Abstract
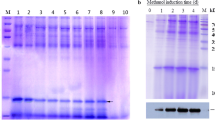
Production of recombinant protein bio-insecticides on a commercial scale can only be cost effective if host strains with very high expression levels are available. A recombinant fusion protein containing an arthropod toxin, ω-hexatoxin-Hv1a, (from funnel web spider Hadronyche versuta) linked to snowdrop lectin (Galanthus nivalis agglutinin; GNA) is an effective oral insecticide and candidate biopesticide. However, the fusion protein was vulnerable to proteolysis during production in the yeast Pichia pastoris. To prevent proteolysis, the Hv1a/GNA fusion expression construct was modified by site-directed mutagenesis to remove a potential Kex2 cleavage site at the C-terminus of the Hv1a peptide. To obtain a high expressing clone of P. pastoris to produce recombinant Hv1a/GNA, a straightforward method was used to produce multi-copy expression plasmids, which does not require multiple integrations to give clones of P. pastoris containing high copy numbers of the introduced gene. Removal of the Kex2 site resulted in increased levels of intact fusion protein expressed in wild-type P. pastoris strains, improving levels of intact recombinant protein recoverable. Incorporation of a C-terminal (His)6 tag enabled single step purification of the fusion protein. These modifications did not affect the insecticidal activity of the recombinant toxin towards lepidopteran larvae. Introduction of multiple expression cassettes increased the amount of secreted recombinant fusion protein in a laboratory scale fermentation by almost tenfold on a per litre of culture basis. Simple modifications in the expression construct can be advantageous for the generation of high expressing P. pastoris strains for production of a recombinant protein, without altering its functional properties.




Similar content being viewed by others
References
Baumgartner P, Harper K, Raemaekers RJ, Durieux A, Gatehouse AMR, Davies HV, Taylor MA (2003) Large-scale production and purification of recombinant Galanthus nivalis agglutinin (GNA) expressed in the methylotrophic yeast Pichia pastoris. Biotech Lett 25:1281–1285
Cino J (1999) High-yield protein production from Pichia pastoris yeast: a protocol for benchtop fermentation. Application Note. New Brunswick Scientific, Edison
Daly R, Hearn MTW (2005) Expression of heterologous proteins in Pichia pastoris: a useful experimental tool in protein engineering and production. J Mol Recog 18:119–138
Down RE, Fitches EC, Wiles DP, Corti P, Bell HA, Gatehouse JA, Edwards JP (2006) Insecticidal spider venom toxin fused to snowdrop lectin is toxic to the peach-potato aphid, Myzus persicae (Hemiptera: Aphididae) and the rice brown planthopper, Nilaparvata lugens (Hemiptera: Delphacidae). Pest Manag Sci 62:77–85
Fitches EC, Bell HA, Powell ME, Back E, Sargiotti C, Weaver RJ, Gatehouse JA (2010) Insecticidal activity of scorpion toxin (ButaIT) and snowdrop lectin (GNA) containing fusion proteins towards pest species of different orders. Pest Manag Sci 66:74–83
Fitches EC, Edwards MG, Mee C, Grishin E, Gatehouse AMR, Edwards JP, Gatehouse JA (2004) Fusion proteins containing insect-specific toxins as pest control agents: snowdrop lectin delivers fused insecticidal spider venom toxin to insect haemolymph following oral ingestion. J Insect Phys 50:61–71
Fitches EC, Gatehouse JA (1998) A comparison of the short and long term effects of insecticidal lectins on the activities of soluble and brush border enzymes of tomato moth larvae (Lacanobia oleracea). J Insect Phys 44:1213–1224
Fitches EC, Pyati PS, King GF, Gatehouse JA (2012) Fusion to snowdrop lectin dramatically enhances the oral activity of the insecticidal peptide ω-hexatoxin-Hv1a by mediating its delivery to the central nervous system. PLoS One 7:e39389
