Abstract
A fermentation system was continuously fed with sugar-cane syrup and operated with recycling of Saccharomyces cerevisiae cells at temperatures varying from 30 to 47°C. The aim of the present work was to obtain and study the colonies of isolates showing elongated cells of yeasts which were sporadically observed at the end of this continuous process. Based on a sequence of assays involving methods of classical taxonomy and RAPD-PCR, two groups of isolates showing characteristics of non-Saccharomyces yeasts were identified in the yeast population where S. cerevisiae was the dominant yeast. The largest group of non-Saccharomyces yeasts, resulting from a slow proliferation over the 2 months, reached a final level of 29.6% at the end of the process. RAPD-PCR profiles obtained for the isolates of this dominant non-Saccharomyces yeast indicated that they were isolates of Issatchenkia orientalis. Pichia membranifaciens was the only species of non-Saccharomyces yeast detected together with I. orientalis but at a very low frequency. The optimum temperature for ethanol formation shown by the isolate 195B of I. orientalis was 42°C. This strain also showed a faster ethanol formation and biomass accumulation than the thermotolerant strain of S. cerevisiae used as the starter of this fermentation process. Some isolates of I. orientalis were also able to grow better at 40°C than at 30°C on plates containing glycerol as carbon source. Yeasts able to grow and produce ethanol at high temperatures can extend the fermentation process beyond the temperature limits tolerated by S. cerevisiae.





Similar content being viewed by others
References
Aidoo KE, Nout MJR, Sarkar PK (2006) Occurrence and function of yeasts in Asian indigenous fermented foods. FEMS Yeast Res 6:30–39
Álvarez-Martín P, Flórez AB, López-Díaz TM, Mayo B (2007) Phenotypic and molecular identification of yeast species associated with Spanish blue-veined Cabrales cheese. Int Dairy J 17:961–967
Barnett JA, Payne RW, Yarrow D (2000) Yeasts: characteristics and identification. Cambridge University Press, Cambridge
Basílio ACM, de Araújo PRL, de Morais JOF, da Silva Filho EA, de Morais MA Jr, Simões DA (2008) Detection and identification of wild yeast contaminants of the industrial fuel ethanol fermentation process. Curr Microbiol 56:322–326
Booth C (1971) Fungal culture media. In: Booth C (ed) Methods in microbiology. Academic Press, London, pp 49–94
Caetano-Anolles G, Bassam B, Gresshoff PM (1992) Primer-template interactions during DNA amplification fingerprinting with single arbitrary oligonucleotides. Mol Gen Genet 235:157–165
Casey GD, Dobson ADW (2003) Molecular detection of Candida krusei contamination in fruit juice using the citrate synthase gene cs1 and a potential role for this gene in the adaptive response to acetic acid. J Appl Microbiol 94:13–22
Ceccato-Antonini SR (2008) Biotechnological implications of filamentation in Saccharomyces cerevisiae. Biotechnol Lett 30:1151–1161
Clemente-Jimenez JM, Mingorance-Cazorla L, Martínez-Rodriguez S, Las Heras-Vázquez FJ, Rodríguez-Vico F (2004) Molecular characterization and oenological properties of wine yeasts isolated during spontaneous fermentation of six varieties of grape must. Food Microbiol 21:149–155
Gomes FCO, Silva CLC, Marini MM, Oliveira ES, Rosa CA (2007) Use of selected indigenous Saccharomyces cerevisiae strains for the production of the traditional cachaça in Brazil. J Appl Microbiol 103:2438–2447
Halm M, Hornbaek T, Arneborg N, Sefa-Dedeh S, Jespersen L (2004) Lactic acid tolerance determined by measurement of intracellular pH of single cells of Candida krusei and Saccharomyces cerevisiae isolated from fermented maize dough. Int J Food Microbiol 94:97–103
Jespersen L (2003) Occurrence and taxonomic characteristics of strains of Saccharomyces cerevisiae predominant in African indigenous fermented foods and beverages. FEMS Yeast Res 3:191–200
Jespersen L, Halm M, Kpodo K, Jakobsen M (1994) Significance of yeasts and moulds occurring in maize dough fermentation for ‘kenkey’ production. Int J Food Microbiol 24:239–248
Kreger-van Rij NJW (1984) The yeasts: a taxomical study. Elsevier Science, Amsterdam
Kudryavtsev VJ (1960) Issatchenkia orientalis. In: Barnett JA, Payne RW, Yarrow D (eds) Yeasts: characteristics and identification. Cambridge University Press, Cambridge, pp 404–405
Kurtzman CP, Fell JW (1998) The yeasts: a taxonomic study. Elsevier Science, Amsterdam, pp 77–100
Kurtzman CP, Robnett CJ, Basehoar-Powers E (2008) Phylogenetic relationships among species of Pichia, Issatchenkia and Williopsis determined from multigene sequence analysis, and the proposal of Barnettozyma gen. nov., Lindnera gen. nov. and Wickerhamomyces gen. nov. FEMS Yeast Res 8:939–954
