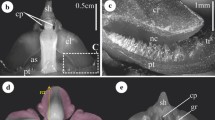
Abstract
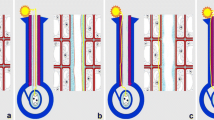
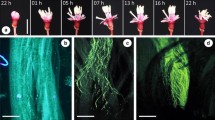
Pollination in Marantaceae is mediated by an explosive style movement. Before release, style tension is held by the hooded staminode. When a pollinator touches the trigger appendage of the hooded staminode the latter deforms and the style rapidly curls upwards. This movement has been interpreted as a turgor movement by some authors, but recent studies clearly indicate that setup, hold and release of tension are purely mechanical processes. However, in view of the high diversity of hooded staminodes, the question arises what keeps the tension in species with very thin staminodes. To test the holding mechanisms, we conducted mechanical and physico-chemical release experiments in four species with robust and four species with thin hooded staminodes in their natural tropical environment. We found almost the same response of all species to mechanical treatments, but species-specific reactions to different physico-chemical conditions. This indicates that style release follows the same mechanical principles in all species, but that the sensitivity of the explosive movement depends on material properties like tissue thickness and turgescence. As to the holding mechanisms, we found different degrees of floral synorganization. The hood of the hooded staminode formerly interpreted as an important holding structure does not play a noteworthy role. Instead, the basal plate of the hooded staminode antagonises the pressure of the style head against the holding point of the hooded staminode in species with robust hooded staminodes and well-developed basal plates. In some species with a thin hooded staminode, the latter is closely attached to the style and most likely stabilises tension by adhesive forces. In another species, a morphologically analogous structure adopts the function of the basal plate. We conclude that the holding mechanism of the style tension diversified during the evolution of Marantaceae whereas the release mechanism itself has been conserved throughout the family.

© M. Jerominek, Mainz




Similar content being viewed by others
Data availabitily
All row data are available from the authors.
References
Andersson L (1981) The neotropical genera of Marantaceae: circumscription and relationships. Nordic J Bot 1:218–245. https://doi.org/10.1111/j.1756-1051.1981.tb00692.x
Andersson L (1998) Marantaceae. In: Kubitzky K (ed) The families and genera of vascular plants. Vol. IV. Flowering plants, Monocotyledons, Alismatanae and Commelinanae (except Gramineae). Springer, Berlin, pp 278–293
Brantjes NBM, de Vos OC (1981) The explosive release of pollen in flowers of Hyptis (Lamiaceae). New Phytol 87:425–430. https://doi.org/10.1111/j.1469-8137.1981.tb03213.x
Claßen-Bockhoff R (1991) Untersuchungen zur Konstruktion des Bestäubungsapparates von Thalia geniculata (Marantaceen). Bot Acta 104:183–193
Claßen-Bockhoff R, Heller A (2008a) Floral synorganization and secondary pollen presentation in four Marantaceae from Costa Rica. Int J Plant Sci 169:745–760. https://doi.org/10.1086/588069
Claßen-Bockhoff R, Heller A (2008b) Style release experiments in four species of Marantaceae from the Golfo Dulce area, Costa Rica. Stapfia 88:557–571
Cocucci A (1989) El mecanismo floral de Schizanthus (Solanaceae). Kurtziana 20:113–132
Dean JA (1999) Lange’s handbook of chemistry. McGraw-Hill, New York
Delpino F (1869) Breve cenno sulle relazioni biologiche e geneologiche delle Matantaceae. Nuovo Giorn Bot Ital 1:293–306
