Abstract
Background
Ramucirumab (RAM) plus paclitaxel (PTX) therapy has shown promising results as a standard second-line treatment for advanced gastric cancer patients. Recently, combined docetaxel (DOC) plus S-1 (DS) therapy could be regarded as the new standard adjuvant chemotherapy for patients with curatively resected stage III gastric cancer. However, the efficacy and safety of RAM plus PTX therapy in patients treated previously with DOC-containing therapy remains unclear.
Methods
This study assessed the clinical outcomes of RAM plus PTX therapy in advanced gastric cancer patients with or without a previous history of treatment with a DOC-containing regimen.
Results
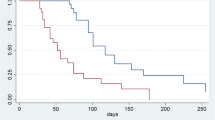
In a series of 107 consecutive patients enrolled for this study, the median PFS and OS were 4.2 and 6.2 months, respectively. Fifty-five patients had a history of prior therapy with DOC and 52 did not. There was no significant difference between with and without DOC groups in the ORR (22.2% vs. 23.5%), PFS (4.2 vs. 5.3 months), or OS (7.2 vs. 6.4 months). In a comparison taking into account the interval from the DOC-containing therapy to the RAM plus PTX therapy, the number of treatment courses was significantly smaller and the PFS significantly shorter in the patient group with an interval of ≤ 6 months (median, 2 vs 4.5 courses, P = 0.033; 3.4 months vs. 5.1 months, P = 0.043).
Conclusions
RAM plus PTX therapy in patients with advanced gastric cancer is effective even in patients who have previously received DOC-containing chemotherapy, especially if the interval is > 6 months.




Similar content being viewed by others
References
Torre LA, Bray F, Siegel RL et al (2012) Global cancer statistics. Cancer J Clin 65:87–108
Sakuramoto S, Sasako M, Yamaguchi T et al (2007) Adjuvant chemotherapy for gastric cancer with S-1, an oral fluoropyrimidine. N Eng J Med 357:1810–1820
Sasako M, Sakuramoto S, Katai H et al (2011) Five-year outcomes of a randomized phase III trial comparing adjuvant chemotherapy with S-1 versus surgery alone in stage II or III gastric cancer. J Clin Oncol 29:4387–4393
Japanese Gastric Cancer Association (2017) Japanese gastric cancer treatment guideline 2014 (ver.4). Gastric Cancer 20:1–19
Wagner AD, Grothe W, Haerting J et al (2006) Chemotherapy in advanced gastric cancer: a systematic review and meta-analysis based on aggregate data. J Clin Oncol 24:2903–2909
Koizumi W, Narahara H, Hata T et al (2008) S-1 plus cisplatin versus S-1 alone for first-line treatment of advanced gastric cancer (SPIRITS trial): a phase III trial. Lancet Oncol 9:215–221
Wilke H, Muro K, Van Cutsem CE et al (2014) Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): a double-blind, randomized phase 3 trial. Lancet Oncol 15:1224–1235
Fuchs CS, Tomasek J, Yong CJ et al (2014) Ramucirumab monotherapy for previously treated advanced gastric or gastro-oesohageal junction adenocarcinoma (REGERD): an international, randomized, multicentre, placebo-controlled, phase 3 trial. Lancet 383:31–39
Shitara K, Muro K, Shimada Y et al (2016) Subgroup analyses of the safety and efficacy of ramucirumab in Japanese and Western patients in RAINBOW: a randomized clinical trial in second-line treatment of gastric cancer. Gastric Cancer 19:927–938
Jung M, Ryu MH, Oh DY et al (2018) Efficacy and tolerability of ramucirumab monotherapy or in combination with paclitaxel in gastric cancer patients from the Expanded Access Program Cohort by the Korean Cancer Study Group (KCSG). Gastric Cancer 21:819–830
Muro K, Oh SC, Shimada Y et al (2016) Subgroup analysis of East Asians in RAINBOW: a phase 3 trial of ramucirumab plus paclitaxel for advanced gastric cancer. J Gastroenterol Hepatol 31:581–589
Murahashi S, Takahari D, Wakatsuki T et al (2018) A retrospective analysis of ramucirumab monotherapy in previously treated Japanese patients with advanced or metastatic gastric adenocarcinoma. Int J Clin Oncol 23:92–97
Yoshida K, Kodera Y, Kochi M et al (2020) Addition of docetaxel to oral fluoropyrimidine improves efficacy in patints with stage III gastric cancer: interim analysis of JACCRO GC-07, a randomized controlled trial. J clin Oncol 37:1296–1304
Migita K, Nashimoto A, Yabusaki H et al (2016) Efficacy of neoadjuvant chemotherapy with docetaxel, cisplatin and S-1 for resectable locally advanced gastric cancer. Int J Clin Oncol 21:102–109
Cancer Therapy Evaluation Program. Common Terminology Criteria for Adverse Events v4.02. (2009) http://ctep.cancer.gov/protocolDevelopment/electronic_applications/ctc.htm#ctc_40. Accessed Jan 2020
Eisenhauer EA, Therasse P, Bogaerts J et al (2009) New response evaluation criteria in solid tumors: revised RESIST guideline (version 1.1). Eur J Cancer 45:228–247
Valero V, Jones SE, Von Hoff DD et al (1998) A phase II study of docetaxel in patients with paclitaxel-resistant metastatic breast cancer. J Clin Oncol 16:3362–3368
Sella A, Yarom N, Zisman A et al (2009) Paclitaxel, estramustine and carboplatin combination chemotherapy after initial docetaxel-based chemotherapy in castration-resistant prostate cancer. Oncology 76:442–446
Kondoh C, Takahari D, Shitara K et al (2012) Efficacy of docetaxel in patients with paclitaxel-resistant advanced gastric cancer. Jpn J Cancer Chemother 39:1511–1515
Shimura T, Kitagawa M, Yamada T et al (2012) The impact of cross-resistance between paclitaxel and docetaxel for metastatic gastric cancer. Onkologie 35:176–183
Ando T, Hosokawa A, Kajiura S et al (2012) Efficacy of weekly paclitaxel in patients with advanced gastric cancer refractory to docetaxel-based chemotherapy. Gastric Cancer 15:427–432
Yamada Y, Boku N, Mizusawa J et al (2019) Docetaxel plus cisplatin and S-1 versus cisplatin and S-1 in patients with advanced gastric cancer (JCOG1013): An open-label, phase 3, randomized controlled trial. Lancet Gastroenterol Hepatol 4:501–510
Nishikawa K, Tsuburaya A, Yoshikawa T et al (2018) A phase II trial of capecitabine plus cisplatin (XP) for patients with advanced gastric cancer with early relapse after S-1 adjuvant therapy: XParTS-1 trial. Gastric Cancer 21:811–818
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Kakuta, T., Yabusaki, H., Bamba, T. et al. Efficacy and safety of ramucirumab plus paclitaxel therapy for advanced gastric cancer patients treated previously with docetaxel-containing chemotherapy. Int J Clin Oncol 26, 684–693 (2021). https://doi.org/10.1007/s10147-020-01845-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-020-01845-0