Abstract
Despite great advances in treatment, 30–40% of patients with DLBCL undergo relapses. Patients with a relapse within 1 year or beyond have a distinct outcome. Few clinical characteristics and survival data in the Chinese population have been published. We aimed to define the incidence and clinical features of DLBCL patients with very early relapse after front-line immunochemotherapy who may benefit greatly from the emerging chimeric antigen receptor T-cell therapy. Data of 564 DLBCL patients were analyzed. Among the 413 patients achieving a first complete remission, 59 underwent relapses: 32 patients (54.2%) relapsed within 1 year, and 27 patients (46.8%) relapsed 1 year or more. Patients relapsing within 1 year, in comparison with the other group, showed an inferior risk profile at diagnosis: elevated lactate dehydrogenase level (P = 0.002), high Eastern Cooperative Oncology Group performance score (P = 0.02), and high international prognosis index (P = 0.004). As expected, a worse overall survival was observed in the early relapse group. Multivariate analysis for OS showed that relapse within 1 year was an independent parameter for reduced overall survival (HR 0.241, P = 0.002).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Diffuse large B cell lymphoma (DLBCL) represents the most common histologic subtype of aggressive non-Hodgkin’s lymphoma (NHL), comprising around 40% of all malignant lymphoid neoplasms [1]. Two dramatic advances have led to great improvements in the outcome and survival of patients with DLBCL. The first advance was the introduction of anthracycline-containing chemotherapy in the mid-1970s [2]. Combination chemotherapy consisting of cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) proves the best available treatment for DLBCL patients in the subsequent clinical trial [3]. The second is the addition of rituximab, the monoclonal anti-CD20 antibody, to the CHOP chemotherapy, which further improves the outcome [4]. At least 50% of DLBCL patients achieved long-term disease-free survival [5].
Despite breakthroughs in treatment, approximately 30–40% of patients with DLBCL undergo relapses eventually [6, 7]. Patients with refractory DLBCL or a relapse within 1 year show dismal outcomes compared to those with a late relapse [8]. According to previous literature, the risk for death doubles for early relapses within 1 year compared to those who relapsed later. Only 13% of patients who relapsed within 1 year were alive after a follow-up of 8 years. Besides, these patients had a lower overall response to salvage regimens [9]. With the advent of CAR-T, the survival of refractory/relapsed DLBCL patients was prolonged significantly compared with standard care, especially those who relapsed within 1 year [10].
Identifying risk factors associated with the very early relapse occurring within 1 year is significant in filtering out potential patients who may benefit from CAR-T therapy. Hitherto, very few series of patients who relapsed within 1 year after complete remission have been published. This study aims to investigate the incidence, clinical features, and overall survival of DLBCL patients with an early relapse in China.
Patients and methods
Study population
All consecutive patients with definite diagnoses of DLBCL between March 2009 and April 2017 at our center were included. Patients with incomplete data on treatment response were excluded. Primary central nervous system lymphomas (PCNSL) at diagnosis were excluded for heterogeneity. The diagnosis of DLBCL was confirmed independently by two experienced pathologists based on the World Health Organization classification criteria for hematopoietic and lymphoid tissue tumors [11]. CR was defined according to the 1999 Cheson criteria [12]. The study was approved by the Ethics Committee of Peking Union Medical College Hospital.
Endpoints and assessment
In our study, the primary endpoint was the occurrence of relapse. Relapse was defined as the recurrence of the disease in patients who achieved CR after the initial treatment. Primarily refractory patients who present progressive disease or stable disease as the best response to chemotherapy were excluded from our study. Relapse was preferably demonstrated by rebiopsy of involved sites. Patients without pathological proof were also considered to relapse if there was evident progression on fluorodeoxyglucose positron-emission tomography (FDG-PET) in concordance with aggressive clinical presentation. Early relapse in our study was defined as relapse within 1 year. The survival endpoint was also assessed. Overall survival (OS) was calculated from the date of diagnosis until the death or date of the last follow-up.
Data collection
Clinical data of relapse patients were collected including age, gender, cell of origin(COO) based on Hans’ classification, Eastern Cooperative Oncology Group performance score (ECOG PS), Ann Arbor stage, international prognostic index (IPI) score, extranodal involvement, bone marrow involvement, lactate dehydrogenase (LDH), β2-microglobulin (β2-MG), EBV DNA, lymphocyte/monocyte ratio (LMR), initial treatment, and relative dose. The relative dose (RD) was calculated as the mean RD for cyclophosphamide and doxorubicin: RD = (actual dose) / (planned dose).
