Abstract
This study describes and characterizes a narrow, hollow tubular structure, termed as duct-like diverticulum (DV), found specifically at the basal midline of papillary craniopharyngiomas (PCPs) located within the third ventricle (3V). The presence of this structure was systematically investigated on autopsy studies and magnetic resonance imaging (MRI) scans of 3536 craniopharyngioma (CP) cases published in the medical literature from 1911 to 2021, as well as in other twelve 3V tumor categories (n = 1470 cases). A basal DV was observed in a total of 50 PCPs, including two of our own cases. This DV corresponds to a tubular-shaped recess invaginated at the midline bottom of the tumor, following the same angled trajectory as the pituitary stalk. It can be easily seen as a hypointense linear structure on T1- and T2-weighted MRI scans, with two main length types: long DVs (74%), which reach the tumor center, and short DVs (26%), which penetrate the tumor only a few millimeters. The DV sign identifies the papillary CP type with a specificity of 100% and a sensitivity of 33% in the overall CP population. This finding also serves to establish the strictly intra-3V location of the lesion with a 95% specificity and 42% sensitivity among papillary CPs. No similar basal DV was found in adamantinomatous CPs nor among other categories of strictly 3V tumors. Consequently, the presence of a diverticulum in a 3V tumor represents a morphological signature pathognomonic of the papillary type and a valuable sign to reliably define the strictly 3V topography.


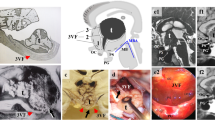
Reproduced from Lei et al. [60], with permission. C H&E-stained slice (× 20) of the tumor specimen from one author’s case showing the typical pseudo-papillary aspect of the sheets of squamous epithelium, with the generation of deep fissures or crevices (black arrow) owing to the normal dehiscence process of the epithelium (without any relation to the DV). D, E Magnetic resonance imaging (MRI) characteristics of the DV in author’s case 1 (number 49; Table 1). The DV is identified as a linear homogeneously hypointense structure which follows a diagonal trajectory along the tumor midline on the T1-weighted MRI scans after gadolinium administration. It is open to the suprasellar cistern and preserves the same signal as the cerebrospinal fluid (black arrows). F Another example of a duct-like basal diverticulum in a solid 3V CP of the squamous-papillary type (author’s case 2, number 50 in Table 1). In this case, the DV ends in a non-bifurcated manner (yellow arrow) as a “cul-de-sac” at the center of the tumor




Similar content being viewed by others
References
Prieto R, Rosdolsky M, Hofecker V, Barrios L, Pascual JM (2020) Craniopharyngioma treatment: an updated summary of important clinicopathological concepts. Expert Rev Endocrinol Metab 15:261–282. https://doi.org/10.1080/17446651.2020.1770081
Louis DN, Perry A, Wesseling P, Brat DJ, Cree IA, Figarella-Branger D et al (2021) The 2021WHO classification of tumors of the central nervous system: a summary. Neuro Oncol 23:1231–1251. https://doi.org/10.1093/neuonc/noab106
Crotty TB, Scheithauer BW, Young WF, David DH, Shaw EG, Miller GM, Burger PC (1995) Papillary craniopharyngioma: a clinicopathological study of 48 cases. J Neurosurg 83:206–214. https://doi.org/10.3171/jns.1995.83.2.0206
Prieto R, Barrios L, Pascual JM (2021) Papillary craniopharyngioma: a type of tumor primarily impairing the hypothalamus. A comprehensive anatomo-clinical characterization of 350 well-described cases. Neuroendocrinology Dec 28 https://doi.org/10.1159/000521652
