Abstract
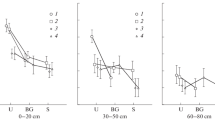
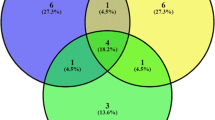
Mines on tree leaves and undamaged leaves were studied to investigate yeast complexes in urban areas (Aesculus hippocastanum, miner — Cameraria ohridella; Betula verrucosa, miner — Caloptilia betulicola; Populus nigra, miner — Lithocolletis populifoliella; Quercus robur, miner — Tischeria companella; Salix caprea, miner — Trachys minuta; Syringa vulgaris, miner — Caloptilia syringella; Tilia cordata, miner — Phyllonorycter issikii; Ulmus laevis, miner — Carpatolechia fugitivella). The abundance and taxonomic structure of yeasts were studied using a surface plating method on solid media (GPY agar). Identification of yeast species was based on the ITS rDNA nucleotide sequence. The average abundance of yeasts during the first stages of mine formation in the internal tissues of leaves was 103 cfu/g. After 23–25 days, during the last stage of larval metamorphosis before mine destruction, the abundance of yeasts in the mines increased by two orders of magnitude to 105 cfu/g. No significant differences were observed in the abundance of yeasts in mines formed by different insects on different trees. A total of twelve yeast species were observed. The fast-growing ascomycetous yeasts Hanseniaspora uvarum and H. occidentalis dominated the mines. On undamaged leaves, the basidiomycetous yeasts Papiliotrema flavescens and Rhodotorula mucilaginosa, typical in the phyllosphere, dominated. The opportunistic yeast Candida parapsilosis was detected in the yeast complexes of all mines examined and was not found on the surface of leaves. Comparison of the relative abundance of yeast species between the studied mines and undamaged leaves using principal component analysis showed that all studied yeast communities in the mines were significantly different from the epiphytic yeast complexes of the undamaged leaves. Thus, miners in urban environments provoke the formation of short-lived endophytic yeast complexes with high abundance of Hanseniaspora. For leaf miners, the yeasts serve primarily as a food source for insect larvae rich in vitamins and amino acids. The adult leaf miners, in turn, contribute to the reproduction of the yeasts and create favorable conditions for their development.



Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Blackwell M (2017) Made for each other: ascomycete yeasts and insects. Microbiol Spectr 5(3):5–3. https://doi.org/10.1128/microbiolspec.FUNK-0081-2016
Blackwell M (2017b) Yeast in insects and other invertebrates. In: Buzzini P, Lachance M-A, Yurkov A.M (eds) Yeasts in natural ecosystems: diversity. Springer International Publishing, Heidelberg: 397–433. https://doi.org/10.1007/978-3-319-62683-3_13
Čadež N, Zupan J, Raspor P (2010) The effect of fungicides on yeast communities associated with grape berries. FEMS Yeast Res 10:619–630. https://doi.org/10.1111/j.1567-1364.2010.00635.x
Camatti-Sartori V, da Silva-Ribeiro R, Valdebenito-Sanhueza R, Pagnocca FC, Echeverrigaray S, Azevedo JL (2005) Endophytic yeasts and filamentous fungi associated with southern Brazilian apple (Malus domestica) orchards subjected to conventional, integrated or organic cultivation. J Basic Microbiol 45:397–402. https://doi.org/10.1002/jobm.200410547
