Abstract
The primary objective of this study was to assess the incidence, timing, risk factors of fungal infections (FIs) within 3 months after liver transplantation (LT). The secondary objective was to evaluate the impact of FIs on outcomes. Four hundred and ten patients undergoing LT from January 2015 until January 2023 in a tertiary university hospital were included in the present retrospective cohort study to investigate the risk factors of FIs and to assess the impacts of FIs on the prognosis of LT recipients using logistic regression. The incidence of FIs was 12.4% (51/410), and median time from LT to the onset of FIs was 3 days. By univariate analysis, advanced recipient age, prolonged hospital stay prior to LT, high Model for End Stage Liver Disease (MELD) score, use of broad-spectrum antibiotics, and elevated white blood cell (WBC) count, increased operating time, massive blood loss and red blood cell transfusion, elevated alanine aminotransferase on day 1 and creatinine on day 3 after LT, prolonged duration of urethral catheter, prophylactic antifungal therapy, the need for mechanical ventilation and renal replacement therapy were identified as factors of increased post-LT FIs risk. Multivariate logistic regression analysis identified that recipient age ≥ 55 years[OR = 2.669, 95%CI: 1.292–5.513, P = 0.008], MELD score at LT ≥ 22[OR = 2.747, 95%CI: 1.274–5.922, P = 0.010], pre-LT WBC count ≥ 10 × 109/L[OR = 2.522, 95%CI: 1.117–5.692, P = 0.026], intraoperative blood loss ≥ 3000 ml [OR = 2.691, 95%CI: 1.262–5.738, P = 0.010], post-LT duration of urethral catheter > 4 d [OR = 3.202, 95%CI: 1.553–6.602, P = 0.002], and post-LT renal replacement therapy [OR = 5.768, 95%CI: 1.822–18.263, P = 0.003] were independently associated with the development of post-LT FIs. Post-LT prophylactic antifungal therapy ≥ 3 days was associated with a lower risk of the development of FIs [OR = 0.157, 95%CI: 0.073–0.340, P < 0.001]. As for clinical outcomes, FIs had a negative impact on intensive care unit (ICU) length of stay ≥ 7 days than those without FIs [OR = 3.027, 95% CI: 1.558–5.878, P = 0.001] but had no impact on hospital length of stay and 1-month all-cause mortality after LT. FIs are frequent complications after LT and the interval between the onset of FIs and LT was short. Risk factors for post-LT FIs included high MELD score at LT, advanced recipient age, pre-LT WBC count, massive intraoperative blood loss, prolonged post-LT duration of urethral catheter, and the need for post-LT renal replacement therapy. However, post-LT prophylactic antifungal therapy was independently associated with the reduction in the risk of FIs. FIs had a significant negative impact on ICU length of stay.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Core tip
Despite advances in technology of liver transplantation, fungal infections remain challenging. Timely prevention of IF is critical. Many risk factors play a crucial role in the occurrence of FIs after LT and determine the prognosis of recipients. This study is one of the comprehensive studies to examine the role of FI in the prognosis of liver transplant recipients and the risk factors for all FIs after liver transplantation and to demonstrate that the risk factors are associated with preoperative, intraoperative and postoperative variables. the identification of these risk factors for FIs provides a basis for the prevention of FIs, which in turn improves the prognosis of LT recipients
Introduction
Liver transplantation (LT) is an effective treatment for end-stage liver diseases and certain malignancies [1]. LT recipients have a significant risk of fungal infections (FIs) which are outcome-determining complications in the early period after LT [2]. The overall incidence of invasive FIs in LT recipients is up to 42%, with an related mortality rate up to 80% [3,4,5,6,7]. LT recipients with invasive FIs were nearly five times more likely to die than those without [1]. The most common fungal pathogens (53%–93%) are Candida spp., followed by Aspergillus spp.(1.4%-19%) and Cryptococcus spp. (0.5%-8%) [8,9,10]. Approximately 34% and 46% of invasive Candida infections occur within one and three months after LT, respectively, earlier than Aspergillus infections do [1, 8].
