Abstract
The aim of this case series was to provide a modern cohort of patients with cerebral aspergillosis and show the effectiveness of modern treatment concepts. In a 10-year period from January 2009 to January 2019, we identified 10 patients (6 male, 4 female) who received surgery or frameless stereotactic drainage of a cerebral aspergilloma at our center. Patients’ and disease characteristics were recorded. The median age was 65 (range 45 to 83). We conducted 133 cranial surgeries in 100 patients due to cerebral brain abscess (BA) during that time, which leads to a percentage of 10% of aspergilloma within BAs in our patient sample. We performed 3.1 surgeries per patient followed by antifungal treatment for 6 months (= median) according to the microbiological findings. Regarding comorbidities, the mean Charlson comorbidity index (CCI) at the time of admission was 5, representing an estimated 10-year survival of 21%. Six (60%) of 10 patients showed conditions of immunosuppression, one suffered endocarditis after replacement of aortic valves. Four patients showed associated frontobasal bone destruction, mycotic aneurysms, or thromboses. The mean duration of hospital stay was 37 days. Mortality was much lower than in literature. Sixty percent of the patients died during the follow-up period. The outcome of the two immunocompetent patients was more favorable. Cerebral aspergillosis is a rare, but still life-threatening, condition, which predominantly occurs in immunosuppressive conditions. Due to radical surgical and antifungal therapy for several months, mortality can be reduced dramatically.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Aspergillosis is a rare type of infection caused by Aspergillus fumigatus, an ubiquitous mold (a fungal organism) of which spores are an inherent component of breathing air [3]. It primarily leads to infection in patients with underlying immunosuppression, such as autoimmune diseases, hematopoietic stem cell or solid organ transplantations, or tumor diseases. Incidence estimates of 1992 in the USA suggested a yearly rate of two cases of aspergillosis per 100,000 population [34]. Aspergillus fumigatus is the most common species in human infections. Once the central nervous system (CNS) is infected, the prognosis is poor [10, 37]. Thereby, brain involvement was only found in 3% of all patients with aspergillosis, and in 20% and up to 42% within the subgroups of previously diagnosed tumor disease, or patients with acute leukemia and hematopoietic stem cell transplantation [4, 19, 28, 40]. Brain involvement most commonly results in brain abscess formation, occasionally accompanied by cerebral infarction due to septic embolisms, or associated mycotic aneurysms [5, 17, 18, 21, 25, 28]. Prognosis of cerebral aspergillosis is poor. A literature review of 1996 revealed a mortality rate of 99% within 141 patients of this particular type of intracerebral mold infection [11]. In 2001, the case fatality rate (CFR) was estimated 88% [23]. Literature shows that therapy concepts of successfully treated aspergillomas contain radical surgical excision of affected tissue and long-term antimycotic therapy [9, 20]. More recent data of patients with cerebral aspergilloma is rare and mostly within case reports.
Pseudallescheria boydii, its asexual form known as Scedosporium apiospermum, is a fungus belonging to the Ascomycota. It is typically found in water and is an emerging opportunistic pathogen. It is the second most common fungus after Aspergillus fumigatus found in patients with cystic fibrosis. In immunocompromised hosts, infection can occur in the lung, skin, bones, and CNS. CNS infection is a very rare incident, and the prognosis is poor [27].
The aim of this case series was to provide a modern cohort of patients with cerebral aspergillosis, assess the clinical characteristics, and reveal the relevant aspects for future therapy for such a condition. Furthermore, we aimed to show the effectiveness of our treatment by analyzing our patients’ clinical outcomes and to identify some risk factors for a poor outcome of cerebral aspergillosis.
Materials and methods
Patient population
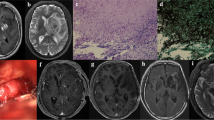
We identified all patients receiving surgery or frameless stereotactic drainage of a cerebral aspergilloma in our center in a 10-year period from January 2009 to January 2019. We included patients with suspect lesions in preoperative MRI or CT scans, which showed isolated fungi of the genus Aspergillus fumigatus from the brain tissue and histopathologically proven signs of fungal infection. The present study was approved by the local ethics committee and performed in accordance with the ethical standards established by the 1964 Declaration of Helsinki and its later amendments [39] (Clinical Trial Registration Number: 217/16S).