Chand-Goyal T, Spotts R (1996) Enumeration of bacterial and yeast colonists of apple fruits and identification of epiphytic yeasts on pear fruits in the Pacific Northwest United States. Microbiol Res 158(4):427–432. https://doi.org/10.1016/S0944-5013(96)80013-9
de Araujo FV, Soares CAG, Hagler AN, Mendonça-Hagler LC (1995) Ascomycetous yeast communities of marine invertebrates in a Southeast Brazilian mangrove ecosystem. Antonie Van Leeuwenhoek 68:91–99. https://doi.org/10.1007/BF00873096
Fonseca Á, Inácio J (2006) Phylloplane yeasts. In: Péter G, Rosa C (eds) Biodiversity and Ecophysiology of Yeasts. Springer, Berlin, Heidelberg: 263–301. https://doi.org/10.1007/3-540-30985-3_13
Gai CS, Lacava PT, Maccheroni W Jr, Glienke C, Araujo WL, Miller TA, Azevedo JL (2009) Diversity of endophytic yeasts from sweet orange and their localization by scanning electron microscopy. J Basic Microbiol 49:441–451. https://doi.org/10.1002/jobm.200800328
Ganter PF (2006) Yeasts and invertebrate associations. In: Péter G, Rosa C (eds) Biodiversity and Ecophysiology of Yeasts. Springer, Berlin, Heidelberg: 303–370. https://doi.org/10.1007/3-540-30985-3_14
García-Martos P, Hernández-Molina JM, Galán F, Ruiz-Henestrosa JR, García-Agudo R, Palomo MJ, Mira J (1999) Isolation of Hanseniaspora uvarum (Kloeckera apiculata) in humans. Mycopathologia 144:73–75. https://doi.org/10.1023/A:1006900909455
Glushakova AM, Chernov IY (2007) Seasonal dynamic of the numbers of epiphytic yeasts. Microbiology 76(5):590–595. https://doi.org/10.1134/S0026261707050128
Glushakova AM, Chernov IY (2010) Seasonal dynamics of the structure of epiphytic yeast communities. Microbiology 79(6):830–839. https://doi.org/10.1134/S0026261710060160
Glushakova AM, Kachalkin AV (2017) Endophytic yeasts in leaf galls. Microbiology 86:250–256. https://doi.org/10.1134/S0026261717020096
Glushakova AM, Kachalkin AV (2017) Endophytic yeasts in Malus domestica and Pyrus communis fruits under anthropogenic impact. Microbiology 86:128–135. https://doi.org/10.1134/S0026261716060102
Glushakova A, Maximova I, Kachalkin A, Yurkov A (2010) Ogataea cecidiorum sp. nov., a methanol-assimilating yeast isolated from galls on willow leaves. Antonie van Leeuwenhoek 98(1):93–101. https://doi.org/10.1007/s10482-010-9433-5
Glushakova AM, Kachalkin AV, Zheltikova TM, Chernov IYu (2015) Yeasts associated with wind-pollinated plants – leading pollen allergens in Central Russia. Microbiology 84(5):722–725. https://doi.org/10.1134/S0026261715050082
Gouka L, Raaijmakers JM, Cordovez V (2022) Ecology and functional potential of phyllosphere yeasts. Trends Plant Sci 27(11):1109–1123. https://doi.org/10.1016/j.tplants.2022.06.007
Hutchison LJ, Sigler L, Hiratsuka Y (1993) Hyphozyma lignicola sp. nov., a yeast-like hyphomycete from black galls and cankers of trembling aspen (Populus tremuloides). Mycol Res 97:1409–1415. https://doi.org/10.1016/S0953-7562(09)80208-6
Isaeva O, Glushakova A, Yurkov A, Chernov IY (2009) The yeast Candida railenensis in the fruits of English oak (Quercus robur L.). Microbiology 78:355–359. https://doi.org/10.1134/S002626170903014X
Kachalkin AV, Glushakova AM, Venzhik AS (2021) Presence of clinically significant endophytic yeasts in agricultural crops: monitoring and ecological safety assessment. IOP Conf Series: Earth Environ Sci 723(042005):1–6. https://doi.org/10.1088/1755-1315/723/4/042005.1
