Abstract
The aim of the study was to evaluate the association between sleep disturbance and headache type and frequency, in a random sample of participants in the third Nord-Trøndelag Health Survey. The headache diagnoses were set by neurologists using the ICHD-2 criteria performing a semi structured face-to-face interview. Sleep problems were measured by the two validated instruments Karolinska Sleep Questionnaire (KSQ) and Epworth Sleepiness Scale (ESS). Among 297 participants, 77 subjects were headache-free, whereas 135 were diagnosed with tension-type headache (TTH), 51 with migraine, and 34 with other headache diagnoses. In the multivariate analyses, using logistic regression, excessive daytime sleepiness, defined as ESS ≥ 10, was three times more likely among migraineurs compared with headache-free individuals (OR = 3.3, 95% CI 1.0–10.2). Severe sleep disturbances, defined as KSQ score in the upper quartile, was five times more likely among migraineurs (OR = 5.4, 95% CI 2.0–15.5), and three times more likely for subjects with TTH (OR = 3.3, 1.4–7.3) compared with headache-free individuals. Subjects with chronic headache were 17 times more likely to have severe sleep disturbances (OR = 17.4, 95% CI 5.1–59.8), and the association was somewhat stronger for chronic migraine (OR = 38.9, 95% CI 3.1–485.3) than for chronic TTH (OR = 18.3, 95% CI 3.6–93.0). In conclusion, there was a significant association between severe sleep disturbances and primary headache disorders, most pronounced for those with chronic headache. Even though one cannot address causality in the present study design, the results indicate an increased awareness of sleep problems among patients with headache.
Similar content being viewed by others
Introduction
Headache and sleep disturbances are both common in the general population [1–3]. Sleep-related symptoms are associated with painful conditions [4], partly because pain may interfere with sleep and vice versa [5]. This dual cause and effect relationship has been known for many years through clinical experience with headache patients [6, 7], and the scientific literature about the comorbidity of headache and sleep disturbances are growing [8].
Many epidemiological studies have evaluated this association in relation to nonspecific headache diagnoses (e.g. headache, morning headache, chronic daily headache) [9], whereas relatively few epidemiological studies have focused on the specific diagnosis of tension-type headache (TTH) [10, 11]. TTH is associated with sleep disorders [12], and subjects with TTH often report of sleep complaints related to insomnia [10, 13].
The impact of sleep complaints in migraineurs is more extensively studied [1]. Migraine is associated with several sleep problems, and this observation is supported by polysomnographic findings [8]. Also, excess of sleep [14], insufficient sleep duration, and poor sleep quality are common triggering factors for migraine attacks [15].
Although many studies have looked at the relationship between headache and sleep disturbances, they have used varying study methodologies. Most studies have been cross-sectional [9, 10, 14, 16–19], but there have also been some case-series [20–24], some with case–control design [13, 25–27], and some prospective studies [11, 28, 29]. However, the existing epidemiologic literature tends to lack diagnostic precision in the classification of headache [8]. Relatively few studies have used face-to-face interviews by a neurologist [10, 30]. Similarly, few previous studies have measured sleep-related symptoms by validated instruments like Karolinska Sleep Questionnaire (KSQ) and Epworth Sleepiness Scale (ESS), both with a good correlation to objective sleep measurements [4, 31–33].
The main object of the present study was to evaluate the association between sleep disturbance and headache type and frequency, in a random sample of participants in a large population-based survey.
Materials and methods
The third Nord-Trøndelag Health Survey (HUNT 3)
Nord-Trøndelag is one of 19 Norwegian counties, located in the central part of Norway [34]. The geography, demography, and inhabitants in Nord-Trøndelag County are fairly representative of Norway, which makes it well suited for a survey like HUNT [35]. There are, however, no cities with more than 25,000 residents [34], and the income and education level among its inhabitants is just below average [35]. The third HUNT study (HUNT = Helseundersøkelsen i Nord-Trøndelag) was carried out between October 2006 and June 2008 [30]. In HUNT 3 all inhabitants >13 years in the Nord-Trøndelag County were invited to participate. Among 94,194 invited adults aged 20 years or more, 50,839 (54%) responded to the first questionnaire that was enclosed with the invitation letter.
Study population of the present study
The method of the present study has been described elsewhere [30], mainly designed to validate questionnaire-based headache diagnoses. A total of eight neurologists performed a semi-structured interview including questions about headache, alcohol consumption, musculoskeletal pain, and sleep-related questions. All invited subjects had previously answered two different large questionnaires in the HUNT 3 study, also covering these topics, but their responses to the questions were unknown for all involved in this study.
Nord-Trøndelag County was divided into 25 study areas by the HUNT study administration. The main survey was performed in two of these study areas, Verdal (14,000 inhabitants) and Stjørdal (20,600 inhabitants), from September to November 2007, and from December 2007 to April 2008, respectively. The present study was performed in December 2007 in Verdal and in February 2008 in Stjørdal. The participation rates in the HUNT 3 study were 52% of the whole adult population in Verdal and 50% in Stjørdal.
