Abstract
The knowledge of understanding the molecular traits of the sterile triploid fish is sparse. Herein, we analyzed the microRNA (miRNA) alternations in the testes of the sterile triploid fish produced by crossing the tetraploid fish with the diploid fish, compared with those of tetraploids and diploids used as the controls. A total of 136, 134, and 142 conserved miRNAs and 105, 112, and 119 novel miRNAs were identified in the diploid, triploid, and tetraploid fish, respectively. The genes targeted by the differentially expressed miRNAs were identified and were enriched in the GO term cell surface receptor signaling pathway, cellular process, G-protein coupled receptor signaling pathway, and metabolic process. KEGG pathway enrichment was also assessed to evaluate the target genes with differentially expressed miRNAs and these genes were enriched in four pathways (synthesis and degradation of ketone bodies, pentose and glucuronate interconversions, cyanoamino acid metabolic process, and ascorbate and aldarate metabolism). Nine differentially expressed miRNAs were verified by quantitative real-time PCR analysis (qPCR). The upregulated miRNAs in triploids, including miR-101a, miR-199-5p, miR-214, miR-222, and miR-193a, showed the same results with high-throughput sequencing. Among the selected downregulated miRNAs, miR-7b and miR-153b had significantly lower expression levels in triploids. Dnah3 and Tekt1 genes targeted by miR-199-5p showed lower expression in triploids by qPCR. These verified differentially expressed miRNAs may participate in testicular development and sperm activity by targeting functional genes, which were identified with differential expression in the triploid. This evidence provides insights into the epigenetic regulatory mechanisms of sterility in triploid cyprinids.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Spermatogenesis is the process by which mature male gametes are generated during reproduction. Germ cells pass through three major phases: mitotic proliferation (spermatogonia), meiosis (spermatocytes), and spermiogenesis, i.e., the transformation of haploid spermatids into flagellated spermatozoa (Schulz et al. 2010). In animals, many factors may cause abnormalities in spermatogenesis, including genetic conditions, disease, endocrine disorders, and nutritional factors (Schulz et al. 2010). Many studies have shown that triploid animals are almost sterile due to the unequal disjunction of homologous chromosomes during meiosis, which generates aneuploid gametes or reduces gonadal development (Benfey 1999; Tiwary et al. 2004). For example, all males are sterile in triploid mollusks (Ruiz-Verdugo et al. 2000; Maldonado-Amparo et al. 2004), crustaceans (Li et al. 2003; Xiang et al. 2006), and fish (Tiwary et al. 2004).
Polyploid fish, which are widespread globally, can be generated from hybridization (Leggatt and Iwama 2003; Seehausen 2004). Most vertebrate hybrids cannot produce offspring due to reproductive isolation (Hendry et al. 2000). Through hybridization, most fish can generate F1 offspring. However, genome incompatibility can be found in the offspring, which usually contain disordered genome recombination, gene deletion, or replication (Stelkens et al. 2010). Additionally, although F1 offspring can be obtained through some distant between-species crosses, the descendants are polyploid with genome duplication (Leggatt and Iwama 2003). This unstable genome may result in failed gamete formation. For instance, polyploid Indian catfish (Heteropneustes fossilis) (Pandian and Koteeswaran 1999), rainbow trout (Oncorhynchus mykiss) (Benfey and Sutterlin 1984), gilthead seabream (Sparus aurata) (Felip et al. 1999), and yellowtail flounder (Limanda ferruginea) (Manning et al. 2004) have abnormal gonadal structures compared to those of diploid fish.
Tetraploidy (4n = 200) was generated through inter-species hybridization from the red crucian carp (Carassius auratus red var., ♀) (2n = 100) and the common carp (Cyprinus carpio L., ♂) (2n = 100) (Liu et al. 2001). Intriguingly, both the male and female tetraploid progenies were fertile. Subsequently, the F4–F27 hybrid allotetraploid line was generated by self-crossing. Using this population, we obtained allotriploids (3n = 150) by crossing the tetraploid males with diploid crucian carp. The triploid fish were sterile and exhibited higher growth rates than the diploid and tetraploid lines. The testes of the tetraploid carp produced normal spermatozoa, whereas the triploid carp did not produce normal sperm (Long et al. 2006). Instead, the germ cells developed into round spermatids and then degenerated (Xu et al. 2015). Thus, cyprinids with different ploidy levels, including diploid, triploid, and tetraploid lines, are suitable models to illustrate differences in reproductive development in polyploid fish.
Next-generation sequencing offers an efficient, low-cost, and high-throughput approach for producing large amounts of digital gene expression profiling (DGE). Using this approach, we showed that a series of important genes (Dnahs, Dnal1, Ifts, and Dnaaf1) that participated in sperm flagellar assembly and motility were downregulated in sterile triploids compared to their expression in fertile diploids (Xu et al. 2015). In addition, some testis-specific candidate markers, including Tcte1, Tekt1, Tekt4, Spag17, Spag5, Spag9a, Spag1b, and Spef2, were also downregulated in the triploid lines (Xu et al. 2015). Based on these findings, we concluded that these differentially expressed genes may lead to failed gamete formation in triploid lines. In addition to affecting the gene levels, epigenetic levels may also affect the appearance of polyploid fish. After genome duplication, epigenetic regulation can also change gene expression and phenotypes in polyploid fish (Inácio et al. 2012). MicroRNAs (miRNAs) regulate gene expression via a well-known molecular mechanism at the epigenetic level (Anderson and Kedersha 2009). By targeting mRNAs, miRNAs regulate more than 60% of human protein-coding genes (He and Hannon 2004). This group of new regulatory molecules has a conservative function in vertebrates, and the gene regulatory mechanism has also been illustrated in teleosts (Tanzer and Stadler 2004; Wienholds et al. 2005; Yang et al. 2015).
