Abstract
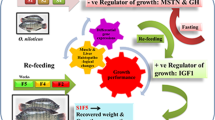
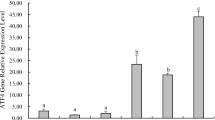
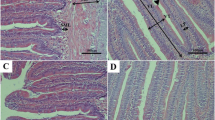
Compensatory growth (CG) in fish is heavily influenced by nutrient metabolism. However, there are limited studies examining how nutrient metabolism is regulated during this process. For silver pomfret, an important commercial marine fish, it's crucial to establish effective starvation and re-feeding strategies to ensure good water quality and fast growth. To identify the complete compensatory growth model of silver pomfret, we conducted an experiment with a control group (normal feeding) and three starvation/re-feeding groups. We observed that the recovery of weight and condition factor in the 14-day starvation and 14-day re-feeding groups was significantly faster than other groups, indicating full compensatory growth. Thus, we selected this group for the next experiment. We performed untargeted metabolomics and transcriptome analysis of muscle tissue on Day 14, 21 and 28 (CG process), and examined the key regulatory genes of nutrient metabolism on Day 0, 7, 14, 21 and 28 (starvation and re-feeding process). Our data revealed that during starvation, silver pomfret first utilized carbohydrates and short-chain lipids, followed by proteins and long-chain lipids. After re-feeding, lipids accumulated first, resulting in rapid growth, followed by the recovery of protein content in muscle. During starvation, the expression of anabolic-related genes such as TER and CALR decreased, and catabolic-related genes such as TSC2 and MLYCD increased, promoting the AMPK pathway. During re-feeding, anabolic-related gene expression increased without AMPK inhibition. Our findings provide insights into the energy utilization strategies of fish and molecular regulation during compensatory growth in fish.






Similar content being viewed by others
Data Availability
We uploaded raw reads to NCBI (BioProject accession numbers: PRJNA818061).
References
Ali M, Nicieza A, Wootton RJ (2003) Compensatory growth in fishes: a response to growth depression. Fish Fish 4:147–190
Atherton PJ, Babraj JA, Smith K, Singh J, Wackerhage H (2005) Selective activation of AMPK-PGC-1alpha or PKB-TSC2-mTOR signaling can explain specific adaptive responses to endurance or resistance training-like electrical muscle stimulation. FASEB J 19:786–788
Bertucci JI, Blanco AM, Sundarrajan L, Rajeswari JJ, Velasco C, Unniappan S (2019) Nutrient regulation of endocrine factors influencing feeding and growth in fish. Front Endocrinol 10:83–96
Campbell P, Creasey N, Parr C (1952) Specificity of muscle phosphorylase. Biochem J 52:448–452
Dobson SH, Holmes RM (2010) Compensatory growth in the rainbow trout, Salmo gairdneri Richardson. J Fish Biol 25:649–656
Grabherr MG, Haas BJ, Yassour M, Levin JZ, Thompson DA, Amit I et al (2011) Full-length transcriptome assembly from RNA-seq data without a reference genome. Nat Biotechnol 29:644–652
Hayward RS, Noltie DB, Wang N (1997) Use of compensatory growth to double hybrid sunfish growth rates. Trans Am Fish Soc 126:316–322
Hu JB, Le QJ, Wang YJ, Yu N, Cao XH, Kuang SW, Yan XJ (2019) Effects of formaldehyde on detoxification and immune responses in silver pomfret (Pampus argenteus). Fish Shellfish Immunol 88:449–457
Jakobsson A, Westerberg R, Jacobsson A (2006) Fatty acid elongases in mammals: Their regulation and roles in metabolism. Prog Lipid Res 45:237–249
Kim MK, Lovell RT (1995) Effect of restricted feeding regimens on compensatory weight gain and body tissue changes in channel catfish Ictalurus punctatus in ponds. Aquaculture 135:285–293
Lee JW, Choe SS, Jang H, Kim J, Jeong HW, Jo H, Kwak TH (2012) AMPK activation with glabridin ameliorates adiposity and lipid dysregulation in obesity. J Lipid Res 53:1277–1286
Li B, Dewey CN (2011) Rsem: accurate transcript quantification from rna-seq data with or without a reference genome. BMC Bioinform 12:93–99
Liao K, Meng R, Ran Z, Cheng G, Wang Y, Xu J, Yan X (2017) Short-term starvation in silver pomfret (Pampus argenteus): molecular effects on lipid mobilization and utilization. Aquac Res 48:4874–4885
Machado CR, Garofaloj MAR, Roselino JES, Kettelhut IC, Migliorini RH (1988) Effects of starvation, refeeding, and insulin on energy-linked metabolic processes in catfish (Rhamdia hilarii) adapted to a carbohydrate-rich diet. Gen Comp Endocrinol 71:429–437
Mao X, Tao C, Olyarchuk JG, Wei L (2005) Automated genome annotation and pathway identification using the kegg orthology (ko) as a controlled vocabulary. Bioinformatics 21:3787–3793
Miglavs I, Jobling M (1989) The effects of feeding regime on proximate body composition and patterns of energy deposition in juvenile Arctic charr, Salvelinus alpinus. J Fish Biol 35:1–11
Mlglavs I, Jobling M (2010) Effects of feeding regime on food consumption, growth rates and tissue nucleic acids in juvenile Arctic charr, Salvelinm alpinus, with particular respect to compensatory growth. J Fish Biol 34:947–957
Ni SZ, Wang YJ, Hu JB, Shi J, Qian D (2020) Identification, histopathology, and phylogenetic analysis of an iridovirus from cultivated silver pomfret in Zhejiang Province, East China. Aquaculture 530:619–735
