Abstract
Patella alta (PA) and patella baja (PB) affect 1–2% of the world population, but are often underreported, leading to potential complications like osteoarthritis. The Insall-Salvati ratio (ISR) is commonly used to diagnose patellar height abnormalities. Artificial intelligence (AI) keypoint models show promising accuracy in measuring and detecting these abnormalities.
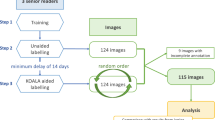
An AI keypoint model is developed and validated to study the Insall-Salvati ratio on a random population sample of lateral knee radiographs. A keypoint model was trained and internally validated with 689 lateral knee radiographs from five sites in a multi-hospital urban healthcare system after IRB approval. A total of 116 lateral knee radiographs from a sixth site were used for external validation. Distance error (mm), Pearson correlation, and Bland–Altman plots were used to evaluate model performance. On a random sample of 2647 different lateral knee radiographs, mean and standard deviation were used to calculate the normal distribution of ISR. A keypoint detection model had mean distance error of 2.57 ± 2.44 mm on internal validation data and 2.73 ± 2.86 mm on external validation data. Pearson correlation between labeled and predicted Insall-Salvati ratios was 0.82 [95% CI 0.76–0.86] on internal validation and 0.75 [0.66–0.82] on external validation. For the population sample of 2647 patients, there was mean ISR of 1.11 ± 0.21. Patellar height abnormalities were underreported in radiology reports from the population sample. AI keypoint models consistently measure ISR on knee radiographs. Future models can enable radiologists to study musculoskeletal measurements on larger population samples and enhance our understanding of normal and abnormal ranges.





Similar content being viewed by others
Data Availability
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
References
Le Hoang Di T, Hoang Ngoc T, Ngo DHA, et al. Evaluation of the Insall-Salvati Ratio Among the Vietnamese Population: Application for Diagnosis of Patellar Malalignment. Orthop Res Rev. 2021;13:57–61.
Hong H-T, Koh Y-G, Nam J-H, Kim PS, Kwak YH, Kang K-T. Gender Differences in Patellar Positions among the Korean Population. NATO Adv Sci Inst Ser E Appl Sci. Multidisciplinary Digital Publishing Institute; 2020;10(24):8842.
Upadhyay S, Raza HKT, Srivastava P. Position of the patella in adults in central India: evaluation of the Insall-Salvati ratio. J Orthop Surg . 2013;21(1):23–27.
Wolfe S, Varacallo M, Thomas JD, Carroll JJ, Kahwaji CI. Patellar instability. 2018. https://europepmc.org/article/nbk/nbk482427.
Biedert RM. Patella Alta: When to Correct and Impact on Other Anatomic Risk Factors for Patellofemoral Instability. Clin Sports Med. 2022;41(1):65–76.
Barth KA, Strickland SM. Surgical Treatment of Iatrogenic Patella Baja. Curr Rev Musculoskelet Med. 2022;15(6):673–679.
Insall J, Salvati E. Patella Position in the Normal Knee Joint. Radiology. 1971. p. 101–104. https://doi.org/10.1148/101.1.101.
Grelsamer RP, Meadows S. The modified Insall-Salvati ratio for assessment of patellar height. Clin Orthop Relat Res. 1992;(282):170–176.
Verhulst FV, van Sambeeck JDP, Olthuis GS, van der Ree J, Koëter S. Patellar height measurements: Insall–Salvati ratio is most reliable method. Knee Surg Sports Traumatol Arthrosc. 2020;28(3):869–875.
Ali SA, Helmer R, Terk MR. Patella alta: lack of correlation between patellotrochlear cartilage congruence and commonly used patellar height ratios. AJR Am J Roentgenol. 2009;193(5):1361–1366.
Hosny A, Parmar C, Quackenbush J, Schwartz LH, Aerts HJWL. Artificial intelligence in radiology. Nat Rev Cancer. 2018;18(8):500–510.
