Abstract
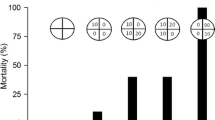
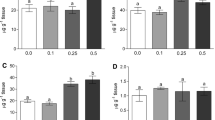
Heavy metal discharges to aquatic environment are of great concern due to their toxicity and accumulative behavior. Zinc is an essential trace element required for different physiological functions and plays important role in cellular metabolism. However, it becomes toxic when elevated concentrations are introduced into the environment. The aim of this work is to analyze zinc induced biochemical changes in the brain tissues of Labeo rohita fingerlings using Fourier Transformation Infrared Spectroscopy. Several important features have been observed in the zinc intoxicated brain tissues, namely, altered membrane lipid, altered protein profile and decreased glycogen content, indicating an alteration in the lipid and protein profiles leading to modification in membrane composition. Further, it is observed that the acute exposure to zinc causes some alteration in protein profile with a decrease in α-helix and an increase in random coil structures.




Similar content being viewed by others
References
Akahori A, Jozwiak Z, Gabryelak T, Gondko R (1999) Effect of zinc on carp (Cyprinus carpio L.) erythrocytes. Comp Biochem Physiol C 123:209–215. doi:10.1016/S0305-0491(99)00063-2
Akkas SB, Severcan M, Yilmaz O, Severcan F (2007) Effects of lipoic acid supplementation on rat brain tissue: an FTIR spectroscopic and neural network study. Food Chem 105:1281–1288. doi:10.1016/j.foodchem.2007.03.015
Alsop DH, Wood CM (2000) Kinetic analysis of zinc accumulation in the gills of juvenile rainbow trout: effects of zinc accumulation and implications for biotic ligand modeling. Environ Toxicol Chem 19:1911–1918. doi :10.1897/1551-5028(2000)019<1911:KAOZAI>2.3.CO;2
APHA (1995) Standard methods for the determination of water and wastes, 19th Edn. American Public Health Association, American Water Works Association, and the Water Environment Federation, Washington, D.C
Begum G (2004) Carbofuran insecticide induced biochemical alterations in liver and muscle tissues of the Clarias bartrachus (linn) and recovery response. Aquat Toxicol 66:83–92. doi:10.1016/j.aquatox.2003.08.002
Benedetti E, Bramanti E, Papineschi F, Rossi I, Benedetti Enzi (1997) Determination of the relative amounts of nucleic acids and proteins in leukemic and normal lymphocytes by means of Fourier transform infrared microscopy. Appl Spectrosc 51:792–797. doi:10.1366/0003702971941304
Cakmak G, Togan I, Severcan F (2006) 17β-Estradiol induced compositional, structural and functional changes in rainbow trout liver, revealed by FT-IR spectroscopy: a comparative study with nonylphenol. Aquat Toxicol 77:53–63. doi:10.1016/j.aquatox.2005.10.015
Chu HL, Liu TY, Lin SY (2001) Effect of cyanide concentrations on the secondary structures of protein in the crude homogenates of the fish gill tissue. Aquat Toxicol 55:171–176. doi:10.1016/S0166-445X(01)00177-1
Filipe PM, Fermandes AC, Manso CF (1995) Effect of zinc on copper-induced and spontaneous lipid peroxidation. Biol Trace Elem Res 17:51–56. doi:10.1007/BF02790100
Finney DJ (1971) Probit analysis. Cambridge University Press, Cambridge, p 333
Hampton MB, Orrenius S (1997) Dual regulation of caspase activity by hydrogen peroxide: implications for apoptosis. FEBS Lett 414:552–556. doi:10.1016/S0014-5793(97)01068-5
Haris PI, Severcan F (1999) FTIR spectroscopic characterization of protein structure in aqueous and non-aqueous media. J Mol Cat Enz 7:207–221. doi:10.1016/S1381-1177(99)00030-2
Hogstrand C, Wood CM (1995) Mechanisms for zinc acclimation in fresh-water rainbow-trout. Mar Environ Res 39:131–135. doi:10.1016/0141-1136(94)00040-V
Kneipp J, Lasch P, Baldauf F, Beeks M, Naumann D (2000) Biochim Biophys Acta 1501:189–199
Liu K, Bose T, Mantsch HH (2002) Infrared spectroscopic study of diabetic platelets. Vib Spectrosc 28:131–136. doi:10.1016/S0924-2031(01)00163-1
Melin A, Perromat A, Deleris G (2000) Pharmacologic application of FT-IR spectroscopy: in vivo toxicity of carbon tetrachloride on rat liver. Biopolymers 57:160–168. doi :10.1002/(SICI)1097-0282(2000)57:3<160::AID-BIP4>3.0.CO;2-1 Bio spectroscopy
Neduka JKC, Conotaues E, Obiakor E (2006) Selective bioaccumulation of metals by different parts of some fish species from crude oil polluted water. Environ Contam Toxicol 77:846–853. doi:10.1007/s00128-006-1221-6
Ping YF, Smerker J, Barrett J, Servey J, Comic A, Margaret H (2007) Characterization of proteins in mouse blood by Fourier transform infrared spectroscopy. Spectrosc Lett 40:465–473. doi:10.1080/00387010701295786
Rice-Evans CA, Diplock AT, Symos MCR (1991) Technique in free radical research. Elsevier, New York, p 207
Roche H, Boge G (1993) Effects of Cu, Zn, and Cr salts on antioxidant enzyme activites in vitro of red blood cells of a marine fish Dicentrarchus labrax. Toxicol In Vitro 7:623–629. doi:10.1016/0887-2333(93)90096-N
Samuvel S, Kathirvel R, Jayavlu T, Chinakkannu P (2005) Protein oxidative damage in arsenic induced rat brain: influence of DL-α-lipoic acid. Toxicol Lett 155:27–34. doi:10.1016/j.toxlet.2004.08.001
Severcan F, Gorgulu G, Gorgulu TS, Guray T (2005) Rapid monitoring of diabetes-induced lipid peroxidation by Fourier transform infrared spectroscopy: evidence from rat liver microsomal membranes. Anal Biochem 339:36–40. doi:10.1016/j.ab.2005.01.011
Sparks T (2000) Statistics in ecotoxicology. Wiley, New York, pp 320
Susi H, Byler DM (1983) Protein structure by Fourier transform infrared spectroscopy: second derivative spectra. Biochem Biophys Res Commun 115:391–397. doi:10.1016/0006-291X(83)91016-1
Toyran N, Zorlu F, Donmez G, Ode K, Severcan F (2004) Chronic hypo perfusion alters the content and structure of proteins and lipids of rat brain homogenates: a Fourier transform infrared spectroscopy study. Eur Biophys J Biophys Lett 33:549–554
Viarengo A (1989) Heavy metals in marine invertebrates: mechanisms of regulation and toxicity at the level. Rev Aquat Sci 1:295–317
Wang J, Chi C, Lin S, Chern Y (1997) Conformational changes in gastric carcinoma cell membrane protein correlated to cell viability after treatment with adamantyl maleimide. Anticancer Res 17:3473–3478
Zago MP, Oteiza PJ (2001) Free Radic Biol Med 31:266–274. doi:10.1016/S0891-5849(01)00583-4
Acknowledgments
The authors are thankful and grateful to the authorities of Annamalai University for providing all necessary facilities to carry out the present work successfully.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Palaniappan, P.R., Pramod, K.S. & Vijayasundaram, V. Effect of acute concentration of zinc on the biochemical contents of brain of Labeo rohita: an FT-IR study. Environ Chem Lett 7, 313–319 (2009). https://doi.org/10.1007/s10311-008-0171-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-008-0171-0