Abstract
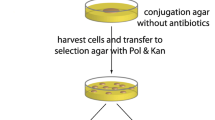
Bacillus sp. strains as attractive hosts for the production of heterologous secretory proteins usually play important roles in bio-industry. However, low transformation efficiency of exogenous plasmids limited the application of Bacillus species. Here, a novel plasmid interspecific transfer system, with high transformation efficiency, high positive rate, and convenient manipulation, has been successfully constructed. A high electrotransformation efficiency strain Bacillus subtilis F-168 containing the counter-selectable marker mazF was used as the plasmid donor strain in this transfer method. A shuttled plasmid pBE980 and its recombinant plasmids pBE980::pulA and pBE980::HSPA were successfully transferred into the recipient Bacillus strains (Bacillus amyloliquefaciens 66, Bacillus licheniformis 124 and Bacillus megaterium 258) by this method. After co-culturing the donor cells (OD600nm = 1.3–1.7) and the recipient cells (OD600nm = 0.5–0.9) for 24 h in 22 °C, more than 1.0 × 105 positive transformants were obtained and a interspecific transformation efficiency of 1.0 × 10−3. It would provide a new approach for genetic manipulation in Bacillus strains and accelerate the research progress of the wild Bacillus strains in bio-industry.






Similar content being viewed by others
References
Akamatsu T, Taguchi H (2012) Plasmid transformation of competent Bacillus subtilis by lysed protoplast DNA. J Biosci Bioeng 114:138–143
Chen Y-YM, Shieh H-R, Lin C-T et al (2011) Properties and construction of plasmid pFW213, a shuttle vector with the oral Streptococcus origin of replication. Appl Environ Microb 77:3967–3974
Chowdhury SP, Hartmann A, Gao X et al (2015) Biocontrol mechanism by root-associated Bacillus amyloliquefaciens FZB42—a review. Front Microbiol 6:780
Gao C, Xue Y, Ma Y (2011) Protoplast transformation of recalcitrant alkaliphilic Bacillus sp. with methylated plasmid DNA and a developed hard agar regeneration medium. PLoS One 6:e28148
Hmani H, Daoud L, Jlidi M et al (2017) A Bacillus subtilis strain as probiotic in poultry: selection based on in vitro functional properties and enzymatic potentialities. J Ind Microbiol Biot 44:1157–1166
Jensen GB, Wilcks A, Petersen SS et al (1995) The genetic basis of the aggregation system in Bacillus thuringiensis subsp. israelensis is located on the large conjugative plasmid pXO16. J Bacteriol 177:2914–2917
Kolodkin-Gal I, Hazan R, Gaathon A et al (2007) A linear pentapeptide is a quorum-sensing factor required for mazEF-mediated cell death in Escherichia coli. Science 318:652–655
Li Y, Gu Z, Zhang L et al (2017) Inducible expression of trehalose synthase in Bacillus licheniformis. Protein Expr Purif 130:115–122
Liu L, Liu Y, Shin HD et al (2013) Developing Bacillus spp. as a cell factory for production of microbial enzymes and industrially important biochemicals in the context of systems and synthetic biology. Appl Microbiol Biot 97:6113–6127
Melo AL, Soccol VT, Soccol CR (2016) Bacillus thuringiensis: mechanism of action, resistance, and new applications: a review. Crit Rev Biotechnol 36:317–326
Naglich JG, Andrews RE Jr (1988) Tn916-dependent conjugal transfer of PC194 and PUB110 from Bacillus subtilis into Bacillus thuringiensis subsp. israelensis. Plasmid 20:113–126
Nguyen HD, Nguyen QA, Ferreira RC et al (2005) Construction of plasmid-based expression vectors for Bacillus subtilis exhibiting full structural stability. Plasmid 54:241–248
Philibert T, Rao Z, Yang T et al (2016) Heterologous expression and characterization of a new heme-catalase in Bacillus subtilis 168. J Ind Microbiol Biot 43:729–740