Patients’ and disease characteristics were recorded, including age, mortality, dates and types of surgical procedures, neurological symptoms, and length and type of antifungal treatment. Germ spectrum and immunosuppressive conditions were also assessed.
Outcome
For all patients, the CCI [7] was calculated at the time of admission. The clinical parameters were the Barthel index (BI) [36] at the time of admission and after surgery, as well as the modified Rankin scale (MRS) [32] at the time of admission, discharge, and several times during the follow-up period (3 months, 1 year, 2 years, and 5 years). Data regarding the antifungal treatment, germ spectrum, and BI was taken from our database. As a retrospective analysis, patients’ informed consent was not necessary.
Statistical data analysis
Statistical analyses, including descriptive data analyses, were performed using IBM SPSS Statistics version 22.0 (IBM Corporation, New York). Associations between all assessed variables were analyzed using chi-square tests. To identify possible risk factors for outcome changes, logistic regression analysis was done. For all analyses, a difference with an error probability of less than 0.05 was considered statistically significant. Descriptive statistics for demographic variables were generated with means and SDs or medians with interquartile ranges as appropriate.
Results
Clinical characteristics of the patients are presented in Table 1.
In a 10-year period from January 2009 to January 2019, we identified 10 patients (6 male, 4 female) who received surgery or frameless stereotactic drainage of a cerebral aspergilloma at our center. We conducted 133 cranial surgeries in 100 patients due to cerebral brain abscess (BA) during that time, which leads to a percentage of 10% of aspergilloma within BAs in our patient sample.
The mean age was 65 years (range 45–83).
Regarding comorbidities, mean CCI at the time of admission was 4.8 representing an estimated 10-year survival of 21% [8]. Six (60%) of 10 patients showed conditions of immunosuppression. Those are presented in Table 2 (B cell lymphoma, renal cell carcinoma, hematopoietic stem cell transplantation, long-term cortisone because of glomerulonephritis, B ALL, and Tolosa-Hunt syndrome). Of the remaining four patients, one suffered severe endocarditis and underwent surgical replacement of aortic and tricuspid valves 1 month before septic cerebral aspergillosis was detected. Relevant comorbidities of this patient were diabetes and COPD. Another patient suffered severe sepsis due to perforation of sigmoid diverticulitis with multiple abdominal operations 3 months before the development of brain abscess. Two patients seemed to be immunocompetent. Further, 5 patients (50%) showed solitaire abscesses, and in 5 patients (50%) multiple BAs could be identified.
Symptoms leading to diagnosis were of wide range: paresis in 40%, visual impairment and headache in 30%, paresis of cranial nerves (oculomotorius), vigilance decrement, and fever in 20% and in 10%, aphasia, disturbances in fine motor skills, loss of olfactory sense, gait disorders, and incidental findings on CT scans.
Infection parameters, measured by C reactive protein (CRP; standard values < 0.5 mg/dl) levels at the time of admission, were elevated in most of the patients (70%), showing mean CRP levels of 1.6 (SD 1.6).
Surgical procedures
In total, we performed 31 surgical procedures (3.1 per patient). Abscess evacuations (23 surgeries) were conducted via open craniotomies in 14 surgeries (61%), VarioGuide aspiration in 5 (22%), transnasal in 4 (17%). Additional four surgeries were performed for treatment of aspergilloma-associated pathologies (2 bypass surgeries for associated aneurysms, 2 frontobasal reconstructions for persisting frontobasal defects). The remaining four procedures were due to complications (3 evacuations of postoperative hematomas, 1 ventriculoperitoneal-shunt-implantation because of hydrocephalus malresorptivus). In 4 cases, craniotomy was chosen because frontobasal reconstruction was needed in addition to abscess evacuation. One case required a split bone graft.
One patient needed endovascular thrombectomy and stenting due to septic embolism of ACI. Initial surgical procedures and patient presentations were emergency procedures because of rapid deterioration of the patients in 80%.
There were no cases of operative mortality.
Microbiological findings
Analysis of the abscess tissue showed Aspergillus fumigatus in nine cases and Pseudallescheria boydii in one case. Four patients showed additional germs (E. coli, Staph aureus, herpes simplex virus). In our patient samples, diagnosis was made through microscopy and culture in 6 patients. Four samples were analyzed via PCR in addition.
In every patient, focus identification was conducted via the consultation of cardiologists (including transesophageal echocardiography); gynecologists or urologists; oral and maxillofacial surgeons; ear, nose, and throat (ENT) physicians; thoracic X-ray; and urine analysis. The focus for aspergilloma was the former surgical site in 3 cases and unknown in 7 cases.
