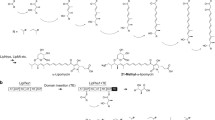
Abstract
Several polyketide secondary metabolites are shown by feeding experiments to incorporate glycerol-derived 3-carbon starter units, 2-carbon extender units, or 3-carbon branches into their hydrocarbon chains. In recent years, genetic studies have begun to elucidate the mechanisms by which this occurs. In this article we review the incorporation of glycerol-derived precursors into polyketides and propose new mechanisms for the incorporation processes.


















Similar content being viewed by others
References
Allen IW, Ritchie DA (1994) Cloning and analysis of DNA sequences from Streptomyces hygroscopicus encoding geldanamycin biosynthesis. Mol Gen Genet 243:593–599
Bentley SD, Brown S, Murphy LD, Harris DE, Quail MA, Parkhill J, Barrell BG, McCormick JR, Santamaria RI, Losick R, Yamasaki M, Kinashi H, Chen CW, Chandra G, Jakimowicz D, Kieser HM, Kieser T, Chater KF (2004) SCP1, a 356 023 bp linear plasmid adapted to the ecology and developmental biology of its host, Streptomyces coelicolor A3(2). Mol Microbiol 51:1615–1628
Bloomer JL, Kappler FE, Pandey GN (1972) Biosynthesis of carolic acid in Penicillium charlesii: the intermediate precursors. J Chem Soc Chem Commun 4:234–243
Byrne KM, Shafiee A, Nielsen JB, Arison BH, Monaghan RL, Kaplan L (1993) The biosynthesis and enzymology of an immunosupressant, immunomycin, produced by Streptomyces hygroscopicus var. ascomyceticus. In: Hamill R (ed) Developments in industrial microbiology series, vol 32. Wm. C. Brown Publisher, Dubuque, Iowa, pp 29–45
Carroll BJ, Moss SJ, Bai L, Kato Y, Toelzer S, Yu T-W, Floss HG (2002) Identification of a set of genes involved in the formation of the substrate for the incorporation of the unusual “glycolate” chain extension unit in ansamitocin biosynthesis. J Am Chem Soc 124:4176–4177
Challis GL, Chater KF (2001) Incorporation of [U–13C]glycerol defines plausible early steps for the biosynthesis of methylenomycin A in Streptomyces coelicolor A3(2). Chem Comm 10:935–936
Chen TSS, Chang CJ, Floss HG (1979) Biosynthesis of the boron-containing macrolide antibiotic asplasmomycin. J Am Chem Soc 101:5826–5827
Chen TSS, Chang CJ, Floss HG (1981) On the biosynthesis of boromycin. J Org Chem 46:2661–2665
Chen TSS, Chang CJ, Floss HG (1981) Biosynthesis of the boron-containing macrolide antibiotic aplasmomycin by Streptomyces griseus . J Am Chem Soc 103:4565–4568
Chijiwa S, Park H-R, Furihata K, Ogata M, Endo T, Kuzuyama T, Hayakawa Y, Shin-ya K (2003) Biosynthetic studies of versipelostatin, a novel 17-membered α-tetronic acid involved macrocyclic compound isolated from Streptomyces versipellis. Tetrahedron Lett 44:5897–5900
Corre C, Challis GL (2005) Evidence for the unusual condensation of a diketide with a pentose in the methylenomycin biosynthetic pathway of Streptomyces coelicolor A3(2). Chembiochem (in press)
Cronin A, Mowbray S, Dürk H, Homburg S, Fleming I, Fisslthaler B, Oesch F, Arand M (2003) The N-terminal domain of mammalian soluble epoxide hydrolase is a phosphatase. Proc Natl Acad Sci USA 100:1552–1557
Dunitz JD, Hawley DM, Miklos D, White DN, Berlin Y, Marusic R, Prelog V (1971) Structure of boromycin. Helv Chim Acta 54:1709–1713