Invitation letters were sent to a random sample from a list of participants in Verdal and Stjørdal (n = 563). The potential participants were selected from the list consecutively in following order: men ≤50 years, men >50 years, women ≤50 years, and women >50 years, to ensure acceptable balanced participation for both genders in all age groups. In the invitation letter they were informed that they would be contacted on telephone by our research assistants to give further information and to make an appointment for the personal interview. The list of participants in Verdal and Stjørdal were received shortly before the time of the interview. Hence, due to lack of time, in case the research assistants were unable to get in contact despite a minimum of two attempts, they were instructed to call the next person on the list.
Headache
The subjects were first asked the following questions:“Have you suffered from headache during the last 12 months?” and “Have you had a headache during the last 12 months?” Subjects who answered yes to the latter question were asked about frequency, intensity, location, aura symptoms, other migraine or cluster headache features, and use of medication. All diagnoses except for medication overuse headache (MOH) were based on ICHD-2 [36]. For MOH the revised version was used [37]. Up to three different headache types were diagnosed in each individual. Subjects who met all, except one, of the diagnostic criteria for migraine and TTH were diagnosed with respectively probable migraine and probable TTH [36].
Sleep related complaints
Karolinska Sleep questionnaire
The first part consisted of a modified short form of the Karolinska Sleep questionnaire (KSQ) and three additional questions about morning headache, after dinner nap and troublesome sweating at night. The KSQ involved eight items which included the following topics: snoring, apnea, insomnia, daytime sleepiness, and restless legs syndrome. Question 1 (snore highly and inconveniently) and question 2 (apnea) were also merged and categorized as “snoring and/or apnea”. Question 3 (difficulty falling asleep), question 4 (many awakenings during the night), and question 5 (too early awakening) were also merged and categorized as “insomnia”. Question 6 (daytime sleepiness) and 10 (very sleepy after awakening) were merged into the category “daytime sleepiness”.
The KSQ is a validated instrument [32] that has been shown to correlate well with the Karolinska Sleep Diary (KSD), which earlier has been found to correlate well with polysomnographic data [31]. Items from the KSQ have been used to measure «disturbed sleep» in other studies [32, 38–42]. The response alternatives to the KSQ were 0 = never, 1 = seldom (some times a year, <one time a month), 2 = sometimes (a few times a month/max one time a week), 3 = several times a week (>once a week), 4 = always.
Each of the eight different KSQ questions were dichotomized and evaluated separately, considering a score of ≥3 as clinically relevant [39, 42]. In addition, we also computed a disturbed sleep index by summing the responses to the eight questions in the KSQ. We defined subjects with a score in the highest quartile to have a severe sleep disturbance. Other studies have used a similar approach, computing sleep index scores from selected KSQ items, where a high score indicated poor sleep quality [40], disturbed sleep [32, 38–42], awakening difficulties, or sleepiness [32]. We used the upper quartile as a cut-off because no validated cut-off score had been reported in those studies [32, 38–42].
The Epworth sleepiness scale
The Epworth sleepiness scale (ESS) is a self-administered eight-item questionnaire [43], which is used as an instrument to evaluate the tendency to doze off during daytime [18]. The ESS looks at sleepiness as a permanent trait component, which is evident as the tendency for falling asleep in low-stimulus situations [44]. The questions in the ESS refer to eight such situations that are more or less soporific [33].
The ESS has been shown to have adequate validity and reliability [4, 33]. It has a significant positive correlation with several different objective sleep measures, including polysomnography [4]. The ESS scores correlated significantly with mean sleep latency in the multiple sleep latency tests in some studies [33, 43, 45], while others show no significant association [46–48].
Responses to each question were ranked from 0 to 3 according to the probability of dozing off/fall asleep in the different situations (0 = never, 1 = low probability, 2 = moderate probability, 3 = high probability). The mean of the responses to the eight items was computed. In addition, a daytime sleepiness index was calculated by summing the questions in the ESS, postulating that a score ≥10 indicates excessive daytime sleepiness [18, 23, 27].
Ethics
The study was included as a part of the HUNT 3 project which has been approved by the Regional Committee for Ethics in Medical Research and the Norwegian Data Inspectorate.
Statistical analyses
Demographic data were compared between headache diagnoses with independent sample Student’s t test for continuous variables (age) and the chi-squared (χ2) test for categorical variables. Regarding mean ESS and mean KSQ scores, non-parametric tests were used for comparisons between headache groups (Kruskal–Wallis test, Mann–Whitney U test) because the distribution of these data was skewed. If statistical significance at the 0.05 level using Kruskal–Wallis test was achieved, Bonferroni correction was used for post hoc comparison of pairs of groups (P < 0.0125).
In the univariate analyses, the χ2 test was also used to compare the index scores of KSQ and ESS between headache-free individuals and the different headache diagnostic groups (TTH, migraine, and “other headaches”). The constructed “other headaches” group included probable TTH (n = 8), probable migraine (n = 5), MOH (n = 5), hangover-headache (n = 6), headache attributed to systemic viral infection (n = 4), rhinosinusitis (n = 2), cervicogenic headache (n = 1), caffeine-withdrawal headache (n = 1), persistent idiopathic facial pain (n = 1), and primary exertional headache (n = 1). Analyses were also performed when probable and definite TTH and probable and definite migraine were merged, respectively. Subjects with chronic headache (n = 22) consisted of subjects with definite TTH (n = 11), definite migraine (n = 4), MOH (n = 5), probable TTH (n = 1), and cerviogenic headache (n = 1).