Treatment was with voriconazole (azole antimycotic) in 6 cases, voriconazole and amphotericin B (streptomycetes) in 3 cases, and in one case with caspofungin (echinocandin). Additional virostatic medication was given in one patient and antibiotics in two patients. The mean duration of treatment was 5.8 months (SD 5.2).
Analysis of blood cultures was done in all cases and turned out to be negative.
A reliable diagnosis of fungal infection is challenging.
Both microscopy and culture should be attempted on appropriate specimens from patients at risk for IA (AII) with a priority for culture in most cases where insufficient material is available. Demonstrating tissue invasion by hyphae through microscopic examination of biopsy or autopsy material provides a diagnosis of proven invasive fungal infection. However, the sensitivity of microscopy for IA is 50% at best [14, 35].
PCR approach can increase the sensitivity to 57–> 90% depending on the amount of fungi in the sample. The culture and subculture of fungi contain Sabouraud dextrose agar; identification is performed by microscopy or sequencing of the fungus. Isolates are screened for azole resistance using supplemented plates with voriconazole and itraconazole. All isolates of Aspergillus fumigatus were azole susceptible. The isolate of Pseudallescheria boydii is known to be amphotericin-resistant; the patient was treated with voriconazole.
Outcome
Figure 1 shows survival curves of our patient collective and the patients with primary brain abscess due to bacteria are treated in our center during the same period. Log-rank test shows no significant differences in survival curves: HR 0.43, 95% CI (0.1053–1.735).
The overall mortality was 60%. Half of the patients died during the first 2 months of treatment. Deaths were all associated with aspergilloma, leading to septic embolisms and multi-organ failure in most cases. All patients could be followed, so there were no patients lost to follow-up.
The mean MRS at the time of admission, discharge, and after 1 year was 3, 4, and 5, respectively. The mean length of hospital stay was 37 days.
Discussion
In this analysis of 10 patients, treated at our center between 2009 and 2019 for cerebral aspergillosis, we assessed clinical characteristics, outcomes, and survival rates of this patient collective.
Cerebral aspergillosis stays a very rare condition. We conducted 133 cranial surgeries in 100 patients due to cerebral brain abscess (BA) during that time, which leads to a percentage of 10% of aspergilloma within BAs in our patient sample.
Regarding comorbidities, the mean CCI at the time of admission was 5, representing an estimated 10-year survival of 21% [8]. Six (60%) of 10 patients showed conditions of immunosuppression. This is in good accordance with the literature, which describes disseminated aspergillosis and CSF infection much more common in an immunocompromised host than in healthy persons [26]. It is seen as an opportunistic infection in AIDS, but also in organ transplantation patients, in neutropenia associated with acute leukemia and chemotherapy, and after long-term steroid therapy [15]. In our patient collective, two patients seemed to be immunocompetent, without any preexisting conditions favoring fungal infections. Abscesses were both located in the maxillar sinus and lead to frontobasal destruction invading the frontal lobe. They both survived after surgical resection and long-term antimycotic therapy. Thus, the mechanisms for those infections stay unclear, the number of patients seems to be rising, while prognosis in those patients is more favorable [6, 12].
The overall mortality of patients with cerebral fungal infection was 60%. During the same period, we analyzed all patients with primary brain abscesses treated in our center. In that collective, 23% died during the follow-up period [21]. This underlines an infection with fungi as a still very severe condition, even though log-rank test could not show a significant difference due to the small sample size. In the literature, mortality with fungal brain abscess ranges between 80 and 99%. This shows that modern treatment strategies as shown in this case series, comprising radical operative resections followed by long-term antimycotic therapies, are able to reduce mortality in this modern era dramatically [1, 11, 23]. Of course, this reduction of mortality can not only be ascribed to radical surgical removal of affected tissue but also to the development of new antifungal therapy as voriconazole. Taking into account some case reports describing patients recovering with long-term antifungal therapy and surgical aspirations only but on the other hand descriptions of voriconazole-refractory invasive aspergillosis, requiring radical surgical resections and even lobectomies, therapy concepts need to be more individual and patient specific [2, 16, 24, 29]. Reduction of infection via stereotactic aspiration or open surgical removal is required in every case. In this collective, every patient with stereotactic biopsy had to undergo open surgical removal of abscess tissue some weeks later due to progression in follow-up MRI despite voriconazole therapy. Nowadays, radical excision of abscess tissue is a safe procedure, leaving patients in a good clinical condition which they need for a long antifungal treatment with many side effects [22]. Finally, diagnosis can be placed fast and earlier due to new imaging techniques and their broad availability.