Multiple previous studies stated many risk factors for invasive FIs, including hospitalization or antiinfective treatment before transplantation, an elevated Model for End Stage Liver Disease (MELD) score and serum creatinine, preexisting end-stage renal disease, fungal colonization prior to LT, live donor, donor derived infections, split-LT, prolonged operation duration, massive transfusion, Roux-en-y choledocho-jejunostomy, postoperative bacterial infection, acute kidney injury, the need for renal replacement therapy, early re-operation, CMV viremia, and biliary leakage, but the consensus is still missing to date [8, 11,12,13,14,15,16,17,18,19].
Although some studies examining the effects of invasive FIs on the prognosis of LT recipients as well as the risk factors for invasive FIs, only a few studies examined these issues in all FIs including invasive and noninvasive FIs. Our present work is one of those studies in an integrative manner to examine the role of all FIs in prognosis of LT recipients as well as the risk factors for all FIs after LT, and demonstrated the risk factors were pre-, intra- and, post-operative variables-related, and LT recipients with FIs had a longer intensive care unit (ICU) stay than those without. We identified these risk factors for FIs to provide a basis of preventing FIs and then to improve the prognosis of LT recipients.
Materials and methods
General information
We conducted a retrospective cohort study including all adult patients who received a LT between January 2015 and January 2023 at the Third Xiangya Hospital, a 1915-bed tertiary care hospital affiliated to Central South University performing liver transplants since 2001. Eventually, 410 LT recipients of grafts from donation after citizens’ death were eligible in this single-center retrospective cohort study. All liver grafts were from donation after brain death with the exception of one from donation after circulatory death. This study received approval by the Ethics Committee of the Third Xiangya Hospital, and written patient consent was waived because of the retrospective nature of the study (no.23776).
Inclusion and exclusion criteria
Inclusion criteria were adult (aged 18 years old and older) LT recipients. We excluded LT recipients who were younger than 18 years old, presented infections within two weeks before LT, or died during the peri-operative period for anesthesia accidents or surgical complications.
Definitions and assessment of FIs
Infection was confirmed based on the criteria of the Centers for Disease Control and Prevention/National Healthcare Safety Network (CDC/NHSN) [20]. FIs were defined according to updated EORTC/MSGERC consensus definitions for non-lung solid organ transplant recipients [21]. Invasive FIs were defined as clinical manifestations and fungal isolation with presence of organ damage by radiology, bronchoscopy or biopsy in the absence of other causes. In noninvasive FIs organ damage was not present [22]. Re-operation implicated in retransplantation or post-LT laparotomy. Acute rejection was T cell- or antibody- mediated and was biopsy-proven.
Treatment strategy
All patients were received a modified piggyback LT. A T-tube was not placed for biliary drainage except for 6 (1.5%) patients. All LT recipients were exposed to antibiotic prophylaxis for 3–5 days with the same third-generation cephalosporin or carbapenem used prior to or during LT according to the protocol of our center. Teicoplanin or other antibiotics against bacterial infections were prescribed with a treatment duration of 5–7 days when needed. The antifungal prophylaxis using caspofungin or another echinocandin for duration of 7–14 days was reserved for LT recipients having at least 2 predefined perioperative risk factors (such as retransplantation, renal dysfunction or fungi colonization prior to LT, choledochojejuno-stomy, massive transfusion, prolonged operating time, and so on), which was recommended by substantial studies [23,24,25].
An intraoperative 500 mg methylprednisolone bolus was administered intravenously as an initial treatment, with subsequent tapering over one week, and basiliximab induction was additionally combined in 217 (52.9%) LT recipients. Maintenance immunosuppressive therapy consisted of a double drug regimen including a calcineurin inhibitor (tacrolimus or ciclosporin A) and corticosteroid with or without an antimetabolite (Mycophenolate mofetil or enteric-coated mycophenolate sodium). Anti-thymocyte globulin was prescribed when needed. All patients after LT were closely monitored in the ICU for 3–7 days.