Pascual JM, Prieto R, Rosdolsky M (2021) Craniopharyngiomas primarily affecting the hypothalamus. In: Swaab DF, Lucassen PJ, Kreier F, Buijs R, Salehi A (ed) The human hypothalamus, Vol 181: Neuroendocrine disorders. In: Aminoff MJ, Voller F, Swaab DF (ed) Handbook of clinical neurology. Third series. Elsevier; pp 75–115. https://doi.org/10.1016/B978-0-12-820683-6.00007-5
Brastianos PK, Taylor-Weiner A, Manley PE, Jones RT, Dias-Santagata D, Thorner AR et al (2014) Exome sequencing identifies BRAF mutations in papillary craniopharyngiomas. Nat Genet 46:161–165. https://doi.org/10.1038/ng.2868
Juratli TA, Jones PS, Wang N, Subramanian M, Aylwin SJB, Odia Y et al (2019) Targeted treatment of papillary craniopharygiomas harboring BRAF V600E mutations. Cancer 125:2910–2914. https://doi.org/10.1002/cncr.32197
Pascual JM, Prieto R, Carrasco R, Barrios L (2013) Displacement of mammillary bodies by craniopharyngiomas involving the third ventricle: surgical-MRI correlation and use in topographical diagnosis. J Neurosurg 119:381–405. https://doi.org/10.3171/2013.1.JNS111722
Prieto R, Pascual JM, Rosdolsky M, Castro-Dufourny I, Carrasco R, Strauss S, Barrios L (2016) Craniopharyngioma adherence: a comprehensive topographical categorization and outcome-related risk stratification model based on the methodical examination of 500 tumors. Neurosurg Focus 41:E13. https://doi.org/10.3171/2016.9.FOCUS16304
Fujio S, Juratli TA, Arita K, Hirano H, Nagano Y, Takajo T et al (2019) A clinical rule for preoperative prediction of BRAF mutation status in craniopharyngiomas. Neurosurgery 85:204–210. https://doi.org/10.1093/neuros/nyy569
Yue Q, Yu Y, Shi Z, Wang Y, Zhu W, Du Z et al (2018) Prediction of BRAF mutation status of craniopharyngioma using magnetic resonance imaging features. J Neurosurg 129:27–34. https://doi.org/10.3171/2017.4.JNS163113
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) PRISMA Group Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLOS Med 6:e1000097. https://doi.org/10.1371/journal.pmed.1000097
Walker CB, Cushing H (1916) Studies of optic-nerve atrophy in association with chiasmal lesions. Arch Ophthal 45:407–437
Hönlinger H, Stricker W (1923) Ein plattenepithelzystopapillom des processus lingualis des hypophysenvorderlappens bei einem falle von hypophysärer kachexie. Frankfurt Zeitschr f Pathol 29:492–518
Cohn H, Goldstein K (1928) Kasuistiche Mitteilungen zur Klinik und pathologischen Anatomie der Nervenkrankheiten. Deutsche Zeitschrift f Nervenheilkunde 103:225–274
Escoroulle R, Poirier J (1971) Tumors arising from embryonal remnants normally absent from the cranial and/or spinal cavity. Tumors of epithelial origin. Craniopharyngiomas. In: Manual of Basic Neuropathology. W. B. Saunders company; pp 63–64.
Kubota T, Yamamoto S, Kohno H, Ito H, Hayashi M (1980) Operative procedures of craniopharyngioma estimated by autopsy findings. Neurol Med Chir (Tokyo) 20:341–354. https://doi.org/10.2176/nmc.20.341
Schmidt B, Gherardi R, Poirier J, Caron JP (1984) Craniopharyngiome pédiculé du troisième ventricule. Rev Neurol 140:281–283
Giangaspero F, Burger PC, Osborne DR, Stein RB (1984) Suprasellar papillary squamous epithelioma (“papillary craniopharyngioma”). Am J Surg Pathol 8:57–64. https://doi.org/10.1097/00000478-198401000-00006
Rubin E, Farber JL (eds) (1988) Pathology. Philadelphia, J.B. Lippincott Company; p. 1444
Kurian K, Moss TH, Camelo-Piragua S (2014) Atlas of gross neuropathology. A practical approach. Cambridge, Cambridge University Press; pp 221.
