Abstract
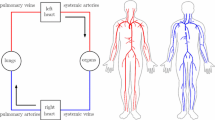
Isolated post-capillary pulmonary hypertension (Ipc-PH) occurs due to left heart failure, which contributes to 1 out of every 9 deaths in the United States. In some patients, through unknown mechanisms, Ipc-PH transitions to combined pre-/post-capillary PH (Cpc-PH) and is associated with a dramatic increase in mortality. Altered mechanical forces and subsequent biological signaling in the pulmonary vascular bed likely contribute to the transition from Ipc-PH to Cpc-PH. However, even in a healthy pulmonary circulation, the mechanical forces in the smallest vessels (the arterioles, capillary bed, and venules) have not been quantitatively defined. This study is the first to examine this question via a computational fluid dynamics model of the human pulmonary arteries, arterioles, venules, and veins. Using this model, we predict temporal and spatial dynamics of cyclic stretch and wall shear stress with healthy and diseased hemodynamics. In the normotensive case for large vessels, numerical simulations show that large arteries have higher pressure and flow than large veins, as well as more pronounced changes in area throughout the cardiac cycle. In the microvasculature, shear stress increases and cyclic stretch decreases as vessel radius decreases. When we impose an increase in left atrial pressure to simulate Ipc-PH, shear stress decreases and cyclic stretch increases as compared to the healthy case. Overall, this model predicts pressure, flow, shear stress, and cyclic stretch that providing a way to analyze and investigate hypotheses related to disease progression in the pulmonary circulation.











Similar content being viewed by others
Notes
Available from open-source Simvascular platform http://simvascular.github.io/clinicalCase4.html.
References
Allen B, van Ooij P, Barker A, Collins J, Carr J, Markl M, Kansal P (2014) Impact of beta-blocker therapy on thoracic aorta 3d wall shear stress in patients with bicuspid aortic valve. J Cardiovasc Magn Reson 16:1–2
Antiga L, Piccinelli M, Botti L, EIordache B, Remuzzi A, Steinman D (2008) An image-based modeling framework for patient-specific computational hemodynamics. Med Biol Eng Comput 46:1097
Banks J, Booth FM, MacKay E, Rajagopalan B, De J, Lee G (1978) The physical properties of human pulmonary arteries and veins. Clin Sci Mol Med 55:477–484
Barron V, Brougham C, Coghlan K, McLucas E, OMahoney D, Stenson-Cox C, McHugh P (2007) The effect of physiological cyclic stretch on the cell morphology, cell orientation and protein expression of endothelial cells. J Mater Sci Mat Med 18:1973–1981
Birukov K (2009) Cyclic stretch, reactive oxygen species, and vascular remodeling. Antioxi Redox Signal 11:1651–1667
Blanco P, Bulant C, Müller L, Talou G, Bezerra C, Lemos P, Feijóo R (2018) Comparison of 1d and 3d models for the estimation of fractional flow reserve. Sci Rep 8:1–12
Blanco P, Feijóo R (2010) A 3d–1d-0d computational model for the entire cardiovascular system. Mecánica Comput 29:5887–5911
Boron W, Boulpaep E (2016) Medical physiology E-book. Elsevier Health Sciences, Amsterdam
Caro C, Saffman P (1965) Extensibility of blood vessels in isolated rabbit lungs. J Physiol 178:193–201
Chaliki H, Hurrell D, Nishimura R, Reinke R, Appleton C (2002) Pulmonary venous pressure: relationship to pulmonary artery, pulmonary wedge, and left atrial pressure in normal, lightly sedated dogs. Catheter Cardiovasc Interv 56:432–438
Chambers M, Colebank M, Qureshi M, Clipp R, Olufsen M (2020) Structural and hemodynamic properties of murine arterial networks under hypoxia-induced pulmonary hypertension. Proc Inst Mech Eng Part H: J Eng Med p. 0954411920944110
Chambers M, Colebank M, Qureshi M, Clipp R, Olufsen M (2020) Structural and hemodynamic properties of murine pulmonary arterial networks under hypoxia-induced pulmonary hypertension. Proc Inst Mech Eng Part H J Eng Med 234:1312–1329
