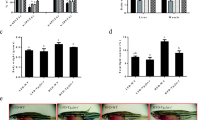
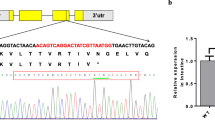
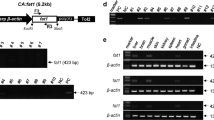
Abstract
Omega-3 long-chain polyunsaturated fatty acid (n-3 LC-PUFA), especially eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), are essential nutrients for human health. However, vertebrates, including humans, have lost the abilities to synthesize EPA and DHA de novo, majorly due to the genetic absence of delta-12 desaturase and omega-3 desaturase genes. Fishes, especially those naturally growing marine fish, are major dietary source of EPA and DHA. Because of the severe decline of marine fishery and the decrease in n-3 LC-PUFA content of farmed fishes, it is highly necessary to develop alternative sources of n-3 LC-PUFA. In the present study, we utilized transgenic technology to generate n-3 LC-PUFA-rich fish by using zebrafish as an animal model. Firstly, fat1 was proved to function efficiently in fish culture cells, which showed an effective conversion of n-6 PUFA to n-3 PUFA with the n-6/n-3 ratio that decreased from 7.7 to 1.1. Secondly, expression of fat1 in transgenic zebrafish increased the 20:5n-3 and 22:6n-3 contents to 1.8- and 2.4-fold, respectively. Third, co-expression of fat2, a fish codon-optimized delta-12 desaturase gene, and fat1 in fish culture cell significantly promoted n-3 PUFA synthesis with the decreased n-6/n-3 ratio from 7.7 to 0.7. Finally, co-expression of fat1 and fat2 in double transgenic zebrafish increased the 20:5n-3 and 22:6n-3 contents to 1.7- and 2.8-fold, respectively. Overall, we generated two types of transgenic zebrafish rich in endogenous n-3 LC-PUFA, fat1 transgenic zebrafish and fat1/fat2 double transgenic zebrafish. Our results demonstrate that application of transgenic technology of humanized fat1 and fat2 in farmed fishes can largely improve the n-3 LC-PUFA production.





Similar content being viewed by others
References
Ahmed AS, Xiong F, Pang SC, He MD, Waters MJ, Zhu ZY, Sun YH (2011) Activation of GH signaling and GH-independent stimulation of growth in zebrafish by introduction of a constitutively activated GHR construct. Transgenic Res 20(3):557–567. doi:10.1007/s11248-010-9439-9
Alimuddin, Yoshizaki G, Kiron V, Satoh S, Takeuchi T (2005) Enhancement of EPA and DHA biosynthesis by over-expression of masu salmon delta6-desaturase-like gene in zebrafish. Transgenic Res 14(2):159–165
Alimuddin, Yoshizaki G, Kiron V, Satoh S, Takeuchi T (2007) Expression of masu salmon delta5-desaturase-like gene elevated EPA and DHA biosynthesis in zebrafish. Mar Biotechnol (NY) 9(1):92–100. doi:10.1007/s10126-006-6003-y
Alimuddin, Kiron V, Satoh S, Takeuchi T, Yoshizaki G (2008) Cloning and over-expression of a masu salmon (Oncorhynchus masou) fatty acid elongase-like gene in zebrafish. Aquaculture 282(1–4):13–18. doi:10.1016/j.aquaculture.2008.06.033
Bousquet M, Gue K, Emond V, Julien P, Kang JX, Cicchetti F, Calon F (2011) Transgenic conversion of omega-6 into omega-3 fatty acids in a mouse model of Parkinson’s disease. J Lipid Res 52(2):263–271. doi:10.1194/jlr.M011692
Buzzi M, Henderson RJ, Sargent JR (1996) The desaturation and elongation of linolenic acid and eicosapentaenoic acid by hepatocytes and liver microsomes from rainbow trout (Oncorhynchus mykiss) fed diets containing fish oil or olive oil. Biochim Biophys Acta 1299(2):235–244
Chen Q, Liu Q, Wu Z, Wang Z, Gou K (2009) Generation of fad2 transgenic mice that produce omega-6 fatty acids. Sci China Ser C Life Sci Chin Acad Sci 52(11):1048–1054. doi:10.1007/s11427-009-0143-z