Histopathologically, single or multiple abscess formations with invasion of blood vessels leading to thromboses, septic embolisms, or mycotic aneurysms are characteristics of aspergillosis [13, 30, 31]. In accordance with this, two of our patients suffered thromboses of internal carotid artery (one needed thromboendarterectomy): one needed bypass surgery for a huge mycotic aneurysm of the anterior communicating artery. The outcome of those patients was fatal.
Aspergillosis usually manifests with acute onset of focal neurologic deficits, confirming that 80% of initial surgical procedures were emergency procedures because of rapid deterioration of the patients. Symptoms are often of wide range, as described previously [33, 38]. Interestingly, one of our patients showed no symptoms at all. Multiple aspergillomas were detected during routine staging. He suffered neutropenia because of ALL and following chemotherapy. Routine surgery was planned, while 1 day prior to surgery date, he developed hemiparesis.
Conclusion
Cerebral aspergillosis is a rare, but still life-threatening, condition, which predominantly occurs in immunosuppressive conditions. Due to radical surgical removal of infected tissue and antifungal therapy for several months, mortality can be reduced dramatically.
References
Alahmari AF (2019) Medical treatment of brain aspergilloma followed by MRI: a case report. Radiology case reports 14:103–111. https://doi.org/10.1016/j.radcr.2018.09.021
Aljuboori Z, Hruska R, Yaseen A, Arnold F, Wojda B, Nauta H (2017) Fungal brain abscess caused by “black mold” (Cladophialophora bantiana) - a case report of successful treatment with an emphasis on how fungal brain abscess may be different from bacterial brain abscess. Surg Neurol Int 8:46. https://doi.org/10.4103/sni.sni_448_16
Barnes PD, Marr KA (2006) Aspergillosis: spectrum of disease, diagnosis, and treatment. Infect Dis Clin N Am 20(545–561):vi–561. https://doi.org/10.1016/j.idc.2006.06.001
Bodey G, Bueltmann B, Duguid W, Gibbs D, Hanak H, Hotchi M, Mall G, Martino P, Meunier F, Milliken S, Naoe S, Okudaira M, Scevola D, Wout J (1992) Fungal infections in cancer patients: an international autopsy survey. Eur J Clin Microbiol Infect Dis 11:99–109
Boes B, Bashir R, Boes C, Hahn F, McConnell JR, McComb R (1994) Central nervous system aspergillosis. Analysis of 26 patients. J Neuroimaging 4:123–129
Bokhari R, Baeesa S, Al-Maghrabi J, Madani T (2014) Isolated cerebral aspergillosis in immunocompetent patients. World Neurosurg 82:e325–e333. https://doi.org/10.1016/j.wneu.2013.09.037
Charlson M, Szatrowski TP, Peterson J, Gold J (1994) Validation of a combined comorbidity index. J Clin Epidemiol 47:1245–1251. https://doi.org/10.1016/0895-4356(94)90129-5
Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40:373–383
Coleman JM, Hogg GG, Rosenfeld JV, Waters KD (1995) Invasive central nervous system aspergillosis: cure with liposomal amphotericin B, itraconazole, and radical surgery--case report and review of the literature. Neurosurgery 36:858–863
Dagenais TR, Keller NP (2009) Pathogenesis of Aspergillus fumigatus in invasive aspergillosis. Clin Microbiol Rev 22:447–465. https://doi.org/10.1128/cmr.00055-08
Denning DW (1996) Therapeutic outcome in invasive aspergillosis. Clin Infect Dis 23:608–615
Ellenbogen JR, Waqar M, Cooke RP, Javadpour M (2016) Management of granulomatous cerebral aspergillosis in immunocompetent adult patients: a review. Br J Neurosurg 30:280–285. https://doi.org/10.3109/02688697.2016.1139046
Forest F, Cinotti E, Habougit C, Ginguene C, Perrot JL, Labeille B, Flori P, Botelho-Nevers E, Peoch M (2016) Rapid characterization of human brain aspergillosis by confocal microscopy on a thick squash preparation. Cytopathol 27:221–222. https://doi.org/10.1111/cyt.12258