Clark A, Tawhai M (2018) Temporal and spatial heterogeneity in pulmonary perfusion: a mathematical model to predict interactions between macro-and micro-vessels in health and disease. The ANZIAM J 59:562–580
Colebank M, Paun L, Qureshi M, Chesler N, Husmeier D, Olufsen M, Fix L (2019) Influence of image segmentation on one-dimensional fluid dynamics predictions in the mouse pulmonary arteries. J Royal Soc Interf 16:20190,284
Colebank M, Qureshi M, Rajagopal S, Krasuski R, Olufsen M (2021) A multiscale model of vascular function in chronic thromboembolic pulmonary hypertension. Am J Physiol. https://doi.org/10.1152/ajpheart.00086.2021
Davies P (2009) Hemodynamic shear stress and the endothelium in cardiovascular pathophysiology. Nature Clin Prac Cardiovasc Med 6:16–26
Eslami P, Tran J, Jin Z, Karady J, Sotoodeh R, Lu M, Hoffmann U, Marsden A (2020) Effect of wall elasticity on hemodynamics and wall shear stress in patient-specific simulations in the coronary arteries. J Biomech Eng 142:024503
Fung Y, Sobin S (1969) Theory of sheet flow in lung alveoli. J Appl Physiol 26:472–488
Gao Y, Raj J (2005) Role of veins in regulation of pulmonary circulation. Am J Physiol 288:L213–L226
Gerges M, Gerges C, Pistritto A, Lang M, Trip P, Jakowitsch J, Binder T, Lang I (2015) Pulmonary hypertension in heart failure. epidemiology, right ventricular function, and survival. Am J Respir Crit Care 192:1234–1246
Ghio S, Gavazzi A, Campana C, Inserra C, Klersy C, Sebastiani R, Arbustini E, Recusani F, Tavazzi L (2001) Independent and additive prognostic value of right ventricular systolic function and pulmonary artery pressure in patients with chronic heart failure. J Am Coll Cardiol 37:183–188
Ghorishi Z, Milstein J, Poulain F, Moon-Grady A, Tacy T, Bennett S, Fineman J, Eldridge M (2007) Shear stress paradigm for perinatal fractal arterial network remodeling in lambs with pulmonary hypertension and increased pulmonary blood flow. Am J Physiol 292:H3006–H3018
Guazzi M, Borlaug B (2012) Pulmonary hypertension due to left heart disease. Circulation 126:975–990
Guglin M, Khan H (2010) Pulmonary hypertension in heart failure. J Card Fail 16:461–474
Hall J, Hall M (2020) Guyton and Hall textbook of medical physiology e-Book. Elsevier Health Sciences, Amsterdam
He X, Ku D (1996) Pulsatile flow in the human left coronary artery bifurcation: average conditions. J Bomech Eng 118:74–82
Huang W, Yen R, McLaurine M, Bledsoe G (1996) Morphometry of the human pulmonary vasculature. J Appl Physiol 81:2123–2133
Kheyfets V, Rios L, Smith T, Schroeder T, Mueller J, Murali S, Lasorda D, Zikos A, Spotti J, Reilly JJ, Finol E (2015) Patient-specific computational modeling of blood flow in the pulmonary arterial circulation. Comp Meth Prog Biomed 120:88–101
Krenz GS, Dawson CA (2003) Flow and pressure distributions in vascular networks consisting of distensible vessels. Am J Physiol 284:H2192–H2203
Lam C, Roger V, Rodeheffer R, Borlaug B, Enders F, Redfield M (2009) Pulmonary hypertension in heart failure with preserved ejection fraction: a community-based stud. J Am Coll Cardiol 53:1119–1126
Lighthill SJ (1975) Mathematical biofluiddynamics. SIAM, Philadelphia, PA
Maloney J, Rooholamini S, Wexler L (1970) Pressure-diameter relations of small blood vessels in isolated dog lung. Microvasc Res 2:1–12
Michel T, Paul MV (2010) Cellular signaling and no production. Eur J Physiol 459:807–816
Miller W, Grill D, Borlaug B (2013) Clinical features, hemodynamics, and outcomes of pulmonary hypertension due to chronic heart failure with reduced ejection fraction: pulmonary hypertension and heart failure. JACC Heart Fail 1:290–299
Moraes D, Loscalzo J (1997) Pulmonary hypertension: newer concepts in diagnosis and management. Clin Cardiol 20:676–682
