polymers is another approach for the modification of polylactic acid to improve mechanical properties as well as degradability. As polylactic acid is hydrophobic in nature, therefore blending it with hydrophilic polymers/polysaccharide has been shown to increase its degradability, attributed to an increase in hydrophilic characteristic of the polymer. For example, Claro et al. (2016) have shown that polylactic acid/chitosan and polylactic acid/cellulose acetate blends allow rapid biodegradation as compared to neat polylactic acid. Lv et al. (2017) studied the biodegradation of polylactic acid blended with starch and wood flour for 105 days under ambient environmental conditions and reported an accelerated degradation of polylactic acid. The authors concluded that the degradation of starch created holes in the polylactic acid matrix which facilitated the diffusion of water, leading to an enhanced hydrolysis of polylactic acid.
In another example, Wilfred et al. (2018) investigated the biodegradation of polylactic acid/starch blend in a commercial compost and soil for 14 and 28 days. The authors reported that the degradation rate of polylactic acid/starch blend was higher as compared to neat polylactic acid. These results also reveal a positive correlation of starch content and degradation rate, as proposed by Yamano et al. (2014) that biodegradability is correlated with hydrophilicity. Likewise, a number of studies have shown that blending of polylactic acid with hydrophilic polymers and or polysaccharides can make them more susceptible to degradation as shown in Table 1.
Blends with hydrophobic polymers
Chuayjuljit et al. (2017) studied the degradation of polylactic acid blended with poly(butylene succinate) in natural environment, buried in soil under ambient environmental temperature for 90 days. The authors reported faster degradation of blended polylactic acid and attributed it to the faster degradation of poly(butylene succinate). Similar results were also reported for blending of polylactic acid with other hydrophobic polymers, for example poly(β-hydroxybutyrate) (Bonartsev et al. 2012) and poly(vinyl acetate) (Haque et al. 2017).
However, not necessarily the blending of polylactic acid with hydrophobic polymers could contribute to their degradability profile. For example, Luzi et al. (2016) reported a decrease in the degradation of polylactic acid blended with poly(butylene succinate), compared to neat polylactic acid, attributed to higher crystallinity induced by poly(butylene succinate). These contradictions in the results, as compared to Chuayjuljit et al. (2017), could be attributed to the difference in the percentage of poly(butylene succinate) content in the blend and composting conditions as well. Furthermore, in some cases the blending of polylactic acid with hydrophobic polymers is shown to have varying effects on the degradation of polylactic acid, as shown in Table 1.
Composites
Compounding of polylactic acid with other materials, having significantly different characteristics, either synthetically or artificially to obtain composites is another approach to improve the degradation rate of polylactic acid. Many studies have reported significant improvements in the degradation rate of the resultant polylactic acid composites. For example, Yaacob et al. (2016) investigated the degradation of paddy straw powder/polylactic acid composite and reported an improved degradation rate of polylactic acid in natural soil burial experiment. The authors attributed the improved degradation rate to the hydrophilic nature of paddy straw. In another work, Zandi et al. (2019) investigated the benzylated pulp (rich in cellulose)/polylactic acid and pulping liquor (rich in lignin)/polylactic acid composites in an indoor soil biodegradation experiment. The authors reported considerable biodegradation of polylactic acid composites, attributed to lower glass transition temperature and higher water absorption, in addition to larger biodegradation of filler. Similar results were also reported by many other authors as shown in Table 1.
Compatibilizers for polymer blend and composite
Many studies have reported that the use of compatibilizers can affect the degradation rate of polylactic acid in blends and composites. For example, Fortunati et al. (2013a) investigated a blend of polylactic acid with ethylene–vinyl acetate-glycidylmethacrylate copolymer, on the degradation rate of polylactic acid. The authors reported a faster degradation of the blend, 71% weight loss, under aerobic conditions in the presence of compost inoculum at 58 °C, as compared to neat polylactic acid. The authors attributed it to the faster diffusion of water in to the polymer mixture. Persenaire et al. (2014) also studied the biodegradation of polylactic acid/poly(butylene succinate) blends in the presence of a compatibilizer, maleic-anhydride-grafted polylactic acid and maleic-anhydride-grafted poly(butylene succinate), under composting conditions, in a test chamber under 50 °C and humidity of 50% for 475 h. The authors reported an enhanced molar mass loss in the presence of compatibilizer as compared to the polylactic acid/poly(butylene succinate) blend. Similar results were also reported by many other studies, including Olewnik-Kruszkowska et al. (2020), Sun et al. (2021), Wan and Zhang (2018) and Wan et al. (2019), while investigating the degradation of polylactic acid after the addition of compatibilizers, as shown in Table 1.
In contrast to these results, a few studies have shown that the use of compatibilizer can also negatively affect the degradation rate of polylactic acid. For example, Carbonell-Verdu et al. (2018) reported that the use of epoxidized cotton seed oil derivatives, as compatibilizer in polylactic acid/poly(butylene adipate-co-terephthalate) binary blends, reduced the disintegration ability of polylactic acid. The authors attributed the reduced disintegration rate of the blend to the lower disintegration rate of poly(butylene adipate-co-terephthalate). However, it is worthy to note that the compatibilizer do improve some of the mechanical properties of the blend as reported by Luzi et al. (2016).
Conversion of polylactic acid into microplastics
The term microplastic was introduced to the scientific literature by Thompson et al. (2004), and defined it as microscopic plastic pieces, while investigating its accumulation in sediments and water of European territory. This definition was further refined by Arthur et al. (2009) as plastic fragments less than 5 mm in size. In term of size, this is the most used definition in the literature with controversy on lower size limit. However, many authors have set different lower size limits ranging from 1 to 20 µm. To address this issue, Frias and Nash (2019) defined microplastic as any synthetic polymeric particle of 1 µm to 5 mm in size, irrespective of shape and source of origin (primary or secondary).
As discussed earlier, polylactic acid is having no or very low biodegradability in the natural environment; as a result it will remain for a long time in the environment. Like petroleum-based plastics, many biotic and abiotic factors will lead to its fragmentation and consequently microplastics will be generated from it. For example, Lambert and Wagner (2016) reported the release of microscopic size particles into the surrounding solution while investigating the biodegradation of polylactic acid along with polyethylene, polyethylene terephthalate, polystyrene and polypropylene in the weathering chamber. The authors also reported that polylactic acid generated significantly higher rate of particles, 11.6 × 106 particles per milliliter, as compared to other polymers, where polyethylene, polyethylene terephthalate, polystyrene and polypropylene generated 8.0 × 106, 9.4 × 106, 9.9 × 106, and 9.8 × 106 particles per milliliter, respectively. This could be attributed to the fact that bioplastics are more susceptible to degradation factors as compared to petroleum-based plastics and consequently bioplastics are having high degradation rate (Napper and Thompson 2019; Wei et al. 2021), leading to the generation of huge number of microplastics.
Niu et al. (2022) assessed the disintegration of polylactic acid and its capacity to form microplastics using sea water and accelerated ultraviolet radiations for 18 months. The authors reported that polylactic acid forms almost 18 times fewer microplastics as compared to the petroleum-based plastic, polypropylene. However, the capacity of polylactic acid to form microplastics was double as compared to polypropylene in control. In contrast to Lambert and Wagner (2016), the number of generated microplastics were very less. The possible reason might be difference in the experimental conditions, for example, temperature which is known to have impact on the degradation of polylactic acid, and chemistry of polylactic acid used in both the studies. In a recent study, Le Gall et al. (2022) studied the formation of microplastics from self-reinforced polylactic acid (homocomposite of two comingled grades of polylactic acid fibers) using artificial sea water and accelerated ultraviolet radiations for 18 months. The authors reported the formation of microplastics in both, control (without ultraviolet radiations exposure and kept in dark) and experimental group. However, the experimental group had double number of microplastics (17 ± 18) as compared to control (9 ± 5).
Many recent research studies including Shruti and Kutralam-Muniasamy (2019), Wei et al. (2021) and Wei et al. (2022) show that other bioplastics also give rise to microplastics under laboratory conditions. The findings of these studies indicate that bioplastics including polylactic acid can give rise to microplastics, similar to petroleum-based plastics. Consequently, very recently, a few studies have identified polylactic acid microplastics in sediments (Bancin et al. 2019), marine ecosystems (Kazour et al. 2019) and wastewater treatment plant (Granberg et al. 2019), which further makes it an alarming issue. Yagi et al. (2012) reported that polylactic acid microplastics degradation rate is much slower as compared to large fragments, which reflects their persistent nature similar to petroleum-based microplastics. Polylactic acid microplastics are also resistant to degradation under normal environmental conditions and will persist in ecosystems, where they will pose potential threat to the natural environment and biota.
Environmental factors affecting formation of microplastics from polylactic acid
The degradation of polylactic acid requires both abiotic and biotic processes therefore the term environmental degradation was suggested for the overall mechanisms of degradation (Nampoothiri et al. 2010). Both types of processes are crucial for the degradation of polylactic acid, as there is evidence that polylactic acid can be degraded by biotic factors, for example enzymes, after decrease in its molecular mass by an abiotic mechanism (Stloukal et al. 2015). Like for all other plastics, there are several factors which affect the degradation of polylactic acid and lead to the formation of microplastics.
Ultraviolet radiations
Ultraviolet radiations can cause changes in the polymer microstructure by different pathways including chain scission and/or cross linking (Kijchavengkul et al. 2010). Considering the latter, polymers exposed to ultraviolet radiations were found to have reduced biodegradation because of higher molecular weight which in turn decreases its assimilation by microorganisms (Kijchavengkul et al. 2008). Jeon and Kim (2013) reported that exposure of polylactic acid to ultraviolet radiations for a long period reduced its biodegradation by microorganisms which suggested the formation of poorly assimilated solids or microplastics. However, Stloukal et al. (2012) reported that exposure of polylactic acid to ultraviolet radiations leads to chain scissions and specific surface area is more important factor than photo-oxidation for degradation. It is very clear from the above discussion that ultraviolet radiations can affect the degradation of polylactic acid and lead to the formation of microplastics.