Guinea J, Verweij PE, Meletiadis J, Mouton JW, Barchiesi F, Arendrup MC (2019) How to: EUCAST recommendations on the screening procedure E.Def 10.1 for the detection of azole resistance in Aspergillus fumigatus isolates using four-well azole-containing agar plates. Clin Microbiol Infect 25:681–687. https://doi.org/10.1016/j.cmi.2018.09.008
Hall WA (1992) Neurosurgical infections in the compromised host. Neurosurg Clin N Am 3:435–442
Hicks JK, Gonzalez BE, Zembillas AS, Kusick K, Murthy S, Raja S, Gordon SM, Hanna R (2016) Invasive aspergillus infection requiring lobectomy in a CYP2C19 rapid metabolizer with subtherapeutic voriconazole concentrations. Pharmacogenomics 17:663–667. https://doi.org/10.2217/pgs-2015-0014
Ho CL, Deruytter MJ (2004) CNS aspergillosis with mycotic aneurysm, cerebral granuloma and infarction. Acta Neurochir 146:851–856. https://doi.org/10.1007/s00701-004-0292-6
Hurst RW, Judkins A, Bolger W, Chu A, Loevner LA (2001) Mycotic aneurysm and cerebral infarction resulting from fungal sinusitis: imaging and pathologic correlation. AJNR Am J Neuroradiol 22:858–863
Jantunen E, Volin L, Salonen O, Piilonen A, Parkkali T, Anttila VJ, Paetau A, Ruutu T (2003) Central nervous system aspergillosis in allogeneic stem cell transplant recipients. Bone Marrow Transplant 31:191–196. https://doi.org/10.1038/sj.bmt.1703812
Koshy R, Malhotra P (2011) Treatment of primary aspergilloma of the central nervous system in a diabetic immunocompetent patient with surgical resection and voriconazole: a case report and review of the literature. Turk Neurosurg 21:641–644. https://doi.org/10.5137/1019-5149.jtn2907-10.1
Lange N, Berndt M, Jorger AK, Wagner A, Wantia N, Lummel N, Ryang YM, Meyer B, Gempt J (2018) Clinical characteristics and course of primary brain abscess. Acta Neurochir 160:2055–2062. https://doi.org/10.1007/s00701-018-3633-6
Levine MT, Chandrasekar PH (2016) Adverse effects of voriconazole: over a decade of use. Clin Transpl 30:1377–1386. https://doi.org/10.1111/ctr.12834
Lin SJ, Schranz J, Teutsch SM (2001) Aspergillosis case-fatality rate: systematic review of the literature. Clin Infect Dis 32:358–366. https://doi.org/10.1086/318483
Malani AN, Kerr LE, Kauffman CA (2015) Voriconazole: how to use this antifungal agent and what to expect. Semin Respir Crit Care Med 36:786–795. https://doi.org/10.1055/s-0035-1562903
Muraoka S, Araki Y, Izumi T, Takeuchi K, Okamoto S, Wakabayashi T (2016) Cerebral infarction and subarachnoid hemorrhage caused by central nervous system aspergillus infection. World Neurosurg 90:705.e709–705.e713. https://doi.org/10.1016/j.wneu.2016.03.021
Nadkarni T, Goel A (2005) Aspergilloma of the brain: an overview. J Postgrad Med 51(Suppl 1):S37–S41
Paajanen J, Halme M, Palomaki M, Anttila VJ (2019) Disseminated Scedosporium apiospermum central nervous system infection after lung transplantation: a case report with successful recovery. Med Mycol Case Rep 24:37–40. https://doi.org/10.1016/j.mmcr.2019.03.003
Pagano L, Ricci P, Montillo M, Cenacchi A, Nosari A, Tonso A, Cudillo L, Chierichini A, Savignano C, Buelli M, Melillo L, La Barbera EO, Sica S, Hohaus S, Bonini A, Bucaneve G, Del Favero A (1996) Localization of aspergillosis to the central nervous system among patients with acute leukemia: report of 14 cases. Gruppo Italiano Malattie Ematologiche dell'Adulto Infect Prog Clin infect Dis 23:628–630
Park SY, Yoon JA, Kim SH (2017) Voriconazole-refractory invasive aspergillosis. Korean J Intern Med 32:805–812. https://doi.org/10.3904/kjim.2017.109
Paulussen C, Hallsworth JE, Alvarez-Perez S, Nierman WC, Hamill PG, Blain D, Rediers H, Lievens B (2017) Ecology of aspergillosis: insights into the pathogenic potency of Aspergillus fumigatus and some other Aspergillus species. Microb Biotechnol 10:296–322. https://doi.org/10.1111/1751-7915.12367