High-throughput sequencing of small RNAs was performed on the testes from diploid, triploid, and tetraploid cyprinids. We focused on identifying differentially expressed miRNAs in these cyprinids with different ploidy levels to provide new insight into the mechanism underlying the sterility of triploid fish from the miRNA perspective.
Materials and Methods
Ethics Statement
All experiments performed from 2015 to 2017 were approved by the Animal Care Committee of Hunan Normal University. The Administration of Affairs Concerning Animal Experimentation guidelines obtained approval from the Science and Technology Bureau of China. The methods were conducted in accordance with the approved guidelines. Experimental individuals were fed in a pool with suitable illumination, water temperature, dissolved oxygen content, and adequate forage in the Center for Polyploidy Fish Genetics Breeding of Hunan Province located at Hunan Normal University, Changsha, China.
Fish and Sample Collection
In June 2015, adult male Carassius auratus red var., triploid Carassius auratus red var. × (Carassius auratus × Cyprinus carpio) and allotetraploid Carassius auratus × Cyprinus carpio (2 years old) were obtained from the Center for Polyploidy Fish Genetics Breeding of Hunan Province, Hunan Normal University, Changsha, China. All individual fish were stocked in a 0.067-ha open pool prior to tissue collection, and the ploidy of all individual fish was confirmed by metaphase chromosome assay of kidney cells. The individuals were euthanized using 2-phenoxyethanol (Sigma, St. Louis, MO, USA). Then, the testes were removed surgically from fish with different ploidy levels and stored in RNAlater (Invitrogen, Carlsbad, CA, USA) for RNA isolation. For each type of fish, three individuals were collected to isolate total RNA.
Preparation and Sequencing of Small RNA Libraries
Total RNA was isolated using the Invitrogen TRIzol® Reagent according to the manufacturer’s instructions. The RNA integrity was analyzed using agarose gel electrophoresis, and the purity was determined using a NanoDrop (Thermo Fisher Scientific, Wilmington, DE, USA). In addition, the RNA integrity number (RIN) was determined using an Agilent 2100 bioanalyzer (Agilent Technologies, Palo Alto, CA, USA). All small RNA libraries were constructed and sequenced by the Novogene Company (Beijing, China). A Small RNA Sample Prep-Kit was used to construct the small RNA libraries (Illumina Inc., San Diego, CA, USA). Briefly, the total RNA was linked with 3′ and 5′ adaptors, and PCR was performed. Subsequently, the PCR products were separated by polyacrylamide gel electrophoresis, and the small RNA fragments were extracted as small RNA libraries. In total, nine small RNA libraries were obtained (n = 3 for each type of fish).
The small RNA libraries were used for next-generation sequencing with the Illumina HiSeq 2500 following the manufacturer’s instructions. The raw data from the Illumina HiSeq 2500 were submitted to the National Center for Biotechnology Information Short Reads Archive and are available for download.
Data Processing
The raw reads were filtered to remove low-quality reads (sequences with N > 10%, more than 50% of nucleotides with Q ≤ 5, or adaptor contamination), and the adaptors were trimmed. Based on the range of small RNAs in the animals (18–35 nt), the reads in this range underwent further analysis. In addition, these clean reads were mapped to the common carp genome using the bowtie software (version 2.0.0-beta2; http://bowtie-bio.sourceforge.net/bowtie2/index.shtml). The mapped small RNAs were used for the subsequent analysis.
Analysis of Conserved and Novel miRNAs
The mapped small RNAs were aligned against miRBase21.0 (http://www.mirbase.org/) to assign the small RNA type and analyze to the secondary structure information. The mapped mature miRNAs, hairpins, and small RNA families, including rRNAs, tRNAs, snRNAs, and snoRNAs, were identified.
The characteristics of the hairpin structures of miRNA precursors can be used to predict novel miRNAs. The available software miREVO v1.2 (http://omictools.com/mirevo-tool) and the miRdeep2 package (https://www.mdc-berlin.de/8551903/en/) were integrated to predict novel miRNAs by exploring the secondary structures, Dicer cleavage sites, and minimum free energy for the remainder of the unmapped small RNAs.
Identification of Differentially Expressed miRNAs
The miRNA expression levels were estimated in transcripts per million using the following formula (Zhou et al. 2010) for normalized expression: (read count × 1,000,000) / (total counts of miRNAs in the sample).
The DESeq2 package (Love et al. 2014) was employed to analyze miRNA expression among the fish with different ploidy levels. In the present analysis, multiple samples were included for each group; thus, we used the adjusted p value for the FDR control (padj) to determine significant differences. The miRNAs were considered significantly different between the groups when padj < 0.05.
