Abstract
Background
Acute kidney injury (AKI) is a globally critical issue. Most studies about AKI have been conducted in limited settings on perioperative or critically ill patients. As a result, there is little information about the epidemiology and risk factors of AKI in the general population.
Methods
We conducted a population-based cohort study using the Shizuoka Kokuho Database. We included subjects with records of health checkup results. The observation period for each participant was defined as from the date of insurance enrollment or April 2012, whichever occurred later, until the date of insurance withdrawal or September 2020, whichever was later. Primary outcome was AKI associated with admission based on the ICD-10 code. We described the incidence of AKI and performed a multivariate analysis using potential risk factors selected from comorbidities, medications, and health checkup results.
Results
Of 627,814 subjects, 8044 were diagnosed with AKI (incidence 251 per 100,000 person-years). The AKI group was older, with more males. Most comorbidities and prescribed medications were more common in the AKI group. As novel factors, statins (hazard ratio (HR) 0.84, 95% confidence interval (CI) 0.80–0.89) and physical activity habits (HR 0.79, 95% CI 0.75–0.83) were associated with reduced incidence of AKI. Other variables associated with AKI were approximately consistent with those from previous studies.
Conclusions
The factors associated with AKI and the incidence of AKI in the general Japanese population are indicated. This study generates the hypothesis that statins and physical activity habits are novel protective factors for AKI.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acute kidney injury (AKI) is defined by a rapid rise in serum creatinine, a decrease in urine output, or both. The “Kidney Disease: Improving Global Outcomes (KDIGO)” diagnostic criteria and severity classification are used widely [1]. AKI is not only a poor prognostic factor in critically ill patients [2] but is also associated with worse long-term survival and renal outcome [3]. AKI is recognized as a critical issue globally, and the International Society of Nephrology has launched the 0by25 initiative, an international cross-sectional study to eliminate preventable deaths from AKI [4]. Patient-related risk factors for AKI have been identified, including nephrotoxic drugs (including drugs aimed to treat other conditions) and various comorbidities [5].
Although many cohort studies have been conducted to date, epidemiological information, such as the true incidence of AKI, is still unclear [6]. Although many studies have investigated the risk factors and prognostic implications of AKI, most have focused on a limited setting, such as critically ill patients, specific comorbidities, or the perioperative period [2, 7, 8]. Few studies conducted in the general population have investigated the effects of modifiable lifestyles.
Identifying the risk factors of AKI (especially modifiable risks) and informing citizens and healthcare providers may reduce the occurrence of AKI. Therefore, this study aims to describe the incidence of AKI associated with hospital admission in the general population and to explore the risk factors.
Materials and methods
Study design and data source
This population-based cohort study analyzed the data obtained from the Shizuoka Kokuho Database (SKDB) [9], an administrative claims database of health insurance subscribers in the municipalities of Shizuoka Prefecture, Japan. This database includes both National Health Insurance (those aged < 75 years) and Latter-Stage Elderly Medical Care System (those aged ≥ 75 years) subscribers. SKDB covers 8.5 years (April 1, 2012 to September 30, 2020) and includes 2,230,848 individuals, and contains basic information (sex, age, observation period, and reason for disenrollment, including death) about individuals, records of health checkup results, and dates of diagnosis and treatment based on the International Statistical Classification of Diseases and Related Health Problems, Tenth Revision (ICD-10), with medications prescribed.
Participant population
The subjects were those registered in SKDB with records of health checkup results. The observation period for each participant was defined as from the date of insurance enrollment or April 2012, whichever occurred later, until the date of insurance withdrawal or September 2020, whichever was later.
We extracted the records of the first health checkup during the observation period. One year before the date of the first health checkup was defined as the baseline period. We extracted information about comorbidities and prescribed medications during the baseline period. We excluded participants who were on maintenance dialysis. Participants who had not received health checkups or who did not realize 1-year baseline periods were also excluded.
