Abstract
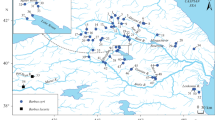
Mitochondrial DNA divergence among populations of the Japanese spinous loach Cobitis shikokuensis, endemic to Shikoku Island, was investigated by restricted fragment length polymorphism analysis. A total of 68 restriction sites on DNA fragments from the cytochrome b to D-loop regions and from the 12S rRNA to 16S rRNA regions, amplified by PCR, were analyzed. A total of 12 haplotypes (plus 6 in outgroups) were detected in 268 specimens collected from 19 localities in seven rivers (and 41 specimens from four localities in three rivers in outgroups). Three of the seven river populations of C. shikokuensis were shown to have unique haplotypes, and four of the seven river populations were monomorphic. The nested structure of the haplotype network for populations of C. shikokuensis exhibited two large clades corresponding to (1) populations from the Shimanto River and its neighbors and (2) two genetically divergent populations in the Shigenobu and Iwamatsu Rivers. The population from the Shimanto River, the largest river inhabited by C. shikokuensis, maintains great haplotype diversity as well as the allozyme diversity previously reported. On the other hand, populations from the Hiji River, the second largest river, which exhibited the highest allozyme diversity, were monomorphic in their mtDNA. The nested clade analysis (NCA) revealed that past fragmentation between the above two clades could occur in the initial distribution process of C. shikokuensis. The large genetic divergence of two river populations from the Shigenobu and Iwamatsu Rivers was inferred to be caused by a process of long distance colonization and fragmentation. MtDNA introgression into the Hiji River population from southern river populations was suggested. Taking genetic divergence into consideration, each river population of C. shikokuensis should be conserved separately as like a distinct species, and conservation programs for the small populations showing less genetic variability should be invoked as soon as possible.




Similar content being viewed by others
References
Aoyagi H (1957) General notes on the freshwater fishes of the Japanese Archipelago. Taishunkan, Tokyo
Avise JC (1994) Molecular markers, natural history and evolution. Chapman & Hall, New York
Crandall KA, Bininda-Emonds ORP, Mace GM, Wayne RK (2000) Considering evolutionary processes in conservation biology. TREE 15:290–295
Efron B (1979) Bootstrap method: Another look at the jacknife. Ann Stat 7:1–26
Gaggiotti OE, Hanski I (2004) Mechanisms of population extinction. In: Hanski I, Gaggiotti OE (eds) Ecology, genetics, and evolution of metapopulations. Elsevier Acad Press, Burlington, pp 337–366
Inoue JG, Miya M, Tsukamoto K, Nishida M (2000) Complete mitochondrial DNA sequence of the Japanese sardine, Sardinops melanostictus. Fish Sci 66:924–932
Kimizuka Y (1987) Cobitidae—Zoogeography of the karyological races. In: Mizuno N, Goto A (eds) Freshwater fishes in Japan. Tokai University Press, Tokyo, pp 61–70
Kimizuka Y, Kobayashi H, Mizuno N (1982) Geographic distributions and karyotypes of Cobitis takatsuensis and Niwaella delicata (Cobitididae). Jpn J Ichthyol 29:305–310
Kitagawa T, Watanabe M, Kobayashi T, Yoshioka M, Kashiwagi M, Okazaki T (2001) Two genetically divergent groups in the Japanese spineds loach, Cobitis takatsuensis, and their phylogenetic relationships among Japanese Cobitis inferred from mitochondrial DNA analysis. Zool Sci 18:249–259
Kitagawa T, Watanabe M, Kitagawa E, Yoshioka M, Kashiwagi M, Okazaki T (2003) Phylogeography and maternal origin of the tetraploid form of the Japanese spined loach, Cobitis biwae, revealed by mitochondrial DNA analysis. Ichthyol Res 50:318–325
McElroy D, Moran P, Bermingham E, Kornfield I (1992) REAP: an integrated environment for the manipulation and phylogenetic analysis of restriction data. J Hered 83:157–158
Moritz C (1994) Defining “Evolutionary Significant Units” for conservation. TREE 9:373–375
Nei M (1987) Molecular evolutionary genetics. Columbia Univ Press, New York
Nei M, Tajima F (1981) DNA polymorphism detectable by restriction endonucleases. Genetics 97:145–163