Target Gene Prediction and Functional Annotation
The target gene predictions for the miRNAs were retrieved based on the genomic sequences and annotation information for the common carp (http://www.carpbase.org/CarpBase/download/V2.0.gtf). We used the miRanda algorithm (parameter: -sc 140 -en -10) to predict the miRNA targets (Betel et al. 2008). PITA was also used in order to detect the putative miRNA target sites (https://genie.weizmann.ac.il/pubs/mir07/mir07_data.html) (Kertesz et al. 2007). The functions of the target genes were annotated using gene ontology (GO) terms and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways. Enriched GO terms were identified using the GOseq method (https://bioconductor.org/packages/release/bioc/html/goseq.html). p < 0.05 confirmed GO terms as enriched. Enrichment of KEGG pathways was determined using a hypergeometric test with a p value cut-off of 0.05.
Quantitative Real-Time PCR Analysis (qPCR)
The miRNA expression analysis was performed using the same total RNA used to generate the RNA for sequencing. Reverse transcription was performed with the miScript II Reverse Transcription Kit (Qiagen, Valencia, CA, USA) according to the manufacturer’s instructions. The qPCR was performed on an ABI 7500 instrument (Applied Biosystems, Foster City, CA, USA) using the miScript SYBR Green PCR Kit (Qiagen). The 20-μl reaction solution included 0.25 μg of equally mixed cDNA (pooled from three testes of the same type of fish), 1 μl of each primer, 4 μl of 5× miScript Hiflex Buffer, and 2 μl of 10× Nucleics Mix. The reaction conditions included a pre-denaturation step for 15 min at 95 °C, followed by 40 cycles at 95 °C for 15 s, 58 °C for 33 s, and 70 °C for 35 s. Nine miRNAs were analyzed using the specific forward primers. The U6 small nuclear RNA was used as an endogenous control for the miRNAs.
Similarly, the mRNA expression analysis was performed using the same total RNA. Reverse transcription was performed with the RevertAid™ First Strand cDNA Synthesis Kit (Thermo, Wilmington, DE, USA) according to the manufacturer’s instructions. The qPCR was performed on above instrument and in a volume of 20 μl containing 10-μl SYBR Green PCR Master Mix, 1 μl of each primers, 2-μl template cDNA and 6-μl ddH2O. The qPCR program was 50 °C for 2 min, 95 °C for 10 min, followed by 40 cycles at 95 °C for 15 s and 60 °C for 45 s. Two most relevant targeted genes (Danh3 and Tekt1) were analyzed using the specific primers. The β-actin was used as an endogenous control for the mRNAs.
To ensure the accuracy of the qPCR results, the analysis of each sample was repeated four times. Each sample was evaluated in quadruplicate, and relative expression was calculated using the 2−ΔΔCt method. All primers for qPCR are listed in Supplemental Table S1.
Statistical Analysis
The qPCR data were expressed as the mean ± standard deviation. Significant differences were determined by one-way ANOVA followed by Tukey’s (HSD) test at 95% confidence levels. The statistical analysis was performed using SPSS v17.0 (SPSS Inc., Chicago, IL, USA). p < 0.05 was confirmed as a significant difference among the groups.
Results
Next-Generation Sequencing of Small RNAs in Fish with Different Ploidy Levels
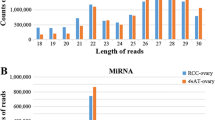
Nine small RNA libraries were constructed from fish with three different ploidy levels, including three diploid, three triploid, and three tetraploid individuals. After next-generation sequencing of the small RNAs, we obtained 87,673,374 raw reads. All of the data were deposited in the Short Read Archive (SRA) under accession numbers SRX1849984, SRX1849985, and SRX1849987. In total, 27,196,671, 29,180,492, and 31,296,211 reads were generated from the diploid, triploid, and tetraploid fish, respectively. After removing the low-quality reads and adaptors, a total of 26,762,693, 28,585,566, and 30,541,328 clean reads were obtained from the diploid, triploid, and tetraploid libraries, respectively (Table 1). For analysis of small RNAs, we chose reads with lengths between 18 and 35 nt. The size distribution of the small RNAs indicated that most were between 21 and 31 nt in length. In total, 2,591,054 (10.03%), 3,026,249 (10.85%), and 6,117,233 (20.68%) small RNAs with lengths between 21 and 22 nt were obtained from the diploid, triploid, and tetraploid libraries, respectively (Fig. 1).
Identification of Conserved and Novel miRNAs
The clean reads were annotated using the common carp reference genome and assigned to different types of small RNAs, including miRNAs, tRNAs, rRNAs, and others, using BLAST with miRBase 21.0 (Table 2). The reads mapped to miRBase were excluded from the subsequent analysis. The unannotated RNAs were used for novel miRNA identification. A total of 136, 134, and 142 conserved miRNAs were identified in the diploid, triploid, and tetraploid libraries, respectively (Supplemental Table S2). A total of 132 miRNAs were identified in fish with all three ploidy levels. The five most highly expressed miRNAs in the diploid library were miR-143, miR-22a, miR-100, miR-101a, and miR-99. miR-143 expression was followed by miR-101a, miR-100, miR-22a, and miR-146a expression in the triploid fish. In the tetraploid fish, the most highly expressed miRNAs were miR-143, miR-22a, miR-7a, miR-21, and miR-146a. Thus, miR-143 expression was the highest among the conserved miRNAs identified from the fish with the three ploidy levels.