Outcome and variables
The primary outcome was AKI associated with admission. We identified this outcome using the ICD-10 codes for AKI (N17, N19) [10,11,12] that are used for diagnosis during hospitalization. Our definition included community-acquired and hospital-acquired AKI. Comorbidities were extracted using ICD-10 codes for diagnoses equivalent to the Charlson Comorbidity Index [13]. Prescribed medications were extracted using the Anatomical Therapeutic Chemical Classification System. In selecting medications, we referred to previous studies and guidelines [14, 15]. We selected medications commonly used to treat chronic disease in the general population among those that might be associated with AKI as covariates. In addition, we extracted the following results from the health checkups: age, sex, body mass index (BMI), blood pressure, questionnaire responses about smoking habits and physical activity (“In your daily life, do you walk or do any equivalent amount of physical activity more than one hour a day?”), and results from laboratory examinations.
Statistical analysis
Continuous and categorical variables were described as mean ± standard deviation and number (%), respectively. We used Fisher’s exact test to compare the proportions of binary variables and Student’s t-test to compare the continuous variables, and log-rank tests to evaluate differences in the cumulative incidence of AKI. We used a cause-specific proportional hazards model to adjust for confounders in the primary outcome. Death was considered as a competing event for AKI. Variables that were significant in the univariate model were included in the multivariate model. Complete case analysis was conducted to construct the multivariate model. Variables with Spearman’s correlation coefficients greater than 0.4 were used as covariates, whichever was considered more clinically important. Factors that were statistically significant in the multivariate model were defined as risk factors. We also performed a sensitivity analysis including interaction terms. Survival time variables were drawn as Kaplan–Meier curves, and log-rank tests were performed for comparisons between groups. The missing covariates used in the analysis did not occur completely randomly among all participants; therefore, we did not impute them. Statistical significance was defined as P < 0.05. We used JMP 16.2 (SAS Institute, Cary, NC, USA) and EZR version 1.55 (Saitama Medical Center, Jichi Medical University, Saitama, Japan) [16] for all statistical analyses.
Results
Characteristics of the patients
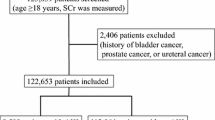
Among all individuals in the SKDB, 627,814 were included in the analysis. Four hundred and nine maintenance dialysis patients were excluded. During the mean 5.1 years of observation, 8044 cases were diagnosed with AKI associated with hospital admission (Fig. 1).
Patients’ characteristics, divided into two groups according to AKI associated with admission (AKI group) or not (No AKI group), are shown in Table 1. The AKI group was older and comprised more males. BMI was higher in the AKI group, but the difference was minimal. Most comorbidities and prescribed medications were more common in the AKI group. The results of health checkups showed that base renal function (serum creatinine and estimated glomerular filtration rate (GFR)) was slightly worse in the AKI group. The AKI group also had a lower hemoglobin level. The number of habits with no physical activity was higher in the AKI group.
Incidence of AKI associated with admission
The incidence of AKI was 251 per 100,000 person-years (mean 5.1 years of observation). The cumulative incidence of AKI is shown in Figs. 2, 3. In the stratified analysis, the incidence was higher in latter-stage older adults (Fig. 2) and males (Fig. 3).
Risk factors for AKI associated with admission
Correlation coefficients between all variables included in the multivariable model were less than 0.4. Multivariate cause-specific proportional hazards models showed that several variables were risk factors (Table 2). The variables with a multivariate hazard ratio (HR) > 1.2 were dementia, chronic pulmonary disease, rheumatic disease, liver disease, diabetes, hemiplegia or paraplegia, renal disease, any malignancy, mineralocorticoid receptor antagonists, calcium channel blockers, and diuretics. The multivariate variables with HR < 0.9 were female sex, statins, fibrates, high estimated GFR, and physical activity habits.
The sensitivity analysis taking into account the interaction terms for statins (cerebrovascular disease and peripheral vascular disease) showed a similar result to that for just statins (HR 0.85, 95% confidence interval 0.79–0.91).
Discussion
We found several factors related to AKI associated with admission. In addition, the incidence of AKI in the general population was estimated. Our study is the first investigation to examine these issues using claims data from an Asian population.
Multivariate analysis results suggested that the administration of statins and physical activity habits are novel protective factors against AKI.
Robust evidence supports the preventive effect of statins on cardiovascular and cerebrovascular disease [17]. Statins are also suggested to benefit kidney disease via mechanisms such as anti-inflammatory, anti-oxidative, and endothelial protective effects [18]. Regarding the association between kidney disease and statins, statins showed beneficial effects in lipid management in patients with chronic kidney disease (CKD) [17, 19]. There is insufficient evidence of an association between statins and AKI. Existing studies suggest that statins both increase the risk [20, 21] and do not affect the risk [22, 23] of AKI. In previous studies unable to indicate the effectiveness of statins, the severity of the underlying disease might have hindered the efficacy of statins [17]. Given that our study was conducted in a healthy general population, we would expect to detect the therapeutic effectiveness of statins.