Temperature
Temperature is another factor which can influence the degradation of polylactic acid. It is shown by many studies that an increase in temperature can accelerate polylactic acid degradation. The possible reason is that high temperature enhances water's affinity for the polymer and increases hydrolysis rate (Goto et al. 2020), resulting in a faster degradation of the polymer. The hydrolysis of polylactic acid starts from its amorphous region (Growney Kalaf et al. 2017), which allows water to diffuse more readily as compared to the crystalline region. Consequently, with the passage of time, portion of crystalline region increases and the rate of degradation decreases (Siparsky et al. 1998). The degradation of polylactic acid was investigated by Le Duigou et al. (2009) considering 20 and 40 °C temperature for 3 months. The authors reported slight change in the molecular weight of polylactic acid at 20 °C and 48% decrease at 40 °C after 3 months.
Lyu et al. (2007) reported significant variations in abiotic hydrolysis of polylactic acid considering a range of temperature from 37 to 90 °C. Similarly, Karamanlioglu et al. (2017) reported that the polylactic acid chains become more flexible at or above glass transition temperature, therefore rate of polylactic acid degradation is higher above this temperature which accelerates both hydrolysis and attachment of microbes. The attachment of microbes to polylactic acid could be very rare in the aquatic environment, as two studies have reported that there is no evidence of microbial degradation after 10 weeks of polylactic acid immersion in static and dynamic seawater (Tsuji and Suzuyoshi 2002a, 2002b). The degraded portion might be that of the amorphous region of polylactic acid and the crystalline region will persist which could lead to the formation of microplastics.
pH
Hydrolysis of polylactic acid in acidic and basic media occurs by bulk erosion and surface erosion, respectively (Rodriguez et al. 2016). There is evidence that pH can also influence the degradation of polylactic acid. The degradation of polylactic acid has been reported under both, acidic and basic media driven by different mechanisms of depolymerization. For example, chain end scission hydrolyzes polylactic acid under acidic medium where protonation activates hydroxyl group, resulting in the depolymerization of polylactic acid to lactic acid (Codari et al. 2012; Lazzari et al. 2014). It should be noted that the rate of degradation was independent of chain length due to high hydrophilicity of chain end and hydrophobicity of polymer chain.
On the other hand, back biting reaction leads to random chain scission under basic medium, which depolymerizes polylactic acid into lactide followed by hydrolysis (de Jong et al. 2001; van Nostrum et al. 2004), as shown in Fig. 4. During hydrolysis, hydroxide ions catalyzed the cleavage of ester. At higher pH, the concentration of hydroxide ions is higher and therefore enhances the degradation of polylactic acid (Cam et al. 1995; Tsuji and Ikada 1998). However, the complete degradation of polylactic acid in natural environment takes time and the resulting disintegrated fragments or microplastics will aid in microplastic pollution before mineralization. Additionally, the reduction in the size of microplastics makes them likely of ingestion by organisms (Naqash et al. 2020). The question is whether these microplastics will pose the same threat to the ecosystems as petroleum-based microplastics or they will have different effects?
Mechanism of polylactic acid degradation. a Back biting reaction, where polylactic acid is depolymerized into lactide followed by hydrolysis. b Chain end scission under acidic environment, where polylactic acid is depolymerized into lactide by the activation of hydroxyl group through protonation. Scheme is adapted from (c) and (d) [de Jong et al. (2001), used with permission from Elsevier]
Impact of polylactic acid microplastics on aquatic biota
Recently some studies have shown that polylactic acid microplastics have a prominent impact on marine biota. Due to their compostable nature, polylactic acid microplastics cannot be degraded and are assimilated as all marine biota do not have the specific enzymes responsible for their degradation. In case of petroleum-based microplastics, it is well known that ingestion of these contaminants can cause a number of adverse effects. However, in the case of polylactic acid microplastics, very recently scientific community has shown an interest in their ecotoxicological evaluation. Very few studies have been conducted on the impact of polylactic acid microplastics on limited number of aquatic species which revealed that in some cases these contaminants can negatively affect the exposed individuals analogous to petroleum-based microplastics. The literature survey reveals that Green et al. (2016) is pioneer in assessing the toxic effects of polylactic acid microplastics on aquatic biota. After this, many research studies focused on the ecotoxicological evaluation of polylactic acid microplastics using a range of experimental model species (Table 2).
Microbial communities
Sediment microbial communities consist of large number of the earth’s biodiversity and which play a key role in biogeochemical cycling of nutrients (Vincent et al. 2021) and ecological purification of pollutants (John et al. 2022). The presence of microplastics in the environment provides a new habitat to these microbes (McCormick et al. 2014), but on the other hand, the degradation of these polymers can produce toxic substances which will have negative effects on these microbes (Kong et al. 2018). A few species of bacteria will be benefited but the others will be negatively affected (Li et al. 2020), as different species of bacteria respond differently to the presence of microplastics (Wang et al. 2020). The impact of petroleum-based microplastics on microbial communities is well studied. However, studies focusing on the impact of polylactic acid microplastics on microbial communities are scarce.
Seeley et al. (2020) have conducted a 2-week microcosm experiment to investigate the effects of petroleum-based and bio-based microplastics on composition and function of sedimentary microbial communities. They reported a significant alteration in microbial communities exposed to petroleum-based microplastics. Surprisingly, polylactic acid microplastics were found to promote nitrification and denitrification. Based on the results, the authors suggested that the microorganisms might have utilized the microplastics as organic carbon for the energy which facilitated these phenomena. However, degradation and assimilation of polylactic acid microplastics in 2 weeks have not been reported in the literature.
Algae
Microalgae are the primary producer of aquatic ecosystems which play an important role in their functioning (Casado et al. 2013). Being a primary producer, microalgae account for 50% of net production (Barbosa 2009), and therefore, any alteration in microalgae population can have serious effects on food webs. Many studies have been carried out to investigate the impact of petroleum-based microplastics on several parameters of microalgae, including growth (Long et al. 2017), morphological changes (Mao et al. 2018), chlorophyll content (Prata et al. 2018), photosynthesis (Zhang et al. 2017) and gene expression (Lagarde et al. 2016). The results of all these studies reveal that petroleum-based microplastics have deleterious effects on microalgae ranging from individual to population level.
The impact of polylactic acid microplastics on microalgae has also been investigated. Su et al. (2022) investigated the effects of petroleum-based microplastics and bio-based microplastics including polylactic acid on marine alga Chlorella vulgaris. The authors reported that both types of microplastics inhibited the growth of microalgae. However, polylactic acid microplastics were reported to have severe effects on growth of algae with highest inhibition rate of 47.95%, as compared to other petroleum-based and bio-based microplastics. The authors attributed these effects to the physiochemical properties and chemical changes of microplastics. The authors also reported that microplastics can stimulate pigments content (chlorophyll a, chlorophyll b and carotenoid), attributed to the cellular defense against stress. These findings are very interesting in two domains. Firstly, in the context of adaptation and or defense mechanisms against microplastics pollution, secondly, in understanding the fact that polylactic acid microplastics appear to be more toxic as compared to petroleum-based microplastics.
Phytoplankton community
Being primary producers, phytoplankton plays a key role in maintaining aquatic ecosystems. Due to their key role in food chains/webs, there is a serious concern about the impact of microplastics on phytoplankton (Koenigstein 2020). Any alteration or threat to the primary producers will have significant effects on the food chains/webs and consequently on the entire ecosystems, therefore, assessing the impact of emerging pollutants on primary producers is crucial. Literature survey revealed that petroleum-based microplastics have deleterious effects on various parameters of phytoplankton including photosynthetic capacity and growth (Sánchez-Fortún et al. 2021).
Studies on the impact of polylactic acid microplastics on phytoplankton are scare. Only a few studies have assessed their impact on phytoplankton. For example, Yokota and Mehlrose (2020) have assessed the impact of polylactic acid microplastics, originated from body wash scrub, on natural phytoplankton communities in a 7-days incubation experiment conducted in temperate mesotrophic lake. The authors reported that polylactic acid microplastics eliminated cryptophytes and increased chrysophytes, resulting in the alteration of taxonomic composition of the phytoplankton in the mesocosms. They suggested that chrysophytes contain a protective siliceous loricae against the polylactic acid microplastics whereas cryptophytes do not have any such protection and thereby got affected by the polylactic acid microplastics.
Zooplankton
Zooplanktons are primary consumers and located at the base of food chains/webs, thereby channeling nutrients and energy from the primary producers to higher trophic levels. Most researchers investigating the effects of microplastics on aquatic biota have focused on the primary consumers. As compared to other species, zooplanktons are more prone to microplastics and therefore documented as potential microplastics consumers (Cole and Galloway 2015). Microplastics act as analogues of zooplankton prey (Gambardella et al. 2017) and can have negative impact on different ecological processes. Recently, many studies have reported adverse effects of petroleum-based microplastics on a range of zooplankton species. The results of those studies reveal that petroleum-based microplastics have adverse effects on survival rate and reproduction (Yu et al. 2020; Zhang et al. 2019a), feeding capacity and selectivity (Cole et al. 2019; Coppock et al. 2019) and behavior (Suwaki et al. 2020). However, studies investigating the effects of polylactic acid microplastics on zooplankton are scare and only a few studies are available.