Novel miRNAs were predicated based on marked hairpin structures of the miRNA precursors. The precursors of the unannotated miRNAs were identified using miREVO v1.2 and the miRdeep2 package. A total of 122 novel miRNAs were identified from the fish with the three ploidy levels (Supplemental Table S3). Among them, 105, 112, and 119 novel miRNAs were identified in the diploid, triploid, and tetraploid fish, respectively (Supplemental Table S4). A total of 100 novel miRNAs were identified in fish with all three ploidy levels. The miRNAs that we named novel_381, novel_361, novel_21, and novel_37 were the four most highly expressed miRNAs in each fish.
Differentially Expressed miRNAs in Fish with Different Ploidy Levels
Transcripts per million (TPM) was used to identify differentially expressed miRNAs among the fish with the three ploidy levels. First, we used Pearson’s correlation analysis to show correlations between biological replicates (Fig. 2). High consistency was found among the individuals for each type of fish.
In total, 75 miRNAs were differentially expressed between the diploid and triploid fish (34 were upregulated and 41 were downregulated). In the comparison between the diploid and tetraploid fish, 67 miRNAs were differentially expressed (22 were upregulated and 45 were downregulated). The most abundant differentially expressed miRNAs were found between the triploid and tetraploid fish, with 53 upregulated and 62 downregulated miRNAs identified (Supplemental Table S5). Among these miRNAs, 47 miRNAs were differentially expressed between the triploid fish and the diploid and tetraploid fish (Supplemental Table S5). The VENN diagrams showed the common and the unique (differential expressed and not) miRNA identified in fish with all three ploidy levels (Fig. 3a, b).
Target Prediction and Gene Annotation
To assess the functions of the miRNAs in the fish with different ploidy levels, candidate target genes were predicted based on the reference genome of the common carp using the miRanda and PITA softwares. We have integrated the putative miRNA target sites using two different software programs. In total, 265 miRNAs were found to target 43,418 unigenes in the common carp genome (Supplemental Table S6).
Then, we focused on the genes targeted by the differentially expressed miRNAs. Enrichment of GO terms was determined using the GOseq method for these genes. Only one biological process term (metabolic process) was enriched in the comparison between the diploid and triploid fish (Fig. 4a). Biological process terms, including surface receptor signaling pathway, cellular process, and G-protein coupled receptor signaling pathway, were enriched in the comparison between the diploid and tetraploid fish (Fig. 4b). When comparing the triploid with the tetraploid fish, the biological process terms biological_process, cellular process, single-organism cellular process, single-organism process, and metabolic process were significantly enriched (Fig. 4c).
KEGG pathway enrichment was also assessed to evaluate the target genes with differentially expressed miRNAs. When comparing the diploid and triploid fish, four pathways were enriched (synthesis and degradation of ketone bodies, pentose and glucuronate interconversions, cyanoamino acid metabolic process, and ascorbate and aldarate metabolism) (Fig. 5a). The same KEGG pathway enrichment was found by comparing the diploid and tetraploid fish (Fig. 5b). When comparing the triploid and tetraploid fish, only three pathways (synthesis and degradation of ketone bodies, pentose and glucuronate interconversions, and cyanoamino acid metabolic process) were significantly enriched (Fig. 5c).
We also wanted to identify the differentially expressed miRNAs that might contribute to the sterile triploid phenotype. Several functional genes that participated in the sperm formation process and sperm vitality were controlled by miRNAs, including axoneme dynein, intraflagellar transport genes, testis-specific genes, transcription factors (TFs), and other genes related to testicular development. Among them, the miRNAs that predictably targeted Dnah3, Dnah9, Tekt1, Rfx3, and Piwil2 were significantly upregulated in the triploid compared to their expression in the diploid and tetraploid fish. In contrast, the miRNAs that targeted eighteen genes (Dnah10, Dnal4, Dnai1, Ift6, Ift43a, Ift46, Ift80, Ift88, Ift140, Ift172, Dnaaf3, Tekt3, Tekt4, Spef1, Zbtb34, Ybx1, Rfx2, and Phb) were downregulated in the triploid compared to their expression in the diploid and tetraploid fish (Supplemental Table S7).
Validation of Differentially Expressed miRNAs and Targeted Genes by qPCR
Furthermore, we focused on the miRNAs that were significantly increased or decreased in the triploid fish compared to their levels in the fish with the other two ploidy levels. Considering the testicular function in fish, nine differentially expressed miRNAs (miR-101a, miR-199-5p, miR-214, miR-222, miR-193a, miR-7b, miR-92b, miR-153b, and novel-39) were identified from the bioinformatics approach and were validated by qPCR (Fig. 6). These miRNAs are candidates for participation in testicular development or sperm vitality. miR-101a, miR-199-5p, miR-214, miR-222, and miR-193a were significantly upregulated in the sterile triploid compared to their levels in the fertile diploid and tetraploid fish; this result was validated by the high-throughput sequencing analysis (Fig. 6). miR-7b and miR-153b expression was lowest in the triploid fish (Fig. 6). No significant differences were found in miR-92b and novel-39 expression among the fish with the three ploidy levels (Fig. 6). The relevant targeted genes (Dnah3 and Tekt1) regulated by upregulated expressed miRNAs in triploids were further tested by qPCR. Triploids showed the lower levels of Dnah3 and Tekt1 expression compared with diploid and tetraploid fish (Fig. 7a, b).