Buls N, Watté N, Nieboer K, Ilsen B, de Mey J. Performance of an artificial intelligence tool with real-time clinical workflow integration--detection of intracranial hemorrhage and pulmonary embolism. Physica Medica: European Journal of Medical Physics. Elsevier; 2021;83:154–160.
Elkorany AS, Elsharkawy ZF. Efficient breast cancer mammograms diagnosis using three deep neural networks and term variance. Sci Rep. 2023;13(1):2663.
Kumar V, Altahan BR, Rasheed T, et al. Improved UNet Deep Learning Model for Automatic Detection of Lung Cancer Nodules. Comput Intell Neurosci. 2023;2023:9739264.
Kanjanasurat I, Tenghongsakul K, Purahong B, Lasakul A. CNN-RNN Network Integration for the Diagnosis of COVID-19 Using Chest X-ray and CT Images. Sensors . 2023;23(3). https://doi.org/10.3390/s23031356.
Lakhani P, Sundaram B. Deep Learning at Chest Radiography: Automated Classification of Pulmonary Tuberculosis by Using Convolutional Neural Networks. Radiology. 2017;284(2):574–582.
Klontzas ME, Stathis I, Spanakis K, Zibis AH, Marias K, Karantanas AH. Deep Learning for the Differential Diagnosis between Transient Osteoporosis and Avascular Necrosis of the Hip. Diagnostics (Basel). 2022;12(8). https://doi.org/10.3390/diagnostics12081870.
Thian YL, Li Y, Jagmohan P, Sia D, Chan VEY, Tan RT. Convolutional Neural Networks for Automated Fracture Detection and Localization on Wrist Radiographs. Radiology: Artificial Intelligence. Radiological Society of North America; 2019;1(1):e180001.
Almășan O, Leucuța D-C, Hedeșiu M, Mureșanu S, Popa Ștefan L. Temporomandibular Joint Osteoarthritis Diagnosis Employing Artificial Intelligence: Systematic Review and Meta-Analysis. J Clin Med Res. 2023;12(3). https://doi.org/10.3390/jcm12030942.
Twinprai N, Boonrod A, Boonrod A, et al. Artificial intelligence (AI) vs. human in hip fracture detection. Heliyon. 2022;8(11):e11266.
Swiecicki A, Li N, O’Donnell J, et al. Deep learning-based algorithm for assessment of knee osteoarthritis severity in radiographs matches performance of radiologists. Comput Biol Med. 2021;133:104334.
Anagnostakos K, Lorbach O, Reiter S, Kohn D. Comparison of five patellar height measurement methods in 90° knee flexion. Int Orthop. 2011;35(12):1791–1797.
Tseng T-W, Chen Y-P, Yeh Y-C, Kuo C-F, Fan T-Y, Lin Y-C. Automatic prosthetic-parameter estimation from anteroposterior pelvic radiographs after total hip arthroplasty using deep learning-based keypoint detection. Int J Med Robot. Wiley; 2022;18(4):e2394.
Yan J-L, Chen Y-L, Chen M-Y, et al. A Robust, Fully Automatic Detection Method and Calculation Technique of Midline Shift in Intracranial Hemorrhage and Its Clinical Application. Diagnostics (Basel). 2022;12(3). https://doi.org/10.3390/diagnostics12030693.
Cha J-Y, Yoon H-I, Yeo I-S, Huh K-H, Han J-S. Peri-Implant Bone Loss Measurement Using a Region-Based Convolutional Neural Network on Dental Periapical Radiographs. J Clin Med Res. 2021;10(5). https://doi.org/10.3390/jcm10051009.
Herdea A, Pencea V, Lungu CN, Charkaoui A, Ulici A. A Prospective Cohort Study on Quality of Life among the Pediatric Population after Surgery for Recurrent Patellar Dislocation. Children. 2021;8(10). https://doi.org/10.3390/children8100830.
Lin C-FJ, Wu J-J, Chen T-S, Huang T-F. Comparison of the Insall-Salvati ratio of the patella in patients with and without an ACL tear. Knee Surg Sports Traumatol Arthrosc. 2005;13(1):8–11.