Mozaffarian D, Benjamin E, Go A, Arnett D, Blaha M, Cushman M, Das S, de Ferranti S, Despres J, Fullerton H, Howard V, Huffman M, Isasi C, Jimenez M, Judd S, Kissela B, Lichtman J, Lisabeth L, Liu S, Mackey R, Magid D, McGuire D, Mohler ER, Moy C, Muntner P, Mussolino M, Nasir K, Neumar R, Nichol G, Palaniappan L, Pandey D, Reeves M, Rodriguez C, Rosamond W, Sorlie P, Stein J, Towfighi A, Turan T, Virani S, Woo D, Yeh R, Turner M, Members WG, Committee AHAS, Subcommittee SS (2016) Heart disease and stroke statistics-2016 update: a report from the american heart association. Circulation 133:e38–e360. https://doi.org/10.1161/CIR.0000000000000350
Mynard J, Smolich J (2015) One-dimensional haemodynamic modeling and wave dynamics in the entire adult circulation. Ann Biomed Eng 43:1443–1460
Nauser TD, Stites SW (2001) Diagnosis and treatment of pulmonary hypertension. Am Fam Phys 63:1789
Nichols W, ORourke M, Kenney W (1991) Mcdonalds blood flow in arteries: theoretical, experimental and clinical principles
Olufsen M, Hill N, Vaughan G, Sainsbury C, Johnson M (2012) Rarefaction and blood pressure in systemic and pulmonary arteries. J Fluid Mech 705:280–305
Olufsen M, Peskin C, Kim W, Pedersen E, Nadim A, Larsen J (2000) Numerical simulation and experimental validation of blood flow in arteries with structured-tree outflow conditions. Ann Biomed Eng 28:1281–1299
Papadaki M, Mclntire L (1999) Quantitative measurement of shear-stress effects on endothelial cells. In: Tissue Engineering Methods and Protocols, pp. 577–593. Springer
Paszkowiak J, Dardik A (2003) Arterial wall shear stress: observations from the bench to the bedside. Vasc Endovasc Surg 37:47–57
Paun L, Colebank M, Olufsen M, Hill N, Husmeier D (2020) Assessing model mismatch and model selection in a bayesian uncertainty quantification analysis of a fluid-dynamics model of pulmonary blood circulation. J Royal Soc Interf 17:20200,886
Pedersen E (1993) In vitro and in vivo studies of blood flow in the normal abdominal aorta and aorta bifurcation. Tech. rep., Technical Report, Århus University Hospital, Skejby, Denmark
Pedersen E, Sung HW, Burlson A, Yoganathan A (1993) Two-dimensional velocity measurements in a pulsatile flow model of the normal abdominal aorta simulating different hemodynamic conditions. J Biomech 26:1237–1247
Postles A, Clark A, Tawhai M (2014) Dynamic blood flow and wall shear stress in pulmonary hypertensive disease. In: 2014 36th Ann Int Conf of the IEEE Eng Med Biol Soc, pp. 5671–5674. IEEE
Pries A, Neuhaus D, Gaehtgens P (1992) Blood viscosity in tube flow: dependence on diameter and hematocrit. Am J Physiol 263:H1770–H1778
Qureshi M, Vaughan G, Sainsbury C, Johnson M, Peskin C, Olufsen M, Hill N (2014) Numerical simulation of blood flow and pressure drop in the pulmonary arterial and venous circulation. Biomech Model Mechanobiol 13:1137–1154
Ramu B, Thenappan T (2016) Evolving concepts of pulmonary hypertension secondary to left heart disease. Curr Heart Fail Rep 13:92–102
Ravi Y, Selvendiran K, Naidu S, Meduru S, Citro L, Bognar B, Khan M, Kalai T, Hideg K, Kuppusamy P, Sai-Sudhakar C (2013) Pulmonary hypertension secondary to left-heart failure involves peroxynitrite-induced downregulation of pten in the lung. Hypertension 61:593–601
Reymond P, Bohraus Y, Perren F, Lazeyras F, Stergiopulos N (2011) Validation of a patient-specific one-dimensional model of the systemic arterial tree. Am J Phsyiol 301:H1173–H1182
Reymond P, Perren F, Lazeyras F, Stergiopulos N (2012) Patient-specific mean pressure drop in the systemic arterial tree, a comparison between 1-d and 3-d models. J Biomech 45:2499–2505
Riches A, Sharp J, Thomas DB, Smith SV (1973) Blood volume determination in the mouse. J Physiol 228:279–284
Roux E, Bougaran P, Dufourcq P, Couffinhal T (2020) Fluid shear stress sensing by the endothelial layer. Front Physiol 11:861
Schäfer M, Ivy D, Barker A, Kheyfets V, Shandas R, Abman S, Hunter K, Truong U (2017) Characterization of cmr-derived haemodynamic data in children with pulmonary arterial hypertension. Eur Heart J Cardiovasc Imag 18:424–431