qPCR verification of miRNA expression among the diploid, triploid, and tetraploid fish. a miR-101a, b miR-199-5p, c miR-214, d miR-222, e miR-193a, f miR-7b, g miR-92b, h miR-153b, i novel-39. The U6 small nuclear RNA was used as an internal control to calibrate the cDNA template for all samples (n = 4 per group). The different letters on each bar indicated significant differences among the groups (p < 0.05)
qPCR verification of targeted gene expression among the diploid, triploid, and tetraploid fish. a Dnah3, b Tekt1. The β-actin was used as an internal control to calibrate the cDNA template for all samples (n = 4 per group). The different letters on each bar indicated significant differences among the groups (p < 0.05)
Discussion
The testicular microstructure and ultrastructure of sterile triploid cyprinid fish were illustrated in our previous studies (Long et al. 2006; Xu et al. 2015). Interestingly, another polyploid fish, the tetraploid cyprinid fish, was fertile (Long et al. 2006). Thus, the appearance of sterility or fertility of the fish is not dependent only on the ploidy level. Previously, one widely recognized interpretation of these results was that multiple ploidy levels, including diploid and tetraploid lines, could successfully produce haplotypes gametes, whereas triploid fish failed to generate gametes with haplotypes gametes. However, in the wild Xiangjiang River, we found that triploid crucian carp could generate sperm with 0.75 times the DNA content of the diploid crucian carp, which suggested that sperm with 1.5 times higher ploidy levels were produced by this triploid crucian carp (Xiao et al. 2011). Triploid Carassius auratus gibelio also successfully produces sperm (Fan and Shen 1990; Zhou and Gui 2002). Thus, the finding that triploids cannot produce gametes is doubtful. Indeed, most triploids cannot complete the reproductive process by generating gametes. Thus, the details of this mechanism should be illustrated at more than the chromosome level. Previous research indicated that the testis of triploid fish develops normally, but could not produce normal sperm that possess normal nuclei and integrated tails. Germ cells of male triploid fish can develop to the stage of round spermatid, then degenerate (Xu et al. 2015). Therefore, we speculated that abnormal regulation during the spermiogenesis in triploids may affect sterility in male triploid fish. Previously, we elucidated the transcriptome changes of these fish with different ploidy levels and found that the genes associated with sperm flagellar assembly and motility and with testis-specific candidate markers were significantly decreased in the testes of triploid fish compared to their levels in other fertile fish, such as diploid and tetraploid fish (Xu et al. 2015). This study provides new insights into the genetic changes that may cause sterility in male triploid fish.
In the present study, we extend this research beyond the genetic level to the epigenetic level. High-throughput sequencing provides abundant data to reflect the profiles of the small RNAs that control mRNA expression. The length distributions of the reads in the three fish represented the majority of the miRNAs in the testes from these fish. Interestingly, in addition to the 21–22-nt range, which was the feature length of the miRNAs, a high ratio of small RNAs ranging from 26 to 29 nt in length was found. To our knowledge, this length range reflects the piRNAs in the testis (Lau et al. 2006). piRNA is another small RNA family that is abundantly expressed in the testis and plays a crucial role in sperm formation (Houwing et al. 2007; Houwing et al. 2008). The peak at 22 nt for tetraploid fish was miRNA peak, while the highest peak was 27 nt in Fig. 1. We proposed that these may be due to the polyploidy levels which resulted in counts of miRNA were higher in 22 nt. However, the details and mechanisms need to be investigated in further study. Herein, we focused on the function of miRNAs in the testes of these fish; thus, we only analyzed the miRNAs.
Using the genome of the common carp as a reference, the miRNAs were mapped to these sequences. In total, 136, 134, and 142 conserved miRNAs were identified from the testes of the diploid, triploid, and tetraploid fish, respectively. Additionally, 105, 112, and 119 novel miRNAs were identified in the diploid, triploid, and tetraploid fish, respectively. Most of the miRNAs were simultaneously identified from all three fish types, which showed similar miRNA expression among fish with different ploidy levels. Further expression analysis suggested that although the expressed miRNAs were similar, the expression levels in these fish were different and that these miRNAs were potentially involved in regulating sperm generation and motility.
The expression of miRNAs in fish with different ploidy levels was similar to the observations in other organisms. Among them, miR-143 had the highest expression level in the testes of each fish type. In tilapia, miR-143 has been reported to be similarly abundant in the testis (Xiao et al. 2014). This miRNA is also upregulated in the mature testes of mammals (Luo et al. 2010; Huang et al. 2011). Other miRNAs, such as miR-99, miR-101a, miR-100, miR-22a, miR-146a, miR-21, and miR-7a, were also highly expressed in the testes. These miRNAs may play crucial roles in essential biological processes in the testes of teleosts.
These differentially expressed miRNAs targeted genes which were enriched in metabolism process, surface receptor signaling pathway, cellular process, G-protein coupled receptor signaling pathway, biological_process, cellular process, single-organism cellular process, single-organism process, and metabolic process. The KEGG pathway enrichment analysis showed that the differentially expressed miRNAs targeted genes were enriched in synthesis and degradation of ketone bodies, pentose and glucuronate interconversions, cyanoamino acid metabolic process, and ascorbate and aldarate metabolism pathways. These GO terms and KEGG pathways were crucial for spermatogenesis. In our previous study, we found that similar GO terms were enriched in comparison between diploid and triploid transcriptomes (Xu et al. 2015). Thus, the miRNA regulated gene expression formed a network for these function categories to affect fertile abilities among different ploidy fishes.