Fletcher JI, Smith R, O’Donoghue SI, Nilges M, Connor M, Howden MEH, Christie MJ, King GF (1997) The structure of a novel insecticidal neurotoxin, ω-atracotoxin-Hv1, from the venom of an Australian funnel web spider. Nat Struct Biol 4:559–566
Gasser B, Prielhofer R, Marx H, Maurer M, Nocon J, Steiger M, Puxbaum V, Sauer M, Mattanovich D (2013) Pichia pastoris: protein production host and model organism for biomedical research. Future Microbiol 8:191–208
Gleeson MAG, White CE, Meininger DP, Komives EA (1998) Generation of protease-deficient strains and their use in heterologous protein expression. Methods Mol Biol 103:81–94
Higgins DR, Cregg JM (1998) Pichia protocols. Humana Press, Totowa
Li P, Anumanthan A, Gao X, Ilangovan K, Suzara VV, Düzgüneş N, Renugopalakrishnan V (2007) Expression of recombinant proteins in Pichia Pastoris. Appl Biochem Biotech 142:105–124
Lõoke M, Kristjuhan K, Kristjuhan A (2011) Extraction of genomic DNA from yeasts for PCR based applications. Biotechniques 50:325–328
Mansur M, Cabello C, Hernandez L, Pais J, Varas L, Valde J, Terrero Y, Hidalgo A, Plana L, Besada V, Garcia L, Lamazares E, Castellanos L, Martınez E (2005) Multiple gene copy number enhances insulin precursor secretion in the yeast Pichia pastoris. Biotech Letts 27:339–345
Raemaekers RJM, deMuro L, Gatehouse JA, Fordham-Skelton AP (1999) Functional phytohemagglutinin (PHA) and Galanthus nivalis agglutinin (GNA) expressed in Pichia pastoris—Correct N terminal processing and secretion of heterologous proteins expressed using the PHA-E signal peptide. Eur J Biochem 265:394–403
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual, 3rd edn. Cold Spring Harbor Laboratory, Spring Harbor, New York
Sinha J, Plantz BA, Inan M, Meagher MM (2005) Causes of proteolytic degradation of secreted recombinant proteins produced in methylotrophic yeast Pichia pastoris: case study with recombinant ovine interferon-tau. Biotechnol Bioeng 89:102–112
Sreekrishna K (2012) Pichia optimization of protein expression. In: Flickinger MC (ed) Encyclopedia of industrial biotechnology: bioprocess, bioseparation, and cell technology. Wiley, New York
Sulan L, Dongting Z, Kexuan T (2005) Functional GNA expressed in E. coli with high efficiency and its effect on Ceratovacuna lanigera Zehntner. Appl Microbiol Biotech 69:184–191
Tedford HW, Gilles N, Ménez A, Doering CJ, Zamponi GW, King GF (2004) Scanning mutagenesis of ω-atracotoxin-Hv1a reveals a spatially restricted epitope that confers selective activity against invertebrate calcium channels. J Biol Chem 279:44133–44140
Trung NP, Fitches EC, Gatehouse JA (2006) A fusion protein containing a lepidopteran-specific toxin from the South Indian red scorpion (Mesobuthus tamulus) and snowdrop lectin shows oral toxicity to target insects. BMC Biotech 6:18
Vassileva A, Chugh DA, Swaminathan S, Khanna N (2001) Effect of copy number on the expression levels of Hepatitif B surface antigen in the methylotrophic yeast Pichia pastoris. Protein Express Purif 21:71–80
Zhu T, Guo M, Tang Z, Zhuang Z, Chu J, Zhang S (2009) Efficient generation of multi-copy strains for optimizing secretory expression of porcine insulin precursor in yeast Pichia pastoris. J Appl Micro 107:954–963
Acknowledgments
This work was supported by funding from the Technology Strategy Board (Project 100865).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pyati, P., Fitches, E. & Gatehouse, J.A. Optimising expression of the recombinant fusion protein biopesticide ω-hexatoxin-Hv1a/GNA in Pichia pastoris: sequence modifications and a simple method for the generation of multi-copy strains. J Ind Microbiol Biotechnol 41, 1237–1247 (2014). https://doi.org/10.1007/s10295-014-1466-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-014-1466-8