Balcázar-Hernandez L, Vargas-Ortega G, Valverde-García Y, Mendoza-Zubieta V, González-Virla B (2017) Anorexia-cachexia syndrome-like hypothalamic neuroendocrine dysfunction in a patient with a papillary craniopharyngioma. Endocrinol Diabetes Metab Case Rep 2017:17–0018. https://doi.org/10.1530/EDM-17-0018
Fukushima T, Hirakawa K, Kimura M, Tomonaga M (1990) Intraventricular craniopharyngioma: its characteristics in magnetic resonance imaging and successful total removal. Surg Neurol 33:22–27. https://doi.org/10.1016/0090-3019(90)90220-j
Yasargil MG (1994) Microneurosurgery, vol IV-A: microneurosurgery of CNS tumors. Georg Thieme Verlag, Sttutgart-New York; pp 202–211.
Sartoretti-Schefer S, Wichmann W, Aguzzi A, Valavanis A (1997) MR differentiation of adamantinous and squamous-papillary craniopharyngiomas. AJNR Am J Neuroradiol 18:77–87
Prasad D, Steiner M, Steiner L (1995) Gamma knife surgery for craniopharyngioma. Acta Neurochir (Wien) 134:167–176. https://doi.org/10.1007/BF01417685
Reul J, Weis J, Spetzger U, Isensee Ch, Thron A (1995) Differential diagnosis of truly suprasellar space-occupying masses: synopsis of clinical findings, CT, and MRI. Eur Radiol 5:224–237
Shibuya M, Takayasu M, Suzuki Y, Saito K, Sugita K (1996) Bifrontal basal interhemispheric approach to craniopharyngioma resection with or without division of the anterior communicating artery. J Neurosurg 84:951–956. https://doi.org/10.3171/jns.1996.84.6.0951
Urbach H, Behrens E, von Deimling A, Reul J (1998) Solides Kraniopharyngiom im III. Ventrikel-Differential-diagnostiche Aspekte. Aktuelle Radiol 8:95–97
Honegger J, Grabenbauer GG, Paulus W, Fahlbusch R (1999) Regression of a large solid papillary craniopharyngioma following fractionated external radiotherapy. J Neurooncol 41:261–266. https://doi.org/10.1023/a:1006112600805
Perneczky A (1999) Keyhole concept in neurosurgery with endoscope-assisted microsurgery and case studies. Perneczky A et al (eds) Thieme, Stuttgart-New York.
Maira G, Anile C, Colosimo C, Cabezas D (2000) Craniopharyngiomas of the third ventricle: trans-lamina terminalis approach. Neurosurgery 47:857–865. https://doi.org/10.1097/00006123-200010000-00014
Barlas O, Bayindir C, Can M (2000) Interstitial irradiation for craniopharyngioma Acta Neurochir (Wien) 142:389–395. https://doi.org/10.1007/s007010050447
Chung WY, Pan DH, Shiau CY, Guo W-Y, Wang L-W (2000) Gamma knife radiosurgery for craniopharyngiomas. J Neurosurg 93(Suppl 3):47–56. https://doi.org/10.3171/jns.2000.93.supplement
Madhavan M, George PJ, Jafri JA, Idris Z (2005) Intraventricular squamous papillary craniopharyngioma: report of a case with intraoperative imprint cytology. Acta Cytol 49:431–434. https://doi.org/10.1159/000326179
Durmaz R, Vural M, Kabukcuoglu S, Atasoy MA (2007) Intraventriküler skuamöz-papiller kraniofarenjioma [Intraventricular squamous-papillary craniopharyngioma]. Osmangazi Tip Dergisi 29:53–59
Burger P, Scheithauer BW (Eds) (2007) Craniopharyngioma. In Burger P, Scheithauer BW (Eds) Tumors of the central nervous system. AFIP Atlas of Tumor Pathology, 4th Series, Fascicle 7. Washington DC, American Registry of Pathology in collaboration with the Armed Forces Institute of Pathology; pp 461–469.