The content and acquisition methods of the data
The electronic medical records were reviewed for all LT recipients over 8 years. We also obtained patients’ information through regular outpatient follow-up. We collected general peri-operative conditions of the patients and all prespecified data possibly related to FIs, and postoperative outcomes (mortality, ICU and hospital length of stay). All LT patients were followed up for 3 months postoperatively for their microbiological data. Samples, including blood, urine, sputum, or abdominal drainage fluid, were routinely subject to bacterial culture once a day for 5–7 days following LT. Thereafter, if infection is suspected, samples were collected for culture within the 3 months after LT. When an infection was suspected, chest film, computerized tomography, blood routine, C-reactive protein, or procalcitonin, one or more of which will also be tested. All infections were distinguished from contamination.
Statistical analyses
All statistical analyses were performed using the SPSS software, version 26.0 statistical (SPSS, Inc., Chicago, IL). Continuous variables were presented as mean ± standard deviation if normally distributed or the median and interquartile range when non-normally distributed. Categorical data were expressed as absolute numbers and their percent. Pearson’s chi-square test or Fisher's exact test was used to test categorical variables, as appropriate. The significant risk factors identified in univariate analysis (P < 0.05) were included in the final multivariate analysis where binary logistic regression on the basis of forward stepwise logistic regression was used to determine risk factors with an odds ratio (OR) value along with a 95% confidence interval (CI). P < 0.05 was determined to represent statistical significance, and statistical assessments were two-tailed.
Results
General characteristics and prognosis of LT recipients
We retrospectively reviewed all patients who received a LT of graft from a deceased donor during the study period. Of 414 patients screened, 4 patients were excluded; 1 patient each died of massive blood loss during the operation and primary graft non-function, and 2 patients were younger than 18 years old. A total of 410 patients were eventually included in this study with median MELD score of 23. The mean age was 47.3 years old (± 10.6), and males accounted for 82.4% of the population. The majority of the patients underwent LT due to hepatitis virus-related cirrhosis/necrosis/tumor (n = 308). The remaining cases involved alcoholic liver disease (n = 31), mixed cirrhosis (n = 19), autoimmune hepatitis (n = 15), primary biliary cirrhosis (n = 11), cryptogenic cirrhosis (n = 9), Budd-Chiari syndrome (n = 5), hepatolenticular degeneration (n = 3), transplanted liver failure (n = 3), drug-induced liver injury (n = 2), polycystic liver (n = 2), and familial hereditary amyloidosis (n = 2). Infections within two months before LT accounted for 40.2% (165/410) of all patients with 45 (35.4%) suffering from pulmonary infections and 13 (3.2%) multiple sites infections, all of whom had pulmonary infections.
It was 0.8 mg/dL, 34.5 g/L, 5.3 × 109/L, 0.8 × 109/L, and 72.0 × 109/L for the median pre-LT creatinine, albumin, WBC count, lymphocyte count, and platelet count, respectively.
The median surgery time, blood loss and RBC transfusion were 378.5 min, 3000.0 ml and 12.0 units, respectively.
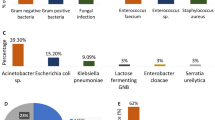
A total of 51 (12.4%) patients were infected with 55 strains of fungus within 3 months. Median time from transplantation to FIs was 3 days. Among these 55 strains of fungus, Candida spp. accounted for 84.3% of the fungi isolated and Aspergillus fumigatus a further 7.8%. Invasive FIs accounted for 45.1% (23/51) of all FIs. Of 410 LT patients involved, 385 (93.9%) were prescribed a carbapenem and the remaining patients (6.1%; 25/410) used a third-generation cephalosporin. After LT, 18 (4.4%) patients were treated with anti-thymocyte immunoglobulin therapy, and 399 (97.3%) with tacrolimus. The median alanine aminotransferase (ALT) and albumin on day 1 and median creatinine on day 3 after LT were 696.0 U/L, 37.2 g/L, and 0.9 mg/dL, respectively. A total 95 patients required mechanical ventilation, 19 needed renal replacement therapy, and 67 arose acute rejection after LT. Moreover, 4.1% (17/410) of cases received re-operation. The median postoperative ICU length of stay was 6.0 days. We observed 1-month mortality of 6.6% (27/410). The baseline demographic, clinical, and laboratory characteristics were described in Table 1.