In the multivariate analyses, using logistic regression with odds ratio (OR) and 95% confidence intervals (CI), we evaluated the association between headache (diagnoses and frequency) and high scores of the KSQ and the ESS. Potential confounding was evaluated by adjusting for age, sex, and sleep medication. Other potential confounding factors such as anxiety and depression (measured by Hospital anxiety and depression scale, HADS), work status, and level of physical exercise were also evaluated. For anxiety and depression the HADS cut-off score of ≥8 were used [49–51].
Results
Among the 563 invited potential participants, 297 subjects participated in the present study. The reasons for not participating were as follows: not able to get in contact (n = 171), not wanting to participate (n = 29), and not able to come (n = 66). Compared with the 266 non-participants, the 297 participants were older (mean age 52.3 vs. 48.6, P = 0.004) and slightly more likely to be men (51 vs. 47%, P = 0.37). No significant difference was found between participants and non-participants with regard to prevalence of headache sufferers (overall 39.5 vs. 37.5%, P = 0.72; men 31.4 vs. 32.9%, P = 0.90; women 46.7 vs. 42.6%, P = 0.55) or prevalence of severe sleep disturbance defined as having a KSQ score in the upper quartile (overall 26.9 vs. 21.9%, P = 0.18; men 23.9 vs. 22.3%, P = 0.76; women 29.9 vs. 21.4%, P = 0.12). The migraine-group patients were slightly younger than the other participants (mean age 44.0 vs. 54.1, P = 0.10), and they were more likely to be women (63 vs. 46%, P = 0.03). Intake of painkillers during the previous month was more common among migraineurs than among individuals with TTH (70 vs. 34%, P < 0.001), whereas use of prophylactic drugs did not differ between these headache types (Table 1). Among the 297 participants, all except one headache-free person completed the KSQ, whereas one headache-free person and one with definite migraine did not answer the ESS questionnaire.
KSQ
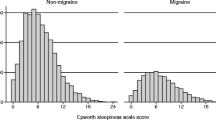
Mean KSQ score was significantly (P < 0.001) higher for patients with definite migraine (n = 51) (12.0) and MOH (n = 5) (19.8) compared with headache-free individuals (n = 76) (8.8) (Fig. 1). In the unadjusted analyses, the prevalence of severe sleep disturbances (defined as a KSQ score the upper quartile) was higher for subjects with definite migraine (n = 51) (33.3%), definite TTH (n = 135) (30.4%), and “other headaches” (n = 34) (32.4%) compared with headache-free individuals (n = 76) (13.2%) (Table 1). Overall, migraineurs (probable migraine included) (n = 56) tended to have a higher prevalence of severe sleep disturbances compared with a merged group of all other participants (n = 240) (35.7 vs. 24.6%, P = 0.09).
Regarding the separate KSQ items, the prevalence of daytime sleepiness was higher for migraineurs (n = 51) (67%) than for subjects with TTH (n = 135) (36%), other headaches (n = 34) (28%) and without headache (n = 76) (17%) (Table 2). For migraineurs (n = 51) also the prevalence of restless legs syndrome (RLS) tended to be somewhat higher compared with headache-free individuals (n = 76) (19.6 vs. 9.1%, P = 0.09). A higher proportion of migraineurs with RLS had severe sleep disturbances compared with migraineurs without RLS (70 vs. 24%, P = 0.01). Subjects with TTH (n = 135) had a higher prevalence of insomnia compared with the subjects without headache, whereas “snoring and/or apnea” were not different among the groups.
In the multivariate analyses, adjusting for age, sex, and sleep medication, severe sleep disturbance was five times more likely among migraineurs (n = 51) (OR = 5.4, 95% CI 2.0–14.5) and three times more likely for individuals with TTH (n = 135) (OR = 3.3, 1.4–7.3) compared with headache-free persons (n = 76) (Table 3).
As demonstrated in Table 4, there was a strong significant trend (P < 0.001) of higher prevalence of severe sleep disturbance with increasing headache frequency. Overall, KSQ score in the upper quartile was 17 times more likely among subjects with chronic headache (n = 22) (OR = 17.4, 95% CI 5.1–59.8) compared with individuals without headache, and somewhat more evident for chronic migraine (n = 4) (OR = 38.9, 95% CI 3.1–485.3) than for chronic TTH (n = 11) (OR = 18.3, 95% CI 3.6–93.0) (Table 4). Adjusted for anxiety, depression, and physical exercise, the ORs decreased slightly for chronic headache (n = 22) (OR = 12.8, 95% CI 3.7–45.0), chronic TTH (n = 11) (OR = 13.0, 95% CI 2.5–68.0), and for chronic migraine (n = 4) (OR = 30.5, 95% CI 1.7–540.9), respectively.