Prieto Gonzalez A (2007) Craneofaringioma. In: Gelabert Gonzalez M (ed) Casos Clínicos en Neurocirugía. Instituto Universitario de Ciencias Neurologicas “Pedro Barrié de la Maza”. Universidad de Santiago de Compostela; p 85.
Stanislavsky A (2010) Craniopharyngioma — 3rd ventricle. Case study, Radiopaedia.org (Accessed on March 31, 2020) https://radiopaedia.org/cases/craniopharyngioma-3rd-ventricle-2
Pan J, Qi S, Lu Y, Fan J, Zhang X, Zhou J, Peng J (2011) Intraventricular craniopharyngioma: morphological analysis and outcome evaluation of 17 cases. Acta Neurochir (Wien) 153:773–784. https://doi.org/10.1007/s00701-010-0938-5
Qi S, Pan J, Lu Y (2017) Craniopharyngiomas — classification and surgical treatment. Frontiers in Neurosurgery. Vol 4. Sharjah, U.A.E., Bentham Science Publishers Ltd.; pp 261–266.
Kenning TJ, Evans JJ (2012) Fractionated stereotactic radiosurgery alone for the treatment of a papillary craniopharyngioma. JHN Journal 7:1–5
Patel GV, Karimi S, Young RJ (2013) Sellar and parasellar masses. In: Karimi S (ed) Atlas of brain and spine oncology imaging, Atlas of Oncology Imaging. Springer Science + Business Media, New York, pp 109–140
Lee CC, Yang HC, Chen CJ, Hung Y-C, Wu H-M, Shiau C-Y et al (2014) Gamma knife surgery for craniopharyngioma: report on a 20-year experience. J Neurosurg 121(Suppl):167–178. https://doi.org/10.3171/2014.8.GKS141411
Lee HJ, Wu CC, Wu HM, Hung S-C, Lirng J-F, Luo C-B et al (2015) Pretreatment diagnosis of suprasellar papillary craniopharyngioma and germ cell tumors of adult patients. AJNR Am J Neuroradiol 36:508–517. https://doi.org/10.3174/ajnr.A4142
Gu Y, Zhang X, Hu F, Yu Y, Xie T, Sun C, Li W (2015) Suprachiasmal translamina terminalis corridor used in endoscopic endonasal approach for resecting third ventricular craniopharyngioma. J Neurosurg 122:1166–1172. https://doi.org/10.3171/2015.1.JNS132842
Tosaka M, Sato K, Amanuma M, Higuchi T, Arai M, Aishima K et al (2015) Superficial siderosis of the central nervous system caused by hemorrhagic intraventricular craniopharyngioma: Case report and literature review. Neurol Med Chir (Tokyo) 55:89–94. https://doi.org/10.2176/nmc.cr.2012-0362
Rambarki O, Rajesh A (2016) Third ventricular craniopharyngioma. Neurol India 64:834–835. https://doi.org/10.4103/0028-3886.185370
Nishioka H, Fukuhara N, Yamaguchi-Okada M, Yamada S (2016) Endoscopic endonasal surgery for purely intrathird ventricle craniopharyngioma. World Neurosurg 91:266–271. https://doi.org/10.1016/j.wneu.2016.04.042
Stoffey RD, Sitton CW, Riascos RF, Bhattacharjee MB (2016) Papillary craniopharyngioma presenting as a purely intraventricular lesion in an adult. 2016, EE-21. https://slideplayer.com/slide/13504069/ Accessed April 2, 2020.