Follow-up
Follow-up was conducted via telephone, review of patient records, and the electronic follow-up system at our hematology department for lymphoma patients. All study subjects were followed from the date of lymphoma diagnosis until death or the end of the study period (Oct 2022), whichever occurred first.
Statistical analysis
Statistics between-group categorical data were compared using the Chi-square test or Fisher’s exact probability test. Factors with p < 0.1 in the univariate analysis were selected for inclusion in the multivariate logistic regression analysis. Survival analysis was performed using the Kaplan–Meier method. Data were analyzed using GraphPad 9.0 (GraphPad, San Diego, CA, USA) and SPSS 22.0 statistical software (IBM Corp., Armonk, NY, USA). Differences with P < 0.05 were considered statistically significant.
Results
Demographic features
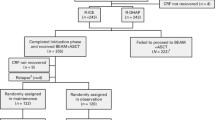
We identified 670 patients diagnosed with DLBCL from our center. Patients with incomplete data on treatment response (n = 65) and patients diagnosed with PCNSL (n = 41) were excluded. Eventually, 564 patients were included. After first-line treatment, 413 patients obtained CR. With a median follow-up of 65.1 months (range: 0.1–159.3 months), 59 patients relapsed: 32 patients (54.2%) relapsed within 1 year, and 27 patients (45.8%) relapsed 1 year or more from CR1(Fig. 1). Patients undergoing an early relapse had a median age of 60 years (range 28–77), while the median age of the patients experiencing relapse after 1 year was 59 years (range 20–81). The male-to-female ratio was 1:1 and 0.6:1, respectively. The difference in age and gender was non-significant between the two groups. Detailed clinical, paraclinical, and treatment features are described in Table 1.
Risk factors
We conducted a comparison of the clinical characteristics between the two groups. Patients relapsing in 1 year showed an inferior risk profile at diagnosis: elevated LDH level (P = 0.002), high ECOG PS (P = 0.02), and high IPI (P = 0.004). No significant difference was identified in cell of origin, Ann Arbor stage, extranodal involvement, bone marrow involvement, β2-MG, and LMR. EBV DNA was available for 37 cases. However, no distinction was detected between two groups.
Initial treatment was similar in the two groups. CHOP or CHOP-like chemotherapy combined with rituximab was adopted with curative intent. Dosage reduction to a relative dose of 75–100% was made in patients who could not tolerate adequate regimens after geriatric assessments. Reduction in dosage was more common in patients with relapse within 1 year (46.9%). In the other group, 25.9% of patients received a reduced regimen. Overall, 13 patients received adjuvant radiotherapy and 8 patients received front-line autologous stem cell transplant (ASCT) as consolidation. Nevertheless, no statistical difference was found.
Logistic regression analysis was conducted to identify the risk factors for early relapses. Factors with P < 0.1 in the univariate analysis were selected for inclusion in the multivariate logistic regression analysis. The results show that a higher ECOG PS and IPI score were closely associated with early relapses (Table 2).
Treatment at relapse
The relapse was biopsy-proven in 25 of 59 patients (42.4%). All of them relapsed with DLBCL histology. Salvage regimens were diverse and are shown in Table 3. Thirty-eight of 59 patients (64.4%) with relapse chose a rituximab-containing regimen as second-line treatment. Chemotherapies such as DHAP (dexamethasone, cisplatin, and cytarabine), GDP (gemcitabine, dexamethasone, and cisplatin), ICE (ifosfamide, carboplatin, and etoposide), ESHAP (etoposide, methylprednisolone, cytarabine, cisplatin), GemOx (gemcitabine, oxaliplatin), and MINE (mesna, ifosfamide, mitoxantrone, etoposide) were given to patients. Only 4 patients in our study received second-line ASCT afterward. A considerable proportion of patients with relapse lost eligibility for ASCT due to a poor response to classic salvage chemotherapy (n = 16) or a rapid decline in clinical status (n = 25). Another barrier to accessing ASCT was the relevant high cost (n = 10). Programmed cell death protein 1 (PD-1) blockade and lenalidomide were attempted in a small fraction of patients with relapse. Since CAR-T therapy was CFDA-approved in 2021 for the treatment of refractory/relapsed DLBCL, only one patient who relapsed within 1 year received CAR-T therapy.
