Abstract
Background
The aim of this study was to evaluate the impact of Surgical Unit volume on the 30-day reoperation rate in patients with CRC.
Methods
Data were extracted from the regional Hospital Discharge Dataset and included patients who underwent elective resection for primary CRC in the Veneto Region (2005–2013). The primary outcome measure was any unplanned reoperation performed within 30 days from the index surgery. Independent variables were: age, gender, comorbidity, previous abdominal surgery, site and year of the resection, open/laparoscopic approach and yearly Surgical Unit volume for colorectal resections as a whole, and in detail for colonic, rectal and laparoscopic resections. Multilevel multivariate regression analysis was used to evaluate the impact of variables on the outcome measure.
Results
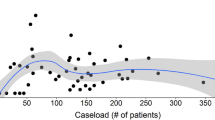
During the study period, 21,797 elective primary colorectal resections were performed. The 30-day reoperation rate was 5.5 % and was not associated with Surgical Unit volume. In multivariate multilevel analysis, a statistically significant association was found between 30-day reoperation rate and rectal resection volume (intermediate-volume group OR 0.75; 95 % CI 0.56–0.99) and laparoscopic approach (high-volume group OR 0.69; 95 % CI 0.51–0.96).
Conclusions
While Surgical Unit volume is not a predictor of 30-day reoperation after CRC resection, it is associated with an early return to the operating room for patients operated on for rectal cancer or with a laparoscopic approach. These findings suggest that quality improvement programmes or centralization of surgery may only be required for subgroups of CRC patients.

Similar content being viewed by others
References
Birkmeyer JD, Dimick JB, Birkmeyer N (2004) Measuring the quality of surgical care: structure, process, or outcomes? J Am Coll Surg 198:626–632
Almoudaris AM, Burns EM, Bottle A et al (2013) Single measures of performance do not reflect overall institutional quality in colorectal cancer surgery. Gut 62:423–429
Surgical Care and Outcomes Assessment Program (SCOAP) (2012) Adoption of laparoscopy for elective colorectal resection: a report from the Surgical Care and Outcomes Assessment Program. J Am Coll Surg 214:909–918
NHS Scotland—Scottish Cancer Taskforce. Colorectal cancer clinical quality performance indicators. Final Publication v2.0. November 2013. http://healthcareimprovementscotland.org/his/idoc.ashx?docid=0213323f-caae-4e15-ace7-f1f252c1e17f&version=-1. Accessed on 31 Mar 2015
Burns EM, Bottle A, Almoudaris AM et al (2013) Hierarchical multilevel analysis of increased caseload volume and postoperative outcome after elective colorectal surgery. Br J Surg 100:1531–1538
Morris AM, Baldwin L-M, Matthews B et al (2007) Reoperation as a quality indicator in colorectal surgery: a population-based analysis. Ann Surg 245:73–79
Krarup PM, Jorgensen LN, Andreasen AH, Harling H, Danish Colorectal Cancer Group (2012) A nationwide study on anastomotic leakage after colonic cancer surgery. Colorectal Dis 14:661–667
Archampong D, Borowski D, Wille-Jørgensen P, Iversen LH (2012) Workload and surgeon’s specialty for outcome after colorectal cancer surgery. Cochrane Database Syst Rev 3:CD005391
Burns EM, Mamidanna R, Currie A et al (2014) The role of caseload in determining outcome following laparoscopic colorectal cancer resection: an observational study. Surg Endosc 28:134–142
Schrag D, Panageas KS, Riedel E et al (2002) Hospital and surgeon procedure volume as predictors of outcome following rectal cancer resection. Ann Surg 236:583–592
Zorzi M, Fedato C, Cogo C et al (2015) I programmi di screening oncologici del Veneto. Rapporto 2012–2013. https://www.registrotumoriveneto.it/screening/presentazione.php
Mahoney FI, Barthel DW (1965) Functional Evaluation: the Barthel Index. Md State Med J 14:61–65
Burns EM, Bottle A, Aylin P, Darzi A, Nicholls RJ, Faiz O (2011) Variation in reoperation after colorectal surgery in England as an indicator of surgical performance: retrospective analysis of Hospital Episode Statistics. BMJ 343:d4836
Wibe A, Eriksen MT, Syse A, Tretli S, Myrvold HE, Søreide O (2005) Norwegian Rectal Cancer Group. Effect of hospital caseload on long-term outcome after standardization of rectal cancer surgery at a national level. Br J Surg 92:217–224
Zorzi M, Fedeli U, Schievano E et al (2014) Impact on colorectal cancer mortality of screening programmes based on the faecal immunochemical test. Gut 64:784–790
Borowski DW, Kelly SB, Bradburn DM et al (2007) Impact of surgeon volume and specialization on short-term outcomes in colorectal cancer surgery. Br J Surg 94:880–889
Borowski DW, Bradburn DM, Mills SJ, Northern Region Colorectal Cancer Audit Group (NORCCAG) et al (2010) Volume-outcome analysis of colorectal cancer-related outcomes. Br J Surg 97:1416–1430