Analysis of the risk factors for FIs after LT
A comparison of patients with and without FIs was performed. By univariate logistic regression, recipient age ≥ 55 years (P = 0.004), MELD score at LT ≥ 22 (P = 0.005), pre-LT hospitalization ≥ 7 days (P = 0.007), pre-LT use of broad-spectrum antibiotics ≥ 3 days (P = 0.012), pre-LT WBC count ≥ 10 × 109/L (P = 0.005), duration of surgery ≥ 450 min (P = 0.017), intraoperative blood loss ≥ 3000 ml (P = 0.004), intraoperative RBC transfusion ≥ 12 U (P = 0.013), ALT on day 1 after LT ≥ 1000 U/L (P = 0.039), creatinine on day 3 after LT ≥ 2 mg/dL (P = 0.023), post-LT duration of urethral catheter ≥ 4 days (P < 0.001), post-LT prophylactic antifungal therapy ≥ 3 days (P < 0.001), and the need for post-LT mechanical ventilation (P < 0.001) and renal replacement therapy (P < 0.001) were associated with an increased risk for post-LT FIs.
Finally, the multivariate analysis identified recipient age ≥ 55 years [OR = 2.669, 95%CI:1.292–5.513, P = 0.008], MELD score at LT ≥ 22 [OR = 2.747, 95%CI:1.274–5.922, P = 0.010], pre-LT WBC count ≥ 10 × 109/L [OR = 2.522, 95%CI:1.117–5.692, P = 0.026], intraoperative blood loss ≥ 3000 ml [OR = 2.691, 95%CI:1.262–5.738, P = 0.010], post-LT duration of urethral catheter > 4 d [OR = 3.202, 95%CI:1.553–6.602, P = 0.002], and post-LT renal replacement therapy [OR = 5.768, 95%CI:1.822–18.263, P = 0.003] as independent risk factors for post-LT FIs, which were reduced in LT recipients with post-LT prophylactic antifungal therapy ≥ 3 days [OR = 0.157, 95%CI:0.073–0.340, P < 0.001]. All data regarding the univariate and multivariate analysis were displayed in Table 2.
Prognosis of patients with FIs after LT
A Pearson’s chi-square test was used to access the effects of FIs on prognosis of LT recipients. Of note, patients with FIs were more likely to stay at ICU ≥ 7 days after LT than those without (P < 0.001). Patients with FIs had higher 1-month all-cause mortality rates than those without (17.6% versus 5.0%; P = 0.001). In contrast, FIs did not show a significant correlation to hospitalization stay after LT ≥ 21 days (P = 0.621) when compared to non-FIs (Table 3). All information was described in Table 3.
A univariate in conjunction with multivariate analysis of those potential risk factors for mortality was performed to decide if FIs were one of those independent risk factors for 1-month all-cause mortality. Recipient age ≥ 55 years [OR = 3.056, 95%CI:1.168–7.994, P = 0.023], creatinine on day 3 after LT ≥ 2 mg/dL [OR = 9.154, 95%CI:3.451–24.280, P < 0.001], post-LT bacterial infection [OR = 7.095, 95%CI:2.553–19.714, P < 0.001], and post-LT mechanical ventilation [OR = 5.742, 95%CI:2.176–15.151, P < 0.001], not FIs, were verified increased risk factors for 1-month mortality after LT. All information was shown in Table 4.
Nonetheless, when performed another analysis of the factors related to prolonged ICU stay, multivariate logistic regression analysis identified FIs [OR = 3.027, 95%CI:1.558–5.878, P = 0.001] as an independent risk factor for ICU stay after LT ≥ 7 days, among other variables such as pre-LT creatinine ≥ 2 mg/dL [OR = 2.843, 95%CI:1.228–6.579, P = 0.015], re-operation [OR = 3.251, 95%CI:1.097–9.628, P = 0.033], post-LT bacterial infection [OR = 1.840, 95%CI:1.150–2.944, P = 0.011], and the need for post-LT mechanical ventilation [OR = 3.029, 95%CI:1.825–5.028, P < 0.001]. All information was summarized in Table 5.