Zimmermann et al. (2020) investigated that how polylactic acid microplastics affected the survival, reproduction, and growth of Daphnia magna in a 21-days experiment. They found that polylactic acid microplastics cause the mortality of 60% individuals exposed to 500 mg/L, while in control the mortality was 5%. The authors also reported decrease in reproductive output and body length in the exposed individuals, induced by the microplastics themselves rather than leachates or additives. Similarly, very recently Di Giannantonio et al. (2022) studied the effects of polylactic acid microplastics on uptake of microplastics, immobility, and behavior of two zooplankton species, the crustacean Artemia franciscana and the cnidarian Aurelia species (common jellyfish) in a 24 h experiment. The authors reported polylactic acid microplastics in the digestive system of A. franciscana and in the gelatinous tissue of Aurelia species exposed to 100 mg/L, with no effects on the immobility of both the species. However, significant alterations were reported in the swimming behavior (pulsation) of Aurelia species at all the exposure concentrations (1, 10 and 100 mg/L), attributed to the direct toxicity of polylactic acid microplastics. It is worthy to note that the concentration of microplastics used in majority of the ecotoxicological studies, to evaluate their potential effects on the exposed organisms, are much higher as compared to their concentrations found in the natural environment.
Annelids
Annelids are invertebrates which play an important role in benthic ecosystems by serving as a link from primary producers to higher trophic levels and in the cycling of minerals (Rafia and Ashok 2014). They are the dominant invertebrates of the deep sea and mostly occupy sediments. Recent studies reported that petroleum-based microplastics can have deleterious effects on various parameters of annelids which include decrease in food intake (Wright et al. 2013), impairment of immune system, physical stress and even death (Browne et al. 2013), leading to a drastic impact on ecological processes (Green et al. 2016). To the best of our knowledge, only three studies have assessed the negative effects of polylactic acid microplastics on annelids. Klein et al. (2021) investigated the impact of polylactic acid microplastics (mixed and or layered on sediment surface) on freshwater worms (Lumbriculus variegatus) under laboratory conditions. The authors reported a significant reduction in the survival of the worms exposed to microplastics mixed with the sediments. However, they attributed the toxicity to the associated chemicals rather than to the polymer.
Similarly, Green et al. (2016) assessed the effects of polylactic acid and petroleum-based microplastics on lugworms (Arenicola marina) using concentrations of 0.02, 0.20 and 2% (wet sediment weight) in a 31-days mesocosm experiment with focus on health, biological activity and nitrogen cycling, in addition to the primary productivity of the sediments. The authors reported a significant impact of both types of microplastics on the health and behavior of the exposed individuals, as well as reduction in the primary productivity of the sediments they inhabited. Polylactic acid microplastics exposure not only reduced the feeding activity of the exposed individuals but also reduced the biomass of the algae on the surface of sediments. They also found that polylactic acid microplastics reduced the concentration of ammonia in pore water, which might be due to the potential of carbonyl and hydroxyl groups of polylactic acid to adsorb cations.
In another study, Green et al. (2017), while investigating the ecological impacts of polylactic acid and petroleum-based microplastics, high-density polyethylene, on the biodiversity and ecosystem functioning, found a difference in faunal invertebrate assemblages in the exposed groups, with less polychaetes and more oligochaetes, highlighting the potential of polylactic acid microplastics to affect ecosystem. These results are quite interesting in the context of species-specific response to microplastics (Bai et al. 2021) or other contaminants, as both the species were exposed to the same types and concentrations of microplastics but showed completely different responses.
Mollusks
Mollusks are a diverse group of filter feeders which can be found in a variety of aquatic habitats. They provide ecological services to a number of organisms ranging from habitat to food (Fernández-Pérez et al. 2018). Being filter feeders, mollusks can accumulate and transfer microplastics to higher trophic levels, which will have detrimental effects on their consumers including humans. Therefore, many ecotoxicological studies have used mollusks as bioindicators of pollution (Capillo et al. 2018). However, there are only a few studies available on the impact of polylactic acid microplastics on mollusks. Green et al. (2017) studied the ecological impacts of polylactic acid and petroleum-based microplastics on the biodiversity and ecosystem functioning of bivalve-dominated European flat oysters (Ostrea edulis) and blue mussels (Mytilus edulis) habitats in outdoor 50-days mesocosms experiment, using two different concentrations of 2.5 and 25 μg/L for each type of microplastics. The authors reported a significant reduction in filtration by M. edulis (exposed to 25 μg/L), while no effects were observed on ecosystem functioning or the associated assemblages of invertebrates. On the other hand, the authors reported a significant increase in filtration by O. edulis after exposure to 2.5 and 25 μg/L and decrease in the pore water ammonium and biomass of benthic cyanobacteria.
Khalid et al. (2021) also studied the effects of polylactic acid microplastics on blue mussels (M. edulis) using two different concentrations, 10 and 100 μg/L, in an 8-days experiment with biochemical endpoints. The authors found no significant effects of polylactic acid microplastics on M. edulis in terms of oxidative stress (catalase, glutathione-S-transferase, and superoxide dismutase activities), neurotoxicity (acetylcholinesterase), and immunotoxicity (lysosomal membrane stability and acid phosphatase activity). In contrast to these results, Green et al. (2019) found a significant alteration in the immunological profile of haemolymph of Mytilus edulis exposed to polylactic acid microplastics in a 52-days mesocosms experiment. However, the authors found no adverse effects of polylactic acid microplastics on the attachment strength of the exposed individuals.
Green (2016) investigated that how polylactic acid and petroleum-based microplastics at low and high concentrations (0.8 and 80 μg/L) affect the health and biological functioning of European flat oysters (Ostrea edulis) along with the impact on structure of associated macro faunal assemblages in a 60-days mesocosm experiment. They reported minimal effects on the exposed individuals, but the associated macro faunal assemblages were significantly altered which were ~ 1.2 and 1.5 times reduced as compared to the control. For instance, the biomass of Scrobicularia plana (peppery furrow shell clam), the abundance of juvenile Littorina sp. (periwinkles) and Idotea balthica (an isopod) were decreased 1.5, 2.0 and 8.0 times in groups exposed to either type of microplastics compared to the control.
Beside the mussels, other filter feeder organisms were also used for the ecotoxicological evaluation of microplastics. For example, Anderson and Shenkar (2021) investigated the impact of polyethylene terephthalate and polylactic acid microplastics on the biological and ecological features of a solitary ascidian (Microcosmus exasperatus). The authors reported that both polylactic acid and petroleum-based microplastics had similar impact on the exposed individuals; for example, both types of microplastics reduced the fertilization rates in the exposed individuals.
Fish
Fish are a good source of unsaturated fatty acids and proteins; therefore, their consumption is recommended in human diet (Ali et al. 2017). Therefore, assessment of microplastics and its consequent impact on fish is of major environmental importance. Many ecotoxicological studies have used fish as bioindicator of water quality and ecosystem health. Fish have the potential to accumulate and magnify pollutants which may have potential impacts on their consumers including humans. Many studies have reported the ingestion and accumulation of petroleum-based microplastics in a range of fish species, while studies on polylactic acid microplastics are very few.
Recently, Chagas et al. (2021) studied the bioaccumulation of polylactic acid microplastics, at a concentration of 2.5 and 5 mg/L, in adult zebrafish and its consequent impact on behavioral, biochemical, and morphological parameters in a 30-days experiment. The authors reported the accumulation of microplastics in the liver, brain, gills, and carcass of the exposed group in addition to behavioral and morphological changes. The reported behavioral and biochemical changes were shoals predictive of co-specific social interaction and an anti-predator defense response defect, attributed to cholinergic changes inferred by an increase in the activity of acetylcholinesterase and redox imbalance whereas the morphological changes were alteration In the pigmentation pattern. However, in contrast to de Oliveira et al. (2021), no locomotor damages or anxiety-like behavior was observed in the exposed individuals. The possible reason might be difference in the life stages of the test organism as early life stages are more sensitive to different contaminants.
Most studies, while investigating the effects of various contaminants on aquatic organisms, have focused on early life stages of fish, for example larvae and embryo (Mu et al. 2022), because of their sensitivity to different contaminants (Schweizer et al. 2018), which are critical on individual and population health's point of view. For instance, zebrafish (Danio rerio) has been widely used as a biological model and/or as a representative of fish group by many researchers to investigate the toxicological impact of microplastics. de Oliveira et al. (2021) investigated the effects of polylactic acid microplastics (3 and 9 mg/L) on zebrafish larvae in a 5-days exposure experiment with behavioral and biochemical endpoints. The authors reported a decrease in the swimming speed and distance of the exposed individuals in open field test. The authors attributed these outcomes as a consequent impact of microplastics on fish locomotor and exploration activities. They also reported anxiety like behavior and accumulation of microplastics, which inhibited the activity of acetylcholinesterase leading to the reinforcement of neurotoxic action in the exposed group.
Similarly, another study also focused on the impact of polylactic acid microplastics (virgin and degraded) on zebrafish larvae (Zhang et al. 2021). They found a slower efflux and detoxification of degraded polylactic acid, mediated by ABC transporters and P450 enzymes, leading to increase in bioaccumulation of microplastics and thereby inhibiting the skeletal development of larvae. They also pointed the higher toxicity of degraded polylactic acid microplastics by identification of crucial mechanisms, for example, mitochondrial structural damage by oxidative stress, apoptosis, depolarization, and fission inhibition. However, no effects were reported on the hatching rate of larvae when exposed to both types of polylactic acid microplastics. The authors attributed these outcomes to the fact that the size of microplastics was larger as compared to the chorionic pore canals and the resistance of the chorionic barrier to polylactic acid microplastics.
