Abstract
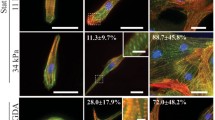
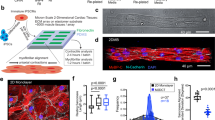
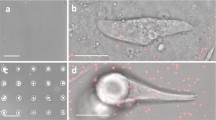
In this review, we discuss recent studies on the mechanosensitive morphology and function of cardiomyocytes derived from embryos and neonates. For early cardiomyocytes cultured on substrates of various stiffnesses, contractile function as measured by force production, work output and calcium handling is optimized when the culture substrate stiffness mimics that of the tissue from which the cells were obtained. This optimal contractile function corresponds to changes in sarcomeric protein conformation and organization that promote contractile ability. In light of current models for myofibillogenesis, a recent mathematical model of striation and alignment on elastic substrates helps to illuminate how substrate stiffness modulates early myofibril formation and organization. During embryonic heart formation and maturation, cardiac tissue mechanics change dynamically. Experiments and models highlighted here have important implications for understanding cardiomyocyte differentiation and function in development and perhaps in regeneration processes.
Similar content being viewed by others
References
Bajaj P, Tang X, Saif TA, Bashir R (2010) Stiffness of the substrate influences the phenotype of embryonic chicken cardiac myocytes. J Biomed Mater Res Part A 95(4): 1261–1269
Bers DM (2001) Excitation-contraction coupling and cardiac contractile force, 2nd ed. Kluwer, Dordrecht
Bhana B, Iyer RK, Chen WLK, Zhao R, Sider KL, Likhitpanichkul M, Simmons CA, Radisic M (2009) Influence of substrate stiffness on the phenotype of heart cells. Biotechnol Bioeng 105(6): 2151–2162
Cadete VJJ, Sawicka J, Polewicz D, Doroszko A, Wozniak M, Sawicki G (2010) Effects of the Rho kinase inhibitor Y-27632 on the proteome of hearts with ischemia-reperfusion injury. Proteomics 10(24): 4377–4385
Engler AJ, Carag-Krieger C, Johnson CP, Raab M, Tang H, Spelcher DW, Sanger JW, Sanger JM, Discher DE (2008) Embryonic cardiomyocytes beat best on a matrix with heart-like elasticity: scar-like rigidity inhibits beating. J Cell Sci 121: 3794–3802
Fila BA, Bayly PV, Taber LA (2010) Mechanical stress as a regulator of cytoskeletal contractility and nuclear shape in embryo epithelia. Ann Biomed Eng 39(1): 443–454
Friedrich BM, Safran SA (2012) How cells feel their substrate: spontaneous symmetry breaking of active surface stresses. Soft Matter 8: 3223–3230
Friedrich BM, Buxboim A, Discher DE, Safran SA (2011) Striated acto-myosin fibers can reorganize and register in response to elastic interactions with the matrix. Biophys J 100: 2706–2715
Gregorio CC, Antin PB (2000) To the heart of myofibril assembly. Trends Cell Biol 10(9): 355–362
Jacot JG, McCulloch AD, Omens JH (2008) Substrate stiffness affects the functional maturation of neonatal rat ventricular myocytes. Biophys J 95: 3479–3487
Jacot JG, Kita-Matsuo H, Wei KA, Chen HSV, Omens JH, Mercola M, McCulloch AD (2010) Cardiac myocyte force development during differentiation and maturation. Ann N Y Acad Sci 1188: 121–127
Johnson CP, Tang H, Carag C, Speicher DW, Discher DE (2007) Forced unfolding of proteins within cells. Science 317(5838): 663–666
Krieg M, Arboleda-Estudillo Y, Puech PH, Kafer J, Graner F, Muller DJ, Heisenberg CP (2008) Tensile forces govern germ-layer organization in zebrafish. Nat Cell Biol 10: 429–436
Lahmers S, Wu Y, Call DR, Labeit S, Granzier H (2004) Developmental control of titin isoform expression and passive stiffness in fetal and neonatal myocardium. Circ Res 94: 505–513
Liu J, Sun N, Bruce MA, Wu JC, Butte MJ (2012) Atomic force mechanobiology of pluripotent stem cell-derived cardiomyocytes. PLoS One 7(5): e37559
McCain ML, Parker KK (2011) Mechanotransduction: the role of mechanical stress, myocyte shape, and cytoskeletal architecture on cardiac function. Eur J Physiol 462: 89–104
McKenna NM, Johnson CS, Wang Y (1986) Formation and alignment of Z lines in living chick myotubes microinjected with rhodamine-labeled alpha-actinin. J Cell Biol 103(6): 2163–2171
Rodriguez AH, Han SJ, Regnier M, Sniadecki NJ (2011) Substrate stiffness increases twitch power of neonatal cardiomyocytes in correlation with changes in myofibril structure and intracellular calcium. Biophys J 101: 2455–2464
Sachinidis A, Fleischmann BK, Kolossov E, Wartenberg M, Sauer H, Hescheler J (2003) Cardiac specific differentiation of mouse embryonic stem cells. Cardiovasc Res 58: 278–291
Sanger JW, Kang S, Siebrands CC, Freeman N, Du A, Wang J, Stout AL, Sanger JM (2005) How to build a myofibril. J Muscle Res Cell Motil 26: 343–354
Sanger JW, Wang J, Fan Y, White J, Sanger JM (2010) Assembly and dynamics of myofibrils. J Biomed Biotechnol. doi:10.1155/2010/858606
Segers VFM, Lee RT (2008) Stem-cell therapy for cardiac disease. Nature 451: 937–942
von Dassow M, Davidson LA (2009) Natural variation in embryo mechanics: gastrulation in Xenopus laevis is highly robust to variation in tissue stiffness. Dev Dyn 238(1): 2–18
Young JL, Engler AJ (2011) Hydrogels with time-dependent material properties enhance cardiomyocyte differentiation in vitro. Biomaterials 32(4): 1002–1009
Zamir EA, Srinivasan V, Perucchio R, Taber LA (2003) Mechanical asymmetry in the embryonic chick heart during looping. Ann Biomech Eng 31: 1327–1336
Zwi L, Caspi O, Arbel G, Huber I, Gepstein A, Park IH, Gepstein L (2009) Cardiomyocyte differentiation of human induced pluripotent stem cells. Circulation 120: 1513–1523
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Majkut, S.F., Discher, D.E. Cardiomyocytes from late embryos and neonates do optimal work and striate best on substrates with tissue-level elasticity: metrics and mathematics. Biomech Model Mechanobiol 11, 1219–1225 (2012). https://doi.org/10.1007/s10237-012-0413-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-012-0413-8