Abstract
Background
Women with a BRCA1 or BRCA2 mutation are at increased risk of developing breast and/or ovarian cancer. This economic modeling study evaluated different preventive interventions for 30-year-old women with a confirmed BRCA (1 or 2) mutation.
Methods
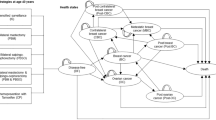
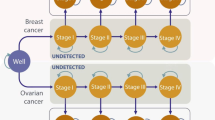
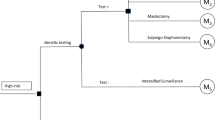
A Markov model was developed to estimate the costs and benefits [i.e., quality-adjusted life years (QALYs), and life years gained (LYG)] associated with prophylactic bilateral mastectomy (BM), prophylactic bilateral salpingo-oophorectomy (BSO), BM plus BSO, BM plus BSO at age 40, and intensified surveillance. Relevant input data was obtained from a large German database including 5902 women with BRCA 1 or 2, and from the literature. The analysis was performed from the German Statutory Health Insurance (SHI) perspective. In order to assess the robustness of the results, deterministic and probabilistic sensitivity analyses were performed.
Results
With costs of €29,434 and a gain in QALYs of 17.7 (LYG 19.9), BM plus BSO at age 30 was less expensive and more effective than the other strategies, followed by BM plus BSO at age 40. Women who were offered the surveillance strategy had the highest costs at the lowest gain in QALYs/LYS. In the probabilistic sensitivity analysis, the probability of cost-saving was 57% for BM plus BSO. At a WTP of 10,000 € per QALY, the probability of the intervention being cost-effective was 80%.
Conclusions
From the SHI perspective, undergoing BM plus immediate BSO should be recommended to BRCA 1 or 2 mutation carriers due to its favorable comparative cost-effectiveness.

Similar content being viewed by others
References
Global Burden of Disease Cancer Collaboration: The global burden of cancer 2013. JAMA Oncol 1(4), 505–527 (2015)
Robert-Koch Institut, Zentrum für Krebsregisterdaten: Brustkrebs (Mammakarzinom). http://www.krebsdaten.de/Krebs/DE/Content/Krebsarten/Brustkrebs/brustkrebs_node.html (2015). Accessed 28 Apr2016
Rhiem, K., Engel, C., Graeser, M., et al.: The risk of contralateral breast cancer in patients from BRCA1/2 negative high risk families as compared to patients from BRCA1 or BRCA2 positive families: a retrospective cohort study. Breast Cancer Res. 14(6), R156 (2012). doi:10.1186/bcr3369
Luengo-Fernandez, R., Leal, J., Gray, A., et al.: Economic burden of cancer across the European Union: a population-based cost analysis. Lancet Oncol 14(12), 1165–1174 (2013). doi:10.1016/S1470-2045(13)70442-X
Willems, R.A., Bolman, C.A., Mesters, I., et al.: Cancer survivors in the first year after treatment: the prevalence and correlates of unmet needs in different domains. Psycho-oncology (2015). doi:10.1002/pon.3870
Sorensen, S.V., Goh, J.W., Pan, F., et al.: Incidence-based cost-of-illness model for metastatic breast cancer in the United States. Int. J. Technol. Assess. Health Care 28(1), 12–21 (2012). doi:10.1017/S026646231100064X
Foster, T.S., Miller, J.D., Boye, M.E., et al.: The economic burden of metastatic breast cancer: a systematic review of literature from developed countries. Cancer Treat. Rev. 37(6), 405–415 (2011). doi:10.1016/j.ctrv.2010.12.008