Historically, healthy lifestyles, including physical activity habits, have prevented several diseases. Studies using data from the Japanese Specified Health Checkups, similar to ours, have shown the association between healthy lifestyles, including physical activity habits, and CKD [24], diabetes, and hypertension [25]. In addition, large prospective studies have shown that lifestyle modifications, including physical activity habits, can prevent cardiovascular disease [26]. Although no previous studies have examined the association between AKI and physical activity habits, a meta-analysis suggested that frailty is a risk factor for AKI [27]. Considering the results of previous studies and our current data, it is suggested that improving physical activity habits may contribute to the prevention of AKI.
Other variables associated with AKI in our study were approximately consistent with those in previous studies [5, 28,29,30,31]. In contrast with previous reports [30], nonsteroidal anti-inflammatory drugs and angiotensin-converting enzyme inhibitors were not a significant risk factor. Although fibrates are generally known to affect kidney function [33], we obtained the contrary result. We assume that these discrepancies are due to the avoidance of prescribing these drugs to patients at high risk for kidney injury.
The incidence of AKI estimated in this study (251 per 100,000 person-years) differs somewhat in comparison with previous studies. Several prospective observational studies in the general population have reported the incidence of severe AKI requiring dialysis as 13–14 cases per 100,000 person-years [33, 34], one of which was conducted in the same region as the present study (Shizuoka, Japan) [34]. Another cohort study in a population with normal renal function reported the incidence of AKI requiring hospitalization as 100 per 100,000 person-years, of which 10 cases in 100,000 were severe AKI requiring dialysis [35]. These results estimate that severe AKI accounts for less than 10% of all AKI. Our study includes patients with impaired baseline renal function and estimated AKI incidence for all severities. Therefore, the higher incidence of AKI than previously reported elsewhere is reasonable. Additionally, we consider that the present results provide useful epidemiological information about AKI in the general population, especially among older people in Japan. Risk factors for AKI are generally similar between our cohort and others, assuming that the populations of these cohorts are comparable. Based on the aforestated, we consider that we extracted AKI from the administrative claims database with a reasonable level of accuracy. Therefore, we were able to explore factors associated with AKI and estimate its incidence in this reliable population.
Although we constructed a linear risk prediction model for the overall population, recent reports have shown that there is heterogeneity in predictive outcomes between individual subgroups in the prediction model and that interactions between risk factors should also be taken into account [36]. The present study attempted to identify risk factors that are independent in nature from linear regression analysis and to suggest the existence of intervening factors that contain bias but have large effects. Moreover, no causal effect estimates for individual factors were made. Therefore, we did not assess predictive performance or examine interactions as a potential risk and confounding factors. Despite these limitations, we believe that the presentation of modifiable factors from simple studies such as ours can help prevent disease.
There are some other limitations to this study. First, only ICD-10 codes were used to define AKI because the data concerning urinary output and changes in renal function around the hospitalization period were unavailable. To preserve the validity of the study, we used ICD-10 codes in accordance with previous studies [10,11,12]. Nevertheless, as a limitation of the coding-based definition of AKI, AKI extraction in our cohort had high specificity, but low sensitivity, and could have been biased to severe cases [37]. Second, we could not use variables not included in the user database; thus, potential risk factors may not have been investigated. We included the major known risk factors for AKI (comorbidities and medications) in multivariate analysis to explore the circumstances as much as possible and conducted a sensitivity analysis with consideration of interaction terms. Third, because the SKDB contains only a Japanese population, caution should be exercised in extrapolating this study’s results to other ethnic groups. Fourth, our dataset may have a time discrepancy between the prescription of medication as risk factors and the onset of AKI. Most medications are regularly prescribed for chronic disease. Therefore, the effect of this limitation is considered to be relatively small. In contrast to previous studies [28], non-steroidal anti-inflammatory drugs were not significant in the multivariate analysis. These types of medications with a shorter prescription duration may have been more affected by this limitation.
