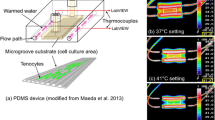
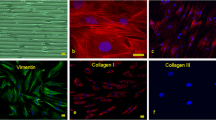
Abstract
Gap junction communication is an essential component in the mechanosensitive response of tenocytes. However, little is known about direct mechanoregulation of gap junction turnover and permeability. The present study tests the hypothesis that mechanical loading alters gap junction communication between tenocyte within tendon fascicles. Viable tenocytes within rat tail tendon fasicles were labelled with calcein-AM and subjected to a fluorescent loss induced by photobleaching (FLIP) protocol. A designated target cell within a row of tenocytes was continuously photobleached at 100% laser power whilst recording the fluorescent intensity of neighbouring cells. A mathematical compartment model was developed to estimate the intercellular communication between tenocytes based upon the experimental FLIP data. This produced a permeability parameter, k, which quantifies the degree of functioning gap functions between cells as confirmed by the complete inhibition of FLIP by the inhibitor 18α-glycyrrhentic acid. The application of 1N static tensile load for 10 min had no effect on gap junction communication. However, when loading was increased to 1 h, there was a statistically significant reduction in gap junction permeability. This coincided with suppression of connexin 43 protein expression in loaded samples as determined by confocal immunofluorescence. However, there was an upregulation of connexin 43 mRNA. These findings demonstrate that tenocytes remodel their gap junctions in response to alterations in mechanical loading with a complex mechanosensitive mechanism of breakdown and remodelling. This is therefore the first study to show that tenocyte gap junctions are not only important in transmitting mechanically activated signals but that mechanical loading directly regulates gap junction permeability.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Abbaci M, Barberi-Heyob M, Stines JR, Blondel W, Dumas D, Guillemin F, Didelon J (2007) Gap junctional intercellular communication capacity by gap-FRAP technique: a comparative study. Biotechnol J 2: 50–61
Abreu E, Leigh D, Derwin K (2008) Effects of altered mechanical load condition on the structure and function of cultured tendon fascicles. J Orthop Res 26: 364–373
Arnoczky SP, Tian T, Lavagnino M, Gardner K (2004) Ex vivo static tensile loading inhibits MMP-1 expression in rat tail tendons cells through a cytoskeletally based mechanotransduction mechanism. J Orthop Res 22: 328–333
Asundi K, Rempel D (2008) Cyclic loading inhibits expression of MMP-3 but not MMP-1 in an in vitro rabbit flexor tendon model. Clin Biomech (Bristol, Avon) 23: 117–121
Banes AJ, Horesovsky G, Tsuzaki M, Boitano S, Lawrence WT, Brown T, Weinhold P, Kenamond C, Benjamin M, Ralphs JR, McNeilly C, Burt J, Miller L (1999a) The connexin 43 gap junction is a mechanosensitive gene in avian flexor tendon cells. In: Caterson B, Archer C, Benjamin M, Ralphs J (eds) The biology of the synovial joint. Harwood Academic Publishers, Netherlands, pp 279–299
Banes AJ, Weinhold P, Yang X, Tsuzaki M, Bynum D, Bottlang M, Brown T (1999b) Gap junctions regulate responses of tendon cells ex vivo to mechanical loading. Clin Orthop Relat Res 367S: 356–370
DePaola N, Davies PF Jr, Pritchard WF, Florez L, Harbeck N, Polacek DC (1999) Spatial and temporal regulation of gap junction connexin 43 in vascular endothelial cells exposed to controlled disturbed flows in vitro. Proc Natl Acad Sci USA 96: 3154–3159
Eckert R (2006) Gap-junctional single-channel permeability for fluorescent tracers in mammalian cell cultures. Biophys J 91: 565–579