Overall survival
A trend of better five-year OS was observed in patients who relapsed after 1 year than those who relapsed within 1 year (median OS, 42.65, and 24.2 months, respectively; log-rank test, P = 0.04; Fig. 2). Multivariate analysis for OS showed that relapse within 1 year was an independent parameter for reduced overall survival (HR 0.241, 95% CI 0.1–0.583, P = 0.002, Table 4). Both ECOG PS and IPI scores failed to be statistically significant in COX regression analysis though they are significant in univariate analysis. We failed to demonstrate a beneficial effect of ASCT on the outcome since only 4 patients (6.7%) received second-line ASCT after salvage chemotherapy. Noticeably, the only patient who adopted CAR-T therapy achieved a second CR and was alive till the last follow-up, indicating the remarkable efficacy of CAR-T therapy on relapsed, especially, early elapsed DLBCL patients.
Discussion
According to previous studies, patients with DLBCL usually relapse early, within 1–2 years from diagnosis [6, 13]. Patients relapsing within 1 year from remission have a poor prognosis resembling refractory patients and are less likely to respond to second-line chemotherapy compared to those relapsing beyond 1 year [8, 9]. The emerging CAR-T therapy shows promise in the front line for these patients and is recommended as the preferred choice for patients who relapsed within 1 year in the latest National Comprehensive Cancer Network guidelines. Besides, CAR-T therapy is demonstrated to be highly effective as part of first-line therapy for high-risk DLBCL, with a manageable safety profile [14, 15]. So it is important to distinguish patients prone to relapse early. Few studies address this issue in the Chinese population in the post-rituximab era.
In our study, patients relapsing within 1 year show different clinical characteristics from those with late relapses at diagnosis. They enjoyed a higher IPI score and worse performance scores. Elevation of LDH was seen more often in them. Other baseline features such as extranodal or bone marrow involvement, EBV DNA, β2-MG, and LMR are also included for analysis. However, no significant difference was demonstrated. All patients received standard R-CHOP therapy. For the elderly intolerant to adequate therapy, a minor reduction was made to a relative dosage of 75–100% with curative intent. We observed a higher percentage of reduced chemotherapy in the early relapse group (46.9% vs. 25.9%) though no significant difference was found between the two groups. In a previous study, radiotherapy was identified to lower the occurrence of relapses within 2 years from diagnosis [16]. However, we failed to verify its effect on the time of relapse in our study, which may be attributed to the limited sample size. Front-line ASCT seems to have a limited protective effect on late relapse. In a multivariate logistic regression analysis, we found that a higher ECOG PS and IPI score were closely associated with early relapses.
As expected, patients relapsing within 1 year have a worse OS than those with late relapse. Moreover, we found that time to relapse was an independent prognostic factor for overall survival, in accordance with the previous study [9]. With regard to second-line therapy, the majority of patients in our study received salvage chemotherapy according to standard care [8, 17]. For those who were not fit for chemotherapy, we provided chemo-free approaches with favorable toxicity profiles. PD-1 inhibitor and lenalidomide were attempted. Secondary ASCT was preferred when a secondary response was achieved for eligible patients. Only one person in our cohort was administered CAR-T therapy. This patient achieved CR and was still alive till the last follow-up, expanding curative options for this population.
In conclusion, patients relapsing within 1 year enjoyed an inferior risk profile at diagnosis: elevated lactate dehydrogenase level, high ECOG score, and high IPI score. In the multivariate logistic regression analysis, ECOG score, and high IPI score are associated with early relapse. Our analysis has shown that the prognosis of early relapsing patients is worse than that of those relapsing later. Time to relapse is an independent prognostic factor for patients with DLBCL. Genetic landscapes associated with early relapse patterns remain to be explored further.