ESS
Mean ESS score did not differ between the groups (Table 1). In the unadjusted analyses the prevalence of an ESS score ≥10 tended to be higher among migraineurs (n = 50) than among the headache-free (n = 76) (20.0 vs. 9.2%, P = 0.08) (Table 1). When including subjects with probable migraine into the migraine group (n = 55) this difference became significant (23.6 vs. 9.2%, P = 0.02). Also the prevalence of an ESS score ≥10 was higher for migraineurs (probable migraine included) (n = 55) compared with a merged group of all other participants (n = 240) (23.6 vs. 10.4%, P = 0.008).
In the multivariate analyses, adjusting for age, sex, and sleep medication, ESS score ≥10 was three times more likely among migraine patients (n = 50) (OR = 3.3, 95% CI 1.04–10.18) than among headache-free individuals (n = 76) (Table 3). No significant association was found for TTH (n = 135). No trend regarding higher prevalence of an ESS score ≥10 with increasing headache-frequency was found (Table 4).
Discussion
In this cross-sectional population-based study, the association between severe sleep disturbances defined as KSQ score in the upper quartile increased markedly with increasing headache frequency and was somewhat stronger for migraine than TTH. The association with excessive daytime sleepiness (ESS ≥ 10) was evident for migraine only.
As far as we know, this is the first population-based study evaluating the relationship between primary headache diagnoses and sleep disturbance using the KSQ. To measure severe sleep disturbances, a KSQ sleep index score was created by summing the different KSQ items. The KSQ includes the topics such as snoring, apnea, insomnia, daytime sleepiness, and restless legs, which means that subjects with a high KSQ sleep index score have difficulties with many aspects of the sleep process. The same approach has been used in other studies with selected items from the KSQ.
According to the present study, severe sleep disturbance were five times more likely among migraineurs, and three times more likely among subjects with TTH. Other studies using different types of questionnaires have also reported a higher prevalence of sleep disturbances among migraineurs [16, 18, 19, 22, 26, 27], and subjects with TTH [10, 13, 25], respectively.
In accordance with the present findings, other studies have reported a higher prevalence of daytime sleepiness among migraineurs [16, 19, 26, 27] and insomnia among subjects with TTH [10, 13, 25]. Also our finding that a higher proportion of migraineurs with RLS had severe sleep disturbances accords with previous studies [52, 53].
The association between headache and severe sleep disturbance increased markedly in relation to the headache frequency. In accordance with the present results, other studies have also reported that the prevalence of sleep disturbances increased with higher frequency of headache-days per month [9, 18] and that subjects with chronic headache [22, 23] had more sleep disturbances compared with subjects with episodic headaches. However, one study did not find any difference in headache frequency among subjects with and without sleep problems [25].
In the present study, migraineurs were three times more likely to have excessive daytime sleepiness (EDS), defined as ESS ≥ 10, whereas no such association was found for TTH. Few other studies have compared sleep disturbances among subjects with different headache diagnoses [18]. No significant differences in sleep disturbances were found between migraineurs and subjects with TTH among children and adolescents in one study [25]. In contrast, sleep problems were positively associated with TTH in a cross-sectional study, whereas migraine was associated with not feeling well rested when waking up [10]. Direct comparison with other studies should be done with caution because of methodological differences. However, the use of validated instruments measuring sleep disturbances and neurologist-based headache diagnoses is a methodological advantage of the present study.
In the present study 20% of the migraine patients had EDS (ESS score ≥10). Somewhat higher prevalence of EDS was found in one uncontrolled clinical-based study (37%) [23] and one controlled cross-sectional study (34%) [18]. The latter did not find the prevalence to be significantly higher among the migraineurs than in the control group, but a case–control study found on the other hand a significant difference with a prevalence of 14% among subjects with migraine [27]. However, none of these studies used participants from a population-based study. In one of the studies [23] an association between the frequency of headache and EDS were found. In contrast to their results, we did not find a higher prevalence of EDS among subjects with chronic headache.
The strengths of the study were the population-based design, and that the headache diagnoses were based on the ICHD-2 criteria set by neurologists with a special competence in headaches performing a face-to-face interview. Furthermore, the information about sleep disturbance and day time sleepiness was collected by using two validated instruments previously used in several studies with a good correlation to objective sleep measurements [4, 31–33]. Other studies have also used the index score we used for EDS [18, 23, 27], and other studies using the KSQ have applied a similar approach for making an index score calculated from the different KSQ items [32, 38–42]. Using two different sleep questionnaires gave us valuable information as they measure different aspects of sleep disturbances. It should be emphasized that the KSQ question regarding daytime sleepiness are linked to a feeling of tiredness, while the ESS measures how likely you are to fall asleep in different situations.
There are limitations of the study that must be taken into account. First, it should be emphasized that the cross-sectional design does not permit any conclusions about causality. Second, in the present study relatively few individuals with headache had other diagnoses than migraine or TTH. Thus, no firm conclusions could be drawn for the specific diagnoses of MOH, chronic migraine, and chronic TTH.