Prieto R, Pascual JM, Barrios L (2017) Topographic diagnosis of craniopharyngiomas: the accuracy of MRI findings observed on conventional T1 and T2 images. AJNR Am J Neuroradiol 38:2073–2080. https://doi.org/10.3174/ajnr.A5361
Conger A, Zhao F, Wang X, Eisenberg A, Griffiths C, Esposito F et al (2018) Evolution of graded repair of CSF leaks and skull base defects in endonasal endoscopic tumor surgery: trends in repair failure and meningitis rates in 509 patients. J Neurosurg 130:861–875. https://doi.org/10.3171/2017.11.JNS172141
Tang B, Xie SH, Xiao LM, Huang GL, Wang ZG, Yang L et al (2018) A novel endoscopic classification for craniopharyngioma based on its origin. Sci Rep 8:10215. https://doi.org/10.1038/s41598-018-28282-4
Concord Hospital. Anatomical Pathological Museum. T9100 – M9351/1 – pituitary – craniopharyngioma (2018); https://www.slhd.nsw.gov.au/concord/Anatpath/content//pdf/ENDOCRINE/T9100_M9351_1.pdf?07042016030448. Accessed April 3, 2020.
Diniz LV, Junior LAJ Jr, Rodrigues LP, Veloso JV, Yamashita S (2018) Intraventricular craniopharyngioma: a case report. J Neurol Stroke 8:185–188
Oh JH, Park ST, Lim HK (2018) Craniopharyngioma with intratumoral hemorrhage and superficial siderosis. iMRI 22: 249–253.
Chakravarthi SS, Kassam AB, Fukui MB, Monroy-Sosa A, Rothong N, Cunningham J et al (2019) Awake surgical management of third ventricular tumors: a preliminary safety, feasibility, and clinical applications study. Oper Neurosurg (Hagerstown) 17:208–226. https://doi.org/10.1093/ons/opy405
Pan J. What role does craniotomy play in the endoscope era? (2019) Lecture read at The International Summit Meeting for Redefining the WHO definition of Craniopharyngioma. Guangzhou, China, Nov 17, 2019.
Lei C, Chuzhong L, Chunhui L, Peng Z, Jiwei B, Xinsheng W, Yazhuo Z, Songbai G (2021) Approach selection and outcomes of craniopharyngioma resection: a single-institute study. Neurosurg Rev 44:1737–1746. https://doi.org/10.1007/s10143-020-01370-8
Gaillard F. Papillary craniopharyngioma – 3rd ventricle (2021) Case study, Radiopaedia.org (Accessed on April 12, 2022) https://doi.org/10.53347/rID-88982
Cao L, Wu W, Kang J, Qiao H, Yang X, Bai J et al (2021) Expanded transsphenoidal trans-lamina terminalis approach to tumors extending into the third ventricle: technique notes and a single institute experience. Front Oncol 11:761281. https://doi.org/10.3389/fonc.2021.761281
Cao L, Wu W, Kang J, Cai K, Li C, Zhu H, Gen S, Zhang Y, Gui S (2022) Feasibility of endoscopic endonasal resection of intrinsic third ventricular craniopharyngioma in adults. Neurosurg Rev May 22. https://doi.org/10.1007/s10143-022-01807-2
Castro-Dufourny I, Carrasco R, Prieto R, Barrios L, Pascual JM (2015) The infundíbulo-tuberal syndrome caused by craniopharyngiomas: clinicopathological evidence from a historical French cohort (1705–1973). Pituitary 18:642–657. https://doi.org/10.1007/s11102-014-0623-4
Pascual JM, Gonzalez-Llanos F, Barrios L, Roda JM (2004) Intraventricular craniopharyngiomas: topographical classification and surgical approach selection based on an extensive overview. Acta Neurochir (Wien) 145:785–802. https://doi.org/10.1007/s00701-004-0295-3
Prieto R, Barrios L, Pascual JM (2022) Strictly third ventricle craniopharyngiomas: pathological verification, anatomo-clinical characterization and surgical results from a comprehensive overview of 245 cases. Neurosurg Rev 45:375–394. https://doi.org/10.1007/s10143-021-01615-0
Pascual JM, Prieto R, Castro-Dufourny I, Mongardi L, Rosdolsky M, Strauss S, Carrasco R, Barrios L (2018) Craniopharyngiomas primarily involving the hypothalamus: a model of neurosurgical lesions to elucidate the neurobiological basis of psychiatric disorders. World Neurosurg 120:e1245–e1278. https://doi.org/10.1016/j.wneu.2018.09.053
Prieto R, Pascual JM, Barrios L (2015) Optic chiasm distortions caused by craniopharyngiomas: clinical and magnetic resonance imaging correlation and influence on visual outcome. World Neurosurg 83:500–529. https://doi.org/10.1016/j.wneu.2014.10.002
Merriam-Webster’s Medical Dictionary. By Merriam-Webster (2016) Merriam-Webster, U.S. An Encyclopedia Britannica Company. Springfield, MA; 2016.