References
Alaggio R, Amador C, Anagnostopoulos I, Attygalle AD, Araujo IBO, Berti E, Bhagat G, Borges AM, Boyer D, Calaminici M, Chadburn A, Chan JKC, Cheuk W, Chng WJ, Choi JK, Chuang SS, Coupland SE, Czader M, Dave SS, de Jong D, Du MQ, Elenitoba-Johnson KS, Ferry J, Geyer J, Gratzinger D, Guitart J, Gujral S, Harris M, Harrison CJ, Hartmann S, Hochhaus A, Jansen PM, Karube K, Kempf W, Khoury J, Kimura H, Klapper W, Kovach AE, Kumar S, Lazar AJ, Lazzi S, Leoncini L, Leung N, Leventaki V, Li XQ, Lim MS, Liu WP, Louissaint A Jr, Marcogliese A, Medeiros LJ, Michal M, Miranda RN, Mitteldorf C, Montes-Moreno S, Morice W, Nardi V, Naresh KN, Natkunam Y, Ng SB, Oschlies I, Ott G, Parrens M, Pulitzer M, Rajkumar SV, Rawstron AC, Rech K, Rosenwald A, Said J, Sarkozy C, Sayed S, Saygin C, Schuh A, Sewell W, Siebert R, Sohani AR, Tooze R, Traverse-Glehen A, Vega F, Vergier B, Wechalekar AD, Wood B, Xerri L, Xiao W. The 5th edition of the World Health Organization classification of haematolymphoid tumours: lymphoid neoplasms. Leukemia. 2022;36(7):1720–48. https://doi.org/10.1038/s41375-022-01620-2.
McKelvey EM, Gottlieb JA, Wilson HE, Haut A, Talley RW, Stephens R, Lane M, Gamble JF, Jones SE, Grozea PN, Gutterman J, Coltman C, Moon TE. Hydroxyldaunomycin (Adriamycin) combination chemotherapy in malignant lymphoma. Cancer. 1976;38(4):1484–93. https://doi.org/10.1002/1097-0142(197610)38:4%3c1484::aid-cncr2820380407%3e3.0.co;2-i.
Fisher RI, Gaynor ER, Dahlberg S, Oken MM, Grogan TM, Mize EM, Glick JH, Coltman CA Jr, Miller TP. Comparison of a standard regimen (CHOP) with three intensive chemotherapy regimens for advanced non-Hodgkin’s lymphoma. N Engl J Med. 1993;328(14):1002–6. https://doi.org/10.1056/nejm199304083281404.
Coiffier B, Lepage E, Briere J, Herbrecht R, Tilly H, Bouabdallah R, Morel P, Van Den Neste E, Salles G, Gaulard P, Reyes F, Lederlin P, Gisselbrecht C. CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large-B-cell lymphoma. N Engl J Med. 2002;346(4):235–42. https://doi.org/10.1056/NEJMoa011795.
Feugier P, Van Hoof A, Sebban C, Solal-Celigny P, Bouabdallah R, Fermé C, Christian B, Lepage E, Tilly H, Morschhauser F, Gaulard P, Salles G, Bosly A, Gisselbrecht C, Reyes F, Coiffier B. Long-term results of the R-CHOP study in the treatment of elderly patients with diffuse large B-cell lymphoma: a study by the Groupe d’Etude des Lymphomes de l’Adulte. J Clin Oncol Off J Am Soc Clin Oncol. 2005;23(18):4117–26. https://doi.org/10.1200/jco.2005.09.131.
Vannata B, Conconi A, Winkler J, Cascione L, Margiotta Casaluci G, Nassi L, Moia R, Pirosa MC, Moccia AA, Stathis A, Rossi D, Gaidano G, Zucca E. Late relapse in patients with diffuse large B-cell lymphoma: impact of rituximab on their incidence and outcome. Br J Haematol. 2019;187(4):478–87. https://doi.org/10.1111/bjh.16106.
Larouche JF, Berger F, Chassagne-Clément C, Ffrench M, Callet-Bauchu E, Sebban C, Ghesquières H, Broussais-Guillaumot F, Salles G, Coiffier B. Lymphoma recurrence 5 years or later following diffuse large B-cell lymphoma: clinical characteristics and outcome. J Clin Oncol Off J Am Soc Clin Oncol. 2010;28(12):2094–100. https://doi.org/10.1200/jco.2009.24.5860.
Gisselbrecht C, Glass B, Mounier N, Singh Gill D, Linch DC, Trneny M, Bosly A, Ketterer N, Shpilberg O, Hagberg H, Ma D, Brière J, Moskowitz CH, Schmitz N. Salvage regimens with autologous transplantation for relapsed large B-cell lymphoma in the rituximab era. J Clin Oncol Off J Am Soc Clin Oncol. 2010;28(27):4184–90. https://doi.org/10.1200/jco.2010.28.1618.
Guglielmi C, Gomez F, Philip T, Hagenbeek A, Martelli M, Sebban C, Milpied N, Bron D, Cahn JY, Somers R, Sonneveld P, Gisselbrecht C, Van Der Lelie H, Chauvin F. Time to relapse has prognostic value in patients with aggressive lymphoma enrolled onto the Parma trial. J Clin Oncol Off J Am Soc Clin Oncol. 1998;16(10):3264–9. https://doi.org/10.1200/jco.1998.16.10.3264.
