Abstract
Background
Hepatocellular carcinoma (HCC) is the most common primary liver cancer in the worldwide. Sorafenib is approved for first-line therapy against advanced HCC, but chemo-resistance is still a leading cause of tumor relapse and treatment failure in HCC. Thus, there is a significant clinical need to identify effective strategies to overcome drug resistance on the disease.
Methods
The protein and mRNA expression of TRIM37 in HCC cell lines and patient tissues were determined using Real-time PCR and Western blot, respectively. HCC tissue samples were analyzed by IHC to investigate the association between TRIM37expression and the clinicopathological characteristics of HCC patients. Functional assays, such as MTT, FACS, and Tunel assay, are used to determine the oncogenic role of TRIM37 in human HCC progression. Furthermore, western blotting and luciferase assay were used to determine the mechanism of TRIM37promotes chemoresistance in HCC.
Results
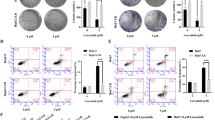
We found that both the mRNA and protein expression of TRIM37 was markedly upregulated in HCC cell lines and tissues, especially in Sorafenib-resistance HCC tissues. Moreover, high TRIM37 expression was associated with poor prognosis with HCC patients. TRIM37 overexpression confers Sorafenib resistance on HCC cells; however, inhibition of TRIM37 sensitized HCC cell lines to Sorafenib cytotoxicity. Additionally, TRIM37 upregulated the levels of AKT activity and phosphorylated AKT, thereby activating canonical AKT signaling.
Conclusion
Our findings suggest that targeting TRIM37 signaling may represent a promising strategy to enhance Sorafenib response in HCC patients with chemoresistant.





Similar content being viewed by others
References
Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F (2015) Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 136:E359–E386
Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, Jemal A, Yu XQ, He J (2016) Cancer statistics in China, 2015. CA Cancer J Clin 66:115–132
Miller KD, Siegel RL, Lin CC, Mariotto AB, Kramer JL, Rowland JH, Stein KD, Alteri R, Jemal A (2016) Cancer treatment and survivorship statistics, 2016. CA Cancer J Clin 66:271–289
Keating GM (2017) Sorafenib: a review in hepatocellular carcinoma. Target Oncol 12:243–253
Kudo M (2018) Systemic therapy for hepatocellular carcinoma: latest advances. Cancers (Basel) 10:6
Giannini EG, Farinati F, Ciccarese F, Pecorelli A, Rapaccini GL, Marco M, Benvegnu L, Caturelli E, Zoli M, Borzio F, Chiaramonte M, Trevisani F (2015) Italian liver cancer, prognosis of untreated hepatocellular carcinoma. Hepatology 61:184–190
Ghouri YA, Mian I, Rowe JH (2017) Review of hepatocellular carcinoma: epidemiology, etiology, and carcinogenesis. J Carcinog 16:1
Brandi G, de Rosa F, Agostini V, di Girolamo S, Andreone P, Bolondi L, Serra C, Sama C, Golfieri R, Gramenzi A, Cucchetti A, Pinna AD, Trevisani F, Biasco G (2013) Italian liver cancer, metronomic capecitabine in advanced hepatocellular carcinoma patients: a phase II study. Oncologist 18:1256–1257
Vivanco I, Sawyers CL (2002) The phosphatidylinositol 3-Kinase AKT pathway in human cancer. Nat Rev Cancer 2:489–501
Qiao M, Sheng S, Pardee AB (2008) Metastasis and AKT activation. Cell Cycle 7:2991–2996
Yuan TL, Cantley LC (2008) PI3K pathway alterations in cancer: variations on a theme. Oncogene 27:5497–5510
Bachman KE, Argani P, Samuels Y, Silliman N, Ptak J, Szabo S, Konishi H, Karakas B, Blair BG, Lin C, Peters BA, Velculescu VE, Park BH (2004) The PIK3CA gene is mutated with high frequency in human breast cancers. Cancer Biol Ther 3:772–775
Rodriguez-Viciana P, Warne PH, Dhand R, Vanhaesebroeck B, Gout I, Fry MJ, Waterfield MD, Downward J (1994) Phosphatidylinositol-3-OH kinase as a direct target of Ras. Nature 370:527–532
Moscatello DK, Holgado-Madruga M, Emlet DR, Montgomery RB, Wong AJ (1998) Constitutive activation of phosphatidylinositol 3-kinase by a naturally occurring mutant epidermal growth factor receptor. J Biol Chem 273:200–206
Downward J (2003) Targeting RAS signalling pathways in cancer therapy. Nat Rev Cancer 3:11–22
Avela K, Lipsanen-Nyman M, Idanheimo N, Seemanova E, Rosengren S, Makela TP, Perheentupa J, Chapelle AD, Lehesjoki AE (2000) Gene encoding a new RING-B-box-Coiled-coil protein is mutated in mulibrey nanism. Nat Genet 25:298–301
Kallijarvi J, Avela K, Lipsanen-Nyman M, Ulmanen I, Lehesjoki AE (2002) The TRIM37 gene encodes a peroxisomal RING-B-box-coiled-coil protein: classification of mulibrey nanism as a new peroxisomal disorder. Am J Hum Genet 70:1215–1228
