Abstract
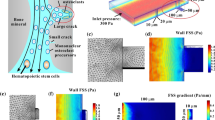
Mechanical loading, such as fluid shear stress (FSS), is regarded as the main factor that regulates the biological responses of bone cells. Our previous studies have demonstrated that the RAW264.7 osteoclast precursors migrate toward the low-FSS region under the gradient FSS field by a cone-and-plate flow chamber, in which the FSS in the outer region is larger than that in the inner region along the radial direction. Whether the FSS distribution on a cell depends on the gradient direction of FSS field should be clarified to explain this experimental observation. In this study, the finite element models of the discretely distributed or closely packed cells adherent on the bottom plate in a cone-and-plate flow chamber were constructed, and cells were regarded as compressible isotropic Hookean solid. Results showed that the average FSS of each discretely distributed cell at the quarter sector far from the center (SFC) was about 0.1% greater than that at the quarter sector near the center (SNC). In the bands with different orientations for a cell, the relative difference between the average FSS in the SFC and the SNC becomes smaller with increased band height. For the hexagonal closely packed cells, the relative value of SFC and SNC increases with increasing cell spacing. The difference between the local wall FSS in the SFC and the SNC may activate mechanosensitive ion channels and further regulate the migration of osteoclast precursors toward the low-FSS region under the gradient FSS field.







Similar content being viewed by others
References
Alcaraz J, Buscemi L, Grabulosa M, Trepat X, Fabry B, Farre R, Navajas D (2003) Microrheology of human lung epithelial cells measured by atomic force microscopy. Biophys J 84(3):2071–2079. https://doi.org/10.1016/S0006-3495(03)75014-0
Attard MM (2003) Finite strain––isotropic hyperelasticity. Int J Solids Struct 40(17):4353–4378. https://doi.org/10.1016/S0020-7683(03)00217-8
Bilodeau GG (1992) Regular pyramid punch problem. J Appl Mech 59(3):519–523. https://doi.org/10.1115/1.2893754
Boudot C, Saidak Z, Boulanouar AK, Petit L, Gouilleux F, Massy Z, Brazier M, Mentaverri R, Kamel S (2010) Implication of the calcium sensing receptor and the phosphoinositide 3-kinase/Akt pathway in the extracellular calcium-mediated migration of RAW 264.7 osteoclast precursor cells. Bone 46(5):1416–1423. https://doi.org/10.1016/j.bone.2010.01.383
Cox DB (1962) Radial flow in the cone-plate viscometer. Nature 193:670
Evans E, Kukan B (1984) Passive material behavior of granulocytes based on large deformation and recovery after deformation tests. Blood 64(5):1028–1035. https://doi.org/10.1182/blood.V64.5.1028.1028
Fabry B, Maksym GN, Butler JP, Glogauer M, Navajas D, Fredberg JJ (2001) Scaling the microrheology of living cells. Phys Rev Lett 87(14):148102. https://doi.org/10.1103/PhysRevLett.87.148102
Gao Y, Li TY, Sun Q, Huo B (2019a) Gradient fluid shear stress regulates migration of osteoclast precursors. Cell Adhes Migr 13(1):183–191. https://doi.org/10.1080/19336918.2019.1619433
Gao Y, Li TY, Sun Q, Ye CY, Guo MM, Chen ZB, Chen J, Huo B (2019b) Migration and differentiation of osteoclast precursors under gradient fluid shear stress. Biomech Model Mechanobiol 18(6):1731–1744. https://doi.org/10.1007/s10237-019-01171-z
George D, Allena R, Rémond Y (2018) A multiphysics stimulus for continuum mechanics bone remodeling. Math Mech Complex Syst 6(4):307–319. https://doi.org/10.2140/memocs.2018.6.307
George D, Allena R, Bourzac C, Pallu S, Bensidhoum M, Portier H, Rémond Y (2020) A new comprehensive approach for bone remodeling under medium and high mechanical load based on cellular activity. Math Mech Complex Syst 8(4):287–306. https://doi.org/10.2140/memocs.2020.8.287
Giorgio I, Spagnuolo M, Andreaus U, Scerrato D, Bersani AM (2020) In-depth gaze at the astonishing mechanical behavior of bone: a review for designing bio-inspired hierarchical metamaterials. Math Mech Solids 26(7):1074–1103. https://doi.org/10.1177/1081286520978516