Chen Y, Mei M, Zhang P, Ma K, Song G, Ma X, Zhao T, Tang B, Ouyang H, Li G, Li Z (2013) The generation of transgenic mice with fat1 and fad2 genes that have their own polyunsaturated fatty acid biosynthetic pathway. Cell Physiol Biochem 32(3):523–532. doi:10.1159/000354456
Deckelbaum RJ (2010) n-6 and n-3 fatty acids and atherosclerosis: ratios or amounts? Arterioscler Thromb Vasc Biol 30(12):2325–2326. doi:10.1161/atvbaha.110.214353
Distel M, Wullimann MF, Koster RW (2009) Optimized Gal4 genetics for permanent gene expression mapping in zebrafish. Proc Natl Acad Sci U S A 106(32):13365–13370. doi:10.1073/pnas.0903060106
FAO (2012) The state of world fisheries and aquaculture. FAO
Fritsche K (2006) Fatty acids as modulators of the immune response. Annu Rev Nutr 26(1):45–73. doi:10.1146/annurev.nutr.25.050304.092610
Hastings N, Agaba M, Tocher DR, Leaver MJ, Dick JR, Sargent JR, Teale AJ (2001) A vertebrate fatty acid desaturase with delta 5 and delta 6 activities. Proc Natl Acad Sci U S A 98(25):14304–14309. doi:10.1073/pnas.251516598
Heckman KL, Pease LR (2007) Gene splicing and mutagenesis by PCR-driven overlap extension. Nat Protoc 2(4):924–932. doi:10.1038/nprot.2007.132
Jump DB (2008) n-3 polyunsaturated fatty acid regulation of hepatic gene transcription. Curr Opin Lipidol 19(3):242–247. doi:10.1097/MOL.0b013e3282ffaf6a
Kabeya N, Takeuchi Y, Yamamoto Y, Yazawa R, Haga Y, Satoh S, Yoshizaki G (2014) Modification of the n-3 HUFA biosynthetic pathway by transgenesis in a marine teleost, nibe croaker. J Biotechnol 172:46–54. doi:10.1016/j.jbiotec.2013.12.004
Kang JX (2007) Fat-1 transgenic mice: a new model for omega-3 research. Prostaglandins Leukot Essent Fat Acids 77(5–6):263–267. doi:10.1016/j.plefa.2007.10.010
Kang ZB, Ge Y, Chen Z, Cluette-Brown J, Laposata M, Leaf A, Kang JX (2001) Adenoviral gene transfer of Caenorhabditis elegans n-3 fatty acid desaturase optimizes fatty acid composition in mammalian cells. Proc Natl Acad Sci U S A 98(7):4050–4054. doi:10.1073/pnas.061040198
Kang JX, Wang J, Wu L, Kang ZB (2004) Transgenic mice: Fat-1 mice convert n-6 to n-3 fatty acids. Nature 427(6974):504–504
Kawakami K, Takeda H, Kawakami N, Kobayashi M, Matsuda N, Mishina M (2004) A transposon-mediated gene trap approach identifies developmentally regulated genes in zebrafish. Dev Cell 7(1):133–144
Kimmel CB, Ballard WW, Kimmel SR, Ullmann B, Schilling TF (1995) Stages of embryonic development of the zebrafish. Dev Dyn 203(3):253–310. doi:10.1002/aja.1002030302
Kris-Etherton PM, Harris WS, Appel LJ, Committee ftN (2003) Fish consumption, fish oil, omega-3 fatty acids, and cardiovascular disease. Arterioscler Thromb Vasc Biol 23(2):e20–e30. doi:10.1161/01.atv.0000038493.65177.94
Lai L, Kang JX, Li R, Wang J, Witt WT, Yong HY, Hao Y, Wax DM, Murphy CN, Rieke A, Samuel M, Linville ML, Korte SW, Evans RW, Starzl TE, Prather RS, Dai Y (2006) Generation of cloned transgenic pigs rich in omega-3 fatty acids. Nat Biotechnol 24(4):435–436, http://www.nature.com/nbt/journal/v24/n4/suppinfo/nbt1198_S1.html
Lieschke GJ, Currie PD (2007) Animal models of human disease: zebrafish swim into view. Nat Rev Genet 8(5):353–367
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2 − ΔΔCT method. Methods 25(4):402–408. doi:10.1006/meth.2001.1262
Marszalek JR, Lodish HF (2005) Docosahexaenoic acid, fatty acid-interacting proteins, and neuronal function: breastmilk and fish are good for you. Annu Rev Cell Dev Biol 21:633–657. doi:10.1146/annurev.cellbio.21.122303.120624
Miller MR, Nichols PD, Carter CG (2008) n-3 oil sources for use in aquaculture—alternatives to the unsustainable harvest of wild fish. Nutr Res Rev 21(2):85–96. doi:10.1017/S0954422408102414