Emmert EAB, Klimowicz AK, Thomas MG, Handelsman J (2004) Genetics of zwittermicin A production by Bacillus cereus. Appl Environ Microbiol 70:104–113
Haber A, Johnson RD, Rinehart KL Jr (1977) Biosynthetic origin of the C2 units of geldanamycin and distribution of label from D-[6−13C]glucose. J Am Chem Soc 99:3541–3544
Haneishi T, Kitahara N, Takiguchi Y, Arai M, Sugawara S (1974a) New antibiotics, methylenomycins A and B. I. Producing organism, fermentation and isolation, biological activities and physical and chemical properties. J Antibiot 27:386–392
Haneishi T, Terahara A, Arai M, Hata T, Tamura C (1974b) New antibiotics, methylenomycins A and B. II. Structures of methylenomycins A and B. J Antibiot 27:393–399
Hatano K, Mizuta E, Akiyama S, Higashide E, Nakao Y (1985) Biosynthetic origin of the ansa-structure of ansamitocin P-3. Agric Biol Chem 49:327–333
Hildebrand M, Waggoner LE, Liu H, Sudek S, Allen S, Anderson C, Sherman DH, Haygood M (2004) bryA: an unusual modular polyketide synthase gene from the uncultivated bacterial symbiont of the marine bryozoan Bugula neritina. Chem Biol 11:1543–1552
Hill AM, Harris JP, Siskos AP (1998) Investigation of glycerol incorporation into soraphen A. Chem Commun 2361–2362
Hill RA, Parker MC (2001) Hot off the press. Nat Prod Rep 18:3–5
Hoffmann L, Grond S (2004) Mixed acetate-glycerol biosynthesis and formation of benzoate directly from shikimate in Streptomyces sp. Eur J Org Chem 4771–4777
Holzbach R, Pape H, Hook D, Kreutzer EF, Chang CJ, Floss HG (1978) Biosynthesis of the macrolide antibiotic chlorothricin: basic building blocks. Biochemistry 17:556–560
Hornemann U, Hopwood DA (1981) Biosynthesis of methylenomycin A: a plasmid-determined antibiotic. In: Corcoran JW (ed) Antibiotics, vol IV. Springer, Berlin Heidelberg New York, pp 123–131
Kakavas SJ, Katz L, Stassi D (1997) Identification and characterization of the niddamycin polyketide synthase genes from Streptomyces caelestis. J Bacteriol 179:7515–7522
Kato Y, Bai L, Xue Q, Revill WP, Yu T-W, Floss HG (2002) Functional expression of genes involved in the biosynthesis of the novel polyketide chain extension unit, methoxymalonyl-acyl carrier protein, and engineered biosynthesis of 2-desmethyl-2-methoxy-6-deoxyerythronolide B. J Am Chem Soc 124:5268–5269
Kerr RG, Lawry J, Gush KA (1996) In vitro biosynthetic studies of the bryostatins, anti-cancer agents from the marine bryozoan Bugula neritina. Tetrahedron Lett 37:8305–8308
Kuhstoss S, Huber M, Turner JR, Paschal JW, Rao RN (1996) Production of a novel polyketide through the construction of a hybrid polyketide synthase. Gene 183:231–236
Lee JJ, Derwick PM, Gorst-Allman CP, Spreafico F, Kowal C, Chang CJ, McInnes AG, Walter JA, Keller PJ, Floss HG (1987) Further studies on the biosynthesis of the boron-containing antibiotic aplasmomycin. J Am Chem Soc 109:5426–5432
Lee JJ, Lee JP, Keller PJ, Cottrell CE, Chang CJ, Zaehner H, Floss HG (1986) Further studies on the biosynthesis of chlorothricin. J Antibiot 39:1123–1134
Ligon J, Hill S, Beck J, Zirkle R, Molnar I, Zawodny J, Money S, Schupp T (2002) Characterization of the biosynthetic gene cluster for the antifungal polyketide soraphen A from Sorangium cellulosum So ce26. Gene 285:257–267