Very recently, Duan et al. (2022) compared the accumulation and toxicity of polylactic acid and poly(ethylene terephthalate) microplastics using zebrafish (Danio rerio) as a model organism. The authors reported 170 times higher polylactic acid microplastics in the fish as compared to poly(ethylene terephthalate) microplastics resulting in intestinal epithelial tract damage followed by affecting the diversity of intestinal microbiota. The authors attributed these results to the depolymerization of polylactic acid in the digestive tract of fish, which decreased the intestinal pH and changed the carbon source structure. These results are quite interesting in understanding the toxicity of polylactic acid microplastics. These findings strongly support the concept that polylactic acid microplastics will have severe effects, similar to petroleum-based plastics, on the exposed individuals.
Conclusion
Due to the persistence and non-biodegradability of most petroleum-based plastics, efforts have been made to develop ecofriendly and environmentally safe substitutes. Polylactic acid is considered as a potential substitute of petroleum-based plastics. A comprehensive literature review shows that polylactic acid is compostable rather than biodegradable in the natural environment, leading to the formation of microplastics. Recent research has clearly identified that microplastics originating from polylactic acid are emerging environmental contaminants similar to microplastics from petroleum-based plastics. They are severely toxic to aquatic biota and might be a threat to human population as well through the food chain.
References
Ainali NM, Kalaronis D, Evgenidou E, Kyzas GZ, Bobori DC, Kaloyianni M, Yang X, Bikiaris DN, Lambropoulou DA (2022) Do poly(lactic acid) microplastics instigate a threat? A perception for their dynamic towards environmental pollution and toxicity. Sci Total Environ 832:155014. https://doi.org/10.1016/j.scitotenv.2022.155014
Ali H, Ali W, Ullah K, Akbar F, Ahrar S, Ullah I, Ahmad I, Ahmad A, Ilahi I, Sajad MA (2017) Bioaccumulation of Cu and Zn in Schizothorax plagiostomus and Mastacembelus armatus from river Swat, river Panjkora and river Barandu in Malakand Division, Pakistan. Pak J Zool 49:1555–1561. https://doi.org/10.17582/journal.pjz/2017.49.5.1555.1561
Almeshal I, Tayeh BA, Alyousef R, Alabduljabbar H, Mustafa Mohamed A, Alaskar A (2020) Use of recycled plastic as fine aggregate in cementitious composites: a review. Constr Build Mater 253:119146. https://doi.org/10.1016/j.conbuildmat.2020.119146
Alomar C, Sanz-Martín M, Compa M, Rios-Fuster B, Álvarez E, Ripolles V, Valencia JM, Deudero S (2021) Microplastic ingestion in reared aquaculture fish: biological responses to low-density polyethylene controlled diets in Sparus aurata. Environ Pollut 280:116960. https://doi.org/10.1016/j.envpol.2021.116960
Anderson G, Shenkar N (2021) Potential effects of biodegradable single-use items in the sea: polylactic acid (PLA) and solitary ascidians. Environ Pollut 268:115364. https://doi.org/10.1016/j.envpol.2020.115364
Arrieta MP, López J, Hernández A, Rayón E (2014a) Ternary PLA–PHB–Limonene blends intended for biodegradable food packaging applications. Eur Polym J 50:255–270. https://doi.org/10.1016/j.eurpolymj.2013.11.009
Arrieta MP, López J, Rayón E, Jiménez A (2014b) Disintegrability under composting conditions of plasticized PLA–PHB blends. Polym Degrad Stab 108:307–318. https://doi.org/10.1016/j.polymdegradstab.2014.01.034
Arrieta MP, Fortunati E, Dominici F, López J, Kenny JM (2015) Bionanocomposite films based on plasticized PLA–PHB/cellulose nanocrystal blends. Carbohydr Polym 121:265–275. https://doi.org/10.1016/j.carbpol.2014.12.056
Arthur C, Baker JE, Bamford HA (2009) Proceedings of the international research workshop on the occurrence, effects, and fate of microplastic marine debris, September 9–11, 2008. University of Washington Tacoma, Tacoma, WA, USA
Bagheri AR, Laforsch C, Greiner A, Agarwal S (2017) Fate of so-called biodegradable polymers in seawater and freshwater. Glob Chall 1:A1700048. https://doi.org/10.1002/gch2.201700048
Bai Z, Wang N, Wang M (2021) Effects of microplastics on marine copepods. Ecotoxicol Environ Saf 217:112243. https://doi.org/10.1016/j.ecoenv.2021.112243
Balart JF, Montanes N, Fombuena V, Boronat T, Sánchez-Nacher L (2018) Disintegration in compost conditions and water uptake of green composites from poly(lactic acid) and hazelnut shell flour. J Polym Environ 26:701–715. https://doi.org/10.1007/s10924-017-0988-3
Bancin LJ, Walther BA, Lee Y-C, Kunz A (2019) Two-dimensional distribution and abundance of micro- and mesoplastic pollution in the surface sediment of Xialiao Beach, New Taipei City, Taiwan. Mar Pollut Bull 140:75–85. https://doi.org/10.1016/j.marpolbul.2019.01.028
Barbosa AB (2009) Dynamics of living phytoplankton: implications for paleoenvironmental reconstructions. IOP Conf Ser Earth Environ Sci 5:012001. https://doi.org/10.1088/1755-1307/5/1/012001
Benninga H (1990) A history of lactic acid making: a chapter in the history of biotechnology, vol 11. Springer, Berlin
Bonartsev AP, Boskhomodgiev AP, Iordanskii AL, Bonartseva GA, Rebrov AV, Makhina TK, Myshkina VL, Yakovlev SA, Filatova EA, Ivanov EA, Bagrov DV, Zaikov GE (2012) Hydrolytic degradation of poly(3-hydroxybutyrate), polylactide and their derivatives: kinetics, crystallinity, and surface morphology. Mol Cryst Liq Cryst 556:288–300. https://doi.org/10.1080/15421406.2012.635982
Browne MA, Niven Stewart J, Galloway Tamara S, Rowland Steve J, Thompson Richard C (2013) Microplastic moves pollutants and additives to worms, reducing functions linked to health and biodiversity. Curr Biol 23:2388–2392. https://doi.org/10.1016/j.cub.2013.10.012
Cam D, Hyon S, Ikada Y (1995) Degradation of high molecular weight poly(l-lactide) in alkaline medium. Biomaterials 16:833–843. https://doi.org/10.1016/0142-9612(95)94144-A
Capillo G, Silvestro S, Sanfilippo M, Fiorino E, Giangrosso G, Ferrantelli V, Vazzana I, Faggio C (2018) Assessment of electrolytes and metals profile of the Faro Lake (Capo Peloro Lagoon, Sicily, Italy) and its impact on Mytilus galloprovincialis. Chem Biodivers 15:1800044. https://doi.org/10.1002/cbdv.201800044
Carbonell-Verdu A, Ferri J, Dominici F, Boronat T, Sanchez-Nacher L, Balart R, Torre L (2018) Manufacturing and compatibilization of PLA/PBAT binary blends by cottonseed oil-based derivatives. Express Polym Lett 12:808–823. https://doi.org/10.3144/expresspolymlett.2018.69
Casado MP, Macken A, Byrne HJ (2013) Ecotoxicological assessment of silica and polystyrene nanoparticles assessed by a multitrophic test battery. Environ Int 51:97–105. https://doi.org/10.1016/j.envint.2012.11.001
Chagas TQ, Araújo APC, Malafaia G (2021) Biomicroplastics versus conventional microplastics: an insight on the toxicity of these polymers in dragonfly larvae. Sci Total Environ 761:143231. https://doi.org/10.1016/j.scitotenv.2020.143231
Chaiwutthinan P, Chuayjuljit S, Srasomsub S, Boonmahitthisud A (2019) Composites of poly(lactic acid)/poly(butylene adipate-co-terephthalate) blend with wood fiber and wollastonite: physical properties, morphology, and biodegradability. J Appl Polym Sci 136:47543. https://doi.org/10.1002/app.47543
Chamas A, Moon H, Zheng J, Qiu Y, Tabassum T, Jang JH, Abu-Omar M, Scott SL, Suh S (2020) Degradation rates of plastics in the environment. ACS Sustain Chem Eng 8:3494–3511. https://doi.org/10.1021/acssuschemeng.9b06635
Chia RW, Lee J-Y, Kim H, Jang J (2021) Microplastic pollution in soil and groundwater: a review. Environ Chem Lett 19:4211–4224. https://doi.org/10.1007/s10311-021-01297-6
Chuayjuljit S, Wongwaiwattanakul C, Chaiwutthinan P, Prasassarakich P (2017) Biodegradable poly(lactic acid)/poly(butylene succinate)/wood flour composites: physical and morphological properties. Polym Compos 38:2841–2851. https://doi.org/10.1002/pc.23886
Claro PIC, Neto ARS, Bibbo ACC, Mattoso LHC, Bastos MSR, Marconcini JM (2016) Biodegradable blends with potential use in packaging: a comparison of PLA/chitosan and PLA/cellulose acetate films. J Polym Environ 24:363–371. https://doi.org/10.1007/s10924-016-0785-4