Lachance MA (1995) Yeast communities in a natural tequila fermentation. Antonie van Leeuwenhoek 68:151–160
Laluce C, Souza CS, Abud CL, Gattas EAL, Walker GM (2002) Continuous ethanol production in a nonconventional five-stage system operating with yeast cell recycling at elevated temperatures. J Ind Microbiol Biotechnol 29:140–144
Laluce C, Tognolli JO, Oliveira KF, Souza CS, Morais MR (2009) Optimization of temperature, sugar concentration and inoculum size to maximize ethanol production without significant decrease in yeast cell viability. Appl Microbiol Biotechnol 83:627–637
Li L, Ye Y, Pan L, Zhu Y, Zheng S, Lin Y (2009) The induction of trehalose and glycerol in Saccharomyces cerevisiae in response to various stresses. Biochem Biophys Res Commun 387:778–783
Limtong S, Sintara S, Suwanarit P, Lotong N (2002) Yeast diversity in Thai traditional fermentation starter (Loog-pang). Kasetsart J (Nat Sci) 36:149–158
Lourens-Hattingh A, Viljoen BC (2002) Survival of dairy-associated yeasts in yoghurt and yoghurt-related products. Food Microbiol 19:597–604
Meroth CB, Hammes WP, Hertel C (2003) Identification and population dynamics of yeasts in sourdough fermentation processes by PCR-denaturing gradient gel electrophoresis. Appl Environ Microbiol 69:7453–7461
Messner R, Prillinger H, Altmann F, Lopandic K, Wimmer K, Molnár O, Weigang F (1994) Molecular characterization and application of random amplified polymorphic DNA analysis on Mrakia and Sterigmatomyces species. Int J Syst Bacteriol 44:694–703
Meyer W, Koch A, Niemann C, Beyermann B, Epplen JT, Börner T (1991) Differentiation of species and strains among filamentous fungi by DNA fingerprinting. Curr Genet 19:239–242
Middelhoven WJ (2001) Identification of yeasts present in sour fermented foods and fodders. In: Spencer JFT, Spencer ALR (eds) Methods in biotechnology: food microbiology protocols, vol 14. Human Press Inc., Totowa, pp 209–224
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
Morais PB, Rosa CA, Linardi VR, Pataro C, Maia ABRA (1997) Short communication: characterization and succession of yeast populations associated with spontaneous fermentations during the production of Brazilian sugar-cane aguardente. World J Microbiol Biotechnol 13:241–243
Omemu AM, Oyewole OB, Bankole MO (2007) Significance of yeasts in the fermentation of maize for ogi production. Food Microbiol 24:571–576
Prillinger H, Molnár O, Eliskases-Lechner F, Lopandic K (1999) Phenotypic and genotypic identification of yeasts from cheese. Antonie van Leeuwenhoek 75:267–283
Rajoka MI, Ferhan M, Khalid AM (2005) Kinetics and thermodynamics of ethanol production by a thermotolerant mutant of Saccharomyces cerevisiae in a microprocessor-controlled bioreactor. Lett Appl Microbiol 40:316–321
Singh D, Banat IM, Nigam P, Marchant R (1998) Industrial scale ethanol production using the thermotolerant yeast Kluyveromyces marxianus IMB3 in an Indian distillery. Biotechnol Lett 20:753–755
Souza CS, Thomaz D, Cides ER, Oliveira KF, Tognolli JO, Laluce C (2007) Genetic and physiological alterations occurring in a yeast population continuously propagated at increasing temperatures with cell recycling. World J Microbiol Biotechnol 23:1667–1677
Tornai-Lehoczki J, Péter G, Dlauchy D (2003) CHROMagar Candida medium as a practical tool for the differentiation and presumptive identification of yeast species isolated from salads. Int J Food Microbiol 86:189–200
van der Aa Kühle A, Jespersen L (1998) Detection and identification of wild yeasts in lager breweries. Int J Food Microbiol 43:205–213
Acknowledgments
The authors are grateful to FAPESP (grants n° 2005/01498-6 and 2008/56247-6) for the fellowship awarded to Karen F. Oliveira (process n° 2008/56587-1) and CAPES for the fellowships awarded to Crisla S. Sousa and Jéssica C. M. Gallardo.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.

Figure 4 S
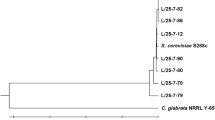
Similarity values shown by dendrograms generated from RAPD–PCR analyses using two sets of primers: (A) among isolates of I. orientalis including the type strain Y01030 using the primer M13; (B) among isolates of P. membranaefaciens including the type Y01044 using the primer M13; (C) among isolates of I. orientalis including the type strain Y01030 using the primer D2; (D) among strain of isolates P. membranaefaciens. (JPEG 525 kb)
Rights and permissions
About this article
Cite this article
Gallardo, J.C.M., Souza, C.S., Cicarelli, R.M.B. et al. Enrichment of a continuous culture of Saccharomyces cerevisiae with the yeast Issatchenkia orientalis in the production of ethanol at increasing temperatures. J Ind Microbiol Biotechnol 38, 405–414 (2011). https://doi.org/10.1007/s10295-010-0783-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-010-0783-9