Cohn, D.E., Kim, K.H., Resnick, K.E., et al.: At what cost does a potential survival advantage of bevacizumab make sense for the primary treatment of ovarian cancer? A cost-effectiveness analysis. J Clin Oncol 29(10), 1247–1251 (2011). doi:10.1200/JCO.2010.32.1075
Familial Breast Cancer: Classification and Care of People at Risk of Familial Breast Cancer and Management of Breast Cancer and Related Risks in People with a Family History of Breast Cancer. National Institute for Health and Clinical Excellence: Guidance. Cardiff (UK) (2013)
Leitlinienprogramm Onkologie (Deutsche Krebsgesellschaft, Deutsche Krebshilfe, AWMF): Interdisziplinäre S3-Leitlinie für die Diagnostik, Therapie und Nachsorge des Mammakarzinoms, Langversion 3.0, Aktualisierung 2012, AWMF-Register-Nummer: 032–045OL
Nelson, H.D., Pappas, M., Zakher, B., et al.: Risk assessment, genetic counseling, and genetic testing for BRCA-related cancer in women: a systematic review to update the U.S. Preventive Services Task Force recommendation. Ann. Intern. Med. 160(4), 255–266 (2014). doi:10.7326/M13-1684
Phillips, K.A., Milne, R.L., Rookus, M.A., et al.: Tamoxifen and risk of contralateral breast cancer for BRCA1 and BRCA2 mutation carriers. J. Clin. Oncol. 31(25), 3091–3099 (2013). doi:10.1200/JCO.2012.47.8313
Grann, V.R., Patel, P.R., Jacobson, J.S., et al.: Comparative effectiveness of screening and prevention strategies among BRCA1/2-affected mutation carriers. Breast Cancer Res. Treat. 125(3), 837–847 (2011). doi:10.1007/s10549-010-1043-4
Anderson, K., Jacobson, J.S., Heitjan, D.F., et al.: Cost-effectiveness of preventive strategies for women with a BRCA1 or a BRCA2 mutation. Ann. Intern. Med. 144(6), 397–406 (2006)
Norum, J., Hagen, A.I., Maehle, L., et al.: Prophylactic bilateral salpingo-oophorectomy (PBSO) with or without prophylactic bilateral mastectomy (PBM) or no intervention in BRCA1 mutation carriers: a cost-effectiveness analysis. Eur. J. Cancer 44(7), 963–971 (2008). doi:10.1016/j.ejca.2008.02.025
Institut für Qualität und Wirtschaftlichkeit im Gesundheitswesen: Allgmeine Methoden. Version 4.2 vom 22.04.2015. https://www.iqwig.de/download/IQWiG_Methoden_Version_4-2.pdf (2015). Accessed 28 Apr 2016
Schad, M., John, J.: Towards a social discount rate for the economic evaluation of health technologies in Germany: an exploratory analysis. Eur. J. Health Econ 13(2), 127–144 (2012). doi:10.1007/s10198-010-0292-9
Chen, T., Jansen, L., Gondos, A., et al.: Survival of ovarian cancer patients in Germany in the early 21st century: a period analysis by age, histology, laterality, and stage. Eur. J. Cancer Prev. 22(1), 59–67 (2013). doi:10.1097/CEJ.0b013e3283552e28
Holleczek, B., Brenner, H.: Trends of population-based breast cancer survival in Germany and the US: decreasing discrepancies, but persistent survival gap of elderly patients in Germany. BMC Cancer 12, 317 (2012). doi:10.1186/1471-2407-12-317
German Consortium for Hereditary Breast and Ovarian Cancer. http://www.krebshilfe.de/wir-helfen/adressen/familiaerer-krebs/brustkrebszentren.html (2016). Accessed 28 Apr 2016
The Cochrane Collaboration. Cochrane Handbook for Systematic Reviews of Interventions. Chapter 13: Including non-randomized studies. http://handbook.cochrane.org/chapter_13/13_including_non_randomized_studies.htm (2011). Accessed 31 Jan 2017