Nishida M, Ohkawa T, Iwata H (1998) Methods of analysis of genetic population structure with mitochondrial DNA markers. Fish Genet Breed Sci 26:81–100
Ochiai A, Fujita S, Hanzawa N (1981) On a rare striped loach closely related to Cobitis takatsuensis from Shimanto river, Kochi Prefecture. Res Rep Kochi Univ 30:47–52
Posada D, Crandall KA, Templeton AR (2000) GEODIS: a program for the cladistic nested analysis of the geographical distribution of genetic haplotypes. Mol Ecol 9:487–488
Roff DA, Bentzen P (1989) The statistical analysis of mitochondrial DNA polymorphisms: χ2 and the problem of small samples. Mol Biol Evol 6:539–545
Ryder OA (1986) Species conservation and systematics: the dilemma of subspecies. TREE 1:9–10
Saitoh K (1989) Sujishima-dojyo agun. In: Kawanabe H, Mizuno N (eds) Freshwater fishes of Japan. Yama-kei Publishers, Tokyo, pp 386–391
Shimizu T (2002) Life history of a Japanese spinous loach, Cobitis takatsuensis, in Shikoku Island. Jpn J Ichthyol 49:33–40
Shimizu T (2003a) Cobitis takatsuensis: Cypriniformes, Cobitidae (EN). In: Ministry of Environment (ed) Threatened wildlife of Japan –Red data book, vol 4, 2nd edn, Pisces—Brackish and freshwater fishes. Japan Wildlife Research Center, Tokyo, pp 104–105
Shimizu T (2003b) Geographic variation of the Japanese spinous loach, Cobitis takatsuensis, inferred from allozyme analysis. Folia Biol (Kraków) 51(Suppl):85–92
Shimizu T (2003c) Present status of Cobitis takatsuensis in Ehime Prefecture, Japan. Jpn J Ichthyol 50:153–158
Shimizu T, Mizuno N (1998) Cobitis takatsuensis. In: Fisheries Agency of Japan (ed) Data book on the rare aquatic species of Japan. Japan Fisheries Resources Conservation Association, Tokyo, pp 150–151
Shimizu T, Suzawa Y, Sakai H (2004) Allozyme divergence between two groups of the Japanese spinous loach, Cobitis takatsuensis. Ichthyol Res 51:241–247
Suzawa Y (2006) A new species of loach, Cobitis shikokuensis (Teleostei: Cobitidae), from Shikoku Island, Japan. Ichthyol Res 53:315–322
Swofford DL (1998) PAUP*: Phylogenetic analysis using parsimony (*and other methods). Version 4. Sinauer Associates, Sunderland, Massachusetts
Takahashi H (2002) Cobitis sp. 1. In: Editorial board of red data book Kochi (ed) Red data book Kochi (animal edition) –Threatened wildlife of Kochi Prefecture. Kochi Prefecture, pp 184–185
Templeton AR (1998) Nested clade analysis of phylogeographic data: testing hypotheses about gene flow and population history. Mol Ecol 7:381–397
Templeton AR, Sing CF (1993) A cladistic analysis of phenotypic associations with haplotypes inferred from restriction endonuclease mapping. IV. Nested analysis with cladogram uncertainty and recombination. Genetics 134:659–669
Umitsu M (1995) Setouchi no minami ni sobieru kewashii yamayama – Shikoku no San-chi. In: Nakamura K, Andoh H, Miyata K, Hori N, Umitsu M, Shinmi O (eds) Nihon no Shizen 6 Chyugoku Shikoku. Iwanamishoten, Tokyo, pp 146–162
Watanabe K (1998) Parsimony analysis of the distribution pattern of Japanese primary freshwater fishes, and its application to the distribution of the bagrid catfishes. Ichthyol Res 45:259–270
Acknowledgments
I am grateful to Dr. H. Sakai (National Fisheries University) for valuable advice and critical reading of the manuscript. Special thanks are given to Drs. M. Nishida (University of Tokyo), K. Watanabe (Kyoto University), and A. Kawaguchi (University of Ryukyus) for teaching me the techniques of mtDNA analysis, and to Dr. Margaret Dooley Ohto for checking the English. Thanks also go to Mr. Y. Suzawa (Institute of River Biology), M. Okamoto (Ecosystem Co., Ltd.), H. Takahashi (Sumiko Techno Research Incorporated), and K. Hashimoto (Osaka Sekkei Co., Ltd.) for collecting specimens. The present study was supported in part by a consignment fund from the Fisheries Agency, Japan, to develop techniques for reproducing endangered freshwater fishes.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Shimizu, T. Geographic differentiation of Cobitis shikokuensis inferred from mtDNA RFLP analysis. Ichthyol Res 55, 101–111 (2008). https://doi.org/10.1007/s10228-007-0013-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10228-007-0013-2