Chun IKH, Ojumah N, Loukas M, Oskouian RJ, Tubbs RS (2018) Martin Heinrich Rathke (1793–1860) and his pouch. Childs Nerv Syst 34:377–379. https://doi.org/10.1007/s00381-017-3482-2
Castellanos LE, Gutierrez C, Smith T, Laws ER, Iorgulesco JB (2022) Epidemiology of common and uncommon adult pituitary tumors in the U.S. according to the 2017 World Health Organization classification. Pituitary 25:201–209. https://doi.org/10.1007/s11102-021-01189-6
Momin AA, Recinos MA, Cioffi G, Patil N, Soni P, Almeida JP et al (2021) Descriptive epidemiology of craniopharyngiomas in the United States. Pituitary 24:517–522. https://doi.org/10.1007/s11102-021-01127-6
Pascual JM, Prieto R, Carrasco R (2011) Infundibulo-tuberal or not strictly intraventricular craniopharyngioma: evidence for a major topographical category. Acta Neurochir (Wien) 153:2403–2426. https://doi.org/10.1007/s00701-011-1149-4
Glastonbury CM, Osborn AG, Salzman KL (2011) Masses and malformations of the third ventricle: normal anatomic relationships and differential diagnoses. Radiographics 31:1889–1905. https://doi.org/10.1148/rg.317115083
Zülch KJ (1961) Die pathologie und biologie der tumoren des dritten ventrikels. Acta Neurochir (Wien) 9:277–296
Jung TY, Jung S, Jang WY, Moon KS, Kim IY, Kang SS (2012) Operative outcomes and adjuvant treatment of purely third ventricle craniopharyngioma after a transcallosal approach. Br J Neurosurg 26:355–360. https://doi.org/10.3109/02688697.2011.631615
Sartor K (1995) MR Imaging of the Skull and Brain. A Correlative Text-Atlas. Berlin, Springer-Verlag; pp 397–403.
Mott FW, Barratt JOW (1899) Three cases of tumor of the third ventricle. Arch Neurol (Lond) 1:417–439
Pascual JM, Prieto R, Rosdolsky M, Strauss S, Castro-Dufourny I, Hofecker V et al (2018) Cystic tumors of the pituitary infundibulum: seminal autopsy specimens (1899 to 1904) that allowed clinical-pathological craniopharyngioma characterization. Pituitary 21:393–405. https://doi.org/10.1007/s11102-018-0889-z
Erdheim J (1904) Über Hypophysengangsgeschwülste und Hirncholesteatome. Sitzungsb Kais Akad Wissen Math Naturw Klasse 113:537–726
Pascual JM, Rosdolsky M, Prieto R, Strauss S, Winter E, Ulrich W (2015) Jakob Erdheim (1874–1937): father of hypophyseal-duct tumors (craniopharyngiomas). Virchows Arch 467:459–469. https://doi.org/10.1007/s00428-015-1798-4
Ciric IS, Cozzens JW (1980) Craniopharyngiomas: transsphenoidal method of approach — for the virtuoso only? Clin Neurosurg 27:169–187
Tilney F (1936) The development and constituents of the human hypophysis. Bull Neurol Inst N.Y. 5: 387–436.