Land, L.H., Dalton, S.O.: Jensen, et al.: impact of comorbidity on mortality: a cohort study of 62,591 Danish women diagnosed with early breast cancer, 1990–2008. Breast Cancer Res. Treat. 131(3), 1013–1020 (2012). doi:10.1007/s10549-011-1819-1
Rebbeck, T.R., Lynch, H.T., Neuhausen, S.L., et al.: Prophylactic oophorectomy in carriers of BRCA1 or BRCA2 mutations. N. Engl. J. Med. 346(21), 1616–1622 (2002). doi:10.1056/NEJMoa012158
Domchek, S.M., Friebel, T.M., Singer, C.F., et al.: Association of risk-reducing surgery in BRCA1 or BRCA2 mutation carriers with cancer risk and mortality. JAMA 304(9), 967–975 (2010). doi:10.1001/jama.2010.1237
Mavaddat, N., Peock, S., Frost, D., et al.: Cancer risks for BRCA1 and BRCA2 mutation carriers: results from prospective analysis of EMBRACE. J. Natl. Cancer Inst. 105(11), 812–822 (2013). doi:10.1093/jnci/djt095
Boughey, J.C., Hoskin, T.L., Degnim, A.C., et al.: Contralateral prophylactic mastectomy is associated with a survival advantage in high-risk women with a personal history of breast cancer. Ann. Surg. Oncol. 17(10), 2702–2709 (2010). doi:10.1245/s10434-010-1136-7
Grann, V.R., Jacobson, J.S., Sundararajan, V., et al.: The quality of life associated with prophylactic treatments for women with BRCA1/2 mutations. Cancer J Sci Am 5(5), 283–292 (1999)
Grann, V.R., Patel, P., Bharthuar, A., et al.: Breast cancer-related preferences among women with and without BRCA mutations. Breast Cancer Res. Treat. 119(1), 177–184 (2010). doi:10.1007/s10549-009-0373-6
Peasgood, T., Ward, S.E., Brazier, J.: Health-state utility values in breast cancer. Expert Rev Pharmacoecon Outcomes Res 10(5), 553–566 (2010). doi:10.1586/erp.10.65
Havrilesky, L.J., Broadwater, G., Davis, D.M., et al.: Determination of quality of life-related utilities for health states relevant to ovarian cancer diagnosis and treatment. Gynecol. Oncol. 113(2), 216–220 (2009). doi:10.1016/j.ygyno.2008.12.026
Ara, R., Wailoo, A.: Using health state utility values in models exploring the cost-effectiveness of health technologies. Value Health 15(6), 971–974 (2012). doi:10.1016/j.jval.2012.05.003
Federal Statistical Office of Germany: Life expectancy 2015. https://www.destatis.de/EN/FactsFigures/SocietyState/Population/Deaths/Deaths.html (2015). Accessed 28 Apr 2016
Metcalfe, K., Lynch, H.T., Ghadirian, P., et al.: Risk of ipsilateral breast cancer in BRCA1 and BRCA2 mutation carriers. Breast Cancer Res. Treat. 127(1), 287–296 (2011). doi:10.1007/s10549-010-1336-7
Litiere, S., Werutsky, G., Fentiman, I.S., et al.: Breast conserving therapy versus mastectomy for stage I-II breast cancer: 20 year follow-up of the EORTC 10801 phase 3 randomised trial. Lancet Oncol. 13(4), 412–419 (2012). doi:10.1016/S1470-2045(12)70042-6
Winter, W.E., Maxwell, G.L., Tian, C., Gynecologic Oncology Group Study, et al.: Prognostic factors for stage III epithelial ovarian cancer: a Gynecologic Oncology Group study. J. Clin. Oncol. 25(24), 3621–3627 (2007)
Rebbeck, T.R., Friebel, T., Lynch, H.T., et al.: Bilateral prophylactic mastectomy reduces breast cancer risk in BRCA1 and BRCA2 mutation carriers: the PROSE Study Group. J. Clin. Oncol. 22(6), 1055–1062 (2004). doi:10.1200/JCO.2004.04.188