Conclusions
The factors associated with AKI and the incidence of AKI associated with hospital admission in the general Japanese population are highlighted. This study generates the hypothesis that statins and physical activity habits are novel protective factors for AKI, although these exploratory results need to be validated in prospective trials. This study of AKI promises to provide essential insights into its etiology and prevention strategy.
References
KDIGO Clinical Practice Guideline for Acute Kidney Injury. Kidney Int Suppl. 2012;2:1–138. https://www.ncbi.nlm.nih.gov/pubmed/25018915
Hoste EAJ, Bagshaw SM, Bellomo R, Cely CM, Colman R, Cruz DN, et al. Epidemiology of acute kidney injury in critically ill patients: the multinational AKI-EPI study. Intensive Care Med. 2015;41:1411–23. https://doi.org/10.1007/s00134-015-3934-7.
See EJ, Jayasinghe K, Glassford N, Bailey M, Johnson DW, Polkinghorne KR, et al. Long-term risk of adverse outcomes after acute kidney injury: a systematic review and meta-analysis of cohort studies using consensus definitions of exposure. Kidney Int [Internet]. 2019;95:160–72. https://doi.org/10.1016/j.kint.2018.08.036.
Mehta RL, Burdmann EA, Cerdá J, Feehally J, Finkelstein F, García-García G, et al. Recognition and management of acute kidney injury in the International Society of Nephrology 0by25 Global Snapshot: a multinational cross-sectional study. Lancet [Internet]. 2016;387:2017–25. https://doi.org/10.1016/S0140-6736(16)30240-9.
Kellum JA, Romagnani P, Ashuntantang G, Ronco C, Zarbock A, Anders H-J. Acute kidney injury. Nat Rev Dis Primers. 2021;7:52. https://doi.org/10.1038/s41572-021-00284-z.
Ronco C, Bellomo R, Kellum JA. Acute kidney injury. Lancet. 2019;394:1949–64. https://doi.org/10.1016/S0140-6736(19)32563-2.
Bıyık M, Ataseven H, Bıyık Z, Asil M, Çifçi S, Sayın S, et al. KDIGO (Kidney Disease: Improving Global Outcomes) criteria as a predictor of hospital mortality in cirrhotic patients. Turk J Gastroenterol. 2016;27:173–9. https://doi.org/10.5152/tjg.2016.15467.
Demirjian S, Allen Bashour C, Shaw A, Schold JD, Simon J, Anthony D, et al. Predictive Accuracy of a Perioperative Laboratory Test–Based Prediction Model for Moderate to Severe Acute Kidney Injury After Cardiac Surgery. JAMA [Internet]. American Medical Association; 2022;327:956–64. Available from: https://jamanetwork.com/journals/jama/article-abstract/2789659?casa_token=g_qtVXpdoQgAAAAA:XfYKdOGKIpfOtGOqvUN2ePxJ9bNvAYARPlhiUpv93-LCtCAp0wa5-HWwyLN2ZjM2D_zbv7XCXoWM. Accessed 2022 Mar 26
Nakatani E, Tabara Y, Sato Y, Tsuchiya A, Miyachi Y. Data resource profile of Shizuoka Kokuho Database (SKDB) using integrated health- and care-insurance claims and health checkups the Shizuoka Study. J Epidemiol. 2021. https://doi.org/10.2188/jea.JE20200480.
Vlasschaert MEO, Bejaimal SAD, Hackam DG, Quinn R, Cuerden MS, Oliver MJ, et al. Validity of administrative database coding for kidney disease: a systematic review. Am J Kidney Dis [Internet]. 2011;57:29–43. https://doi.org/10.1053/j.ajkd.2010.08.031.
Rampersad C, Kraut E, Whitlock RH, Komenda P, Woo V, Rigatto C, et al. Acute Kidney Injury Events in Patients With Type 2 Diabetes Using SGLT2 Inhibitors Versus Other Glucose-Lowering Drugs: A Retrospective Cohort Study. Am J Kidney Dis [Internet]. 2020;76:471-479.e1. https://doi.org/10.1053/j.ajkd.2020.03.019.
Jain A, McDonald HI, Nitsch D, Tomlinson L, Thomas SL. Risk factors for developing acute kidney injury in older people with diabetes and community-acquired pneumonia: a population-based UK cohort study. BMC Nephrol [Internet]. 2017;18:142. https://doi.org/10.1186/s12882-017-0566-x.