Hu X, Xiang D, Xie Y, Tao L, Zhang Y, Jin Y, Pinello L, Wan Y, Yuan GC, Li Z (2019) Suppresses invasion migration and metastasis of luminal breast cancer cells via activation of GATA3 and repression of TRIM37 expression. Oncogene 7:234
Han C, Xia X, Jiao S, Li G, Ran Q, Yao S (2019) Tripartite motif containing protein 37 involves in thrombin stimulated BV-2 microglial cell apoptosis and interleukin 1beta release. Biochem Biophys Res Commun 516:1252–1257
Brigant B, Metzinger-Le Meuth V, Rochette J, Metzinger L (2018) TRIMming down to TRIM37: relevance to inflammation, cardiovascular disorders, and cancer in MULIBREY nanism. Int J Mol Sci 20:9
Li Y, Deng L, Zhao X, Li B, Ren D, Yu L, Pan H, Gong Q, Song L, Zhou X, Dai T (2018) Tripartite motif-containing 37 (TRIM37) promotes the aggressiveness of non-small-cell lung cancer cells by activating the NF-kappaB pathway. J Pathol 246:366–378
Tao Y, Xin M, Cheng H, Huang Z, Hu T, Zhang T, Wang J (2017) TRIM37 promotes tumor cell proliferation and drug resistance in pediatric osteosarcoma. Oncol Lett 14:6365–6372
Dong S, Pang X, Sun H, Yuan C, Mu C, Zheng S (2018) TRIM37 targets AKT in the growth of lung cancer cells. Onco Targets Ther 11:7935–7945
Tang SL, Gao YL, Wen-Zhong H (2018) Knockdown of TRIM37 suppresses the proliferation, migration and invasion of glioma cells through the inactivation of PI3K/Akt signaling pathway. Biomed Pharmacother 99:59–64
Chan TO, Rittenhouse SE, Tsichlis PN (1999) AKT/PKB and other D3 phosphoinositide-regulated kinases: kinase activation by phosphoinositide-dependent phosphorylation. Annu Rev Biochem 68:965–1014
Potter CJ, Pedraza LG, Xu T (2002) Akt regulates growth by directly phosphorylating Tsc2. Nat Cell Biol 4:658–665
Fu HW, Lin X, Zhu YX, Lan X, Kuang Y, Wang YZ, Ke ZG, Yuan T, Chen P (2019) Circ-IGF1R has pro-proliferative and anti-apoptotic effects in HCC by activating the PI3K/AKT pathway. Gene 716:144031
Jondal DE, Thompson SM, Butters KA, Knudsen BE, Anderson JL, Roberts LR, Callstrom MR, Woodrum DA (2019) Single-dose neoadjuvant AKT pathway inhibitor reduces growth of hepatocellular carcinoma after laser thermal ablation in small-animal model. Radiology 292:752–759
Han P, Li H, Jiang X, Zhai B, Tan G, Zhao D, Qiao H, Liu B, Jiang H, Sun X (2017) Dual inhibition of Akt and c-Met as a second-line therapy following acquired resistance to sorafenib in hepatocellular carcinoma cells. Mol Oncol 11:320–334
Tang W, Yang L, Yang T, Liu M, Zhou Y, Lin J, Wang K, Ding C (2019) INPP4B inhibits cell proliferation, invasion and chemoresistance in human hepatocellular carcinoma. Onco Targets Ther 12:3491–3507
Pretre V, Wicki A (2018) Inhibition of Akt and other AGC kinases: a target for clinical cancer therapy? Semin Cancer Biol 48:70–77
Jiang J, Yu C, Chen M, Tian S, Sun C (2015) Over-expression of TRIM37 promotes cell migration and metastasis in hepatocellular carcinoma by activating Wnt/β-catenin signaling. Biochem Biophys Res Commun 464:1120–1127
Pretre V, Wicki A (2017) Epithelial-to-mesenchymal transition: a mediator of sorafenib resistance in advanced hepatocellular carcinoma. Curr Cancer Drug Targets 17:698–706
Wang W, Xia ZJ, Farre JC, Subramani S (2017) TRIM37, a novel E3 ligase for PEX5-mediated peroxisomal matrix protein import. J Cell Biol 216:2843–2858
Bhatnagar S, Gazin C, Chamberlain L, Ou J, Zhu X, Tushir JS, Virbasius CM, Lin L, Zhu LJ, Wajapeyee N, Green MR (2014) TRIM37 is a new histone H2A ubiquitin ligase and breast cancer oncoprotein. Nature 516:116–120
Kallijarvi J, Lahtinen U, Hamalainen R, Lipsanen-Nyman M, Palvimo JJ, Lehesjoki AE (2005) TRIM37 defective in mulibrey nanism is a novel RING finger ubiquitin E3 ligase. Exp Cell Res 308:146–155
Acknowledgment
Natural Science Foundation of China (No. 81602701, No.81974443); Natural Science Foundation of Guangdong Province (No. 2017A030313547, No. 2018A030313176); Science and Technology Projects Foundation of Guangdong Province (No. 2015A070710006, No. 2016A020215053); Science and Technology Projects Foundation of Guangzhou City (No. 201507020037, No. 201607010260); Research project of Gannan Medical University, Gannan Medical University Key Cultivation project (No:ZD201802); Natural Science Foundation of Jiangxi Province (No:20202BAB206040)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Tan, G., Xie, B., Yu, N. et al. TRIM37 overexpression is associated with chemoresistance in hepatocellular carcinoma via activating the AKT signaling pathway. Int J Clin Oncol 26, 532–542 (2021). https://doi.org/10.1007/s10147-020-01832-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-020-01832-5