Among the differentially expressed miRNAs, we found several miRNAs that targeted key genes, including axoneme dynein, intraflagellar transport, testis-specific, and TF genes, as well as other genes related to testicular development. Previous RNA-seq results showed that a series of functional genes involved in sperm flagellar assembly and motility (Dnahs, Dnal1, Ifts, and Dnaaf1) as well as testis-specific candidate markers (Tcte1, Tekt1, Tekt4, Spag17, Spag5, Spag9a, Spag1b, and Spef2) had significantly lower expression levels in the testes of triploid fish than in diploid fish (Xu et al. 2015). The present study proved that the miRNAs that targeted these genes were also differentially expressed in the triploid fish, which suggested that these weakly expression genes might be under the control of miRNAs. For instance, miR-199-5p, which targeted Dnah3 and Tekt1, was upregulated, whereas Dnah3 and Tekt1 were downregulated in the triploid testes (Xu et al. 2015), and the qPCR further verified the higher expression of miR-199-5p and lower expression of Dnah3 and Tekt1 in triploids. Tekt1, a member of the tektin family, is involved in the formation of sperm flagellum and may be associated with flagellar stability and sperm motility (Amos 2008). The qPCR results also demonstrated that miR-101a, miR-214, miR-222, and miR-193a were significantly upregulated in the sterile triploid fish. These expression changes of mRNA and miRNA may be due to the dosage compensation which was contributed by genome duplication. Unfortunately, the mechanism and pattern of gene expression changes in polyploid in both plants and animals are not clear. The clues which could be confirmed are that the different ploidy affects gene expression in different patterns depended on genes.
miR-101a is one of the most abundant miRNAs in the carp ovary (Wang et al. 2017) and plays a key role in the response to nonylphenol exposure in mouse Sertoli cells (Madison-Villar and Michalak 2011), suggesting a function of this miRNA in gonad development. miR-199-5p was also abundantly expressed in the ovaries of the common carp, which indicated a crucial role in gonad development (Wang et al. 2017). miR-214 and miR-199-5p are in the same cluster in humans and are regulated by Twist-1 (Youn-Bok et al. 2009). In addition, miR-199-5p represses DNMT1, which is responsible for DNA methylation (Chen et al. 2014). miR-222 is a well-known miRNA that regulates the undifferentiated state in mammalian male germ cells (Yang et al. 2013). miR-193a also functions in DNA methylation (Heller et al. 2012). These clues indicate that these upregulated miRNAs may repress key functional genes in the testes in triploid fish and lead to sterility. Only miR-7b and miR-153b were confirmed to be downregulated miRNAs in the triploid fish by both high-throughput sequencing and qPCR. miR-7b is a hormonally sensitive testicular miRNA (Nicholls et al. 2009). miR-153b has been reported to be highly expressed in channel catfish (Ictalurus punctatus). However, the function of this miRNA in fish has not been elucidated.
To date, epigenetic control by miRNAs in sterile triploids is largely unknown. The hormonal regulation and transcriptome profiles were reported previously. Herein, we analyzed miRNA profiles at the epigenetic level to perform a pioneering study on fish with different ploidy levels. Based on the present results, we propose the following three regulatory levels in the testes of fish with different ploidy levels: first, miRNAs represent epigenetic regulation and mainly mediate post-transcriptional regulation; second, gene expression regulates key proteins that participate in testicular development and sperm activity; and third, hormonal regulation controls testicular maturation. In male triploid fish, these levels are abnormal and ultimately lead to sterility. Further studies should focus on verification of miRNA targets and the functional details of the miRNA-mRNA pairs. Taken together, our results indicate that the miRNAs that contribute to testicular development and sperm activity are abnormally expressed in triploid fish, which may account for the failure of the triploid testis.