Codari F, Lazzari S, Soos M, Storti G, Morbidelli M, Moscatelli D (2012) Kinetics of the hydrolytic degradation of poly(lactic acid). Polym Degrad Stab 97:2460–2466. https://doi.org/10.1016/j.polymdegradstab.2012.06.026
Cole M, Galloway TS (2015) Ingestion of nanoplastics and microplastics by Pacific oyster larvae. Environ Sci Technol 49:14625–14632. https://doi.org/10.1021/acs.est.5b04099
Cole M, Coppock R, Lindeque PK, Altin D, Reed S, Pond DW, Sørensen L, Galloway TS, Booth AM (2019) Effects of nylon microplastic on feeding, lipid accumulation, and moulting in a coldwater copepod. Environ Sci Technol 53:7075–7082. https://doi.org/10.1021/acs.est.9b01853
Coppock RL, Galloway TS, Cole M, Fileman ES, Queirós AM, Lindeque PK (2019) Microplastics alter feeding selectivity and faecal density in the copepod, Calanus helgolandicus. Sci Total Environ 687:780–789. https://doi.org/10.1016/j.scitotenv.2019.06.009
de Jong SJ, Arias ER, Rijkers DTS, van Nostrum CF, Kettenes-van den Bosch JJ, Hennink WE (2001) New insights into the hydrolytic degradation of poly(lactic acid): participation of the alcohol terminus. Polymer 42:2795–2802. https://doi.org/10.1016/S0032-3861(00)00646-7
de Oliveira JPJ, Estrela FN, Rodrigues ASL, Guimarães ATB, Rocha TL, Malafaia G (2021) Behavioral and biochemical consequences of Danio rerio larvae exposure to polylactic acid bioplastic. J Hazard Mater 404:124152. https://doi.org/10.1016/j.jhazmat.2020.124152
Di Giannantonio M, Gambardella C, Miroglio R, Costa E, Sbrana F, Smerieri M, Carraro G, Utzeri R, Faimali M, Garaventa F (2022) Ecotoxicity of polyvinylidene difluoride (PVDF) and polylactic acid (PLA) microplastics in marine zooplankton. Toxics 10:479. https://doi.org/10.3390/toxics10080479
Duan Z, Cheng H, Duan X, Zhang H, Wang Y, Gong Z, Zhang H, Sun H, Wang L (2022) Diet preference of zebrafish (Danio rerio) for bio-based polylactic acid microplastics and induced intestinal damage and microbiota dysbiosis. J Hazard Mater 429:128332. https://doi.org/10.1016/j.jhazmat.2022.128332
Eltemsah YS, Bøhn T (2019) Acute and chronic effects of polystyrene microplastics on juvenile and adult Daphnia magna. Environ Pollut 254:112919. https://doi.org/10.1016/j.envpol.2019.07.087
Eom H-J, Nam S-E, Rhee J-S (2020) Polystyrene microplastics induce mortality through acute cell stress and inhibition of cholinergic activity in a brine shrimp. Mol Cell Toxicol 16:233–243. https://doi.org/10.1007/s13273-020-00088-4
EuropeanBioplastics (2022) Market—European bioplastics. european-bioplastics.org. Accessed July 2022
Fernández-Pérez J, Nantón A, Arias-Pérez A, Freire R, Martínez-Patiño D, Méndez J (2018) Mitochondrial DNA analyses of Donax trunculus (Mollusca: Bivalvia) population structure in the Iberian Peninsula, a bivalve with high commercial importance. Aquat Conserv Mar Freshw Ecosyst 28:1139–1152. https://doi.org/10.1002/aqc.2929
Filachione E, Costello E (1952) Lactic esters by reaction of ammonium lactate with alcohols. Ind Eng Chem 44:2189–2191
Fortunati E, Puglia D, Kenny JM, Minhaz-Ul Haque M, Pracella M (2013a) Effect of ethylene-co-vinyl acetate-glycidylmethacrylate and cellulose microfibers on the thermal, rheological and biodegradation properties of poly(lactic acid) based systems. Polym Degrad Stab 98:2742–2751. https://doi.org/10.1016/j.polymdegradstab.2013.10.007
Fortunati E, Puglia D, Monti M, Santulli C, Maniruzzaman M, Foresti ML, Vazquez A, Kenny JM (2013b) Okra (Abelmoschus esculentus) fibre based PLA composites: mechanical behaviour and biodegradation. J Polym Environ 21:726–737. https://doi.org/10.1007/s10924-013-0571-5
Fortunati E, Luzi F, Puglia D, Dominici F, Santulli C, Kenny JM, Torre L (2014) Investigation of thermo-mechanical, chemical and degradative properties of PLA-limonene films reinforced with cellulose nanocrystals extracted from Phormium tenax leaves. Eur Polym J 56:77–91. https://doi.org/10.1016/j.eurpolymj.2014.03.030
Frias JPGL, Nash R (2019) Microplastics: finding a consensus on the definition. Mar Pollut Bull 138:145–147. https://doi.org/10.1016/j.marpolbul.2018.11.022
Gambardella C, Morgana S, Ferrando S, Bramini M, Piazza V, Costa E, Garaventa F, Faimali M (2017) Effects of polystyrene microbeads in marine planktonic crustaceans. Ecotoxicol Environ Saf 145:250–257. https://doi.org/10.1016/j.ecoenv.2017.07.036
Gardella L, Forouharshad M, Pastorino L, Monticelli O (2017) Hyperbranched PDLA-polyglicerol: a novel additive for tuning PLLA electrospun fiber degradation and properties. Eur Polym J 91:21–30. https://doi.org/10.1016/j.eurpolymj.2017.03.054
Garlotta D (2001) A literature review of poly(lactic acid). J Polym Environ 9:63–84. https://doi.org/10.1023/A:1020200822435
Goto T, Kishita M, Sun Y, Sako T, Okajima I (2020) Degradation of polylactic acid using sub-critical water for compost. Polymers 12:2434. https://doi.org/10.3390/polym12112434
Granberg M, von Friesen LW, Bach L, Collard F, Gabrielsen GW, Strand J (2019) Anthropogenic microlitter in wastewater and marine samples from Ny-Ålesund, Barentsburg and Signehamna, Svalbard
Green DS (2016) Effects of microplastics on European flat oysters, Ostrea edulis and their associated benthic communities. Environ Pollut 216:95–103. https://doi.org/10.1016/j.envpol.2016.05.043
Green DS, Boots B, Sigwart J, Jiang S, Rocha C (2016) Effects of conventional and biodegradable microplastics on a marine ecosystem engineer (Arenicola marina) and sediment nutrient cycling. Environ Pollut 208:426–434. https://doi.org/10.1016/j.envpol.2015.10.010
Green DS, Boots B, O’Connor NE, Thompson R (2017) Microplastics affect the ecological functioning of an important biogenic habitat. Environ Sci Technol 51:68–77. https://doi.org/10.1021/acs.est.6b04496
Green DS, Colgan TJ, Thompson RC, Carolan JC (2019) Exposure to microplastics reduces attachment strength and alters the haemolymph proteome of blue mussels (Mytilus edulis). Environ Pollut 246:423–434. https://doi.org/10.1016/j.envpol.2018.12.017
Grigoras AG (2021) Natural and synthetic polymeric antimicrobials with quaternary ammonium moieties: a review. Environ Chem Lett 19:3009–3022. https://doi.org/10.1007/s10311-021-01215-w
Growney Kalaf EA, Hixon KR, Kadakia PU, Dunn AJ, Sell SA (2017) Electrospun biomaterials for dermal regeneration. In: Uyar T, Kny E (eds) Electrospun materials for tissue engineering and biomedical applications. Woodhead Publishing, Sawston, pp 179–231. https://doi.org/10.1016/B978-0-08-101022-8.00005-3
Haider TP, Völker C, Kramm J, Landfester K, Wurm FR (2019) Plastics of the future? The impact of biodegradable polymers on the environment and on society. Angew Chem Int Ed 58:50–62. https://doi.org/10.1002/anie.201805766
Hammiche D, Boukerrou A, Azzeddine B, Guermazi N, Budtova T (2019) Characterization of polylactic acid green composites and its biodegradation in a bacterial environment. Int J Polym Anal Charact 24:236–244. https://doi.org/10.1080/1023666X.2019.1567083
Haque MM-U, Puglia D, Fortunati E, Pracella M (2017) Effect of reactive functionalization on properties and degradability of poly(lactic acid)/poly(vinyl acetate) nanocomposites with cellulose nanocrystals. React Funct Polym 110:1–9. https://doi.org/10.1016/j.reactfunctpolym.2016.11.003
Hu Y, Jiang X, Ding Y, Zhang L, Yang C, Zhang J, Chen J, Yang Y (2003) Preparation and drug release behaviors of nimodipine-loaded poly(caprolactone)–poly(ethylene oxide)–polylactide amphiphilic copolymer nanoparticles. Biomaterials 24:2395–2404. https://doi.org/10.1016/S0142-9612(03)00021-8
Hubbe MA, Lavoine N, Lucia LA, Dou C (2021) Formulating bioplastic composites for biodegradability, recycling, and performance: a review. BioResources 16:2021–2083
Ismail RF, Saleh NE, Sayed AE-DH (2021) Impacts of microplastics on reproductive performance of male tilapia (Oreochromis niloticus) pre-fed on Amphora coffeaeformis. Environ Sci Pollut Res 28:68732–68744. https://doi.org/10.1007/s11356-021-14984-2
Jeon HJ, Kim MN (2013) Biodegradation of poly(L-lactide) (PLA) exposed to UV irradiation by a mesophilic bacterium. Int Biodeterior Biodegrad 85:289–293. https://doi.org/10.1016/j.ibiod.2013.08.013
John J, Nandhini AR, Velayudhaperumal Chellam P, Sillanpää M (2022) Microplastics in mangroves and coral reef ecosystems: a review. Environ Chem Lett 20:397–416. https://doi.org/10.1007/s10311-021-01326-4