Ockner RK, Manning JA, Poppenhausen RB, Ho WKL (1972) A binding protein for fatty acids in cytosol of intestinal mucosa, liver, myocardium and other tissues. Science 177:56–58
Reimers E, Kjørrefjord AG, Stavøstrand SM (2010) Compensatory growth and reduced maturation in second sea winter farmed Atlantic salmon following starvation in February and March. J Fish Biol 43:805–810
Schultze SM, Hemmings BA, Markus N, Oliver T (2012) PI3K/AKT, MAPK and AMPK signalling: protein kinases in glucose homeostasis. Expert Rev Mol Med 14:e1
Shrago E (2000) Long-chain acyl-CoA as a multi-effector ligand in cellular metabolism. J Nutr 130:290–293
Stewart JM (2000) The cytoplasmic fatty-acid-binding proteins: thirty years and counting. Cel Mol Life Sci 57:1345–1359
Sullivan A, Singh M, Srere P, Glusker JP (1977) Reactivity and inhibitor potential of hydroxycitrate isomers with citrate synthase, citrate lyase, and ATP citrate lyase. J Biol Chem 252:7583–7590
Tao Z, Shen C, Zhou SM, Yang N, Wang GL, Wang YJ, Xu SL (2018) An outbreak of Photobacterium damselae subsp. damselae infection in cultured silver pomfret Pampus argenteus in Eastern China. Aquaculture 492:201–205
Tee A, Fingar D, Manning B, Kwiatkowski D, Cantley L, Blenis J (2002) Tuberous sclerosis complex-1 and -2 gene products function together to inhibit mammalian target of rapamycin (mTOR)-mediated downstream signaling. Proc Natl Acad Sci USA 99:13571–13576
Watkins PA (1997) Fatty acid activation. Prog Lipid Res 36:55–83
Wieser W, Krumschnabel G, Ojwang-Okwor JP (1992) The energetics of starvation and growth after refeeding in juveniles of three cyprinid species. Environ Biol Fishes 33:63–71
Won ET, Borski RJ (2013) Endocrine regulation of compensatory growth in fish. Front Endocrinol 4:74
Xie S, Zhu X, Cui Y, Wootton RJ, Lei W, Yang Y (2010) Compensatory growth in the gibel carp following feed deprivation: temporal patterns in growth, nutrient deposition, feed intake and body composition. J Fish Biol 58:999–1009
Xu S, Qiu C, Gu J, Wang D (2013) The difference in ultrastructure changes of esophageal sac and liver in normal and hunger juvenile of Pampus argenteus. Oceanol Limnol Sin 44:1016–1023
Yonezawaa K (2004) mTOR signaling pathway. Hepatol Res 30:9–13
Young MD, Wakefield MJ, Smyth GK, Oshlack A (2010) Gene ontology analysis for RNA-seq: accounting for selection bias. Genome Biol 11:1–12
Zamal H, Ollevier F (2010) Effect of feeding and lack of food on the growth, gross biochemical and fatty acid composition of juvenile catfish. J Fish Biol 46:404–414
Zhang L, Wu G, Tate CG, Lookene A, Olivecrona G (2003) Calreticulin promotes folding/dimerization of human lipoprotein lipase expressed in insect cells (Sf21). J Biol Chem 278:29344–29351
Zhang Y, Hu J, Li Y, Zhang M, Jacques KJ, Gu W, Xu S (2021) Immune response of silver pomfret (Pampus argenteus) to Amyloodinium ocellatum infection. J Fish Dis 44:2111–2123
Funding
This work was funded by the Ningbo 2025 Major Project of Science Technology and Innovation (2021Z003), the Natural Science Foundation of Zhejiang (LY18C190008 and LY18C1900013), the Natural Science Foundation of China (42306114, 31872195, 42076118 and 31872586), the Ningbo Public Welfare Science and Technology Project (2021S061), the Ningbo Natural Science Foundation (202003N4121), China Postdoctoral Science Foundation (2022M721729), General scientific research projects of Zhejiang Provincial Department of Education (Y202249062), and the Ningbo Yongjiang Talent Introduction Programme.
Author information
Authors and Affiliations
Contributions
Jiabao Hu: Methodology, Writing- Original draft preparation and Reviewing, Investigation. Man Zhang: Investigation. Kaiheng Yan: Investigation. Yaya Li: Investigation. Youyi Zhang: Investigation. Yuanbo Li: Investigation. Weiwei Gu: Investigation. Shanliang Xu: Methodology, Investigation. Yajun Wang: Methodology. Xiaojun Yan: Supervision,Writing- Reviewing.
Corresponding authors
Ethics declarations
Ethics Approval
The Animal Care and Use Committee of Ningbo University approved the protocols (NBU20220079) in this study.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Figure S1
Scatter plot of PLS-DA score in muscle metabolome of silver pomfret. a: Day 14, b: Day 21 and c: Day 28. The X axis is the score of the sample on the first principal component; The Y axis is the score of the sample on the second principal component. PIM: positive ion mode, NIM: negative ion mode. (TIF 6761 KB)
Figure S2
KEGG pathway (a), HMDB classification (b) and lipid maps annotation (c). The X axis represents the number of metabolites and the Y axis represents the classification annotated. PIM: positive ion mode, NIM: negative ion mode. (TIF 8368 KB)
Figure S3
Correlation analysis of gene expression. (TIF 2826 KB)
Figure S4
Histogram of go enrichment in muscle of Pampus argenteus. The X axis is the GO term of the next level of the three go categories, and the Y axis is the number of differential genes annotated to the term (including the sub term of the term). (TIF 3182 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, J., Zhang, Y., Yan, K. et al. Change and Regulation of Nutritional Metabolism in Silver Pomfret During Compensatory Growth. Mar Biotechnol 25, 1085–1098 (2023). https://doi.org/10.1007/s10126-023-10261-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10126-023-10261-0