References
Amos LA (2008) The tektin family of microtubule-stabilizing proteins. Genome Biol 9:229
Anderson P, Kedersha N (2009) RNA granules: post-transcriptional and epigenetic modulators of gene expression. Nat Rev Mol Cell Biol 10:430–436
Benfey TJ (1999) The physiology and behavior of triploid fishes. Rev Fish Sci 7:39–67
Benfey TJ, Sutterlin AM (1984) Growth and gonadal development in triploid landlocked Atlantic salmon (Salmo salar). Can J Fish Aquat Sci 41:1387–1392
Betel D, Wilson M, Gabow A, Marks DS, Sander C (2008) The microRNA.org resource: targets and expression. Nucleic Acids Res 36:D149–D153
Chen BF, Suen YK, Gu S, Li L, Chan WY (2014) A mir-199a/mir-214 self-regulatory network via PSMD10, TP53 and DNMT1 in testicular germ cell tumor. Sci Rep 4:6413
Fan ZT, Shen J (1990) Studies on the evolution of bisexual reproduction in crucian carp (Carassius auratus gibelio Bloch). Aquaculture 84:235–244
Felip A, Zanuy S, Carrillo M, Piferrer F (1999) Growth and gonadal development in triploid sea bass (Dicentrarchus labrax L.) during the first two years of age. Aquaculture 173:389–399
He L, Hannon GJ (2004) MicroRNAs: small RNAs with a big role in gene regulation. Nat Rev Genet 5:522–531
Heller G, Weinzierl M, Noll C, Babinsky V, Ziegler B, Altenberger C, Minichsdorfer C, Lang G, Döme B, End-Pfützenreuter A, Arns B-M, Grin Y, Klepetko W, Zielinski CC, Zöchbauer-Müller S (2012) Genome-wide miRNA expression profiling identifies mir-9-3 and mir-193a as targets for DNA methylation in non–small cell lung cancers. Clin Cancer Res 18:1619–1629
Hendry AP, Wenburg JK, Bentzen P, Volk EC, Quinn TP (2000) Rapid evolution of reproductive isolation in the wild: evidence from introduced salmon. Science 290:516–518
Houwing S, Berezikov E, Ketting RF (2008) Zili is required for germ cell differentiation and meiosis in zebrafish. EMBO J 27:2702–2711
Houwing S, Kamminga LM, Berezikov E, Cronembold D, Girard A, Elst HVD, Filippov D, Blaser H, Raz E, Moens CB, Plasterk RHA, Hannon GJ, Draper BW, Ketting RF (2007) A role for Piwi and piRNAs in germ cell maintenance and transposon silencing in zebrafish. Cell 129:69–82
Huang JM, Ju ZH, Li QL, Hou QL, Wang CF, Li JB, Li RL, Wang LL, Sun T, Huang SQ, Gao YD, Hou MH, Zhong JF (2011) Solexa sequencing of novel and differentially expressed microRNAs in testicular and ovarian tissues in Holstein cattle. Int J Biol Sci 7:1016–1026
Inácio A, Pinho J, Pereira PM, Comai L, Coelho MM (2012) Global analysis of the small RNA transcriptome in different ploidies and genomic combinations of a vertebrate complex–the Squalius alburnoides. PLoS One 7:e41158
Kertesz M, Iovino N, Unnerstall U, Gaul U, Segal E (2007) The role of site accessibility in microRNA target recognition. Nat Genet 39:1278–1284
Lau NC, Seto AG, Kim J, Kuramochi-Miyagawa S, Nakano T, Bartel DP, Kingston RE (2006) Characterization of the piRNA complex from rat testes. Science 313:363–367
Leggatt RA, Iwama GK (2003) Occurrence of polyploidy in the fishes. Rev Fish Biol Fish 13:237–246
Li FH, Xiang JH, Zhang XJ, Zhou LH, Zhang CS, Wu CG (2003) Gonad development characteristics and sex ratio in triploid chinese shrimp (Fenneropenaeus chinensis). Mar Biotechnol (NY) 5:528–535
Liu SJ, Liu Y, Zhou GJ, Zhang XJ, Luo C, Feng H, He XX, Zhu GH, Yang H (2001) The formation of tetraploid stocks of red crucian carp × common carp hybrids as an effect of interspecific hybridization. Aquaculture 192:171–186
Long Y, Liu SJ, Huang WR, Zhang J, Sun YD, Zhang C, Chen S, Liu JH, Liu Y (2006) Comparative studies on histological and ultra-structure of the pituitary of different ploidy level fishes. Sci China C Life Sci 49:446–453
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15:1–21
Luo LF, Ye LZ, Liu G, Shao GC, Zheng R, Ren ZQ, Zuo B, Xu DQ, Lei MG, Jiang SW, Deng CY, Xiong YZ, Li FG (2010) Microarray-based approach identifies differentially expressed microRNAs in porcine sexually immature and mature testes. PLoS One 5:e11744
Maldonado-Amparo R, RamiRez JL, Ávila S, Ibarra AM (2004) Triploid lion-paw scallop (Nodipecten subnodosus Sowerby); growth, gametogenesis, and gametic cell frequencies when grown at a high food availability site. Aquaculture 235:185–205
Manning AJ, Burton MP, Crim LW (2004) Reproductive evaluation of triploid yellowtail flounder, Limanda ferruginea (Storer). Aquaculture 242:625–640
Madison-Villar MJ, Michalak P (2011) Misexpression of testicular microRNA in sterile Xenopus hybrids points to tetrapod-specific microRNAs associated with male fertility. J Mol Evol 73:316–324
Nicholls PK, Harrison CA, O'Donnell L, Stanton PG (2009) Hormonal regulation of miRNA in the testis. Reprod Fert Develop 21:66–66
Pandian T, Koteeswaran R (1999) Polyploids in the Indian catfish, Heteropneustes fossilis. Curr Sci 76:1134–1137