Degnan AJ, Maldjian C, Adam RJ, Fu FH, Di Domenica M. Comparison of Insall-Salvati ratios in children with an acute anterior cruciate ligament tear and a matched control population. AJR Am J Roentgenol. 2015;204(1):161–166.
Sahiner B, Chen W, Samala RK, Petrick N. Data drift in medical machine learning: implications and potential remedies. Br J Radiol. 2023;20220878.
Rahmani K, Thapa R, Tsou P, et al. Assessing the effects of data drift on the performance of machine learning models used in clinical sepsis prediction. Int J Med Inform. 2023;173:104930.
Miller TT, Staron RB, Feldman F. Patellar height on sagittal MR imaging of the knee. AJR Am J Roentgenol. 1996;167(2):339–341.
Schlenzka D, Schwesinger G. The height of the patella: an anatomical study. Eur J Radiol. 1990;11(1):19–21.
Leung YF, Wai YL, Leung YC. Patella alta in southern China. A new method of measurement. Int Orthop. 1996;20(5):305–310.
Althani S, Shahi A, Tan TL, Al-Belooshi A. Position of the Patella among Emirati Adult Knees. Is Insall-Salvati Ratio Applicable to Middle-Easterners? Arch Bone Jt Surg. 2016;4(2):137–140.
He K, Gkioxari G, Dollár P, Girshick R. Mask R-CNN. arXiv [cs.CV]. 2017. http://arxiv.org/abs/1703.06870.
Liu Z, Mao H, Wu C-Y, Feichtenhofer C, Darrell T, Xie S. A ConvNet for the 2020s. arXiv [cs.CV]. 2022. p. 11976–11986. http://openaccess.thecvf.com/content/CVPR2022/html/Liu_A_ConvNet_for_the_2020s_CVPR_2022_paper.html. Accessed April 7, 2023.
Russakovsky O, Deng J, Su H, et al. ImageNet Large Scale Visual Recognition Challenge. Int J Comput Vis. 2015;115(3):211–252.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Clinical Impact
Artificial intelligence algorithms can measure the Insall-Salvati ratio and detect patellar height abnormalities, which are likely underreported in the clinical setting.
Highlights
Key Finding: Artificial intelligence algorithms can measure the Insall-Salvati ratio and detect patellar height abnormalities, which are likely underreported.
Importance: Accurate diagnosis of patellar height abnormalities can help prevent early-onset osteoarthritis of the knee.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix
Appendix
Neural Network Architecture
The Mask R-CNN architecture was used, with standard modifications for keypoint detection [35]. This network is based on a Convolutional Neural Network (CNN) with additional components for detecting, localizing, and classifying objects on an image. The complete model architecture is detailed in Figure S1. Briefly, an input image is first processed by a base CNN, which consists of convolutional and max-pooling layers, to produce feature maps. After the last convolutional layer, a regional proposal network is trained to propose candidate keypoint detections on the image on the basis of a fixed set of anchors on each position of the feature maps. The location and size of each anchor are fine-tuned with bounding-box regression. Candidate keypoints are filtered by performing nonmaximum suppression which removes redundant overlapping candidate regions. Region proposals are then sent to a region-of-interest pooling layer, which resamples the feature maps inside each proposal and is fed to another branch of the network that predicts the confidence scores. The base CNN used in our model was ConvNeXt_Large, which was set with pretrained weights from ImageNet, a large-scale object detection, segmentation, and captioning dataset [36, 37]. Ultimately, the keypoint model outputs three sets of x and y coordinates (keypoints) for each image: one set for the superior aspect of the patella, one set for the inferior aspect of the patella, and a third for the tibial tubercle, where the patellar tendon attaches. This network was implemented via the PyTorch v2.0.0 software package with Python 3.6.5, and training was done on Nvidia K80 and Nvidia P100 GPUs (Nvidia, Inc, Santa Clara, CA).
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Adleberg, J., Benitez, C.L., Primiano, N. et al. Fully Automated Measurement of the Insall-Salvati Ratio with Artificial Intelligence. J Digit Imaging. Inform. med. 37, 601–610 (2024). https://doi.org/10.1007/s10278-023-00955-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10278-023-00955-1