Sambrock J, Russel D (2001) Molecular cloning: a laboratory manual, 3rd edn. Cold Spring Harbor Laboratory Press, New York
Sella SR, Vandenberghe LP, Soccol CR (2015) Bacillus atrophaeus: main characteristics and biotechnological applications—a review. Crit Rev Biotechnol 35:533–545
Song P, Liu S, Guo X et al (2015) Scarless gene deletion in methylotrophic Hansenula polymorpha by using mazF as counter-selectable marker. Anal Biochem 468:66–74
Spizizen J (1958) Transformation of biochemically deficient strains of Bacillus subtilis by deoxyribonucleate. P Natl Acad Sci USA 44:1072–1078
Strohmaier H, Noiges R, Kotschan S et al (1998) Signal transduction and bacterial conjugation: characterization of the role of ArcA in regulating conjugative transfer of the resistance plasmid R1. J Mol Biol 277:309–316
Titok MA, Chapuis J, Selezneva YV et al (2003) Bacillus subtilis soil isolates: plasmid replicon analysis and construction of a new theta-replicating vector. Plasmid 49:53–62
Tjalsma H, Koetje EJ, Kiewiet R et al (2004) Engineering of quorum-sensing systems for improved production of alkaline protease by Bacillus subtilis. J Appl Microbiol 96:569–578
Tominaga Y, Ohshiro T, Suzuki H (2016) Conjugative plasmid transfer from Escherichia coli is a versatile approach for genetic transformation of thermophilic Bacillus and Geobacillus species. Extremophiles 20:375–381
Wu SC, Wong SL (1999) Development of improved pUB110-based vectors for expression and secretion studies in Bacillus subtilis. J Biotechnol 72:185–195
Xue G-P, Johnson JS, Dalrymple BP (1999) High osmolarity improves the electro-transformation efficiency of the gram-positive bacteria Bacillus subtilis and Bacillus licheniformis. J Microbiol Methods 34:183–191
Yzturk S, Yalik P, Yzdamar TH (2016) Fed-batch biomolecule production by Bacillus subtilis: a state of the art review. Trends Biotechnol 34:329–345
Zhang K, Duan X, Wu J (2016) Multigene disruption in undomesticated Bacillus subtilis ATCC 6051a using the CRISPR/Cas9 system. Sci Rep UK 6:27943
Zhang XZ, Yan X, Cui ZL et al (2006) mazF, a novel counter-selectable marker for unmarked chromosomal manipulation in Bacillus subtilis. Nucleic Acids Res 34:e71
Zhang XZ, You C, Zhang YH (2014) Transformation of Bacillus subtilis. In: Sun L, Shou W (eds) Engineering and analyzing multicellular systems. Methods in molecular biology (methods and protocols), vol 1151. Humana Press, New York
Zhang Z, Ding ZT, Shu D et al (2015) Development of an efficient electroporation method for iturin A-producing Bacillus subtilis ZK. Int J Mol Sci 16:7334–7351
Zheng H, Liu Y, Liu X et al (2012) Overexpression of a Paenibacillus campinasensis xylanase in Bacillus megaterium and its applications to biobleaching of cotton stalk pulp and saccharification of recycled paper sludge. Bioresour Technol 125:182–187
Zhou C, Shi L, Ye B et al (2017) pheS (*), an effective host-genotype-independent counter-selectable marker for marker-free chromosome deletion in Bacillus amyloliquefaciens. Appl Microbiol Biot 101:217–227
Acknowledgements
This work was supported by the National Natural Science Fund of China (Grant 31701534), the Key Deployment Project in Chinese Academy of Sciences (Grant KFJ-STS-ZDTP-016-1, KFZD-SW-211-2), and the Tianjin Science & Technology Planning Project (Grant 16YFZCSY00790,16YFXTSY00530 and 15YFYssy00040).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no competing interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhao, X., Xu, J., Tan, M. et al. Construction of a plasmid interspecific transfer system in Bacillus species with the counter-selectable marker mazF. J Ind Microbiol Biotechnol 45, 417–428 (2018). https://doi.org/10.1007/s10295-018-2038-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-018-2038-0