Marusch F, Koch A, Schmidt U, Pross M, Gastinger I, Lippert H (2001) Hospital caseload and the results achieved inpatients with rectal cancer. Br J Surg 88:1397–1402
Marusch F, Koch A, Schmidt U et al (2001) Effect of caseload on the short-term outcome of colon surgery: results of a multicenter study. Int J Colorectal Dis 16:362–369
Harling H, Bülow S, Møller LN, Jørgensen T, Danish Colorectal Cancer Group (2005) Hospital volume and outcome of rectal cancer surgery in Denmark 1994–1999. Colorectal Dis 7:90–95
Kressner M, Bohe M, Cedermark B et al (2009) The impact of hospital volume on surgical outcome in patients with rectal cancer. Dis Colon Rectum 52:1542–1549
Association of Coloproctology of Great Britain and Ireland, Clinical Effectiveness Unit at The Royal College of Surgeons of England, The Health and Social Care Information Centre. National Bowel Cancer Audit Report 2014. http://www.hqip.org.uk/assets/NCAPOP-Library/NCAPOP-2014-15/nati-clin-audi-supp-prog-bowe-canc-2014-rep1.pdf. Accessed 02/04/2015
Moghadamyeghaneh Z, Masoomi H, Mills SD et al (2014) Outcomes of conversion of laparoscopic colorectal surgery to open surgery. JSLS 18 pii:e2014.00230
Delaney CP, Chang E, Senagore AJ, Broder M (2008) Clinical outcomes and resource utilization associated with laparoscopic and open colectomy using a large national database. Ann Surg 247:819–824
Faiz O, Warusavitarne J, Bottle A, Tekkis PP, Darzi AW, Kennedy RH (2009) Laparoscopically assisted vs. open elective colonic and rectal resection: a comparison of outcomes in English National Health Service Trusts between 1996 and 2006. Dis Colon Rectum 52:1695–1704
Bilimoria KY, Bentrem DJ, Merkow RP et al (2008) Laparoscopic-assisted vs. open colectomy for cancer: comparison of short-term outcomes from 121 hospitals. J Gastrointest Surg 12:2001–2009
Schwenk W, Haase O, Neudecker J, Müller JM (2005) Short term benefits for laparoscopic colorectal resection. Cochrane Database Syst Rev 20:CD003145
Damle RN, Macomber CW, Flahive JM et al (2014) Surgeon volume and elective resection for colon cancer: an analysis of outcomes and use of laparoscopy. J Am Coll Surg 218:1223–1230
Aylin P, Bottle A, Elliott P, Jarman B (2007) Surgical mortality: hospital episode statistics v central cardiac audit database. BMJ 335:839
Garout M, Tilney HS, Tekkis PP, Aylin P (2008) Comparison of administrative data with the Association of Coloproctology of Great Britain and Ireland (ACPGBI) colorectal cancer database. Int J Colorectal Dis 23:155–163
Burns EM, Rigby E, Mamidanna R et al (2012) Systematic review of discharge coding accuracy. J Public Health (Oxf) 34:138–148
Almoudaris AM, Burns EM, Mamidanna R et al (2011) Value of failure to rescue as a marker of the standard of care following reoperation for complications after colorectal resection. Br J Surg 98:1775–1783
Merkow RP, Hall BL, Cohen ME et al (2013) Validity and feasibility of the American College of Surgeons colectomy composite outcome quality measure. Ann Surg 257:483–489
Mamidanna R, Burns EM, Bottle A et al (2012) Reduced risk of medical morbidity and mortality in patients selected for laparoscopic colorectal resection in England: a population-based study. Arch Surg 147:219–227
Mamidanna R, Eid-Arimoku L, Almoudaris AM et al (2012) Poor 1-year survival in elderly patients undergoing nonelective colorectal resection. Dis Colon Rectum 55:788–796
Nachiappan S, Burns EM, Faiz O (2013) Validity and Feasibility of the American College of Surgeons Colectomy composite outcome quality measure. Ann Surg 261:e158
Renzulli P, Laffer UT (2005) Learning curve: the surgeon as a prognostic factor in colorectal cancer surgery. Recent Results Cancer Res 165:86–104
Ghaferi AA, Birkmeyer JD, Dimick JB (2009) Variation in hospital mortality associated with inpatient surgery. N Eng J Med 361:1368–1375
Howell AM, Panesar SS, Burns EM, Donaldson LJ, Darzi A (2014) Reducing the burden of surgical harm: a systematic review of the interventions used to reduce adverse events in surgery. Ann Surg 259:630–641
Acknowledgments
This study was supported in part by a grant from the AIRC Foundation and in part by a grant from Italian Ministry of Health (RF-2011-02349645). The article was reviewed and edited for English language usage by American Journal Experts.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Informed consent was not required for this type of study.
Appendix
Appendix
See Table 5.
Rights and permissions
About this article
Cite this article
Pucciarelli, S., Chiappetta, A., Giacomazzo, G. et al. Surgical Unit volume and 30-day reoperation rate following primary resection for colorectal cancer in the Veneto Region (Italy). Tech Coloproctol 20, 31–40 (2016). https://doi.org/10.1007/s10151-015-1388-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10151-015-1388-0