Dodson CH (1962) Pollination and variation in the subtribe Catasetinae (Orchidaceae). Ann Missouri Bot Garden 49:35–56. https://doi.org/10.2307/2394740
Edwards J, Whitaker D, Klionsky S, Laskowski MJ (2005) A record-breaking pollen catapult. Nature 435:164. https://doi.org/10.1038/435164a
Eichler AW (1884) Beiträge zur Morphologie und Systematik der Marantaceen. Kgl. Akad. Wiss., Berlin
Forterre Y, Skotheim JM, Dumais J, Mahadevan L (2005) How the Venus flytrap snaps. Nature 433:421–425
Glinos E, Cocucci AA (2011) Pollination biology of Canna indica (Cannaceae) with particular reference to the functional morphology of the style. Plant Syst Evol 291:49–58. https://doi.org/10.1007/s00606-010-0379-x
Govaerts R, Kennedy H (2016) World checklist of Marantaceae. Facilitated by the royal botanic gardens, Kew. https://apps.kew.org/wcsp/. Retrieved 23 Jan 2016
Grant V (1994) Modes and origins of mechanical and ethological isolation in angiosperms. Proc Natl Acad Sci USA 91:3–10. https://doi.org/10.1073/pnas.91.1.3
Harkins WD, McLaughlin HM (1925) The structure of films of water on salt solutions I. Surface tension and adsorption for aqueous solutions of sodium chloride. J Am Chem Soc 47:2083–2089
Jerominek M, Claßen-Bockhoff R (2015) Electrical signals in prayer plants (Marantaceae)? Insights into the trigger mechanism of the explosive style movement. PLoS ONE 10:e0126411. https://doi.org/10.1371/journal.pone.0126411
Jerominek M, Will M, Claßen-Bockhoff R (2018) Insights into the inside—a quantitative histological study of the explosively moving style in Marantaceae. Front Plant Sci 9:1695. https://doi.org/10.3389/fpls.2018.01695
Jerominek M (2015) Experimental investigations on the explosive pollination mechanism in Marantaceae. Dissertation, Johannes Gutenberg-University Mainz, Germany
Kennedy K (1978) Systematics and pollination of the “cloed flowered” species of Calathea (Marantaceae). Univ Calif Publ Bot 71:1–90
Kennedy H (2000) Diversification in pollination mechanisms in the Marantaceae. In: Wilson KL, Morrison DA (eds) Monocots: systematics and evolution. CSIRO, Melbourne, pp 335–343
Kunze H (1984) Vergleichende Studien an Cannaceen- und Marantaceenblüten. Flora 175:301–318. https://doi.org/10.1016/S0367-2530(17)31453-6
Ley AC, Claßen-Bockhoff R (2010) Parallel evolution in plant-pollinator interaction in African Marantaceae. In: van der Burgt X, van der Maesen J, Onana JM (eds) Systematics and conservation of African plants. Royal Botanic Gardens, Kew, pp 847–854
Ley AC, Claßen-Bockhoff R (2011) Ontogenetic and phylogenetic diversification in Marantaceae. In: Wanntorp L, Ronse de Craene L (eds) Flowers on the tree of life. Cambridge University Press, Cambridge, pp 239–255
Ley AC, Claßen-Bockhoff R (2012) Floral synorganization and its influence on mechanical isolation and autogamy in Marantaceae. Bot J Linn Soc 168:300–322. https://doi.org/10.1111/j.1095-8339.2011.01202.x
Locatelli E, Machado IC, Medeiros P (2004) Saranthe klotzschiana (KOER.) EICHL. (Marantaceae) e seu mecanismo explosive de polinização. Rev Bras Bot 27:757–765
El Ottra JHL, Toni JFG, Thaowetsuwan P, Dos Santos P, Jeiter J, Ronse De Craene L, Bull-Hereñu K, Claβen-Bockhoff R (2023) Pollen transfer within flowers: how pollen is secondarily presented. Int J Plant Sci 185:15–31
Pischtschan E, Claßen-Bockhoff R (2008) Setting-up tension in the style of Marantaceae. Plant Biol 10:441–450. https://doi.org/10.1111/j.1438-8677.2008.00051.x
Pischtschan E, Claßen-Bockhoff R (2010) Anatomic insights into the thigmonastic style tissue in Marantaceae. Plant Syst Evol 286:91–102. https://doi.org/10.1007/s00606-010-0282-5