The participation rate was good among individuals we were able to get in contact with, but moderate (53%) when the total invited group was considered [30]. Because the overall participation rate of this study was only 27% when the total invited group in HUNT 3 in Verdal/Stjørdal were considered (all inhabitants >13 years), generalization of our results to the whole population must be done with caution [30]. However, among the invited individuals selective participation due to headache status or sleep complaints seems less likely, because the prevalence of headache sufferers and severe sleep disturbance were quite similar among participants and non-participants.
ESS and KSQ are both subjective measurements of EDS and sleep disturbances, respectively. The main problem for subjective measures is that they are vulnerable to environmental and motivational variables. Individuals often tend to overestimate their own disorders, and self-evaluations risk giving an exaggerated impression of the subject’s psycho-physical complaints [44]. Some studies do, however, point out that the ESS [33] and KSQ [31] correlate well with objective sleep measurements, which could be an indication that subjects can give reliable self reports about this aspect of their life [33].
Since the study is cross-sectional, it cannot be determined whether frequent headache causes sleep disturbance or vice versa, or whether other risk factors or shared susceptibility causes this association. One possible explanation might be that pain gives arousals which prevent initiation of sleep or alters the sleep architecture so that it is more fragmented, which lead to shorter sleep duration and daytime sleepiness [54]. An alternative theory is the other way around, postulating that poor sleep can alter pain processing and therefore causes pain [55, 56]. Results from several studies indicate that sleep deprivation might give a transient disturbance of the descending pain inhibitory control system [56]. These two perspectives do not exclude each other, as it could be a dual relationship between sleep deprivation and the process of pain [55]. A study on the chronification of headache postulates that this relationship might become a vicious circle, which could explain why episodic headache changes to chronic headache in some individuals [57].
A third perspective is that neither sleep-disturbances cause pain, nor vice versa, but that both could be secondary phenomena due to a common neurobiological dysfunction [55]. Several authors have suggested the hypothalamus as the location from which this dysfunction might originate because of its connection to the brainstem and its important role in both pain and sleep regulation [6, 8, 58, 59]. Moreover, studies using MRI have reported activation of the brainstem [60, 61] and the hypothalamus [62] during headache attacks. Although the role of hypothalamus in migraine remains puzzling [63], the present results may suggest a stronger link to hypothalamus for migraine than TTH, because the association with severe sleep disturbances and excessive day time sleepiness was most evident for migraine.
Psychological factors may also be a common denominator for chronic headache and sleep disturbance, as both are strongly associated with anxiety and depression [1, 8, 64]. However, the strengths of the association were only marginally reduced after adjusting for anxiety and depression measured by HADS in the multivariate analyses. In addition, the analyses were also adjusted for possible contributing factors like age, sex, sleep medications, exercise, and work status. However, we cannot rule out that other unmeasured factors related to sleep disturbance may influence the association with chronic headache.
In conclusion, the strongest association was found between sleep disturbance and chronic headache. A close attention to sleep problems among patients with headache may have implication for the choice of treatment and the prognosis, possibly preventing chronification of the headache disorder.
References
Alberti A (2006) Headache and sleep. Sleep Med Rev 10:431–437, 10.1016/j.smrv.2006.03.003, 16872851
Leger D, Poursain B, Neubauer D, Uchiyama M (2008) An international survey of sleeping problems in the general population. Curr Med Res Opin 24:307–317, 10.1185/030079907X253771, 1:STN:280:DC%2BD1c%2FgvF2mtQ%3D%3D, 18070379