Omura S, Nakagawa A, Takeshima H, Atusmi K, Miyazawa J, Piriou F, Lukacs G (1975) Biosynthetic studies using carbon-13 enriched precursors on the 16-membered macrolide antibiotic leucomycin A3. J Am Chem Soc 97:6600–6602
Mascaretti OA, Chang CJ, Hook D, Otsuka H, Kreutzer EF, Floss HG (1981) Biosynthesis of the macrolide antibiotic chlorothricin. Biochemistry 20:919–924
Mashimo Y, Sekiyama Y, Araya H, Fujimoto Y, (2004) Biosynthesis of agglomerin A: stereospecific incorporation of pro-R- and pro-S-hydrogens at sn-C-3 of glycerol into the branched C3 moiety. Bioorg Med Chem Lett 14:649–651
Midland SL, Keen NT, Sims JJ, Midland MM, Stayton MM, Burton V, Smith MJ, Mazzola EP, Graham KJ, Clardy J (1993) The structures of syringolides 1 and 2, novel C-glycosidic elicitors from Pseudomonas syringae pv. Tomato. J Org Chem 58:2940–2945
Moore BS (1999) Biosynthesis of marine natural products: microorganisms and macroalgae. Nat Prod Rep 16:653–674
Moore BS, Hertweck C (2002) Biosynthesis and attachment of novel bacterial polyketide synthase starter units. Nat Prod Rep 19:70–99
Motamedi H, Shafiee A (1998) The biosynthetic gene cluster for the macrolactone ring of the immunosuppressant FK506. Eur J Biochem 256:528–534
Newman JW, Morisseau C, Harris TR, Hammock BD (2003) The soluble epoxide hydrolase encoded by EPXH2 is a bifunctional enzyme with novel lipid phosphate phosphatase activity. Proc Natl Acad Sci USA 100:1558–1563
Omura S, Tsuzuki K, Nakagawa A, Lukacs G (1983) Biosynthetic origin of carbon-3 and carbon-4 of leucomycin aglycone. J Antibiot 36:611–613
Ono M, Sakuda S, Ikeda H, Furihata K, Nakayama J, Suzuki A, Isogai A (1998) Structures and biosynthesis of aflastatins: novel inhibitors of aflatoxin production by Aspergillus parasiticus. J Antibiot 51:1019–1028
Patel K, Piagentini M, Rascher A, Tian Z-Q, Buchanan GO, Regentin R, Hu Z, Hutchinson CR, McDaniel R (2004) Engineered biosynthesis of geldanamycin analogs for Hsp90 inhibition. Chem Biol 11:1625–1633
Pettit GR, Herald CL, Doubek DL, Herald DL, Arnold E, Clardy J (1982) Isolation and structure of bryostatin 1. J Am Chem Soc 104:6846–6848
Piel J (2002) A polyketide synthase-peptide synthetase gene cluster from an uncultured bacterial symbiont of Paederus beetles. Proc Natl Acad Sci USA 99:14002–14007
Rascher A, Hu Z, Viswanathan N, Schirmer A, Reid R, Nierman WC et al (2003) Cloning and characterization of a gene cluster for geldanamycin production in Streptomyces hygroscopicus NRRL 3602. FEMS Microbiol Lett 218:223–230
Rawlings BJ (1997) Biosynthesis of polyketides. Nat Prod Rep 14:523–556
Ridder IS, Dijkstra BW (1999) Identification of the Mg2+-binding site in the P-type ATPase and phosphatase members of the HAD (haloacid dehalogenase) superfamily by structural similarity to the response regulator protein CheY. Biochem J 339:223–226
Rohmer M, Seemann M, Horbach S, Bringer-Meyer S, Sahm H (1996) Glyceraldehyde 3-phosphate and pyruvate as precursors of isoprenic units in an alternative non-mevalonate pathway for terpenoid biosynthesis. J Am Chem Soc 118:2564–2566
Sakuda S, Ono M, Ikeda H, Inagaki Y, Nakayama J, Suzuki A, Isogai A (1997) Structure of blasticidin A. Tetrahedron Lett 38:7399–7402
Sakuda S, Higashi A, Tanaka S, Nihira T, Yamada Y (1992) Biosynthesis of virginiae butanolide A, a butyrolactone autoregulator from Streptomyces. J Am Chem Soc 114:663–668