Segers P, Dubois F, De Wachter D, Verdonck P (1998) Role and relevancy of a cardiovascular simulator. Cardiovasc Eng 3:48–56
Selzer A (1992) Understanding heart disease. University of California Press, Berkeley, CA
Sicard D, Fredenburgh L, Tschumperlin D (2017) Measured pulmonary arterial tissue stiffness is highly sensitive to afm indenter dimensions. J Mech Behavior Biomed Mater 74:118–127
Simonneau G, Montani D, Celermajer D, Denton C, Gatzoulis M, Krowka M, Williams P, Souza R (2019) Haemodynamic definitions and updated clinical classification of pulmonary hypertension. Eur Respir J 53:1801,913
Sobin S, Lindal R, Bernick S (1977) The pulmonary arteriole. Microvasc Res 14:227–239
Spilker R, Feinstein J, Parker D, Reddy V, Taylor C (2007) Morphometry-based impedance boundary conditions for patient-specific modeling of blood flow in pulmonary arteries. Ann Biomed Eng 35:546–559
Stack A, Derksen F, Williams K, Robinson N, Jackson W (2014) Lung region and racing affect mechanical properties of equine pulmonary microvasculature. J Appl Physiol 117:370–376
Stalder A, Russe M, Frydrychowicz A, Bock J, Hennig J, Markl M (2008) Quantitative 2d and 3d phase contrast mri: optimized analysis of blood flow and vessel wall parameters. Magnetic Reson Med 60:1218–1231
Stergiopulos N, Young D, Rogge T (1992) Computer simulation of arterial flow with applications to arterial and aortic stenoses. J Biomech 25:1477–1488
Tang B, Fonte T, Chan F, Tsao P, Feinstein J, Taylor C (2011) Three-dimensional hemodynamics in the human pulmonary arteries under resting and exercise conditions. Ann Biomed Eng 39:347–358
Tang B, Pickard S, Chan F, Tsao P, Taylor C, Feinstein J (2012) Wall shear stress is decreased in the pulmonary arteries of patients with pulmonary arterial hypertension: an image-based, computational fluid dynamics study. Pulm Circ 2:470–476
Townsley M (2012) Structure and composition of pulmonary arteries, capillaries and veins. Comprehen Physiol 2:675
Truong U, Fonseca B, Dunning J, Burgett S, Lanning C, Ivy D, Shandas R, Hunter K, Barker A (2013) Wall shear stress measured by phase contrast cardiovascular magnetic resonance in children and adolescents with pulmonary arterial hypertension. J Cardiovasc Magn Reson 15:1–9
van Duin R, Stam K, Cai Z, Uitterdijk A, Garcia-Alvarez A, Ibanez B, Danser A, Reiss I, Duncker D, Merkus D (2019) Transition from post-capillary pulmonary hypertension to combined pre-and post-capillary pulmonary hypertension in swine: a key role for endothelin. J Physiol 597:1157–1173
Van de Vosse F, Stergiopulos N (2011) Pulse wave propagation in the arterial tree. Ann Rev Fluid Mech 43:467–499
Wenger N, Boden W, Carabello B, Carney R, Cerqueira M, Criqul M (2010) Cardiovascular disability: updating the social security listings. Nat Acad Sci
Westerhof N, Bosman F, De Vries C, Noordergraaf A (1969) Analog studies of the human systemic arterial tree. J Biomech 2:121–143
Wiener F, Morkin E, Skalak R, Fishman A (1966) Wave propagation in the pulmonary circulation. Circ Res 19:834–850
Wieslander B, Ramos J, Ax M, Petersson J, Ugander M (2019) Supine, prone, right and left gravitational effects on human pulmonary circulation. J Cardiovasc Magn Reson 21:1–15
Wilson N, Ortiz A, Johnson A (2013) The vascular model repository: a public resource of medical imaging data and blood flow simulation results. J Med Dev 7:040923
Yang W, Dong M, Rabinovitch M, Chan FP, Marsden A, Feinstein J (2019) Evolution of hemodynamic forces in the pulmonary tree with progressively worsening pulmonary arterial hypertension in pediatric patients. Biomech Model Mechanobiol 18:779–796
Yen R, Foppiano L (1981) Elasticity of small pulmonary veins in the cat. J Biomech Eng 103:38–42