Grover WH, Bryan AK, Diez-Silva M, Suresh S, Higgins JM, Manalis SR (2011) Density-measuring single-cell density. Proc Natl Acad Sci USA 108(27):10992–10996. https://doi.org/10.1073/pnas.1104651108
Guilak F, Mow VC (2000) The mechanical environment of the chondrocyte: a biphasic nite element model of cell matrix interactions in articular cartilage. J Biomech Eng 130(6):061009. https://doi.org/10.1115/1.2978991
Ishii M, Kikuta J, Shimazu Y, Meier-Schellersheim M, Germain RN (2010) Chemorepulsion by blood S1P regulates osteoclast precursor mobilization and bone remodeling in vivo. J Exp Med 207(13):2793–2798. https://doi.org/10.1084/jem.20101474
Jones WR, Ting-Beall PH, Lee GM, Kelley SS, Hochmuth RM, Guilak F (1999) Alterations in the Young’s modulus and volumetric properties of chondrocytes isolated from normal and osteoarthritic human cartilage. J Biomech 32(2):119–127. https://doi.org/10.1016/s0021-9290(98)00166-3
Koay EJ, Shieh AC, Athanasiou KA (2003) Creep indentation of single cells. J Biomech Eng 125(3):334–341. https://doi.org/10.1115/1.1572517
Koizumi K, Saitoh Y, Minami T, Takeno N, Tsuneyama K, Miyahara T, Nakayama T, Sakurai H, Takano Y, Nishimura M, Imai T, Yoshie O, Saiki I (2009) Role of CX3CL1/fractalkine in osteoclast differentiation and bone resorption. J Immunol 183(12):7825–7831. https://doi.org/10.4049/jimmunol.0803627
Li P, Hu M, Sun S, Zhang Y, Gao Y, Long M, Huo B, Zhang D (2012) Fluid flow-induced calcium response in early or late differentiated osteoclasts. Ann Biomed Eng 40(9):1874–1883. https://doi.org/10.1007/s10439-012-0554-z
Li P, Liu C, Hu M, Long M, Zhang D, Huo B (2014) Fluid flow-induced calcium response in osteoclasts: signaling pathways. Ann Biomed Eng 42(6):1250–1260. https://doi.org/10.1007/s10439-014-0984-x
Li P, Bian X, Liu C, Wang S, Guo M, Tao Y, Huo B (2018) STIM1 and TRPV4 regulate fluid flow-induced calcium oscillation at early and late stages of osteoclast differentiation. Cell Calcium 71:45–52. https://doi.org/10.1016/j.ceca.2017.12.001
Liu CL, Li SN, Ji BH, Huo B (2014) Flow-induced migration of osteoclasts and regulations of calcium signaling pathways. Cell Mol Bioeng 8(1):213–223. https://doi.org/10.1007/s12195-014-0372-5
Mahaffy RE, Shih CK, MacKintosh FC, Kas J (2000) Scanning probe-based frequency-dependent microrheology of polymer gels and biological cells. Phys Rev Lett 85(4):880–883. https://doi.org/10.1103/PhysRevLett.85.880
Maksym GN, Fabry B, Butler JP, Navajas D, Tschumperlin DJ, Laporte JD, Fredberg JJ (2000) Mechanical properties of cultured human airway smooth muscle cells from 0.05 to 0.4 Hz. J Appl Physiol 89(4):1619–1632. https://doi.org/10.1152/jappl.2000.89.4.1619
McAllister TN, Du T, Frangos JA (2000) Fluid shear stress stimulates prostaglandin and nitric oxide release in bone marrow-derived preosteoclast-like cells. Biochem Biophys Res CO 270(2):643–648. https://doi.org/10.1006/bbrc.2000.2467
McGarry, JG, Prendergast, PJ (2004) A three-dimensional finite element model of an adherent eukaryotic cell. Eur Cells Mater 16(7):27–34. https://doi.org/10.22203/eCM.v007a03
Mijailovich SM, Kojic M, Zivkovic M, Fabry B, Fredberg JJ (2002) A finite element model of cell deformation during magnetic bead twisting. J Appl Physiol 93(4):1429–1436. https://doi.org/10.1152/japplphysiol.00255.2002
Petersen NO, McConnaughey WB, Elson EL (1982) Dependence of locally measured cellular deformability on position on the cell. Proc Natl Acad Sci USA 79(17):5327–5331. https://doi.org/10.1073/pnas.79.17.5327
Robling AG, Castillo AB, Turner CH (2006) Biomechanical and molecular regulation of bone remodeling. Annu Rev Biomed Eng 8:455–498. https://doi.org/10.1146/annurev.bioeng.8.061505.095721
Sdougos HP, Bussolari SR, Dewey CF (2006) Secondary flow and turbulence in a cone-and-plate device. J Fluid Mech 138:379–404. https://doi.org/10.1017/s0022112084000161
Sebastian JM, Franziska W, Carina B, Christian B, Albrecht K, Gekle S (2020) A hyperelastic model for simulating cells in flow. Biomech Model Mechanobiol 20(2):509–520. https://doi.org/10.1007/s10237-020-01397-2
Shieh AC, Athanasiou KA (2002) Biomechanics of single chondrocytes and osteoarthritis. Crit Rev Biomed Eng 30(4–6):307–343. https://doi.org/10.1615/critrevbiomedeng.v30.i456.40