Stovner LJ, Hagen K (2006) Prevalence, burden, and cost of headache disorders. Curr Opin Neurol 19:281–285, 10.1097/01.wco.0000227039.16071.92, 16702836
Menefee LA, Frank ED, Doghramji K, Picarello K, Park JJ, Jalali S, Perez-Schwartz L (2000) Self-reported sleep quality and quality of life for individuals with chronic pain conditions. Clin J Pain 16:290–297, 10.1097/00002508-200012000-00003, 1:STN:280:DC%2BD3M7jvVKhuw%3D%3D, 11153783
Moldofsky H (2001) Sleep and pain. Sleep Med Rev 5:385–396, 10.1053/smrv.2001.0179, 12531004
Poceta JS (2003) Sleep-related headache syndromes. Curr Pain Headache Rep 7:281–287, 10.1007/s11916-003-0048-7, 12828877
Jennum P, Jensen R (2002) Sleep and headache. Sleep Med Rev 6:471–479, 10.1053/smrv.2001.0223, 12505479
Rains JC, Poceta JS, Penzien DB (2008) Sleep and headaches. Curr Neurol Neurosci Rep 8:167–175, 10.1007/s11910-008-0027-9, 18460287
Boardman HF, Thomas E, Millson DS, Croft PR (2005) Psychological, sleep, lifestyle, and comorbid associations with headache. Headache 45:657–669, 10.1111/j.1526-4610.2005.05133.x, 15953298
Rasmussen BK (1993) Migraine and tension-type headache in a general population: precipitating factors, female hormones, sleep pattern and relation to lifestyle. Pain 53:65–72, 10.1016/0304-3959(93)90057-V, 1:STN:280:DyaK3szgt12mug%3D%3D, 8316392
Lyngberg AC, Rasmussen BK, Jorgensen T, Jensen R (2005) Prognosis of migraine and tension-type headache: a population-based follow-up study. Neurology 65:580–585, 10.1212/01.wnl.0000172918.74999.8a, 16116119
Langemark M, Olesen J, Poulsen DL, Bech P (1988) Clinical characterization of patients with chronic tension headache. Headache 28:590–596, 10.1111/j.1526-4610.1988.hed2809590.x, 1:STN:280:DyaL1M3lsVCmtw%3D%3D, 3248935
Ong JC, Stepanski EJ, Gramling SE (2009) Pain coping strategies for tension-type headache: possible implications for insomnia? J Clin Sleep Med 5:52–56, 19317381
Gilman DK, Palermo TM, Kabbouche MA, Hershey AD, Powers SW (2007) Primary headache and sleep disturbances in adolescents. Headache 47:1189–1194, 10.1111/j.1526-4610.2007.00885.x, 17883524
Miller VA, Palermo TM, Powers SW, Scher MS, Hershey AD (2003) Migraine headaches and sleep disturbances in children. Headache 43:362–368, 10.1046/j.1526-4610.2003.03071.x, 1:STN:280:DC%2BD3s7kt1arsg%3D%3D, 12656707
Isik U, Ersu RH, Ay P, Save D, Arman AR, Karakoc F, Dagli E (2007) Prevalence of headache and its association with sleep disorders in children. Pediatr Neurol 36:146–151, 10.1016/j.pediatrneurol.2006.11.006, 17352946
Sand T, Hagen K, Schrader H (2003) Sleep apnoea and chronic headache. Cephalalgia 23:90–95, 10.1046/j.1468-2982.2003.00460.x, 1:STN:280:DC%2BD3s%2Fpt1Wjtg%3D%3D, 12603364
Seidel S, Hartl T, Weber M, Matterey S, Paul A, Riederer F, Gharabaghi M, Wober-Bingol C, Wober C (2009) Quality of sleep, fatigue and daytime sleepiness in migraine—a controlled study. Cephalalgia. doi:10.1111/j.1468-2982.2008.01784.x
Gupta R, Bhatia MS, Dahiya D, Sharma S, Sapra R, Semalti K, Dua RP (2008) Impact of primary headaches on subjective sleep parameters among adolescents. Ann Indian Acad Neurol 11:164–169, 10.4103/0972-2327.42936, 19893663
Blau JN (1982) Resolution of migraine attacks: sleep and the recovery phase. J Neurol Neurosurg Psychiatry 45:223–226, 10.1136/jnnp.45.3.223, 1:STN:280:DyaL383hsF2jtA%3D%3D, 7086442
Goder R, Fritzer G, Kapsokalyvas A, Kropp P, Niederberger U, Strenge H, Gerber WD, Aldenhoff JB (2001) Polysomnographic findings in nights preceding a migraine attack. Cephalalgia 21:31–37, 10.1046/j.1468-2982.2001.00141.x, 1:STN:280:DC%2BD3M3htVWhtw%3D%3D, 11298661
Kelman L, Rains JC (2005) Headache and sleep: examination of sleep patterns and complaints in a large clinical sample of migraineurs. Headache 45:904–910, 10.1111/j.1526-4610.2005.05159.x, 15985108
Peres MF, Stiles MA, Siow HC, Silberstein SD (2005) Excessive daytime sleepiness in migraine patients. J Neurol Neurosurg Psychiatry 76:1467–1468, 10.1136/jnnp.2005.062497, 1:STN:280:DC%2BD2MvpslygtQ%3D%3D, 16170102
Spierings EL, Ranke AH, Honkoop PC (2001) Precipitating and aggravating factors of migraine versus tension-type headache. Headache 41:554–558, 10.1046/j.1526-4610.2001.041006554.x, 1:STN:280:DC%2BD38%2FhvFKnsA%3D%3D, 11437890
Bruni O, Fabrizi P, Ottaviano S, Cortesi F, Giannotti F, Guidetti V (1997) Prevalence of sleep disorders in childhood and adolescence with headache: a case-control study. Cephalalgia 17:492–498, 10.1046/j.1468-2982.1997.1704492.x, 1:STN:280:DyaK2szltlGhtQ%3D%3D, 9209768
