Abstract
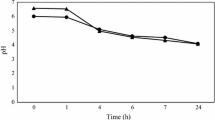
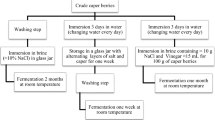
In 2004, Leuconostoc mesenteroides DRC was first used as a starter culture for achieving higher organoleptic effects in Korean kimchi manufacture. For a better understanding of starter growth in a mixed culture system, and for predicting starter predominance in kimchi, a monitoring system for the starter was established. The chloramphenicol resistance marker gene (cat) was randomly integrated into chromosomal DNA of L. mesenteroides DRC using a viral transposon and transposase. The DRC mutant, tDRC2, had a similar growth pattern to the host strain, with no major alteration in phenotypic characteristics. The mutant strain was inoculated into real kimchi, and monitoring of the starter population was successfully achieved. The overall predominance of Leuconostoc in kimchi inoculated with DRC followed the general growth pattern of this genus during kimchi fermentation. Our results also demonstrate the competitive ability of the DRC starter against Leuconostoc from natural flora, maintaining its predominance above 88% during the whole fermentation period. Based on this experiment, the random gene integration method using a transposon was shown to be of utility in transferring any commercial starter into a selectable and monitorable strain for simulation purposes.




Similar content being viewed by others
References
Amann RI, Binder BJ, Olson RJ, Chisholm SW, Devereux R, Stahl DA (1990) Combination of 16S rRNA-targeted oligonucleotide probes with flow cytometry for analysing mixed microbial populations. Appl Environ Microbiol 56:1919–1925
Beimfohr C, Krause A, Amann RI, Ludwig W, Schleifer KH (1993) In situ identification of Lactococci, Enterococci and Streptococci. Syst Appl Microbiol 16:450–456
Cho EJ, Park KY, Rhee SH (1997) Standardization of ingredient ratios of Chinese cabbage kimchi. Korean J Food Sci Technol 29:1228–1235
Choi IK, Jung SH, Kim BJ, Park SY, Kim J, Han HU (2003) Novel Leuconostoc citreum starter culture system for the fermentation of kimchi, a fermented cabbage product. Antonie Van Leeuwenhoek 84:247–253. doi:10.1023/A:1026050410724
Cogan TM, Jordan KN (1994) Metabolism of Leuconostoc bacteria. J Dairy Sci 77:2704–2717
Cooper NS, Brown ME, Caulcott CA (1987) A mathematical method for analysing plasmid stability in micro-organisms. J Gen Microbiol 133:1871–1880
Corthier G, Delorme C, Ehrlich SD, Renault P (1998) Use luciferase genes as biosensors to study bacterial physiology in the digestive tract. Appl Environ Microbiol 64:2721–2722
Duncan S, Glover LA, Killham K, Prosser JI (1994) Luminescence-based detection of activity of starved and viable but nonculturable bacteria. Appl Environ Microbiol 60:1308–1316
Eom HJ, Seo DM, Han NS (2007) Selection of psychrotrophic Leuconostoc spp. producing highly active dextransucrase from lactate fermented vegetables. Int J Food Microbiol 10:61–67. doi:10.1016/j.ijfoodmicro.2007.02.027
Geoffroy MC, Guyard C, Quatannens B, Pavan S, Lange M, Mercenier A (2000) Use of green fluorescent protein to tag lactic acid bacterium strains under development as life vaccine vectors. Appl Environ Microbiol 66:383–391
Giraffa G, Neviani E (2000) Molecular identification and characterisation of food-associated Lactobacilli. Ital J Food Sci 12:403–423
Giraffa G, Rossetti L (2004) Monitoring of the bacterial composition of dairy starter cultures by RAPD-PCR. FEMS Microbiol Lett 237:133–138. doi:10.1111/j.1574-6968.2004.tb09688.x
Gory L, Montel MC, Zagorec M (2001) Use of green fuorescent protein to monitor Lactobacillus sakei in fermented meat products. FEMS Microbiol Lett 194:127–133. doi:10.1111/j.1574-6968.2001.tb09457.x
Goryshin IY, Reznikoff WS (1998) Tn5 in vitro transposition. J Biol Chem 273:7367–7374. doi:10.1074/jbc.273.13.7367
Hamer L, DeZwaan TM, Montenegro-Chamorro MV, Frank SA, Hamer JE (2001) Recent advances in large-scale transposon mutagenesis. Curr Opin Chem Biol 5:67–73. doi:10.1016/S1367-5931(00)00162-9
