Abstract
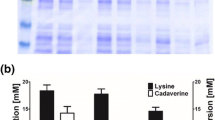
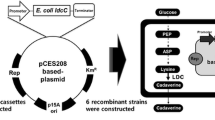
Cadaverine (1,5-pentanediamine, diaminopentane), the desired raw material of bio-polyamides, is an important industrial chemical with a wide range of applications. Biosynthesis of cadaverine in Corynebacterium glutamicum has been a competitive way in place of petroleum-based chemical synthesis method. To date, the cadaverine exporter has not been found in C. glutamicum. In order to improve cadaverine secretion, the cadaverine–lysine antiporter CadB from Escherichia coli was studied in C. glutamicum. Fusion expression of cadB and green fluorescent protein (GFP) gene confirmed that CadB could express in the cell membrane of C. glutamicum. Co-expression of cadB and ldc from Hafnia alvei in C. glutamicum showed that the cadaverine secretion rate increased by 22 % and the yield of total cadaverine and extracellular cadaverine increased by 30 and 73 %, respectively. Moreover, the recombinant strain cultured at acid and neutral pH separately hardly had any difference in cadaverine concentrations. These results suggested that CadB could be expressed in the cell membrane of C. glutamicum and that recombinant CadB could improve cadaverine secretion and the yield of cadaverine. Moreover, the pH value did not affect the function of recombinant CadB. These results may be a promising metabolic engineering strategy for improving the yield of the desired product by enhancing its export out of the cell.





Similar content being viewed by others
References
Buschke N, Becker J, Wittmann C et al (2013) Systems metabolic engineering of xylose-utilizing Corynebacterium glutamicum for production of 1, 5-diaminopentane. Biotechnol J 8:557–570
Buschke N, Schroder H, Wittmann C (2011) Metabolic engineering of Corynebacterium glutamicum for production of 1,5-diaminopentane from hemicellulose. Biotechnol J 6:306–317
Cassan F, Maiale S, Masciarelli O et al (2009) Cadaverine production by Azospirillum brasilense and its possible role in plant growth promotion and osmotic stress mitigation. Eur J Soil Biol 45:12–19
Casalino M, Latella MC, Prosseda G et al (2005) Molecular evolution of the lysine decarboxylase defective phenotype in Shigella sonnei. Int J Med Microbiol 294:503–512
Fecker LF, Beier H, Berlin J (1986) Cloning and characterization of a lysine decarboxylase gene from Hafnia alvei. Mol Gen Genet MGG 203(1):177–184
Haneburger I, Fritz G, Jurkschat N et al (2012) Deactivation of the E. coli pH stress sensor CadC by cadaverine. J Mol Biol 424(1–2):15–27
Igarashi K, Kashiwagi K (2010) Modulation of cellular function by polyamines. Int J Biochem Cell B 42:39–51
Kind S, Jeong WK, Schroder H et al (2010) Systems-wide metabolic pathway engineering in Corynebacterium glutamicum for bio-based production of diaminopentane. Metab Eng 12:341–351
Kind S, Jeong WK, Schröder H et al (2010) Identification and elimination of the competing N-acetyl-diaminopentane pathway for improved production of diaminopentane by Corynebacterium glutamicum. Appl Environ Microbiol 76:5175–5180
Kind S, Kreye S, Wittmann C (2011) Metabolic engineering of cellular transport for overproduction of the platform chemical 1,5-diaminopentane in Corynebacterium glutamicum. Metab Eng 13:617–627
Lemonnier M, Lane D (1998) Expression of the second lysine decarboxylase gene of Escherichia coli. Microbiology 144:751–760
Meng SY, Bennett GN (1992) Nucleotide sequence of the Escherichia coli cad operon: a system for neutralization of low extracellular pH. J Bacteriol 174:2659–2669
Mimitsuka T, Sawai H, Hatsu M, Yamada K (2007) Metabolic engineering of Corynebacterium glutamicum for cadaverine fermentation. Biosci Biotechnol Biochem 71:2130–2135
Neely MN, Olson ER (1996) Kinetics of expression of the Escherichia coli cad operon as a function of pH and lysine. J Bacteriol 178:5522–5528
Qian ZG, Xia XX, Lee SY (2011) Metabolic engineering of Escherichia coli for the production of cadaverine: a five-carbon diamine. Biotechnol Bioeng 108:93–103
Tabor CW, Tabor H (1985) Polyamines in microorganisms. Microbiol Rev 49:81–99
Tateno T, Okada Y, Tsuchidate T et al (2009) Direct production of cadaverine from soluble starch using Corynebacterium glutamicum coexpressing α–amylase and lysine decarboxylase. Appl Microbiol Biotechnol 82:115–121
Van der Rest ME, Lange C, Molenaar D (1999) A heat shock following electroporation induces highly efficient transformation of Corynebacterium glutamicum with xenogenetic plasmid DNA. Appl Microbiol Biotechnol 52:541–545
Soksawatmaekhin W, Kuraishi A, Sakata K et al (2004) Excretion and uptake of cadaverine by CadB and its physiological functions in Escherichia coli. Mol Microbiol 51:1401–1412
Schneider J, Wendisch VF (2011) Biotechnological production of polyamines by bacteria: recent achievements and future perspectives. Appl Microbiol Biotechnol 91(1):17–30
Soksawatmaekhin W, Uemura T, Fukiwake N et al (2006) Identification of the cadaverine recognition site on the cadaverine-lysine antiporter CadB. J Biol Chem 281(39):29213–29220
Tomitori H, Kashiwagi K, Igarashi K (2012) Structure and function of polyamine-amino acid antiporters CadB and PotE in Escherichia coli. Amino Acids 42(2–3):733–740
Watson N, Dunyak DS, Rosey EL, Slonczewski JL, Olson ER (1992) Identification of elements involved in transcriptional regulation of the Escherichia coli cad operon by external pH. J Bacteriol 174:530–540
Yamamoto Y, Miwa Y, Miyoshi K et al (1997) The Escherichia coli ldcC gene encodes another lysine decarboxylase probably a constitutive enzyme. Genes Genet Syst 72:167–172
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 21176190), Key Technology Research and Development Program of Tianjin, China (No. 11ZCKFSY00900) and Tianjin Research Program of Application Foundation and Advanced Technology (No. 11JCYBJC09600).
Conflict of Interest
We declare that we have no financial or personal relationships with other people or organizations that could inappropriately influence our work. There is no professional or other personal interest of any nature or kind in any product, service and/or company that could be construed as influencing the position presented in, or the review of, the manuscript entitled ‘Improving the secretion of cadaverine in Corynebacterium glutamicum by co-expressing lysine decarboxylase and cadaverine–lysine antiporter’.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, M., Li, D., Huang, Y. et al. Improving the secretion of cadaverine in Corynebacterium glutamicum by cadaverine–lysine antiporter. J Ind Microbiol Biotechnol 41, 701–709 (2014). https://doi.org/10.1007/s10295-014-1409-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-014-1409-4