Juodeikiene G, Vidmantiene D, Basinskiene L, Cernauskas D, Bartkiene E, Cizeikiene D (2015) Green metrics for sustainability of biobased lactic acid from starchy biomass vs chemical synthesis. Catal Today 239:11–16. https://doi.org/10.1016/j.cattod.2014.05.039
Karamanlioglu M, Preziosi R, Robson GD (2017) Abiotic and biotic environmental degradation of the bioplastic polymer poly(lactic acid): a review. Polym Degrad Stab 137:122–130. https://doi.org/10.1016/j.polymdegradstab.2017.01.009
Kazour M, Jemaa S, Issa C, Khalaf G, Amara R (2019) Microplastics pollution along the Lebanese coast (Eastern Mediterranean Basin): occurrence in surface water, sediments and biota samples. Sci Total Environ 696:133933. https://doi.org/10.1016/j.scitotenv.2019.133933
Kelly A, Lannuzel D, Rodemann T, Meiners KM, Auman HJ (2020) Microplastic contamination in east Antarctic sea ice. Mar Pollut Bull 154:111130. https://doi.org/10.1016/j.marpolbul.2020.111130
Kfoury G, Raquez J-M, Hassouna F, Odent J, Toniazzo V, Ruch D, Dubois P (2013) Recent advances in high performance poly(lactide): from “green” plasticization to super-tough materials via (reactive) compounding. Front Chem 1:32. https://doi.org/10.3389/fchem.2013.00032
Khalid A, Zalouk-Vergnoux A, Benali S, Mincheva R, Raquez J-M, Bertrand S, Poirier L (2021) Are bio-based and biodegradable microplastics impacting for blue mussel (Mytilus edulis)? Mar Pollut Bull 167:112295. https://doi.org/10.1016/j.marpolbul.2021.112295
Kijchavengkul T, Auras R, Rubino M, Ngouajio M, Fernandez RT (2008) Assessment of aliphatic–aromatic copolyester biodegradable mulch films. Part I: field study. Chemosphere 71:942–953. https://doi.org/10.1016/j.chemosphere.2007.10.074
Kijchavengkul T, Auras R, Rubino M, Alvarado E, Camacho Montero JR, Rosales JM (2010) Atmospheric and soil degradation of aliphatic–aromatic polyester films. Polym Degrad Stab 95:99–107. https://doi.org/10.1016/j.polymdegradstab.2009.11.048
Klein K, Piana T, Lauschke T, Schweyen P, Dierkes G, Ternes T, Schulte-Oehlmann U, Oehlmann J (2021) Chemicals associated with biodegradable microplastic drive the toxicity to the freshwater oligochaete Lumbriculus variegatus. Aquat Toxicol 231:105723. https://doi.org/10.1016/j.aquatox.2020.105723
Kliem S, Kreutzbruck M, Bonten C (2020) Review on the biological degradation of polymers in various environments. Materials 13:4586. https://doi.org/10.3390/ma13204586
Koenigstein S (2020) Arctic marine ecosystems, climate change impacts, and governance responses: an integrated perspective from the Barents Sea. In: Pongrácz E, Pavlov V, Hänninen N (eds) Arctic marine sustainability: arctic maritime businesses and the resilience of the marine environment. Springer, Cham, pp 45–71. https://doi.org/10.1007/978-3-030-28404-6_3
Kong X, Jin D, Jin S, Wang Z, Yin H, Xu M, Deng Y (2018) Responses of bacterial community to dibutyl phthalate pollution in a soil-vegetable ecosystem. J Hazard Mater 353:142–150. https://doi.org/10.1016/j.jhazmat.2018.04.015
Kumar A, Tumu VR, Ray Chowdhury S, Ramana Reddy SVS (2019) A green physical approach to compatibilize a bio-based poly (lactic acid)/lignin blend for better mechanical, thermal and degradation properties. Int J Biol Macromol 121:588–600. https://doi.org/10.1016/j.ijbiomac.2018.10.057
Lagarde F, Olivier O, Zanella M, Daniel P, Hiard S, Caruso A (2016) Microplastic interactions with freshwater microalgae: Hetero-aggregation and changes in plastic density appear strongly dependent on polymer type. Environ Pollut 215:331–339. https://doi.org/10.1016/j.envpol.2016.05.006
Lambert S, Wagner M (2016) Formation of microscopic particles during the degradation of different polymers. Chemosphere 161:510–517. https://doi.org/10.1016/j.chemosphere.2016.07.042
Lazzari S, Codari F, Storti G, Morbidelli M, Moscatelli D (2014) Modeling the pH-dependent PLA oligomer degradation kinetics. Polym Degrad Stab 110:80–90. https://doi.org/10.1016/j.polymdegradstab.2014.08.012
Le Duigou A, Davies P, Baley C (2009) Seawater ageing of flax/poly(lactic acid) biocomposites. Polym Degrad Stab 94:1151–1162. https://doi.org/10.1016/j.polymdegradstab.2009.03.025
Le Gall M, Niu Z, Curto M, Catarino AI, Demeyer E, Jiang C, Dhakal H, Everaert G, Davies P (2022) Behaviour of a self-reinforced polylactic acid (SRPLA) in seawater. Polym Test 111:107619. https://doi.org/10.1016/j.polymertesting.2022.107619
Li C, Gan Y, Dong J, Fang J, Chen H, Quan Q, Liu J (2020) Impact of microplastics on microbial community in sediments of the Huangjinxia reservoir—water source of a water diversion project in western China. Chemosphere 253:126740. https://doi.org/10.1016/j.chemosphere.2020.126740
Long M, Paul-Pont I, Hégaret H, Moriceau B, Lambert C, Huvet A, Soudant P (2017) Interactions between polystyrene microplastics and marine phytoplankton lead to species-specific hetero-aggregation. Environ Pollut 228:454–463. https://doi.org/10.1016/j.envpol.2017.05.047
Luzi F, Fortunati E, Jiménez A, Puglia D, Pezzolla D, Gigliotti G, Kenny JM, Chiralt A, Torre L (2016) Production and characterization of PLA_PBS biodegradable blends reinforced with cellulose nanocrystals extracted from hemp fibres. Ind Crops Prod 93:276–289. https://doi.org/10.1016/j.indcrop.2016.01.045
Lv S, Zhang Y, Gu J, Tan H (2017) Biodegradation behavior and modelling of soil burial effect on degradation rate of PLA blended with starch and wood flour. Colloids Surf B Biointerfaces 159:800–808. https://doi.org/10.1016/j.colsurfb.2017.08.056
Lyu SJ, Loy B, Lind D, Hobot C, Sparer R, Untereker D (2007) Kinetics and time−temperature equivalence of polymer degradation. Biomacromol 8:2301–2310. https://doi.org/10.1021/bm070313n
MacDonald RT, McCarthy SP, Gross RA (1996) Enzymatic degradability of poly(lactide): effects of chain stereochemistry and material crystallinity. Macromolecules 29:7356–7361. https://doi.org/10.1021/ma960513j
Mao Y, Ai H, Chen Y, Zhang Z, Zeng P, Kang L, Li W, Gu W, He Q, Li H (2018) Phytoplankton response to polystyrene microplastics: perspective from an entire growth period. Chemosphere 208:59–68. https://doi.org/10.1016/j.chemosphere.2018.05.170
Martin RT, Camargo LP, Miller SA (2014) Marine-degradable polylactic acid. Green Chem 16:1768–1773. https://doi.org/10.1039/C3GC42604A
Masutani K, Kimura Y (2015) Chapter 1 PLA synthesis. From the monomer to the polymer. In: Jiménez A, Peltzer M, Ruseckaite R (eds) Poly(lactic acid) science and technology: processing, properties, additives and applications. The Royal Society of Chemistry, Cambridge, pp 1–36. https://doi.org/10.1039/9781782624806-00001
Matijaković Mlinarić N, Selmani A, Brkić AL, Njegić Džakula B, Kralj D, Kontrec J (2022) Exposure of microplastics to organic matter in waters enhances microplastic encapsulation into calcium carbonate. Environ Chem Lett 20:2235–2242. https://doi.org/10.1007/s10311-022-01433-w
McCormick A, Hoellein TJ, Mason SA, Schluep J, Kelly JJ (2014) Microplastic is an abundant and distinct microbial habitat in an urban river. Environ Sci Technol 48:11863–11871. https://doi.org/10.1021/es503610r
Meimoun J, Sutapin C, Stoclet G, Favrelle A, Roussel P, Bria M, Chirachanchai S, Bonnet F, Zinck P (2021) Lactide lactone chain shuttling copolymerization mediated by an aminobisphenolate supported aluminum complex and Al(OiPr)3: access to new polylactide based block copolymers. J Am Chem Soc 143:21206–21210. https://doi.org/10.1021/jacs.1c09744
Miao Y, Rousseau C, Mortreux A, Martin P, Zinck P (2011) Access to new carbohydrate-functionalized polylactides via organocatalyzed ring-opening polymerization. Polymer 52:5018–5026. https://doi.org/10.1016/j.polymer.2011.08.040
Msuya N, Katima J, Masanja E, Temu A (2017) Poly (lactic-acid) production from monomer to polymer: a review. SciFed J Polym Sci 1:1000002
Mu X, Qi S, Liu J, Yuan L, Huang Y, Xue J, Qian L, Wang C, Li Y (2022) Toxicity and behavioral response of zebrafish exposed to combined microplastic and bisphenol analogues. Environ Chem Lett 20:41–48. https://doi.org/10.1007/s10311-021-01320-w
Nair NR, Nampoothiri KM, Pandey A (2012) Preparation of poly(l-lactide) blends and biodegradation by Lentzea waywayandensis. Biotechnol Lett 34:2031–2035. https://doi.org/10.1007/s10529-012-1005-5