Han NS, Jung YS, Eom HJ, Koh YH, Robyt JF, Seo JH (2002) Simultaneous biocatalytic synthesis of panose during lactate fermentation in kimchi. J Microbiol Biotechnol 12:46–52. doi:10.1159/000070151
Han HU, Lim CR, Park HK (1990) Determination of microbial community as an indicator of kimchi fermentation. Korean J Food Sci Technol 22:26–32
Hattemer-Frey HA, Brandt EJ, Travis CC (1990) Small-scale field test of the genetically engineered lacZY marker. Regul Toxicol Pharmacol 11:253–261. doi:10.1016/0273-2300(90)90025-7
Hols P, Ferain T, Garmyn D, Bernard N, Delcour J (1994) Use of homologous expression-secretion signals and vector-fress stable chromosomal integration in engineering of Lactobacillus plantarum for α-amylase and levanase expression. Appl Environ Microbiol 60:1401–1413
Johanningsmeier SD, Fleming HP, Breidt F Jr (2004) Malolactic activity of lactic acid bacteria during sauerkraut fermentation. J Food Sci 69:222–227
Koo YJ, Choi SY (1991) Science and Technology of Kimchi, 2nd edn. Korea Food Research Institute, Seoul
Lee KH, Park WJ, Kim JY, Kim HG, Lee JM, Kim JH, Park JW, Lee JH, Chung SK, Chung DK (2007) Development of a monitoring vector for Leuconostoc mesenteroides using the green fluorescent protein. J Microbiol Biotechnol 17:1213–1216
McDonald LC, Fleming HP, Hassan HM (1990) Acid tolerance of Leuconostoc mesenteroides and Lactobacillus plantarum. Appl Environ Microbiol 56:2120–2124
Olive DM, Bean P (1999) Principles and applications of methods for DNA-based typing of microbial organisms. J Clin Microbiol 37:1661–1669
Park MS, Shin DW, Lee KH, Ji GE (1999) Sequence analysis of plasmid pKJ50 from Bifidobacterium longum. Microbiol 145:585–592
Prosser JI (1994) Molecular marker systems for detection of genetically engineered micro-organisms in the environment. Microbiol 140:5–17
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Scott KP, Mercer DK, Richardson AJ, Melville CM, Glover LA, Flint HJ (2000) Chromosomal integration of the green fluorescent protein gene in lactic acid bacteria and the survival of marked strains in human gut simulations. FEMS Microbiol Lett 182:23–27. doi:10.1111/j.1574-6968.2000.tb08867.x
Tamminen M, Joutsjoki T, Sjöblom M, Joutsen M, Palva A, Ryhänen EL, Joutsjoki V (2004) Screening of lactic acid bacteria from fermented vegetables by carbohydrate profiling and PCR–ELISA. Lett Appl Microbiol 39:439–444. doi:10.1111/j.1472-765X.2004.01607.x
de Valdez GF, de Giore GS, Garro M, Mozzi F, Oliver G (1990) Lactic acid bacteria from naturally fermented vegetables. Microbiol Aliment Nutr 8:175–179
Vogel RF, Ehrmann MA, Gänzle MG (2002) Development and potential of starter Lactobacilli resulting from exploration of the sourdough ecosystem. Antonie Van Leeuwenhoek 81:631–638. doi:10.1023/A:1020530227192
Wagner M, Amann R, Kämpfer P, Assmus B, Hartmann A, Hutzler P, Springer N, Schleifer KH (1994) Identification and in situ detection of gram-negative filamentous bacteria in activated sludge. Syst Appl Microbiol 17:405–417
Wyckoff HA, Sandine WE (1991) Transformation of dairy Leuconostoc using plasmid vectors from Bacillis, Escherichia, and Lactococcus hosts. J Dairy Sci 74:1454–1460
Youngman P (1993) Transposons and their applications. In: Sonenshein AL, Hoch JA, Losick R (eds) Bacillus subtilis and other gram-positive bacteria, American Society for Microbiology, Washington, DC, pp 585–596
Acknowledgments
This work was supported by the fund of Research Center for Bioresource and Health (RCBH) at Chungbuk National University and ITEP & MOCIE of Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Eom, HJ., Park, J.M., Seo, M.J. et al. Monitoring of Leuconostoc mesenteroides DRC starter in fermented vegetable by random integration of chloramphenicol acetyltransferase gene. J Ind Microbiol Biotechnol 35, 953–959 (2008). https://doi.org/10.1007/s10295-008-0369-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-008-0369-y