Locke FL, Miklos DB, Jacobson CA, Perales MA, Kersten MJ, Oluwole OO, Ghobadi A, Rapoport AP, McGuirk J, Pagel JM, Muñoz J, Farooq U, van Meerten T, Reagan PM, Sureda A, Flinn IW, Vandenberghe P, Song KW, Dickinson M, Minnema MC, Riedell PA, Leslie LA, Chaganti S, Yang Y, Filosto S, Shah J, Schupp M, To C, Cheng P, Gordon LI, Westin JR. Axicabtagene ciloleucel as second-line therapy for large B-cell lymphoma. N Engl J Med. 2022;386(7):640–54. https://doi.org/10.1056/NEJMoa2116133.
Baram DV, Asaulenko ZP, Spiridonov IN, Krivolapov YA. WHO classification of tumors of hematopoietic and lymphoid tissues, 2022 (5th edition): lymphoid tumors. Arkh Patol. 2023;85(4):24–31. https://doi.org/10.17116/patol20238504124.
Cheson BD, Pfistner B, Juweid ME, Gascoyne RD, Specht L, Horning SJ, Coiffier B, Fisher RI, Hagenbeek A, Zucca E, Rosen ST, Stroobants S, Lister TA, Hoppe RT, Dreyling M, Tobinai K, Vose JM, Connors JM, Federico M, Diehl V. Revised response criteria for malignant lymphoma. J Clin Oncol Off J Am Soc Clin Oncol. 2007;25(5):579–86. https://doi.org/10.1200/jco.2006.09.2403.
Lee AY, Connors JM, Klimo P, O’Reilly SE, Gascoyne RD. Late relapse in patients with diffuse large-cell lymphoma treated with MACOP-B. J Clin Oncol Off J Am Soc Clin Oncol. 1997;15(5):1745–53. https://doi.org/10.1200/jco.1997.15.5.1745.
Abramson JS, Palomba ML, Gordon LI, Lunning MA, Wang M, Arnason J, Mehta A, Purev E, Maloney DG, Andreadis C, Sehgal A, Solomon SR, Ghosh N, Albertson TM, Garcia J, Kostic A, Mallaney M, Ogasawara K, Newhall K, Kim Y, Li D, Siddiqi T. Lisocabtagene maraleucel for patients with relapsed or refractory large B-cell lymphomas (TRANSCEND NHL 001): a multicentre seamless design study. Lancet (London, England). 2020;396(10254):839–52. https://doi.org/10.1016/s0140-6736(20)31366-0.
Neelapu SS, Dickinson M, Munoz J, Ulrickson ML, Thieblemont C, Oluwole OO, Herrera AF, Ujjani CS, Lin Y, Riedell PA, Kekre N, de Vos S, Lui C, Milletti F, Dong J, Xu H, Chavez JC. Axicabtagene ciloleucel as first-line therapy in high-risk large B-cell lymphoma: the phase 2 ZUMA-12 trial. Nat Med. 2022;28(4):735–42. https://doi.org/10.1038/s41591-022-01731-4.
Modvig L, Vase M, d’Amore F. Clinical and treatment-related features determining the risk of late relapse in patients with diffuse large B-cell lymphoma. Br J Haematol. 2017;179(1):75–82. https://doi.org/10.1111/bjh.14822.
Thieblemont C, Gisselbrecht C. Second-line treatment paradigms for diffuse large B-cell lymphomas. Curr Oncol Rep. 2009;11(5):386–93. https://doi.org/10.1007/s11912-009-0052-0.
Acknowledgements
Thanks to all the patients and their families and thanks to Dr. Yuping Zhou for her help in statistics.
Author information
Authors and Affiliations
Contributions
HYC wrote the first draft of the manuscript; JRZ and DQZ designed the work; WW, CW, and ZW provided the data; DBZ and WZ revised the final draft. All authors reviewed and edited the final manuscript before submission.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of Peking Union Medical College Hospital.
Consent to participate
Informed consent was obtained from the patient in this case report.
Consent to publish
Informed consent was obtained to publish our work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, H., Zhao, J., Zhao, D. et al. Lymphoma relapse 1 year or later after immunochemotherapy in DLBCL patients: clinical features and outcome. Clin Exp Med 24, 48 (2024). https://doi.org/10.1007/s10238-024-01306-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10238-024-01306-2