Zambrano B, McLean N, Zhao X, Tan JL, Zhong L, Figueroa C, Lee L, Baek S (2018) Image-based computational assessment of vascular wall mechanics and hemodynamics in pulmonary arterial hypertension patients. J Biomech 68:84–92
Zamir M, Budwig R (2002) Physics of pulsatile flow. Appl Mech Rev 55:B35–B35
Acknowledgements
This work was supported in part by the National Institute of Health (NIH-HLBI 5R01HL147590-02 and NIH-NIAID 1R01AI139085-01), the National Science Foundation (NSF-DMS 1615820) and the American Heart Association (AHA 19PRE34380459).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
As described in detail by Qureshi et al. (2014), for the arterial and venous networks we define a relation between pressure and flow at both ends of each vessel, and derive a boundary condition that matches pressure and flow at the terminal large arteries and veins. To do so, we compute the admittance of each structured tree, relating pressure and flow at the outlet of each large terminal artery to pressure and flow at the inlet to the corresponding large terminal vein. At each junction, we set up an admittance matrix satisfying continuity of pressure and conservation of volume flux. The total admittance is calculated by joining “junction” admittances in series and parallel as illustrated in Fig. 12.
Relationship between flow and pressure via admittance \(\mathbf {Y}\), \(\mathbf {Y}^{\Vert }\) and \(\mathbf {Y}^{\Leftrightarrow }\) for a single vessel (a), vessels connected in parallel (b), and vessels connected in series (c). Note that \(\mathbf {Y}\) is the admittance for a single vessel, \(\mathbf {Y}^S\) is the admittance for vessel S, and \(\mathbf {Y}^S\) is the admittance for vessel T. Here, the color blue represents deoxygenated arteries, and red represents oxygen-rich veins
Given the proximal and distal flow \(Q_{1}=Q(0, \omega )\) and \(Q_{2}=Q(L, \omega )\), the admittance matrix is obtained by relating the flow and pressure at the proximal, \(x=0,\) and distal, \(x=L,\) ends of a vessel of radius \(r_0\) yielding
where \(C_{L} \equiv \cos (\omega L / c), S_{L} \equiv \sin (\omega L / c),\) and
is the admittance matrix for any one artery or vein when \(\omega \ne 0.\) For \(\omega =0,\) , we have
and therefore
For two vessels (S and T) in parallel, continuity of pressure between the inlet and outlet and conservation of volume flux across the junction gives
where \(Q_{1}\) and \(Q_{2}\) denote the vessel inflow and outflow, \(P_1\) and \(P_2\) are the corresponding inlet and outlet pressure, and \(\mathbf {Y}^{\Vert }=\mathbf {Y}^{S}+\mathbf {Y}^{T}\). We denote \(\mathbf {Y}^{S}\) and \(\mathbf {Y}^{T}\) as the admittances for vessel S and T, respectively.
Similarly, for two vessels (S and T) connected in series flow and pressure are related by
where \(i=S, T\) and \(k=1,2\) with \(Y_{k l}\) being the components of the \(2 \times 2\) admittance matrix. Assuming that \(P=P_{2}^{S}=P_{1}^{T}\) and \(Q_{2}^{S}=-Q_{1}^{T}\) at the junction of two vessels in series, the system for the vessels is represented by
where
is the admittance matrix. The symbol \(\Leftrightarrow\) denotes that the vessels are joined in series.
To compute pressure and flow in the arterioles and venules, we first prescribe the pressure from the terminal large arteries or veins at the start of the structured tree, i.e. \(P^A(0,\omega )\) and \(P^V(0,\omega )\). The corresponding flows are calculated using the grand admittance,
The small vessel pressure and flow at \(x=L\) are determined by
for \(j=A,V\) and \(\omega \ne 0\), whereas the zeroth frequency solutions are
Rights and permissions
About this article
Cite this article
Bartolo, M.A., Qureshi, M.U., Colebank, M.J. et al. Numerical predictions of shear stress and cyclic stretch in pulmonary hypertension due to left heart failure. Biomech Model Mechanobiol 21, 363–381 (2022). https://doi.org/10.1007/s10237-021-01538-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-021-01538-1