Monroig O, Rotllant J, Sanchez E, Cerda-Reverter JM, Tocher DR (2009) Expression of long-chain polyunsaturated fatty acid (LC-PUFA) biosynthesis genes during zebrafish Danio rerio early embryogenesis. Biochim Biophys Acta 1791(11):1093–1101. doi:10.1016/j.bbalip.2009.07.002
Monroig Ó, Zheng X, Morais S, Leaver MJ, Taggart JB, Tocher DR (2010) Multiple genes for functional ∆6 fatty acyl desaturases (Fad) in Atlantic salmon (Salmo salar L.): gene and cDNA characterization, functional expression, tissue distribution and nutritional regulation. Biochim Biophys Acta (BBA) - Mol Cell Biol Lipids 1801(9):1072–1081. doi:10.1016/j.bbalip.2010.04.007
Monroig Ó, Navarro JC, Tocher DR (2011) Long-chain polyunsaturated fatty acids in fish: recent advances on desaturases and elongases involved in their byosinthesis. Avances Nutr Acuícola 11:257–283
Morais S, Pratoomyot J, Taggart JB, Bron JE, Guy DR, Bell JG, Tocher DR (2011) Genotype-specific responses in Atlantic salmon (Salmo salar) subject to dietary fish oil replacement by vegetable oil: a liver transcriptomic analysis. BMC Genomics 12:255. doi:10.1186/1471-2164-12-255
Morimoto KC, Van Eenennaam AL, DePeters EJ, Medrano JF (2005) Endogenous production of n-3 and n-6 fatty acids in mammalian cells. J Dairy Sci 88(3):1142–1146
Nakamura MT, Nara TY (2004) Structure, function, and dietary regulation of delta6, delta5, and delta9 desaturases. Annu Rev Nutr 24:345–376. doi:10.1146/annurev.nutr.24.121803.063211
Okuley J, Lightner J, Feldmann K, Yadav N, Lark E, Browse J (1994) Arabidopsis FAD2 gene encodes the enzyme that is essential for polyunsaturated lipid synthesis. Plant Cell Online 6(1):147–158. doi:10.1105/tpc.6.1.147
Özogul Y, Özogul F, Alagoz S (2007) Fatty acid profiles and fat contents of commercially important seawater and freshwater fish species of Turkey: a comparative study. Food Chem 103(1):217–223. doi:10.1016/j.foodchem.2006.08.009
Pei DS, Sun YH, Chen CH, Chen SP, Wang YP, Hu W, Zhu ZY (2008) Identification and characterization of a novel gene differentially expressed in zebrafish cross-subfamily cloned embryos. BMC Dev Biol 8:29. doi:10.1186/1471-213X-8-29
Peyou-Ndi MM, Watts JL, Browse J (2000) Identification and characterization of an animal delta(12) fatty acid desaturase gene by heterologous expression in Saccharomyces cerevisiae. Arch Biochem Biophys 376(2):399–408. doi:10.1006/abbi.2000.1733
Ren H-t, Zhang G-q, Li J-l, Tang Y-k, Li H-x, Yu J-h, Xu P (2013) Two Δ6-desaturase-like genes in common carp (Cyprinus carpio var. Jian): structure characterization, mRNA expression, temperature and nutritional regulation. Gene 525(1):11–17. doi:10.1016/j.gene.2013.04.073
Rosen ED, Walkey CJ, Puigserver P, Spiegelman BM (2000) Transcriptional regulation of adipogenesis. Genes Dev 14(11):1293–1307. doi:10.1101/gad.14.11.1293
Saeki K, Matsumoto K, Kinoshita M, Suzuki I, Tasaka Y, Kano K, Taguchi Y, Mikami K, Hirabayashi M, Kashiwazaki N, Hosoi Y, Murata N, Iritani A (2004) Functional expression of a Delta12 fatty acid desaturase gene from spinach in transgenic pigs. Proc Natl Acad Sci U S A 101(17):6361–6366. doi:10.1073/pnas.0308111101
Senadheera SD, Turchini GM, Thanuthong T, Francis DS (2011) Effects of dietary alpha-linolenic acid (18:3n-3)/linoleic acid (18:2n-6) ratio on fatty acid metabolism in Murray cod (Maccullochella peelii peelii). J Agric Food Chem 59(3):1020–1030. doi:10.1021/jf104242y
Spychalla JP, Kinney AJ, Browse J (1997) Identification of an animal omega-3 fatty acid desaturase by heterologous expression in Arabidopsis. Proc Natl Acad Sci U S A 94(4):1142–1147