Discussion
LT recipients are susceptible to opportunistic infections due to complex surgical procedures placing a greater number of patients at risk [26]. According to a recent study, half of the early fatal infections were mainly fungi-related [1]. The incidence of invasive FIs within 90 days after LT remains very high, reaching 12%, which was rather similar to our overall incidence of FIs (12.4%) which included all invasive and non-invasive FIs [1, 27,28,29] Candida represented the most frequently isolated species in LT recipients with invasive FIs in previous studies, also in line with our present cohort (84.3%) [1].
As previously reported, we observed that recipient age ≥ 55 years was independently associated with post-LT FIs. This goes along with recent findings by Breitkopf R, et al. that recipient age had an increased hazard ratio for breakthrough or whole invasive FIs within 90 days in a multivariate Cox regression model [2, 30].
We found that MELD score at LT ≥ 22 was association with the development of post-LT FIs, in accordance with the studies from in Saliba et al. and Utsumi et al. who concluded the association between high MELD score and post-LT invasive FIs [12, 19, 31].
We also established the association between pre-LT WBC count ≥ 10 × 109/L and post-LT FIs. No previous studies reported elevated WBC influenced FIs development after LT. Hu et al. (2021) performed a meta analysis by combining data from 14 relevant studies enrolling 4,284 patients and concluded that bacterial infection was one of strong risk factors for invasive FIs after LT, which may partially explain why an elevated WBC count, in most cases, indicating the presence of an infection, was associated with FIs in our cohort [17].
As for intraoperative variables, our results suggest that blood loss ≥ 3000 ml was independently associated with post-LT FIs, in line with a previous study suggesting that excessive blood loss was one of risk factors for fungal infections in LT recipients [32].
We revealed post-LT duration of urethral catheter > 4 days as an independent risk factor for post-LT FIs. In a univariate analysis of bacterial and fungal infections after LT, Zhang W and colleagues found the association between urinary catheterization and infections; however, the significance did not remain in further multivariate analysis [33]. We should realize there is a reciprocal relationship between urinary catheterization and FIs. One the one hand, prolonged urinary catheterization may increase the risk for FIs; one the other hand, the FIs can also aggravate the disease and prolong the duration of urinary catheterization.
We confirmed that post-LT renal replacement therapy was correlated with FIs, consistent with a previous work [3]. A recent prospective multicenter study investigating the risk factors for breakthrough-invasive FI after LT also showed patients with and without breakthrough-invasive FI were significantly different for the need for renal replacement therapy after LT [29]. We confirmed that the risk for FIs was significantly reduced when a post-LT antifungal prophylaxis was implemented ≥ 3 d, in line with previous studies claiming that receipt of an antifungal agent was associated with decreased risk of post-LT invasive FIs after LT [12, 34,35,36,37].
Previous reports demonstrated that although most of mortality were unrelated to FIs, a high mortality was expected among immunocompromised patients with FIs [38, 39]. However, the effect of FIs on outcome of LT recipients is still a subject of discussion. Our present study revealed that FIs had a negative impact on ICU length of stay but had no impact on hospitalization stay after LT and 1-month all-cause mortality. Breitkopf R, et al. also confirmed the impact of invasive FIs on longer ICU stay and in contrast to our findings, they has linked invasive FIs and breakthrough invasive FIs with a significant impact on 1-year mortality [2, 30]. Now that the morbidity and mortality of invasive candidiasis and aspergillosis were high in LT recipients, targeted prophylaxis for Candida spp. and Aspergillus spp. was recommended since 2009 [40].
Limits of the study
This study has several limitations. First, the retrospective single-center design implicates inherent selection bias and a limited generalizability, and only association rather than causation can be inferred. However, all consecutive LT recipients over a period of 8 years were investigated which represents one of the largest recently published studies in this field to examine the role of all FIs in outcomes as well as the risk factors for all FIs in an integrative manner. Second, the information of fungal colonization prior to LT would have provided insight into potential effects on the development of post-LT FIs which many studies stated, unfortunately these data were not available in our center [17]. Third, the heterogeneity of types of infection may lead to an inaccurate analysis of risk factors for IFs.
Conclusion
All in all, although our study comprised a comparably large cohort of LT recipients, the burden of these infections on the fate of LT recipients emphasises the need for further prospective studies to identify these risk factors. Knowledge of these information aids in a successful LT as a result of prevention of FIs.