Shieh AC, Athanasiou KA (2003) Principles of cell mechanics for cartilage tissue engineering. Ann Biomed Eng 31(1):1–11. https://doi.org/10.1114/1.1535415
Shin D, Athanasiou K (1999) Cytoindentation for obtaining cell biomechanical properties. J Orthop Res 17(6):880–890. https://doi.org/10.1002/jor.1100170613
Simo JC, Pister KS (1984) Remarks on rate constitutive equations for finite deformation problems: computational implications. Comput Methods Appl Mech Eng 46:201–205. https://doi.org/10.1016/0045-7825(84)90062-8
Theret DP, Levesque MJ, Sato M, Nerem RM, Wheeler LT (1988) The application of a homogeneous half-space model in the analysis of endothelial cell micropipette measurements. J Biomech Eng 110(3):190–199. https://doi.org/10.1115/1.3108430
Wheal BD, Beach RJ, Tanabe N, Dixon SJ, Sims SM (2014) Subcellular elevation of cytosolic free calcium is required for osteoclast migration. J Bone Miner Res 29(3):725–734. https://doi.org/10.1002/jbmr.2068
Xue F, Lennon AB, McKayed KK, Campbell VA, Prendergast PJ (2015) Effect of membrane stiffness and cytoskeletal element density on mechanical stimuli within cells: an analysis of the consequences of ageing in cells. Comput Methods Biomech Biomed Eng 18(5):468–476. https://doi.org/10.1080/10255842.2013.811234
Yan WW, Cai B, Liu Y, Fu BM (2012) Effects of wall shear stress and its gradient on tumor cell adhesion in curved microvessels. Biomech Model Mechan 11(5):641–653. https://doi.org/10.1007/s10237-011-0339-6
Ye CY, Ali S, Sun Q, Guo MM, Liu Y, Gao Y, Huo B (2019) Novel cone-and-plate flow chamber with controlled distribution of wall fluid shear stress. Comput Biol Med 106:140–148. https://doi.org/10.1016/j.compbiomed.2019.01.014
Yoshikawa N, Ariyoshi H, Ikeda M, Sakon M, Kawasaki T, Monden M (1997) Shear-stress causes polarized change in cytoplasmic calcium concentration in human umbilical vein endothelial cells. Cell Calcium 22(3):189–197. https://doi.org/10.1016/S0143-4160(97)90012-9
Acknowledgements
This work was supported by the National Natural Science Foundation of China [12072034 and 11572043 (BH)].
Author information
Authors and Affiliations
Contributions
BH, YG and XZ designed the research. XZ, YG, QS, CYY and TYL performed the numerical simulation. XZ, YG and BH draft the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
We declare that we have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10237_2022_1574_MOESM1_ESM.docx
Supplementary file1 (DOCX 1199 kb)—Figure S1. Average FSS in the SFC and the SNC on discretely distributed cells along the radial direction when the secondary flow is not considered. (A–C) Average FSS in the SFC and the SNC in a cell when 1, 3, or 5 cells are placed on the plate along the radial direction. (D) Relative average FSS between the SFC and the SNC. Figure S2. Average FSS within the bands with different orientations in the SFC and the SNC on discretely distributed cells when the secondary flow is not considered. (A–I) Average FSS within the bands with different orientations in the SFC and the SNC in a cell when 1, 3, or 5 cells are placed on the plate along the radial direction. (J) Relative average FSS within the bands between the SFC and the SNC. Figure S3. Average FSS in the SFC and the SNC on hexagonal closely packed cells when the secondary flow is not considered. (A–C) Average FSS in the SFC and the SNC in 1, 3, and 5 cell groups. (D) Relative average FSS between the SFC and the SNC. Figure S4. Average FSS within bands with different orientations in the SFC and the SNC on hexagonal closely packed cells when the secondary flow is not considered. (A-I) Average FSS within the bands with different orientations in the SFC and the SNC in 1, 3, and 5 cell groups. (J) Relative average FSS within bands between the SFC and the SNC.
Rights and permissions
About this article
Cite this article
Zhang, X., Sun, Q., Ye, C. et al. Finite element analysis on mechanical state on the osteoclasts under gradient fluid shear stress. Biomech Model Mechanobiol 21, 1067–1078 (2022). https://doi.org/10.1007/s10237-022-01574-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-022-01574-5