Sullivan, P.W., Lawrence, W.F., Ghushchyan, V.: A national catalog of preference-based scores for chronic conditions in the United States. Med. Care 43(7), 736–749 (2005)
Dawood, S.: Triple-negative breast cancer: epidemiology and management options. Drugs 70(17), 2247–2258 (2010). doi:10.2165/11538150-000000000-00000
Holleczek, B., Jansen, L., Brenner, H.: Breast cancer survival in Germany: a population-based high resolution study from Saarland. PLoS One 8(7), e70680 (2013). doi:10.1371/journal.pone.0070680
Hess, K.R., Esteva, F.J.: Effect of HER2 status on distant recurrence in early stage breast cancer. Breast Cancer Res. Treat. 137(2), 449–455 (2013). doi:10.1007/s10549-012-2366-0
Inwald, E.C., Ortmann, O., Zeman, F., et al.: Guideline concordant therapy prolongs survival in HER2-positive breast cancer patients: results from a large population-based cohort of a cancer registry. Biomed. Res. Int. 2014, 137304 (2014). doi:10.1155/2014/137304
DeKoven, M., Bonthapally, V., Jiao, X., et al.: Treatment pattern by hormone receptors and HER2 status in patients with metastatic breast cancer in the UK, Germany, France, Spain and Italy (EU-5): results from a physician survey. J Comp Eff Res 1(5), 453–463 (2012). doi:10.2217/cer.12.43
Dufresne, A., Pivot, X., Tournigand, C., et al.: Impact of chemotherapy beyond the first line in patients with metastatic breast cancer. Breast Cancer Res. Treat. 107(2), 275–279 (2008). doi:10.1007/s10549-007-9550-7
Martin, L.P., Schilder, R.J.: Management of recurrent ovarian carcinoma: current status and future directions. Semin. Oncol. 36(2), 112–125 (2009). doi:10.1053/j.seminoncol.2008.12.003
Modesitt, S.C., Jazaeri, A.A.: Recurrent epithelial ovarian cancer: pharmacotherapy and novel therapeutics. Expert Opin. Pharmacother. 8(14), 2293–2305 (2007). doi:10.1517/14656566.8.14.2293
Lux, M.P., Reichelt, C., Wallwiener, D., et al.: Results of the Zometa cost-utility model for the German healthcare system based on the results of the ABCSG-12 study. Onkologie 33(7), 360–368 (2010). doi:10.1159/000315699
Universitätsklinikum Münster DRG Research Group Medizinisches Management: Webgrouper. http://drg.uni-muenster.de/index.php?option=com_webgrouper&view=webgrouper&Itemid=26 (2015). Accessed 28 Apr 2016
Rote Liste® Service GmbH: Rote Liste. http://www.rote-liste.de/ (2015). Accessed 28 Apr 2016
Kassenärztliche Bundesvereinigung: Uniform value scale. http://www.kbv.de/html/ebm.php (2015). Accessed 28 Apr 2016
Arican, A., Bozkurt, T., Bozcuk, H., et al.: A cross-sectional survey of the diagnosis and management of bone metastasis in breast cancer patients in Turkey. Support. Care Cancer 22(10), 2629–2634 (2014). doi:10.1007/s00520-014-2253-9
Verband der Ersatzkassen: Rahmenverträge und Vergütungslisten bei Heilmitteln.. http://www.vdek.com/vertragspartner/heilmittel/rahmenvertrag.html (2015). Accessed 28 Apr 2016
IntelliMed GmbH.:Catalog of therapeutic products. http://www.heilmittelkatalog.de/heilmittelkatalog-online.html (2014). Accessed 28 Apr 2016
Statistisches Bundesamt. GENESIS-Online Datenbank. https://www-genesis.destatis.de/genesis/online;jsessionid=131E6809880313E767FC8E952532F25D.tomcat_GO_1_3?operation=previous&levelindex=2&levelid=1479294919749&step=2 (2016). Accessed 31 Jan 2017
Fryback, D.G., Chinnis Jr., J.O., Ulvila, J.W.: Bayesian cost-effectiveness analysis. An example using the GUSTO trial. Int. J. Technol. Assess. Health Care 17(1), 83–97 (2001)