Nair AB, Sivasubramanian P, Balakrishnan P, Ajith Kumar KAN, Sreekala MS (2013) Environmental effects, biodegradation, and life cycle analysis of fully biodegradable “green” composites. In: Thomas S, Joseph K, Malhotra SK, Goda K, Sreekala MS (eds) Polymer composites. Wiley, Weinheim, pp 515–568. https://doi.org/10.1002/9783527674220.ch15
Nampoothiri KM, Nair NR, John RP (2010) An overview of the recent developments in polylactide (PLA) research. Bioresour Technol 101:8493–8501. https://doi.org/10.1016/j.biortech.2010.05.092
Nanda S, Patra BR, Patel R, Bakos J, Dalai AK (2022) Innovations in applications and prospects of bioplastics and biopolymers: a review. Environ Chem Lett 20:379–395. https://doi.org/10.1007/s10311-021-01334-4
Nandhini R, Sivaprakash B, Rajamohan N, Vo D-VN (2022) Lignin and polylactic acid for the production of bioplastics and valuable chemicals. Environ Chem Lett. https://doi.org/10.1007/s10311-022-01505-x
Napper IE, Thompson RC (2019) Environmental deterioration of biodegradable, oxo-biodegradable, compostable, and conventional plastic carrier bags in the sea, soil, and open-air over a 3-year period. Environ Sci Technol 53:4775–4783. https://doi.org/10.1021/acs.est.8b06984
Naqash N, Prakash S, Kapoor D, Singh R (2020) Interaction of freshwater microplastics with biota and heavy metals: a review. Environ Chem Lett 18:1813–1824. https://doi.org/10.1007/s10311-020-01044-3
Narancic T, Verstichel S, Reddy Chaganti S, Morales-Gamez L, Kenny ST, De Wilde B, Babu Padamati R, O’Connor KE (2018) Biodegradable plastic blends create new possibilities for end-of-life management of plastics but they are not a panacea for plastic pollution. Environ Sci Technol 52:10441–10452. https://doi.org/10.1021/acs.est.8b02963
Naser AZ, Deiab I, Darras BM (2021) Poly (lactic acid)(PLA) and polyhydroxyalkanoates (PHAs), green alternatives to petroleum-based plastics: a review. RSC Adv 11:17151–17196. https://doi.org/10.1039/D1RA02390J
Niu Z, Catarino AI, Le Gall M, Curto M, Demeyer E, Hom D, Davies P, Everaert G (2022) Release of microplastics from a bio-based composite after ultraviolet irradiation. Copernicus Meetings
Obbard RW, Sadri S, Wong YQ, Khitun AA, Baker I, Thompson RC (2014) Global warming releases microplastic legacy frozen in Arctic Sea ice. Earth’s Future 2:315–320. https://doi.org/10.1002/2014EF000240
Olewnik-Kruszkowska E, Burkowska-But A, Tarach I, Walczak M, Jakubowska E (2020) Biodegradation of polylactide-based composites with an addition of a compatibilizing agent in different environments. Int Biodeterior Biodegrad 147:104840. https://doi.org/10.1016/j.ibiod.2019.104840
Persenaire O, Quintana R, Lemmouchi Y, Sampson J, Martin S, Bonnaud L, Dubois P (2014) Reactive compatibilization of poly(l-lactide)/poly(butylene succinate) blends through polyester maleation: from materials to properties. Polym Int 63:1724–1731. https://doi.org/10.1002/pi.4700
Pinto VC, Ramos T, Alves S, Xavier J, Tavares P, Moreira PMGP, Guedes RM (2015) Comparative failure analysis of PLA, PLA/GNP and PLA/CNT-COOH biodegradable nanocomposites thin films. Procedia Eng 114:635–642. https://doi.org/10.1016/j.proeng.2015.08.004
Prata JC, Lavorante BRBO, Montenegro MdC BSM, Guilhermino L (2018) Influence of microplastics on the toxicity of the pharmaceuticals procainamide and doxycycline on the marine microalgae Tetraselmis chuii. Aquat Toxicol 197:143–152. https://doi.org/10.1016/j.aquatox.2018.02.015
Qi X, Ren Y, Wang X (2017) New advances in the biodegradation of poly(lactic) acid. Int Biodeterior Biodegrad 117:215–223. https://doi.org/10.1016/j.ibiod.2017.01.010
Qin M, Chen C, Song B, Shen M, Cao W, Yang H, Zeng G, Gong J (2021) A review of biodegradable plastics to biodegradable microplastics: Another ecological threat to soil environments? J Clean Prod 312:127816. https://doi.org/10.1016/j.jclepro.2021.127816
Rafia R, Ashok KP (2014) Macroinvertebrates (oligochaetes) as indicators of pollution: a review. J Ecol Nat Environ 6:140–144
Ramos M, Fortunati E, Peltzer M, Dominici F, Jiménez A, Garrigós MC, Kenny JM (2014) Influence of thymol and silver nanoparticles on the degradation of poly(lactic acid) based nanocomposites: thermal and morphological properties. Polym Degrad Stab 108:158–165. https://doi.org/10.1016/j.polymdegradstab.2014.02.011
Reddy RL, Reddy VS, Gupta GA (2013) Study of bio-plastics as green and sustainable alternative to plastics. Int J Emerg Technol Adv Eng 3:76–81
Reeve MS, McCarthy SP, Downey MJ, Gross RA (1994) Polylactide stereochemistry: effect on enzymic degradability. Macromolecules 27:825–831
Ribba L, Lopretti M, Montes de Oca-Vásquez G, Batista D, Goyanes S, Vega-Baudrit JR (2022) Biodegradable plastics in aquatic ecosystems: latest findings, research gaps, and recommendations. Environ Res Lett 17:033003. https://doi.org/10.1088/1748-9326/ac548d
Rodriguez EJ, Marcos B, Huneault MA (2016) Hydrolysis of polylactide in aqueous media. J Appl Polym Sci 133:44152. https://doi.org/10.1002/app.44152
Sánchez-Fortún A, Fajardo C, Martín C, D’ors A, Nande M, Mengs G, Costa G, Martín M, Sánchez-Fortún S (2021) Effects of polyethylene-type microplastics on the growth and primary production of the freshwater phytoplankton species Scenedesmus armatus and Microcystis aeruginosa. Environ Expe Bot 188:104510. https://doi.org/10.1016/j.envexpbot.2021.104510
Sarker S, Huda ANMS, Niloy MNH, Chowdhury GW (2022) Trophic transfer of microplastics in the aquatic ecosystem of Sundarbans mangrove forest, Bangladesh. Sci Total Environ 838:155896. https://doi.org/10.1016/j.scitotenv.2022.155896
Schweizer M, Dieterich A, Corral Morillas N, Dewald C, Miksch L, Nelson S, Wick A, Triebskorn R, Köhler H-R (2018) The importance of sediments in ecological quality assessment of stream headwaters: embryotoxicity along the Nidda River and its tributaries in Central Hesse, Germany. Environ Sci Eur 30:22. https://doi.org/10.1186/s12302-018-0150-4
Seeley ME, Song B, Passie R, Hale RC (2020) Microplastics affect sedimentary microbial communities and nitrogen cycling. Nat Commun 11:2372. https://doi.org/10.1038/s41467-020-16235-3
Sharma S, Majumdar A, Butola BS (2021) Tailoring the biodegradability of polylactic acid (PLA) based films and ramie- PLA green composites by using selective additives. Int J Biol Macromol 181:1092–1103. https://doi.org/10.1016/j.ijbiomac.2021.04.108
Shruti VC, Kutralam-Muniasamy G (2019) Bioplastics: missing link in the era of Microplastics. Sci Total Environ 697:134139. https://doi.org/10.1016/j.scitotenv.2019.134139
Siparsky GL, Voorhees KJ, Miao F (1998) Hydrolysis of polylactic acid (PLA) and polycaprolactone (PCL) in aqueous acetonitrile solutions: autocatalysis. J Environ Polym Degrad 6:31–41. https://doi.org/10.1023/A:1022826528673
Stloukal P, Verney V, Commereuc S, Rychly J, Matisova-Rychlá L, Pis V, Koutny M (2012) Assessment of the interrelation between photooxidation and biodegradation of selected polyesters after artificial weathering. Chemosphere 88:1214–1219. https://doi.org/10.1016/j.chemosphere.2012.03.072
Stloukal P, Pekařová S, Kalendova A, Mattausch H, Laske S, Holzer C, Chitu L, Bodner S, Maier G, Slouf M, Koutny M (2015) Kinetics and mechanism of the biodegradation of PLA/clay nanocomposites during thermophilic phase of composting process. Waste Manag 42:31–40. https://doi.org/10.1016/j.wasman.2015.04.006
Su Y, Cheng Z, Hou Y, Lin S, Gao L, Wang Z, Bao R, Peng L (2022) Biodegradable and conventional microplastics posed similar toxicity to marine algae Chlorella vulgaris. Aquat Toxicol 244:106097. https://doi.org/10.1016/j.aquatox.2022.106097
Sun Y, Lee D, Wang Y, Li S, Ying J, Liu X, Xu G, Gwon J, Wu Q (2021) Thermal decomposition behavior of 3D printing filaments made of wood-filled polylactic acid/starch blend. J Appl Polym Sci 138:49944. https://doi.org/10.1002/app.49944
Suwaki CH, De-La-Cruz LT, Lopes RM (2020) Impacts of microplastics on the swimming behavior of the copepod Temora turbinata (Dana, 1849). Fluids 5:103. https://doi.org/10.3390/fluids5030103
Taib N-AAB, Rahman MR, Huda D, Kuok KK, Hamdan S, Bakri MKB, Julaihi MRMB, Khan A (2022) A review on poly lactic acid (PLA) as a biodegradable polymer. Polym Bull. https://doi.org/10.1007/s00289-022-04160-y