Heng K, Wirrell E (2006) Sleep disturbance in children with migraine. J Child Neurol 21:761–766, 10.1177/08830738060210092201, 16970882
Barbanti P, Fabbrini G, Aurilia C, Vanacore N, Cruccu G (2007) A case-control study on excessive daytime sleepiness in episodic migraine. Cephalalgia 27:1115–1119, 10.1111/j.1468-2982.2007.01399.x, 1:STN:280:DC%2BD2srmslGntw%3D%3D, 17725651
Boardman HF, Thomas E, Millson DS, Croft PR (2006) The natural history of headache: predictors of onset and recovery. Cephalalgia 26:1080–1088, 10.1111/j.1468-2982.2006.01166.x, 1:STN:280:DC%2BD28vpsVaisg%3D%3D, 16919058
Wober C, Brannath W, Schmidt K, Kapitan M, Rudel E, Wessely P, Wober-Bingol C (2007) Prospective analysis of factors related to migraine attacks: the PAMINA study. Cephalalgia 27:304–314, 10.1111/j.1468-2982.2007.01279.x, 1:STN:280:DC%2BD2s7ns1egtQ%3D%3D, 17376107
Hagen K, Zwart JA, Aamodt AH, Nilsen KB, Brathen G, Helde G, Stjern M, Tronvik EA, Stovner LJ (2008) A face-to-face interview of participants in HUNT 3: the impact of the screening question on headache prevalence. J Headache Pain 9:289–294, 10.1007/s10194-008-0062-6, 18690490
Axelsson J, Kecklund G, Åkerstedt T, Ekstedt M, Menenga J (2002) A comparison of the Karolinska Sleep Questionnaire and the Karolinska Sleep Diary: a methodological study [abstract]. J Sleep Res 11:1–8
Eriksen CA, Kecklund G (2007) Sleep, sleepiness and health complaints in police officers: the effects of a flexible shift system. Ind Health 45:279–288, 10.2486/indhealth.45.279, 17485872
Johns MW (1991) A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep 14:540–545, 1:STN:280:DyaK387ovVSksw%3D%3D, 1798888
Hagen K, Zwart JA, Vatten L, Stovner LJ, Bovim G (2000) Head-HUNT: validity and reliability of a headache questionnaire in a large population-based study in Norway. Cephalalgia 20:244–251, 10.1046/j.1468-2982.2000.00049.x, 1:STN:280:DC%2BD3cvkslWjsA%3D%3D, 10999674
Ebbesen MH, Hannestad YS, Midthjell K, Hunskaar S (2009) Diabetes related risk factors did not explain the increased risk for urinary incontinence among women with diabetes. The Norwegian HUNT/EPINCONT study. BMC Urol 9:11, 10.1186/1471-2490-9-11, 19740449
Silberstein SD, Olesen J, Bousser MG, Diener HC, Dodick D, First M, Goadsby PJ, Gobel H, Lainez MJ, Lance JW, Lipton RB, Nappi G, Sakai F, Schoenen J, Steiner TJ (2005) The international classification of headache disorders, 2nd edition (ICHD-II)—revision of criteria for 8.2 Medication-overuse headache. Cephalalgia 25:460–465, 10.1111/j.1468-2982.2005.00878.x, 1:STN:280:DC%2BD2M3mvVGiug%3D%3D, 15910572
Olesen J, Bousser MG, Diener HC, Dodick D, First M, Goadsby PJ, Gobel H, Lainez MJ, Lance JW, Lipton RB, Nappi G, Sakai F, Schoenen J, Silberstein SD, Steiner TJ (2006) New appendix criteria open for a broader concept of chronic migraine. Cephalalgia 26:742–746, 10.1111/j.1468-2982.2006.01172.x, 1:STN:280:DC%2BD283mt1Gruw%3D%3D, 16686915
Akerstedt T, Ingre M, Broman JE, Kecklund G (2008) Disturbed sleep in shift workers, day workers, and insomniacs. Chronobiol Int 25:333–348, 10.1080/07420520802113922, 18484368
Akerstedt T, Knutsson A, Westerholm P, Theorell T, Alfredsson L, Kecklund G (2002) Sleep disturbances, work stress and work hours: a cross-sectional study. J Psychosom Res 53:741–748, 10.1016/S0022-3999(02)00333-1, 1:STN:280:DC%2BD38vlvFyiug%3D%3D, 12217447
Leineweber C, Kecklund G, Akerstedt T, Janszky I, Orth-Gomer K (2003) Snoring and the metabolic syndrome in women. Sleep Med 4:531–536, 10.1016/S1389-9457(03)00160-6, 14607347
Leineweber C, Kecklund G, Orth-Gomer K (2007) Prediction of cardiocerebrovascular and other significant disease from disturbed sleep and work strain. Scand J Work Environ Health 33:215–222, 17572831
Sterud T, Hem E, Ekeberg O, Lau B (2008) Health problems and help-seeking in a nationwide sample of operational Norwegian ambulance personnel. BMC Public Health 8:3, 10.1186/1471-2458-8-3, 18177497
Johns MW (1992) Reliability and factor analysis of the Epworth Sleepiness Scale. Sleep 15:376–381, 1:STN:280:DyaK38zptVKmsg%3D%3D, 1519015
Curcio G, Casagrande M, Bertini M (2001) Sleepiness: evaluating and quantifying methods. Int J Psychophysiol 41:251–263, 10.1016/S0167-8760(01)00138-6, 1:STN:280:DC%2BD3MzpslCkug%3D%3D, 11448507
Johns MW (1994) Sleepiness in different situations measured by the Epworth Sleepiness Scale. Sleep 17:703–710, 1:STN:280:DyaK2M3hsFKqtQ%3D%3D, 7701181