Stinnett, A.A., Mullahy, J.: Net health benefits: a new framework for the analysis of uncertainty in cost-effectiveness analysis. Med. Decis. Making 18(2 Suppl), S68–S80 (1998)
Geiger, A.M., Nekhlyudov, L., Herrinton, L.J., et al.: Quality of life after bilateral prophylactic mastectomy. Ann. Surg. Oncol. 14(2), 686–694 (2007). doi:10.1245/s10434-006-9206-6
Rebbeck, T.R., Mitra, N., Wan, F., et al.: Association of type and location of BRCA1 and BRCA2 mutations with risk of breast and ovarian cancer. JAMA 313(13), 1347–1361 (2015). doi:10.1001/jama.2014.5985
Chai, X., Friebel, T.M., Singer, C.F., et al.: Use of risk-reducing surgeries in a prospective cohort of 1,499 BRCA1 and BRCA2 mutation carriers. Breast Cancer Res. Treat. 148(2), 397–406 (2014). doi:10.1007/s10549-014-3134-0
Cappelli, M., Surh, L., Humphreys, L., et al.: Measuring women’s preferences for breast cancer treatments and BRCA1/BRCA2 testing. Qual. Life Res. 10(7), 595–607 (2001)
Hildebrandt, T., Thiel, F.C., Fasching, P.A., et al.: Health utilities in gynecological oncology and mastology in Germany. Anticancer Res. 34(2), 829–835 (2014)
Parkin, D., Devlin, N.: Is there a case for using visual analogue scale valuations in cost-utility analysis? Health Econ. 15(7), 653–664 (2006). doi:10.1002/hec.1086
Greiner, W., Claes, C., Busschbach, J.J., von der Schulenburg, J.M.: Validating the EQ-5D with time trade off for the German population. Eur. J. Health Econ. 6(2), 124–130 (2005)
Chan, Y.M., Ngan, H.Y., Li, B.Y., et al.: A longitudinal study on quality of life after gynecologic cancer treatment. Gynecol. Oncol. 83(1), 10–19 (2001). doi:10.1006/gyno.2001.6345
den Heijer, M., Seynaeve, C., Timman, R., et al.: Body image and psychological distress after prophylactic mastectomy and breast reconstruction in genetically predisposed women: a prospective long-term follow-up study. Eur. J. Cancer 48(9), 1263–1268 (2012). doi:10.1016/j.ejca.2011.10.020
den Heijer, M., Seynaeve, C., Vanheusden, K., et al.: Long-term psychological distress in women at risk for hereditary breast cancer adhering to regular surveillance: a risk profile. Psycho-Oncology 22(3), 598–604 (2013). doi:10.1002/pon.3039
Hooker, G.W., King, L., Vanhusen, L., et al.: Long-term satisfaction and quality of life following risk reducing surgery in BRCA1/2 mutation carriers. Hered Cancer Clin Pract 12(1), 9 (2014). doi:10.1186/1897-4287-12-9
Author information
Authors and Affiliations
Corresponding author
Additional information
Statement
The analysis was part of the More-Risk Study, an evaluation of the economical, legal, ethical and risk-communicative implications of a risk-adapted screening strategy to detect mamma- and ovarian cancer. The More-Risk Study was funded by the “Federal Ministry of Education and Research” (NKP-332-019).
Authors S. Stock and K. Rhiem are contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Müller, D., Danner, M., Rhiem, K. et al. Cost-effectiveness of different strategies to prevent breast and ovarian cancer in German women with a BRCA 1 or 2 mutation. Eur J Health Econ 19, 341–353 (2018). https://doi.org/10.1007/s10198-017-0887-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10198-017-0887-5