Data sharing statement
The original anonymous dataset is available on request from the corresponding author at eraldo.occhetta@maggioreosp.novara.it.
Data availability
No datasets were generated or analyzed during the current study.
References
Breitkopf R, Treml B, Bukumiric Z, et al. Invasive fungal infections: the early killer after liver transplantation. J Fungi (Basel), 2023;9(6).[PMID: 37367592 https://doi.org/10.3390/jof9060655]
Breitkopf R, Treml B, Senoner T, et al. Invasive fungal breakthrough infections under targeted echinocandin prophylaxis in high-risk liver transplant recipients. J Fungi (Basel), 2023;9(2).[PMID: 36836384 https://doi.org/10.3390/jof9020272]
Nagao M, Fujimoto Y, Yamamoto M, et al. Epidemiology of invasive fungal infections after liver transplantation and the risk factors of late-onset invasive aspergillosis. J Infect Chemother, 2016;22(2): 84–9.[PMID: 26683245 https://doi.org/10.1016/j.jiac.2015.11.005]
Nieto-Rodriguez JA, Kusne S, Manez R, et al. Factors associated with the development of candidemia and candidemia-related death among liver transplant recipients. Ann Surg, 1996;223(1): 70–6.[PMID: 8554421 https://doi.org/10.1097/00000658-199601000-00010]
Senoner T, Breitkopf R, Treml B, et al. Invasive fungal infections after liver transplantation. J Clin Med, 2023;12(9).[PMID: 37176678 https://doi.org/10.3390/jcm12093238]
Neofytos D, Chatzis O, Nasioudis D, et al. Epidemiology, risk factors and outcomes of invasive aspergillosis in solid organ transplant recipients in the Swiss Transplant Cohort Study. Transpl Infect Dis, 2018;20(4):e12898.[PMID: 29668068 https://doi.org/10.1111/tid.12898]
Barchiesi F, Mazzocato S, Mazzanti S, et al. Invasive aspergillosis in liver transplant recipients: epidemiology, clinical characteristics, treatment, and outcomes in 116 cases. Liver Transpl, 2015;21(2): 204–12.[PMID: 25348192 https://doi.org/10.1002/lt.24032]
Scolarici M, Jorgenson M, Saddler C, et al. Fungal Infections in Liver Transplant Recipients. J Fungi (Basel), 2021;7(7).[PMID: 34210106 https://doi.org/10.3390/jof7070524]
Pappas P G, Alexander B D, Andes D R, et al. Invasive fungal infections among organ transplant recipients: results of the Transplant-Associated Infection Surveillance Network (TRANSNET). Clin Infect Dis, 2010;50(8):1101–11.[PMID: 20218876 https://doi.org/10.1086/651262]
van Delden C, Stampf S, Hirsch H H, et al. Burden and timeline of infectious diseases in the first year after solid organ transplantation in the swiss transplant cohort study. J Clin Infect Dis, 2020;71(7): e159-e69.[PMID: 31915816 https://doi.org/10.1093/cid/ciz1113]
Husain S, Tollemar J, Dominguez E A, et al. Changes in the spectrum and risk factors for invasive candidiasis in liver transplant recipients: prospective, multicenter, case-controlled study. Transplantation, 2003;75(12): 2023–9.[PMID: 12829905 https://doi.org/10.1097/01.TP.0000065178.93741.72]
Saliba F, Delvart V, Ichai P, et al. Fungal infections after liver transplantation: outcomes and risk factors revisited in the MELD era. Clin Transplant, 2013;27(4): E454–61.[PMID: 23656358 https://doi.org/10.1111/ctr.12129]