Benbadis SR, Mascha E, Perry MC, Wolgamuth BR, Smolley LA, Dinner DS (1999) Association between the Epworth sleepiness scale and the multiple sleep latency test in a clinical population. Ann Intern Med 130:289–292, 1:STN:280:DyaK1M7kvF2ltw%3D%3D, 10068387
Chervin RD, Aldrich MS (1999) The Epworth Sleepiness Scale may not reflect objective measures of sleepiness or sleep apnea. Neurology 52:125–131, 1:STN:280:DyaK1M7hvVygsA%3D%3D, 9921859
Furuta H, Kaneda R, Kosaka K, Arai H, Sano J, Koshino Y (1999) Epworth Sleepiness Scale and sleep studies in patients with obstructive sleep apnea syndrome. Psychiatry Clin Neurosci 53:301–302, 10.1046/j.1440-1819.1999.00511.x, 1:STN:280:DyaK1MzoslCktg%3D%3D, 10459718
Augestad LB, Slettemoen RP, Flanders WD (2008) Physical activity and depressive symptoms among Norwegian adults aged 20–50. Public Health Nurs 25:536–545, 18950418
Baekken PM, Skorpen F, Stordal E, Zwart JA, Hagen K (2008) Depression and anxiety in relation to catechol-O-methyltransferase Val158Met genotype in the general population: the Nord-Trondelag Health Study (HUNT). BMC Psychiatry 8:48, 10.1186/1471-244X-8-48, 18578865
Panicker V, Evans J, Bjoro T, Asvold BO, Dayan CM, Bjerkeset O (2009) A paradoxical difference in relationship between anxiety, depression and thyroid function in subjects on and not on T4: findings from the HUNT study. Clin Endocrinol (Oxf) 71:574–580, 10.1111/j.1365-2265.2008.03521.x
Chen PK, Fuh JL, Chen SP, Wang SJ (2009) Association between Restless Legs Syndrome and Migraine. J Neurol Neurosurg Psychiatry
d’Onofrio F, Bussone G, Cologno D, Petretta V, Buzzi MG, Tedeschi G, Bonavita V, Cicarelli G (2008) Restless legs syndrome and primary headaches: a clinical study. Neurol Sci 29(1):S169–S172, 10.1007/s10072-008-0916-3, 18545926
Smith MT, Haythornthwaite JA (2004) How do sleep disturbance and chronic pain inter-relate? Insights from the longitudinal and cognitive-behavioral clinical trials literature. Sleep Med Rev 8:119–132, 10.1016/S1087-0792(03)00044-3, 15033151
Foo H, Mason P (2003) Brainstem modulation of pain during sleep and waking. Sleep Med Rev 7:145–154, 10.1053/smrv.2002.0224, 1:STN:280:DC%2BD3s7hs1Wrtg%3D%3D, 12628215
Lautenbacher S, Kundermann B, Krieg JC (2006) Sleep deprivation and pain perception. Sleep Med Rev 10:357–369, 10.1016/j.smrv.2005.08.001, 16386930
Rains JC (2008) Chronic headache and potentially modifiable risk factors: screening and behavioral management of sleep disorders. Headache 48:32–39, 10.1111/j.1526-4610.2008.01119_1.x, 18184283
Dodick DW, Eross EJ, Parish JM, Silber M (2003) Clinical, anatomical, and physiologic relationship between sleep and headache. Headache 43:282–292, 10.1046/j.1526-4610.2003.03055.x, 12603650
Montagna P (2006) Hypothalamus, sleep and headaches. Neurol Sci 27(2):S138–S143, 10.1007/s10072-006-0589-8, 16688618
Afridi SK, Giffin NJ, Kaube H, Friston KJ, Ward NS, Frackowiak RS, Goadsby PJ (2005) A positron emission tomographic study in spontaneous migraine. Arch Neurol 62:1270–1275, 10.1001/archneur.62.8.1270, 16087768
Weiller C, May A, Limmroth V, Juptner M, Kaube H, Schayck RV, Coenen HH, Diener HC (1995) Brain stem activation in spontaneous human migraine attacks. Nat Med 1:658–660, 10.1038/nm0795-658, 1:CAS:528:DyaK2MXms1Kgsbs%3D, 7585147
Denuelle M, Fabre N, Payoux P, Chollet F, Geraud G (2007) Hypothalamic activation in spontaneous migraine attacks. Headache 47:1418–1426, 18052951
Alstadhaug KB (2009) Migraine and the hypothalamus. Cephalalgia 29:809–817, 10.1111/j.1468-2982.2008.01814.x, 1:STN:280:DC%2BD1MvoslWitw%3D%3D, 19604254
Vgontzas A, Cui L, Merikangas KR (2008) Are sleep difficulties associated with migraine attributable to anxiety and depression? Headache 48:1451–1459, 10.1111/j.1526-4610.2008.01175.x, 18624714
Acknowledgments
The Nord-Trøndelag Health Study (The HUNT study) is a collaboration between The HUNT Research Centre, Faculty of Medicine, The Norwegian University of Science and Technology (NTNU), and the Nord-Trøndelag County Council.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License ( https://creativecommons.org/licenses/by-nc/2.0 ), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Ødegård, S.S., Engstrøm, M., Sand, T. et al. Associations between sleep disturbance and primary headaches: the third Nord-Trøndelag Health Study. J Headache Pain 11, 197–206 (2010). https://doi.org/10.1007/s10194-010-0201-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10194-010-0201-8