Quan H, Sundararajan V, Halfon P, Fong A, Burnand B, Luthi J-C, et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care [Internet]. 2005;43:1130–9. https://doi.org/10.1097/01.mlr.0000182534.19832.83.
[Practice guidelines for drug-induced kidney disease 2016]. Nihon Jinzo Gakkai Shi [Internet]. 2016;58:477–555. Available from: https://www.ncbi.nlm.nih.gov/pubmed/27416697. Accessed 18 Nov 2022.
Usui J, Yamagata K, Imai E, Okuyama H, Kajiyama H, Kanamori H, et al. Clinical practice guideline for drug-induced kidney injury in Japan 2016: digest version. Clin Exp Nephrol [Internet]. 2016;20:827–31. https://doi.org/10.1007/s10157-016-1334-0.
Kanda Y. Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transplant [Internet]. 2013;48:452–8. https://doi.org/10.1038/bmt.2012.244.
Newman CB, Preiss D, Tobert JA, Jacobson TA, Page RL 2nd, Goldstein LB, et al. Statin Safety and Associated Adverse Events: A Scientific Statement From the American Heart Association. Arterioscler Thromb Vasc Biol [Internet]. 2019;39:e38-81. https://doi.org/10.1161/ATV.0000000000000073.
Epstein M, Campese VM. Pleiotropic effects of 3-hydroxy-3-methylglutaryl coenzyme a reductase inhibitors on renal function. Am J Kidney Dis [Internet]. 2005;45:2–14. https://doi.org/10.1053/j.ajkd.2004.08.040.
Wanner C, Tonelli M. Kidney Disease Improving Global Outcomes Lipid Guideline Development Work Group Members. KDIGO Clinical Practice Guideline for Lipid Management in CKD: summary of recommendation statements and clinical approach to the patient. Kidney Int [Internet]. 2014;85:1303–9. https://doi.org/10.1038/ki.2014.31.
Dormuth CR, Hemmelgarn BR, Paterson JM, James MT, Teare GF, Raymond CB, et al. Use of high potency statins and rates of admission for acute kidney injury: multicenter, retrospective observational analysis of administrative databases. BMJ [Internet]. 2013;346:880. https://doi.org/10.1136/bmj.f880.
Tonelli M, Lloyd AM, Bello AK, James MT, Klarenbach SW, McAlister FA, et al. Statin use and the risk of acute kidney injury in older adults. BMC Nephrol [Internet]. 2019;20:103. https://doi.org/10.1186/s12882-019-1280-7.
Ridker PM, Danielson E, Fonseca FAH, Genest J, Gotto AM Jr, Kastelein JJP, et al. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N Engl J Med [Internet]. 2008;359:2195–207. https://doi.org/10.1056/NEJMoa0807646.
Zhao B-C, Shen P, Liu K-X. Perioperative Statins Do Not Prevent Acute Kidney Injury After Cardiac Surgery: A Meta-analysis of Randomized Controlled Trials. J Cardiothorac Vasc Anesth [Internet]. 2017;31:2086–92. https://doi.org/10.1053/j.jvca.2017.04.038.
Wakasugi M, Kazama JJ, Yamamoto S, Kawamura K, Narita I. A combination of healthy lifestyle factors is associated with a decreased incidence of chronic kidney disease: a population-based cohort study. Hypertens Res [Internet]. 2012;36:328–33. https://doi.org/10.1038/hr.2012.186.
Wakasugi M, Narita I, Iseki K, Asahi K, Yamagata K, Fujimoto S, et al. Healthy Lifestyle and Incident Hypertension and Diabetes in Participants with and without Chronic Kidney Disease: The Japan Specific Health Checkups (J-SHC) Study. Intern Med. 2022. https://doi.org/10.2169/internalmedicine.8992-21.
Chiuve SE, McCullough ML, Sacks FM, Rimm EB. Healthy lifestyle factors in the primary prevention of coronary heart disease among men: benefits among users and nonusers of lipid-lowering and antihypertensive medications. Circulation [Internet]. 2006;114:160–7. https://doi.org/10.1161/CIRCULATIONAHA.106.621417.
