Abstract
Background
Disseminated intravascular coagulation (DIC) associated with solid tumors (DIC-ST) is often encountered in clinical practice. Patients with DIC-ST are usually in poor condition and have bleeding diathesis due to advanced or metastatic diseases. Although some affected patients are treated with heparin, this strategy has not been prospectively studied. Recombinant human soluble thrombomodulin (thrombomodulin alfa, TM-α) is a new anticoagulant developed in Japan. We conducted a prospective study that evaluated the efficacy and safety of TM-α in patients with DIC-ST.
Methods
A prospective one-arm study with TM-α was conducted for DIC-ST. TM-α (380 U/kg) was given for 30 min intravenously once daily for 6–14 days. The primary efficacy endpoint was the DIC resolution rate. Change in DIC scores and improvement in bleeding symptoms and outcomes were also evaluated. Safety endpoints included the incidence of bleeding-related adverse events.
Results
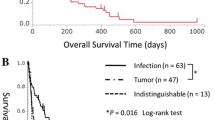
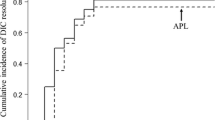
A total of 101 patients were treated with TM-α. The three main underlying malignant diseases were lung, stomach, and breast cancer, which accounted for 60 % of all patients. The DIC resolution rate was 34.0 % at the end of TM-α treatment. Improvement in DIC scores was seen in 55.2 % of patients, while only 22.9 % of patients had worsening of DIC scores. The overall survival rate was 55.4 % on day 28. The incidence of hemorrhage related to TM-α was 12.9 % until day 28. Cases of severe hemorrhage related to TM-α did not occur.
Conclusions
TM-α is effective and safe for DIC-ST. This agent is the treatment of choice for the management of DIC-ST.


Similar content being viewed by others
References
Sallah S, Wan JY, Nguyen NP et al (2001) Disseminated intravascular coagulation in solid tumors: clinical and pathologic study. Thromb Haemost 86:828–833
Saba HI, Morelli GA, Saba RI (2009) Disseminated intravascular coagulation (DIC) in cancer. Cancer Treat Res 148:137–156
Saito H, Maruyama I, Shimazaki S et al (2007) Efficacy and safety of recombinant human soluble thrombomodulin (ART-123) in disseminated intravascular coagulation: results of a phase III, randomized, double-blind clinical trial. J Thromb Haemost 5(1):31–41
Kobayashi N, Maegawa K, Takada M et al (1983) Criteria for diagnosis of DIC based on the analysis of clinical and laboratory findings in 345 DIC patients collected by the Research Committee on DIC in Japan. Bibl Haematol 49:265–275
Tsuruta K, Yamada Y, Serada M et al (2011) Model-based analysis of covariate effects on population pharmacokinetics of thrombomodulin alfa in patients with disseminated intravascular coagulation and normal subjects. J Clin Pharmacol 51(9):1276–1285
Levi M (2009) Disseminated intravascular coagulation in cancer patients. Best Pract Res Clin Haematol 22(1):129–136
Lebelle CA, Kitchens CS (2005) Disseminated intravascular coagulation: treat the cause, not the lab values. Cleve Clin J Med 72:377–397
Rhee J, Han SW, Oh DY et al (2010) Clinicopathologic features and clinical outcomes of gastric cancer that initially presents with disseminated intravascular coagulation: a retrospective study. J Gastroenterol Hepatol 25(9):1537–1542
Lin PH, Lu YS, Lin CH et al (2010) Vinorelbine plus 24-hour infusion of high-dose 5-fluorouracil and leucovorin as effective palliative chemotherapy for breast cancer patients with acute disseminated intravascular coagulation. Anticancer Res 30(7):3087–3091
Tokar M, Bobilev D, Ariad S et al (2006) Disseminated intravascular coagulation at presentation of advanced gastric cancer. Isr Med Assoc J 8(12):853–855
Pasquini E, Gianni L, Aitini E et al (1995) Acute disseminated intravascular coagulation syndrome in cancer patients. Oncology 52(6):505–508
Voulgaris E, Pentheroudakis G, Vassou A et al (2009) Disseminated intravascular coagulation (DIC) and non-small cell lung cancer (NSCLC): report of a case and review of the literature. Lung Cancer 64(2):247–249
Acknowledgments
This work was supported by Asahi Kasei Pharma Corporation (AKP). In addition to the authors, 34 institutions participated in this study. The institutions and investigators are shown in Table 7. The authors have access to all primary data, which can be made available to the journal editors upon request.
Conflict of interest
K. Tamura, H. Saito and N. Aoki have received consultancy funding from AKP. H. Asakura and K. Okamoto have received lecture fees from AKP. J. Tagawa and T. Hayakawa are employees of AKP.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Tamura, K., Saito, H., Asakura, H. et al. Recombinant human soluble thrombomodulin (thrombomodulin alfa) to treat disseminated intravascular coagulation in solid tumors: results of a one-arm prospective trial. Int J Clin Oncol 20, 821–828 (2015). https://doi.org/10.1007/s10147-014-0768-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-014-0768-1