Stoletov K, Fang L, Choi SH, Hartvigsen K, Hansen LF, Hall C, Pattison J, Juliano J, Miller ER, Almazan F, Crosier P, Witztum JL, Klemke RL, Miller YI (2009) Vascular lipid accumulation, lipoprotein oxidation, and macrophage lipid uptake in hypercholesterolemic zebrafish. Circ Res 104(8):952–960. doi:10.1161/CIRCRESAHA.108.189803
Thanuthong T, Francis DS, Senadheera SP, Jones PL, Turchini GM (2011) LC-PUFA biosynthesis in rainbow trout is substrate limited: use of the whole body fatty acid balance method and different 18:3n-3/18:2n-6 ratios. Lipids 46(12):1111–1127. doi:10.1007/s11745-011-3607-4
Wan JB, Huang LL, Rong R, Tan R, Wang J, Kang JX (2010) Endogenously decreasing tissue n-6/n-3 fatty acid ratio reduces atherosclerotic lesions in apolipoprotein E-deficient mice by inhibiting systemic and vascular inflammation. Arterioscler Thromb Vasc Biol 30(12):2487–2494. doi:10.1161/ATVBAHA.110.210054
Wei C-Y, Wang H-P, Zhu Z-Y, Sun Y-H (2014) Transcriptional factors Smad1 and Smad9 act redundantly to mediate zebrafish ventral specification downstream of Smad5. J Biol Chem. doi:10.1074/jbc.M114.549758
Whelan J, Rust C (2006) Innovative dietary sources of n-3 fatty acids. Annu Rev Nutr 26(1):75–103. doi:10.1146/annurev.nutr.25.050304.092605
Wu X, Ouyang H, Duan B, Pang D, Zhang L, Yuan T, Xue L, Ni D, Cheng L, Dong S, Wei Z, Li L, Yu M, Sun QY, Chen DY, Lai L, Dai Y, Li GP (2012) Production of cloned transgenic cow expressing omega-3 fatty acids. Transgenic Res 21(3):537–543. doi:10.1007/s11248-011-9554-2
Xiong F, Wei ZQ, Zhu ZY, Sun YH (2013) Targeted expression in zebrafish primordial germ cells by Cre/loxP and Gal4/UAS systems. Mar Biotechnol (NY) 15(5):526–539. doi:10.1007/s10126-013-9505-4
Yamashita A, Kawana K, Tomio K, Taguchi A, Isobe Y, Iwamoto R, Masuda K, Furuya H, Nagamatsu T, Nagasaka K, Arimoto T, Oda K, Wada-Hiraike O, Yamashita T, Taketani Y, Kang JX, Kozuma S, Arai H, Arita M, Osuga Y, Fujii T (2013) Increased tissue levels of omega-3 polyunsaturated fatty acids prevents pathological preterm birth. Sci Rep 3:3113. doi:10.1038/srep03113
Zhang P, Zhang Y, Dou H, Yin J, Chen Y, Pang X, Vajta G, Bolund L, Du Y, Ma RZ (2012) Handmade cloned transgenic piglets expressing the nematode fat-1 gene. Cell Reprogram 14(3):258–266. doi:10.1089/cell.2011.0073
Zhang P, Liu P, Dou H, Chen L, Chen L, Lin L, Tan P, Vajta G, Gao J, Du Y, Ma RZ (2013) Handmade cloned transgenic sheep rich in omega-3 fatty acids. PLoS One 8(2):e55941. doi:10.1371/journal.pone.0055941
Zheng X, Tocher DR, Dickson CA, Bell JG, Teale AJ (2004) Effects of diets containing vegetable oil on expression of genes involved in highly unsaturated fatty acid biosynthesis in liver of Atlantic salmon (Salmo salar). Aquaculture 236(1–4):467–483. doi:10.1016/j.aquaculture.2004.02.003
Zhu ZY, Sun YH (2000) Embryonic and genetic manipulation in fish. Cell Res 10(1):17–27
Acknowledgments
The authors thank Hanhua Hu, Xian-Tao Fang, Chun Cai, Junjie Zhang, and Guangzhao Chen for their technical support throughout the research project. This work was supported by the China 863 High-Tech Program Grant 2011AA100404, the China 973 Basic Research Program Grants 2010CB126306 and 2012CB944504, the National Science Fund for Excellent Young Scholars of NSFC Grant 31222052, and FEBL Grant 2011FBZ23 to Y. H. S.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Table S1
(DOCX 26 kb)
Supplemental Table S2
(DOCX 26 kb)
Rights and permissions
About this article
Cite this article
Pang, SC., Wang, HP., Li, KY. et al. Double Transgenesis of Humanized fat1 and fat2 Genes Promotes Omega-3 Polyunsaturated Fatty Acids Synthesis in a Zebrafish Model. Mar Biotechnol 16, 580–593 (2014). https://doi.org/10.1007/s10126-014-9577-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10126-014-9577-9