Jiesisibieke ZL, Tung T-H, Xu Q-Y, Chen P-E, Hsu S-Y, Liu Y, et al. Association of acute kidney injury with frailty in elderly population: a systematic review and meta-analysis. Ren Fail [Internet]. 2019;41:1021–7. https://doi.org/10.1080/0886022X.2019.1679644.
Lapi F, Azoulay L, Yin H, Nessim SJ, Suissa S. Concurrent use of diuretics, angiotensin converting enzyme inhibitors, and angiotensin receptor blockers with non-steroidal anti-inflammatory drugs and risk of acute kidney injury: nested case-control study. BMJ [Internet]. 2013;346:e8525. https://doi.org/10.1136/bmj.e8525.
Secora AM, Shin J-I, Qiao Y, Alexander GC, Chang AR, Inker LA, et al. Hyperkalemia and Acute Kidney Injury with Spironolactone Use Among Patients with Heart Failure. Mayo Clin Proc [Internet]. 2020;95:2408–19. https://doi.org/10.1016/j.mayocp.2020.03.035.
Dreischulte T, Morales DR, Bell S, Guthrie B. Combined use of nonsteroidal anti-inflammatory drugs with diuretics and/or renin-angiotensin system inhibitors in the community increases the risk of acute kidney injury. Kidney Int [Internet]. 2015;88:396–403. https://doi.org/10.1038/ki.2015.101.
Rey A, Batteux B, Laville SM, Marienne J, Masmoudi K, Gras-Champel V, et al. Acute kidney injury associated with febuxostat and allopurinol: a post-marketing study. Arthritis Res Ther [Internet]. 2019;21:229. https://doi.org/10.1186/s13075-019-2011-y.
Emami F, Hariri A, Matinfar M, Nematbakhsh M. Fenofibrate-induced renal dysfunction, yes or no? J Res Med Sci [Internet]. 2020;25:39. https://doi.org/10.4103/jrms.JRMS_772_19.
Feest TG, Round A, Hamad S. Incidence of severe acute renal failure in adults: results of a community based study. BMJ [Internet]. 1993;306:481–3. https://doi.org/10.1136/bmj.306.6876.481.
Yasuda H, Kato A, Fujigaki Y, Hishida A, Shizuoka Kidney Disease Study Group. Incidence and clinical outcomes of acute kidney injury requiring renal replacement therapy in Japan. Ther Apher Dial [Internet]. 2010;14:541–6. https://doi.org/10.1111/j.1744-9987.2010.00826.x.
James MT, Hemmelgarn BR, Wiebe N, Pannu N, Manns BJ, Klarenbach SW, et al. Glomerular filtration rate, proteinuria, and the incidence and consequences of acute kidney injury: a cohort study. Lancet. 2010;376:2096–103.
Liu K, Zhang X, Chen W, Yu ASL, Kellum JA, Matheny ME, et al. Development and Validation of a Personalized Model With Transfer Learning for Acute Kidney Injury Risk Estimation Using Electronic Health Records. JAMA Netw Open. 2022;5:2219776. https://doi.org/10.1001/jamanetworkopen.2022.19776.
Carrero JJ, Fu EL, Vestergaard SV, Jensen SK, Gasparini A, Mahalingasivam V, et al. Defining measures of kidney function in observational studies using routine healthcare data: methodological and reporting considerations. Kidney Int. 2022. https://doi.org/10.1016/j.kint.2022.09.020.
Acknowledgements
We used prescription drug information in Japan (which is connected to ATC code data) provided by DATA iNDEX, Tokyo, Japan. We thank Hugh McGonigle, from Edanz (https://www.jp.edanz.com/ac), for editing a draft of the manuscript.
Author information
Authors and Affiliations
Contributions
All authors have contributed significantly. HD, KM, and AS conceived the study. HD and EN extracted the data from the SKDB. HD and EN provided statistical advice on study design and analyzed the data. HD drafted the article. All authors contributed substantially to its revision, took responsibility for the paper as a whole, and agreed with the content of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee at which the studies were conducted and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This study was approved by the ethics board of Shizuoka Graduate University of Public Health (SGUPH_2021_001_024).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Dote, H., Nakatani, E., Mori, K. et al. Factors associated with incidence of acute kidney injury: a Japanese regional population-based cohort study, the Shizuoka study. Clin Exp Nephrol 27, 321–328 (2023). https://doi.org/10.1007/s10157-022-02310-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-022-02310-0