Pischtschan E, Ley AC, Classen-Bockhoff R (2010) Ontogenetic and phylogenetic diversification of the hooded staminode in Marantaceae. Taxon 59:1111–1125. https://doi.org/10.1002/tax.594011
Prince LM, Kress J (2006) Phylogenetic relationsships and classification in Marantaceae: insights from plastid DNA sequence data. Taxon 55:281–296. https://doi.org/10.2307/25065578
Raju AJS, Rao SP (2006) Explosive pollen release and pollination as a function of nectar-feeding activity of certain bees in the biodiesel plant, Pongamia pinnata (L.) Pierre (Fabaceae). Curr Sci 90:960–967
Schumann K (1902) Marantaceae. In: Engler A (ed) Das Pflanzenreich, vol IV. W. Engelmann, Leipzig, pp 1–184
Skotheim JM, Mahadevan L (2005) Physical limits and design principles for plant and fungal movements. Science 308:1308–1310. https://doi.org/10.1126/science.1107976
Steffens J, Harel E, Hunt M (1994) Polyphenol Oxidase. In: Ellis B, Kuroki G, Stafford H (eds) Genetic engineering o plant secondary metabolism, vol 28. Springer, Berlin, pp 275–312
Westerkamp C (1993) The co-operation between the asymmetric flower of Lathyrus latifolius (Fabaceae-Vicieae) and its visitors. Phyton 33:121–137
Acknowledgements
We thank Dr. Piyakaset Suksathan and Dr. Suyanee Vessabutr for local assistance and arrangements in the Queen Sirikit Botanic Garden (Thailand), and Dr. Zak Zahawi in the Wilson Botanical Garden (Costa Rica). We are grateful to Rodolfo Quirós and Pattarin Nunthamontree for their technical assistance. Thanks are also given to the editor of this special issue, Akitoshi Iwamoto, and to two unknown reviewers for their helpful comments.
Funding
Travel and accommodation expenses were funded by the German Academic Exchange Service (DAAD; D/09/49132).
Author information
Authors and Affiliations
Contributions
The project was initiated by RC-B. Both authors contributed to the study conception and design. Material preparation and data collection were performed by MJ. Data analysis and interpretation were conducted by MJ and RC-B. The first draft of the manuscript was written by MJ, the final version by RC-B. Both authors approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
There are no competing interests.
Ethics approval
There are no humans or animals involved in the research.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
V1: Trigger deflection (Goeppertia ecuadoriana) (MP4 9861 KB)
V2: Basal plate slipped away (Goeppertia zebrina var. humilior) (MP4 1174 KB)
V3: Longitudinal cut in distal hood (Goeppertia zebrina var. humilior) (MP4 3020 KB)
V4: Stinging style (Calathea crotalifera) (MP4 10215 KB)
V5: Transverse cut and downward buckling of the style (Goeppertia zebrina var. humilior) (MP4 2206 KB)
V6: Transverse cut and rupture of the style (Goeppertia zebrina var. zebrina) (MP4 5270 KB)
V7: Longitudinal cut at lower side of the style (Calathea crotalifera) (MP4 5383 KB)
V8: Time-lapse (20 min) black colouration after wounding (Goeppertia zebrina var. humilior) (MP4 5546 KB)
V9: Time-lapse (20 min) black colouration in 70% ethanol (Goeppertia zebrina var. zebrina) (MP4 5190 KB)
V10: Time-lapse (10 min) fixation chloroform steam (Stromanthe sanguinea) (MP4 5149 KB)
V11: Time-lapse (30 min) fixation in NaCl (Goeppertia zebrina var. zebrina) (MP4 7539 KB)
V12: Time-lapse (20 min) black colouration after freezing (Goeppertia zebrina var. humilior) (MP4 3036 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jerominek, M., Claßen-Bockhoff, R. What keeps the style under tension? Experimental tests to understand the biomechanics of the explosive style movement in Marantaceae. J Plant Res (2024). https://doi.org/10.1007/s10265-024-01535-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10265-024-01535-2