Schuhmann T, Grond S (2004) Biosynthetic investigations of the V-type ATPase inhibitors bafilomycin A1, B1 and concanamycin A. J Antibiot 57:655–661
Schummer D, Schomburg D, Irschik H, Reichenbach H, Höfle G (1996) Antibiotics from gliding bacteria. Part LXXV. Absolute configuration and biosynthesis of tartrolon B, a boron-containing macrolide from Sorangium cellulosum. Liebigs Ann 6:965–969
Schupp T, Toupet C, Cluzel B, Neff S, Hill S, Beck JJ, Ligon JM (1995) A Sorangium cellulosum (myxobacterium) gene cluster for the biosynthesis of the macrolide antibiotic soraphen A: cloning, characterization, and homology to polyketide synthase genes from actinomycetes. J Bacteriol 177:3673–3679
Sekiyama Y, Araya H, Hasumi K, Endo A, Fujimoto Y (1998) Biosynthesis of acaterin: incorporation of glycerol into the C3 branched unit. Tetrahedron Lett 39:6233–6236
Sekiyama Y, Hasumi K, Endo A, Fujimoto Y (1999) Biosynthesis of acaterin: metabolic fate of sn-3 hydrogens of glycerol during the formation of 4-dehydroacaterin. Tetrahedron Lett 40:4223–4226
Sekiyama Y, Fujimoto Y, Hasumi K, Endo A, (2001) Biosynthesis of acaterin: coupling of C5 unit with octanoate. J Org Chem 66:5649–5654
Stohl EA, Milner JL, Handelsman J (1999) Zwittermicin A biosynthetic cluster. Gene 237:403–411
Suga T, Hirata T (1982) The biosynthesis of protoanemonin in Ranunculus glaber. The pivotal biosynthetic intermediate and the stereospecific hydrogen elimination from the intermediate. Bull Chem Soc Jpn 55:1584–1587
Uramoto M, Otake N, Cary L, Tanabe M (1978) Biosynthetic studies with carbon-13. Lankacidin group of antibiotics. J Am Chem Soc 100:3616–3617
Watanabe K, Khosla C, Stroud RM, Tsai S-C (2003) Crystal structure of an acyl-ACP dehydrogenase from the FK520 polyketide biosynthetic pathway: insights into extender unit biosynthesis. J Mol Biol 334:435–444
Wu K, Chung L, Revill WP, Katz L, Reeves CD (2000) The FK520 gene cluster of Streptomyces hygroscopicus var. ascomyceticus (ATCC 14891) contains genes for biosynthesis of unusual polyketide extender units. Gene 251:81–90
Yamada Y (1999) In: England R, Hobbs G, Bainton N, Roberts D (eds) Microbial signalling and communication. Cambridge University Press, Cambridge, pp 177–196
Yu T-W, Bai L, Clade D, Hoffmann D, Toelzer S, Trinh KQ, Xu J, Moss SJ, Leistner E, Floss HG (2002) The biosynthetic gene cluster of the maytansinoid antitumor agent ansamitocin from Actinosynnema pretiosum. Proc Natl Acad Sci USA 99:7968–7973
Yucel I, Midland SL, Sims JJ, Keen NT (1994) Class I and Class II avrD alleles direct the production of different products in gram-negative bacteria. Mol Plant Microbe Interact 7:148–150
Acknowledgements
Research in the corresponding author’s lab is supported by the UK BBSRC and the European Community.
Author information
Authors and Affiliations
Corresponding author
Additional information
Laura J. Walton, Christophe Corre contributed equally to this article
Rights and permissions
About this article
Cite this article
Walton, L.J., Corre, C. & Challis, G.L. Mechanisms for incorporation of glycerol-derived precursors into polyketide metabolites. J IND MICROBIOL BIOTECHNOL 33, 105–120 (2006). https://doi.org/10.1007/s10295-005-0026-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-005-0026-7