Perry JJ, Rech RR, Camus MS, Howerth EW (2010) Pathology in practice. Disseminated aspergillosis J Am Vet Med Assoc 236:173–175. https://doi.org/10.2460/javma.236.2.173
Quinn TJ, Dawson J, Walters MR, Lees KR (2009) Reliability of the modified Rankin scale: a systematic review. Stroke 40:3393–3395. https://doi.org/10.1161/strokeaha.109.557256
Rajshekhar V (2007) Surgical management of intracranial fungal masses. Neurol India 55:267–273
Rees JR, Pinner RW, Hajjeh RA, Brandt ME, Reingold AL (1998) The epidemiological features of invasive mycotic infections in the San Francisco Bay area, 1992-1993: results of population-based laboratory active surveillance. Clin Infect Dis 27:1138–1147
Ruchel R, Schaffrinski M (1999) Versatile fluorescent staining of fungi in clinical specimens by using the optical brightener Blankophor. J Clin Microbiol 37:2694–2696
Shah S, Vanclay F, Cooper B (1989) Improving the sensitivity of the Barthel index for stroke rehabilitation. J Clin Epidemiol 42:703–709. https://doi.org/10.1016/0895-4356(89)90065-6
Ullmann AJ, Aguado JM, Arikan-Akdagli S, Denning DW, Groll AH, Lagrou K, Lass-Florl C, Lewis RE, Munoz P, Verweij PE, Warris A, Ader F, Akova M, Arendrup MC, Barnes RA, Beigelman-Aubry C, Blot S, Bouza E, Bruggemann RJM, Buchheidt D, Cadranel J, Castagnola E, Chakrabarti A, Cuenca-Estrella M, Dimopoulos G, Fortun J, Gangneux JP, Garbino J, Heinz WJ, Herbrecht R, Heussel CP, Kibbler CC, Klimko N, Kullberg BJ, Lange C, Lehrnbecher T, Loffler J, Lortholary O, Maertens J, Marchetti O, Meis JF, Pagano L, Ribaud P, Richardson M, Roilides E, Ruhnke M, Sanguinetti M, Sheppard DC, Sinko J, Skiada A, Vehreschild M, Viscoli C, Cornely OA (2018) Diagnosis and management of Aspergillus diseases: executive summary of the 2017 ESCMID-ECMM-ERS guideline. Clin Microbiol Infect 24(Suppl 1):e1–e38. https://doi.org/10.1016/j.cmi.2018.01.002
Walsh TJ, Hier DB, Caplan LR (1985) Aspergillosis of the central nervous system: clinicopathological analysis of 17 patients. Ann Neurol 18:574–582. https://doi.org/10.1002/ana.410180511
World-Medical-Association (2013) Declaration of Helsinki: ethical principles for medical research involving human subjects. Jama 310:2191–2194. https://doi.org/10.1001/jama.2013.281053
Yamazaki T, Kume H, Murase S, Yamashita E, Arisawa M (1999) Epidemiology of visceral mycoses: analysis of data in annual of the pathological autopsy cases in Japan. J Clin Microbiol 37:1732–1738
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
Nicole Lange: Conceptualization, methodology, and drafting the manuscript
Nina Wantia: Review and editing manuscript
Ann-Kathrin Jorger: Review and editing manuscript
Arthur Wagner: Review and editing manuscript
Friederike Liesche: Review and editing manuscript
Bernhard Meyer: Validation, investigation, and supervision
Jens Gempt: Validation, investigation, and supervision
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest affecting this study. The study was completely financed by the Department of Neurosurgery.
Ethics approval
The present study was approved by the local ethics committee and performed in accordance with the ethical standards established by the 1964 Declaration of Helsinki and its later amendments (Clinical Trial Registration Number: 217/16S).
Informed consent
As a retrospective analysis, informed consent was not necessary.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lange, N., Wantia, N., Jörger, AK. et al. Fungal brain infection—no longer a death sentence. Neurosurg Rev 44, 2239–2244 (2021). https://doi.org/10.1007/s10143-020-01410-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-020-01410-3