Viehman J A, Clancy C J, Clarke L, et al. Surgical site infections after liver transplantation: emergence of multidrug-resistant bacteria and implications for prophylaxis and treatment strategies. Transplantation, 2016;100(10): 2107–14.[PMID: 27479167 https://doi.org/10.1097/TP.0000000000001356]
Winston DJ, Pakrasi A, Busuttil RW. Prophylactic fluconazole in liver transplant recipients. A randomized, double-blind, placebo-controlled trial. Ann Intern Med, 1999; 131(10): 729–37.[PMID: 10577295 https://doi.org/10.7326/0003-4819-131-10-199911160-00003]
Rogers J, Rohal S, Carrigan D R, et al. Human herpesvirus-6 in liver transplant recipients: role in pathogenesis of fungal infections, neurologic complications, and outcome. Transplantation, 2000;69(12): 2566–73.[PMID: 10910278 https://doi.org/10.1097/00007890-200006270-00016]
Karchmer A W, Samore M H, Hadley S, et al. Fungal infections complicating orthotopic liver transplantation. Trans Am Clin Climatol Assoc, 1995;106: 38–47; discussion -8.[PMID: 7483177
Liu M, Zhu Z, Sun L. Risk Factors of invasive fungal infection in recipients after liver transplantation: a systematic review and meta-analysis. Front Med (Lausanne), 2021;8: 687028.[PMID: 34671611 https://doi.org/10.3389/fmed.2021.687028]
Eschenauer G A, Kwak E J, Humar A, et al. Targeted versus universal antifungal prophylaxis among liver transplant recipients. Am J Transplant, 2015;15(1): 180–9.[PMID: 25359455 https://doi.org/10.1111/ajt.12993]
Utsumi M, Umeda Y, Yagi T, et al. Risk analysis for invasive fungal infection after living donor liver transplantation: Which patient needs potent prophylaxis? Dig Surg, 2019;36(1): 59–66.[PMID: 29649828 https://doi.org/10.1159/000486548]
Horan T C, Andrus M, Dudeck M A. CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control, 2008;36(5): 309–32.[PMID: 18538699 https://doi.org/10.1016/j.ajic.2008.03.002]
Donnelly J P, Chen S C, Kauffman C A, et al. Revision and update of the consensus definitions of invasive fungal disease from the european organization for research and treatment of cancer and the mycoses study group education and research consortium. Clin Infect Dis, 2020;71(6): 1367–76.[PMID: 31802125 https://doi.org/10.1093/cid/ciz1008]
Gavalda J, Meije Y, Fortun J, et al. Invasive fungal infections in solid organ transplant recipients. Clin Microbiol Infect, 2014;20 Suppl 7: 27–48.[PMID: 24810152 https://doi.org/10.1111/1469-0691.12660]
Breitkopf R, Treml B, Bukumiric Z, Innerhofer N, Fodor M, Rajsic S. Invasive fungal infections: the early killer after liver transplantation. J Fungi (Basel). 2023;9(6):655.
Breitkopf R, Treml B, Simmet K, Bukumiric´ Z, Fodor M, Senoner T, Rajsi S. Incidence of invasive fungal infections in liver transplant recipients under targeted echinocandin prophylaxis. J Clin Med 2023;12:1520
Breitkopf R, Treml B, Senoner T, Bukumiric´ Z, Rajsic S. Invasive fungal breakthrough infections under targeted echinocandin prophylaxis in high-risk liver transplant recipients. J Fungi 2023;9:272.