Ruiz-Verdugo CA, RamiRezm JL, SKA JR, Ibarra AM (2000) Triploid catarina scallop (Argopecten ventricosus Sowerby II, 1842): growth, gametogenesis, and suppression of functional hermaphroditism. Aquaculture 186:13–32
Schulz RW, de França LR, Lareyre JJ, LeGac F, Chiarini-Garcia H, Nobrega RH, Miura T (2010) Spermatogenesis in fish. Gen Comp Endocrinol 165:390–411
Seehausen O (2004) Hybridization and adaptive radiation. Trends Ecol Evol 19:198–207
Stelkens RB, Young KA, Seehausen O (2010) The accumulation of reproductive incompatibilities in African cichlid fish. Evolution 64:617–633
Tanzer A, Stadler PF (2004) Molecular evolution of a microRNA cluster. J Mol Biol 339:327–335
Tiwary BK, Kirubagaran R, Raym AK (2004) The biology of triploid fish. Rev Fish Biol Fish 14:391–402
Wang F, Jia YF, Wang P, Yang QW, Du QY, Chang ZJ (2017) Identification and profiling of Cyprinus carpio, microRNAs during ovary differentiation by deep sequencing. BMC Genomics 18:333
Wienholds E, Kloosterman WP, Miska E, Alvarez-Saavedra E, Berezikov E, de Bruijin E, Horvitz HR, Kauppinen S, Plasterk RHA (2005) MicroRNA expression in zebrafish embryonic development. Science 309:310–311
Xiang JH, Li FH, Zhang CS, Zhang XJ, Yu KJ, Zhou LH, Wu CG (2006) Evaluation of induced triploid shrimp Penaeus, (Fenneropenaeus) chinensis, cultured under laboratory conditions. Aquaculture 259:108–115
Xiao J, Zhong H, Zhou Y, Yu F, Gao Y, Luo YJ, Tang ZY, Guo ZB, Guo EY, Gan X, Zhang M, Zhang YP (2014) Identification and characterization of microRNAs in ovary and testis of Nile tilapia (Oreochromis niloticus) by using solexa sequencing technology. PLoS One 9:e86821
Xiao J, Zou TM, Chen YB, Chen L, Liu SJ, Tao M, Zhang C, Zhao RR, Zhou Y, Long Y, You CP, Yan JP, Liu Y (2011) Coexistence of diploid, triploid and tetraploid crucian carp (Carassius auratus) in natural waters. BMC Genet 12:1–15
Xu K, Wen M, Duan W, Ren L, Hu FZ, Xiao J, Wang J, Tao M, Zhang C, Wang J, Zhou Y, Zhang Y, Liu Y, Liu SJ (2015) Comparative analysis of testis transcriptomes from triploid and fertile diploid cyprinid fish. Biol Reprod 92(95):1–12
Yang LD, Irwin DM, He SP (2015) Genome-wide identification and characterization of teleost-specific microRNAs within zebrafish. Gene 561:181–189
Yang Q, Racicot KE, Kaucher AV, Oatley MJ, Oatley JM (2013) MicroRNAs 221 and 222 regulate the undifferentiated state in mammalian male germ cells. Development 140:280–290
Youn-Bok L, Ioannis B, Do-Young L, Leonidas P, Caldwell MA, Uney JB (2009) Twist-1 regulates the mir-199a/214 cluster during development. Nucleic Acids Res 37:123
Zhou L, Chen JH, Li ZZ, Li XX, Hu XD, Huang Y, Zhao XK, Liang CZ, Wang Y, Sun L, Shi M, Xu XH, Shen F, Chen MS, Han ZJ et al (2010) Integrated profiling of microRNAs and mRNAs: microRNAs located on Xq27.3 associate with clear cell renal cell carcinoma. Plos One 5:e15224
Zhou L, Gui JF (2002) Karyotypic diversity in polyploid gibel carp, Carassius auratus gibelio Bloch. Genetica 115:223–232
Funding
This research was supported by the National Natural Science Foundation of China (Grant Nos. 31430088, 91631305, 31672627), the earmarked fund for China Agriculture Research System (Grant No. CARS-45), and the Major Program of the Educational Commission of Hunan Province (Grant No. 15A116).
Author information
Authors and Affiliations
Contributions
MT, YZ, and SJL designed the research. MT, SNL, and ML contributed to the qPCR. ML, JC, HH, LY, and CZ contributed to sampling and species identification. MT, YZ, LR, and JL analyzed the data. MT, YZ, SNL, and HZ wrote the paper.
Corresponding author
Ethics declarations
All experiments performed from 2015 to 2017 were approved by the Animal Care Committee of Hunan Normal University.
Competing Interests
The authors declare that they have no competing interests.
Additional information
Min Tao, Yi Zhou, Shengnan Li, and Huan Zhong are co-first authors.
Electronic Supplementary Material
Additional information noted in the text includes Supplemental Tables.
Supplemental Table S1
qPCR primers used in the present study (XLSX 12 kb)
Supplemental Table S2
Identified conserved miRNAs in different ploidy fishes (XLSX 20 kb)
Supplemental Table S3
Sequences of the novel miRNA (XLSX 15 kb)
Supplemental Table S4
Identified novel miRNAs in different ploidy fishes (XLSX 18 kb)
Supplemental Table S5
Differentially expressed miRNAs among the samples (XLSX 33 kb)
Supplemental Table S6
Prediction of miRNA target by matching the genome of common carp (XLS 10521 kb)
Supplemental Table S7
Testicular functional genes which targeted by differentially expressed miRNAs in triploid (XLSX 14 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Tao, M., Zhou, Y., Li, S. et al. MicroRNA Alternations in the Testes Related to the Sterility of Triploid Fish. Mar Biotechnol 20, 739–749 (2018). https://doi.org/10.1007/s10126-018-9845-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10126-018-9845-1