Thery J, Bialais C, Kazour M, Moreau M, Dufour D, Benali S, Amara R, Monchy S, Raquez JM, Souissi S (2022) A new method for microplastics identification in copepods. Front Environ Chem 3:905303. https://doi.org/10.3389/fenvc
Thompson RC, Olsen Y, Mitchell RP, Davis A, Rowland SJ, John AWG, McGonigle D, Russell AE (2004) Lost at sea: Where is all the plastic? Science 304:838–838. https://doi.org/10.1126/science.1094559
Tsuji H, Ikada Y (1998) Properties and morphology of poly(L-lactide). II. Hydrolysis in alkaline solution. J Polym Sci Part A Polym Chem 36:59–66. https://doi.org/10.1002/(SICI)1099-0518(19980115)36:1%3c59::AID-POLA9%3e3.0.CO;2-X
Tsuji H, Suzuyoshi K (2002a) Environmental degradation of biodegradable polyesters 1. Poly(ε-caprolactone), poly[(R)-3-hydroxybutyrate], and poly(l-lactide) films in controlled static seawater. Polym Degrad Stab 75:347–355. https://doi.org/10.1016/S0141-3910(01)00240-3
Tsuji H, Suzuyoshi K (2002b) Environmental degradation of biodegradable polyesters 2. Poly(ε-caprolactone), poly[(R)-3-hydroxybutyrate], and poly(l-lactide) films in natural dynamic seawater. Polym Degrad Stab 75:357–365. https://doi.org/10.1016/S0141-3910(01)00239-7
van Nostrum CF, Veldhuis TFJ, Bos GW, Hennink WE (2004) Hydrolytic degradation of oligo(lactic acid): a kinetic and mechanistic study. Polymer 45:6779–6787. https://doi.org/10.1016/j.polymer.2004.08.001
Vardar S, Demirel B, Onay TT (2022) Degradability of bioplastics in anaerobic digestion systems and their effects on biogas production: a review. Rev Environ Sci Bio/technol 21:205–223. https://doi.org/10.1007/s11157-021-09610-z
Vincent SGT, Jennerjahn TC, Ramasamy K (2021) Microbial communities in coastal sediments: structure and functions. Elsevier, Amsterdam
Wan L, Zhang Y (2018) Jointly modified mechanical properties and accelerated hydrolytic degradation of PLA by interface reinforcement of PLA-WF. J Mech Behav Biomed Mater 88:223–230. https://doi.org/10.1016/j.jmbbm.2018.08.016
Wan L, Zhou S, Zhang Y (2019) Parallel advances in improving mechanical properties and accelerating degradation to polylactic acid. Int J Biol Macromol 125:1093–1102. https://doi.org/10.1016/j.ijbiomac.2018.12.148
Wang J, Huang M, Wang Q, Sun Y, Zhao Y, Huang Y (2020) LDPE microplastics significantly alter the temporal turnover of soil microbial communities. Sci Total Environ 726:138682. https://doi.org/10.1016/j.scitotenv.2020.138682
Wang C, Yu J, Lu Y, Hua D, Wang X, Zou X (2021) Biodegradable microplastics (BMPs): A new cause for concern? Environ Sci Pollut Res 28:66511–66518. https://doi.org/10.1007/s11356-021-16435-4
Wei X-F, Bohlén M, Lindblad C, Hedenqvist M, Hakonen A (2021) Microplastics generated from a biodegradable plastic in freshwater and seawater. Water Res 198:117123. https://doi.org/10.1016/j.watres.2021.117123
Wei X-F, Capezza AJ, Cui Y, Li L, Hakonen A, Liu B, Hedenqvist MS (2022) Millions of microplastics released from a biodegradable polymer during biodegradation/enzymatic hydrolysis. Water Res 211:118068. https://doi.org/10.1016/j.watres.2022.118068
Weinstein JE, Dekle JL, Leads RR, Hunter RA (2020) Degradation of bio-based and biodegradable plastics in a salt marsh habitat: another potential source of microplastics in coastal waters. Mar Pollut Bull 160:111518. https://doi.org/10.1016/j.marpolbul.2020.111518
Wilfred O, Tai H, Marriott R, Liu Q, Tverezovskiy V, Curling S, Fan Z, Wang W (2018) Biodegradation of polyactic acid and starch composites in compost and soil. Int J Nano Res 1:1–11
Wright SL, Rowe D, Thompson RC, Galloway TS (2013) Microplastic ingestion decreases energy reserves in marine worms. Curr Biol 23:R1031–R1033. https://doi.org/10.1016/j.cub.2013.10.068
Wu C-S (2012) Preparation, characterization, and biodegradability of renewable resource-based composites from recycled polylactide bioplastic and sisal fibers. J Appl Polym Sci 123:347–355. https://doi.org/10.1002/app.34223
Xie L, Xu H, Wang Z-P, Li X-J, Chen J-B, Zhang Z-J, Yin H-M, Zhong G-J, Lei J, Li Z-M (2014) Toward faster degradation for natural fiber reinforced poly(lactic acid) biocomposites by enhancing the hydrolysis-induced surface erosion. J Polym Res 21:357. https://doi.org/10.1007/s10965-014-0357-z
Yaacob ND, Ismail H, Ting SS (2016) Soil burial of polylactic acid/paddy straw powder biocomposite. BioResources 11:1255–1269
Yagi H, Ninomiya F, Funabashi M, Kunioka M (2012) Anaerobic biodegradation of poly (lactic acid) film in anaerobic sludge. J Polym Environ 20:673–680. https://doi.org/10.1007/s10924-012-0472-z
Yamano N, Kawasaki N, Oshima M, Nakayama A (2014) Polyamide 4 with long-chain fatty acid groups—suppressing the biodegradability of biodegradable polymers. Polym Degrad Stab 108:116–122. https://doi.org/10.1016/j.polymdegradstab.2014.06.011
Yang X, Hakkarainen M (2015) Migration resistant glucose esters as bioplasticizers for polylactide. J Appl Polym Sci 132:41928. https://doi.org/10.1002/app.41928
Yokota K, Mehlrose M (2020) Lake phytoplankton assemblage altered by irregularly shaped PLA body wash microplastics but not by PS calibration beads. Water 12:2650. https://doi.org/10.3390/w12092650
Yu J, Tian J-Y, Xu R, Zhang Z-Y, Yang G-P, Wang X-D, Lai J-G, Chen R (2020) Effects of microplastics exposure on ingestion, fecundity, development, and dimethylsulfide production in Tigriopus japonicus (Harpacticoida, copepod). Environ Pollut 267:115429. https://doi.org/10.1016/j.envpol.2020.115429
Zaaba NF, Jaafar M (2020) A review on degradation mechanisms of polylactic acid: hydrolytic, photodegradative, microbial, and enzymatic degradation. Polym Eng Sci 60:2061–2075. https://doi.org/10.1002/pen.25511
Zandi A, Zanganeh A, Hemmati F, Mohammadi-Roshandeh J (2019) Thermal and biodegradation properties of poly(lactic acid)/rice straw composites: effects of modified pulping products. Iran Polym J 28:403–415. https://doi.org/10.1007/s13726-019-00709-3
Zhang R, Ma PX (2004) Biomimetic polymer/apatite composite scaffolds for mineralized tissue engineering. Macromol Biosci 4:100–111. https://doi.org/10.1002/mabi.200300017
Zhang C, Chen X, Wang J, Tan L (2017) Toxic effects of microplastic on marine microalgae Skeletonema costatum: interactions between microplastic and algae. Environ Pollut 220:1282–1288. https://doi.org/10.1016/j.envpol.2016.11.005
Zhang C, Jeong C-B, Lee J-S, Wang D, Wang M (2019a) Transgenerational proteome plasticity in resilience of a marine copepod in response to environmentally relevant concentrations of microplastics. Environ Sci Technol 53:8426–8436. https://doi.org/10.1021/acs.est.9b02525
Zhang P, He Y-Y, Gao D, Cai Y, Liu B (2019b) Hydrolytic and thermal degradation of polyethylene glycol compatibilized poly(lactic acid)-nanocrystalline cellulose bionanocomposites. J Appl Polym Sci 136:46933. https://doi.org/10.1002/app.46933
Zhang X, Xia M, Su X, Yuan P, Li X, Zhou C, Wan Z, Zou W (2021) Photolytic degradation elevated the toxicity of polylactic acid microplastics to developing zebrafish by triggering mitochondrial dysfunction and apoptosis. J Hazard Mater 413:125321. https://doi.org/10.1016/j.jhazmat.2021.125321
Zimmermann L, Göttlich S, Oehlmann J, Wagner M, Völker C (2020) What are the drivers of microplastic toxicity? Comparing the toxicity of plastic chemicals and particles to Daphnia magna. Environ Pollut 267:115392. https://doi.org/10.1016/j.envpol.2020.115392
Acknowledgements
The authors are thankful to Program for Early-Stage Researchers in Lille (PEARL), coordinated by the Foundation I-SITE ULNE, for the funding under the project ‘Assessing the toxicity of plastic fragments on zooplankton ecology via video tracking and behavioral analysis’ TOPAZ. Sami SOUISSI acknowledges the French CNRS delegation program providing him one sabbatical semester in 2022–2023. The authors are also grateful to the Editor for his valuable comments on the earlier versions of the manuscript, which really helped to substantially improve the quality of the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest regarding the publication of this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ali, W., Ali, H., Gillani, S. et al. Polylactic acid synthesis, biodegradability, conversion to microplastics and toxicity: a review. Environ Chem Lett 21, 1761–1786 (2023). https://doi.org/10.1007/s10311-023-01564-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-023-01564-8