Flick J, Davkota A, Tsuzaki M, Almekinders L, Weinhold P (2006) Cyclic loading alters biomechanical properties and secretion of PGE2 and NO from tendon explants. Clin Biomech (Bristol, Avon) 21: 99–106
Garcia M, Knight MM (2010) Cyclic Loading Opens Hemichannels to Release ATP as Part of a Chondrocyte Mechanotransduction Pathway. J Orthop Res 28: 510–515
Goldberg GS, Lampe PD, Nicholson BJ (1999) Selective transfer of endogenous metabolites through gap junctions composed of different connexins. Nat Cell Biol 1: 457–459
Kannus P (2000) Structure of the tendon connective tissue. Scand J Med Sci Sports 10: 312–320
Lavagnino M, Arnoczky SP, Tian T, Vaupel Z (2003) Effect of amplitude and frequency of cyclic tensile strain on the inhibition of MMP-1 mRNA expression in tendon cells: an in vitro study. Connect Tissue Res 44: 181–187
Leigh DR, Abreu EL, Derwin KA (2008) Changes in gene expression of individual matrix metalloproteinases differ in response to mechanical unloading of tendon fascicles in explant culture. J Orthop Res 26: 1306–1312
Maeda E, Shelton JC, Bader DL, Lee DA (2007) Time dependence of cyclic tensile strain on collagen production in tendon fascicles. Biochem Biophys Res Commun 362: 399–404
Maeda E, Shelton JC, Bader DL, Lee DA (2009) Differential regulation of gene expression in isolated tendon fascicles exposed to cyclic tensile strain in vitro. J Appl Physiol 106: 506–512
Maeda E, Fleischmann C, Mein CA, Shelton JC, Bader DL, Lee DA (2010) Functional analysis of tenocytes gene expression in tendon fascicles subjected to cyclic tensile strain. Connect Tissue Res 51: 434–444
Marsolais D, Duchesne E, Cote CH, Frenette J (2007) Inflammatory cells do not decrease the ultimate tensile strength of intact tendons in vivo and in vitro: protective role of mechanical loading. J Appl Physiol 102: 11–17
McNeilly CM, Banes AJ, Benjamin M, Ralphs JR (1996) Tendon cells in vivo form a three dimensional network of cell process linked by gap junctions. J Anat 189: 593–600
Pan J, Zhou X, Li W, Novotny JE, Doty SB, Wang L (2009) In situ measurement of transport between subchondral bone and articular cartilage. J Orthop Res 27: 1347–1352
Screen HRC, Lee DA, Bader DL, Shelton JC (2004) An investigation into the effects of the hierarchical structure of tendon fascicles on micromechanical properties. Proc Inst Mech Eng H 218H: 109–119
Segretain D, Falk MM (2004) Regulation of connexin biosynthesis, assembly, gap junction formation, and removal. Biochim Biophys Acta 1662: 3–21
Wagett AD, Benjamin M, Ralphs JR (2006) Connexin 32 and 43 gap junctions differentially modulate tenocyte response to cyclic mechanical load. Eur J Cell Biol 85: 1145–1154
Wall ME, Banes AJ (2005) Early responses to mechanical load in tendon: role for calcium signaling, gap junctions and intercellular communication. J Musculoskelet Neuronal Interact 5: 70–84
Wang JHC (2006) Mechanobiology of tendon. J Biomech 39: 1563–1582
Yamamoto E, Iwanaga W, Miyazaki H, Hayashi K (2002) Effects of static stress on the mechanical properties of cultures collagen fascicles from the rabbit patellar tendon. J Biomech Eng 124: 85–93
Zhu LJ, Altmann S (2005) mRNA and 18S-RNA coapplication-reverse transcription for quantitative gene expression analysis. Anal Biochem 1: 102–109
Acknowledgments
The present study was funded by an the UK Engineering and Physical Science Research Council (EPSRC) Platform Grant (No. EP/E046975/1).
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
The Below is the Electronic Supplementary Material.
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Maeda, E., Ye, S., Wang, W. et al. Gap junction permeability between tenocytes within tendon fascicles is suppressed by tensile loading. Biomech Model Mechanobiol 11, 439–447 (2012). https://doi.org/10.1007/s10237-011-0323-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-011-0323-1