Kollias SS, Ball WS, Prenger EC (1995) Review of the embryologic development of the pituitary gland and report of a case with hypophyseal duplication detected by MRI. Neuroradiology 37:3–12. https://doi.org/10.1007/BF00588511
Martinez-Barbera JP, Andoniaou CL (2016) Concise review: paracrine role of stem cell in pituitary tumors: a focus on adamantinomatous craniopharyngioma. Stem Cells 34:268–276. https://doi.org/10.1002/stem.2267
Harrison MJ, Morgello S, Post KD (1994) Epithelial cystic lesions of the sellar and parasellar region: a continuum of ectodermal derivatives? J Neurosurg 80:1018–1025. https://doi.org/10.3171/jns.1994.80.6.1018
Prieto R, Pascual JM (2013) Craniopharyngiomas with a mixed histological pattern: the missing link to the intriguing pathogenesis of adamantinomatous and squamous-papillary varieties? Neuropathology 33:682–686. https://doi.org/10.1111/neup.12030
Abele TA, Salzman KL, Harnsberger HR, Glastonbury CM (2013) Craniopharyngeal canal and its spectrum of pathology. AJNR Am J Neuroradiol 35:772–777. https://doi.org/10.3174/ajnr.A3745
Belotti F, Lupi I, Cosottini M, Ambrosi C, Gasparotti R, Bogazzi F et al (2018) Persisting embryonal infundibular recess (PEIR): two case reports and systematic literature review. J Clin Endocrinol Metab 103:2424–2429. https://doi.org/10.1210/jc.2018-00437
Pascual JM, Prieto R (2022) Letter to the Editor: Craniopharyngioma in the elderly: the crucial influence of tumor topography on surgical risk assessment. Neuroendocrinology 112:196–200. https://doi.org/10.1159/000516172
Prieto R, Pascual JM (2018) Can tissue biomarkers reliably predict the biological behavior of craniopharyngiomas? A comprehensive overview. Pituitary 21:431–442. https://doi.org/10.1007/s11102-018-0890-6
Acknowledgements
The authors wish to especially thank Crystal Smith and Liliya Gusakova, Reference Librarians of the National Library of Medicine, National Institutes of Health (Bethesda, MD), for their kind assistance during the process of searching and retrieving articles and monographs used in this study. We are also grateful to Melissa Grafe, Librarian for Medical History Library, Yale University (New Haven, CT), and to Lucretia MacLure, Jack Eckert, and the staff at the Francis Countway Medical Library at Harvard Medical School (Boston, MA) for their invaluable help in obtaining some of the original research material used for this study. We thank Professor Kenneth Lee, Head of Anatomical Pathology Department at Concord Repatriation General Hospital, who has gently permitted us to reproduce the autopsy image of the papillary CP shown in Figure 2A. Finally, we are grateful to George Hamilton for his critical review of the language and style of the manuscript.
Author information
Authors and Affiliations
Contributions
Conception and design: José M. Pascual. Acquisition of data: José M. Pascual, Ruth Prieto and Rodrigo Carrasco. Analysis of data: José M. Pascual, Laura Barrios; drafting the article: José M. Pascual, Ruth Prieto; critically revising the article: all authors.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable as this study consists of a review of previously published literature.
Human and animal ethics
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Authors’ information
José María Pascual, M.D., Ph.D.
Department of Neurosurgery, La Princesa University Hospital, Madrid, Spain.
e-mail: jmpasncj@hotmail.com.
Ruth Prieto, M.D., Ph.D.
Department of Neurosurgery, Puerta de Hierro University Hospital, Madrid Spain.
e-mail: rprieto29@hotmail.com.
Rodrigo Carrasco, M.D.
Department of Neurosurgery, Ramón y Cajal University Hospital, Madrid, Spain.
e-mail: rocamo@gmail.com.
Laura Barrios, B.S.
Statistics Department, Computing Center, C.S.I.C. Madrid, Spain.
e-mail: laura.barrios@csic.es.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pascual, J.M., Prieto, R., Carrasco, R. et al. Duct-like diverticulum at the base of third ventricle tumors: a morphological signature diagnostic of papillary craniopharyngioma. Neurosurg Rev 45, 3361–3379 (2022). https://doi.org/10.1007/s10143-022-01848-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-022-01848-7