Kim SI. Bacterial infection after liver transplantation. World J Gastroenterol, 2014;20(20): 6211–20.[PMID: 24876741 https://doi.org/10.3748/wjg.v20.i20.6211]
Neyra KM, Brizendine K D. Retrospective study evaluating the performance of risk factors for invasive mold infections in liver transplantation. Transpl Infect Dis, 2020;22(1):e13223.[PMID: 31782873 https://doi.org/10.1111/tid.13223]
Kang WH, Song GW, Lee SG, et al. A multicenter, randomized, open-label study to compare micafungin with fluconazole in the prophylaxis of invasive fungal infections in living-donor liver transplant recipients. J Gastrointest Surg, 2020;24(4): 832–40.[PMID: 31066013 https://doi.org/10.1007/s11605-019-04241-w]
Rinaldi M, Bartoletti M, Ferrarese A, et al. Breakthrough invasive fungal infection after liver transplantation in patients on targeted antifungal prophylaxis: A prospective multicentre study. Transpl Infect Dis, 2021;23(4): e13608.[PMID: 33768656 https://doi.org/10.1111/tid.13608]
Breitkopf R, Treml B, Simmet K, et al. Incidence of invasive fungal infections in liver transplant recipients under targeted echinocandin prophylaxis. J Clin Med, 2023;12(4).[PMID: 36836055 https://doi.org/10.3390/jcm12041520]
Lichtenstern C, Hochreiter M, Zehnter V D, et al. Pretransplant model for end stage liver disease score predicts posttransplant incidence of fungal infections after liver transplantation. Mycoses, 2013;56(3): 350–7.[PMID: 23346877 https://doi.org/10.1111/myc.12041]
Fung JJ. Fungal infection in liver transplantation.J Transpl Infect Dis, 2002;4 Suppl 3: 18–23.[PMID: 12486788 https://doi.org/10.1034/j.1399-3062.4.s3.3.x]
Zhang W, Wang W, Kang M, et al. Bacterial and fungal infections after liver transplantation: microbial epidemiology, risk factors for infection and death with infection. Ann Transplant, 2020;25: e921591.[PMID: 32424111 https://doi.org/10.12659/AOT.921591]
Sharpe MD, Ghent C, Grant D, et al. Efficacy and safety of itraconazole prophylaxis for fungal infections after orthotopic liver transplantation: a prospective, randomized, double-blind study. Transplantation, 2003;76(6): 977–83.[PMID: 14508365 https://doi.org/10.1097/01.TP.0000085653.11565.52]
Cruciani M, Mengoli C, Malena M, et al. Antifungal prophylaxis in liver transplant patients: a systematic review and meta-analysis. Liver Transpl, 2006;12(5): 850–8.[PMID: 16628697 https://doi.org/10.1002/lt.20690]
Evans J D, Morris P J, Knight S R. Antifungal prophylaxis in liver transplantation: a systematic review and network meta-analysis. Am J Transplant, 2014;14(12): 2765–76.[PMID: 25395336 https://doi.org/10.1111/ajt.12925]
Chen Y C, Huang T S, Wang Y C, et al. Effect of prophylactic antifungal protocols on the prognosis of liver transplantation: a propensity score matching and multistate model approach. Biomed Res Int, 2016;2016: 6212503.[PMID: 27747235 https://doi.org/10.1155/2016/6212503]
Samanta P, Clancy C J, Marini R V, et al. Isavuconazole Is as effective as and better tolerated than voriconazole for antifungal prophylaxis in lung transplant recipients. Clin Infect Dis, 2021;73(3): 416–26.[PMID: 32463873 https://doi.org/10.1093/cid/ciaa652]
Herrera S, Davoudi S, Farooq A, et al. Late onset invasive pulmonary aspergillosis in lung transplant recipients in the setting of a targeted prophylaxis/preemptive antifungal therapy strategy. Transplantation, 2020;104(12): 2575–81.[PMID: 32080158 https://doi.org/10.1097/TP.0000000000003187]
Pappas P G, Kauffman C A, Andes D, et al. Clinical practice guidelines for the management of candidiasis: 2009 update by the Infectious Diseases Society of America. Clin Infect Dis, 2009;48(5): 503–35.[PMID: 19191635 https://doi.org/10.1086/596757]
Acknowledgements
We are grateful to Hunan Provincial Health Commission for providing funding and all the patients who collected data for their co-operation
Funding
The Key project of Hunan Provincial Health Commission, China (grant numbers 202217012851).
Author information
Authors and Affiliations
Contributions
JJ, QW collected and analyzed the data; QW, PP wrote the original manuscript, revised the paper, and approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
All the Authors have no conflict of interest related to the manuscript.
Institutional review board statement
The study was reviewed and approved for publication by our Institutional Reviewer.
Informed consent statement
All study participants or their legal guardian provided informed written consent about personal and medical data collection prior to study enrolment.
STROBE statement
We have read the STROBE Statement—checklist of items, and the manuscript was prepared and revised according to the STROBE Statement—checklist of items.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jiang, J., Peng, P. & Wan, Q. The predictors of fungal infections after liver transplantation and the influence of fungal infections on outcomes. Clin Exp Med 24, 144 (2024). https://doi.org/10.1007/s10238-024-01419-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10238-024-01419-8