Kurtzman CP, Fell JW, Boekhout T (2011) The yeasts, a taxonomic study. Elsevier: 1–2080
Lachance MA (1981) Hanseniaspora nodinigri, a new yeast species found in black knots (Dibotryon morbosum) of Prunus virginiana. Can J Microbiol 27(7):651–653. https://doi.org/10.1139/m81-100
Matosevic D (2007) First record of Phyllonorycter issikii and distribution of invasive leaf-miner species from the family Gracillariidae in Croatia. Rad Sumar inst Jastrebar 42(2):127–142. https://hrcak.srce.hr/en/clanak/41056
Medina K, Boido E, Dellacassa E, Carrau F (2012) Growth of non-Saccharomyces yeasts affects nutrient availability for Saccharomyces cerevisiae during wine fermentation. Int J Food Microbiol 157:245–250. https://doi.org/10.1016/j.ijfoodmicro.2012.05.012
Melenti VO, Lezhenina IP, Stankevych SV, Shapetko EV, Matsyura AV, Zabrodina IV, Filatov MO, Molchanova OA (2020) Entomophages of spruce bud scales (Hemiptera: Coccidae) in the Ukrainian eastern forest–steppe. Ukrainian J Ecol 10(6):219–224
Melenti VO, Molchanova OA, Zhukova LV, Golovan LV, Klymenko IV (2020) Prognostication in plant protection. Review of the past, present and future of nonliner dynamics method. Ukrainian J Ecol 10(4):225–234
Milevoj L (2004) The occurrence of some pests and diseases on horse chestnut, plane tree and Indian bean tree in urban areas of Slovenia. Acta Agriculturae Slovenica 83(2):297–300
Miller MW, Phaff HJ (1962) Successive microbial populations in Calimyrna figs. Appl Microbiol 10:394–400. https://doi.org/10.1128/am.10.5.394-400.1962
Mittelbach M, Yurkov AM, Stoll R, Begerow D (2016) Inoculation order of nectar-borne yeasts opens a door for transient species and changes nectar rewarded to pollinators. Fungal Ecol 22:90–97. https://doi.org/10.1016/j.funeco.2015.12.003
Mittelbach M, Vannette RL (2017). Mutualism in yeasts. In: Buzzini P, Lachance M-A, Yurkov A. (eds) Yeasts in Natural Ecosystems: Ecology. Springer International Publishing, Cham. pp. 155–178. https://doi.org/10.1007/978-3-319-61575-2_6
Morais PB, Hagler AN, Rosa CA, Mendonça-Hagler LC (1992) Yeast associated with Drosophila in tropical forests of Rio de Janeiro, Brazil. Can J Microbiol 38:1150–1155. https://doi.org/10.1139/m92-188
Morais PB, Martins MB, Klaczko LB, Mendonça-Hagler LC, Hagler AN (1995) Yeast succession in the Amazon fruit Parahancornia amapa as resource partitioning among Drosophila species. Appl Environ Microbiol 61:4251–4257. https://doi.org/10.1128/aem.61.12.4251-4257.1995
Peay KG, Belisle M, Fukami T (2012) Phylogenetic relatedness predicts priority effects in nectar yeast communities. Proc R Soc B 279:749–758. https://doi.org/10.1098/rspb.2011.1230
Pozo MI, Bartlewicz J, van Oystaeyen A, Benavente A, van Kemenade G, Wäckers F, Jacquemyn H (2018) Surviving in the absence of flowers: do nectar yeasts rely on overwintering bumblebee queens to complete their annual life cycle? FEMS Microbiol Ecol 94:fiy196. https://doi.org/10.1093/femsec/fiy196
Pozo MI, Van Kemenade G, Van Oystaeyen A, Aledón-Catalá T, Benavente A, Van den Ende W, Wäckers F, Jacquemyn H (2020) The impact of yeast presence in nectar on bumble bee behavior and fitness. Ecological Monographs. 90(1):e01393. https://doi.org/10.1002/ecm.1393
Salleo S (2003) Effects of defoliation caused by the leaf miner Cameraria ohridella on wood production and efficiency in Aesculus hippocastanum growing in north-eastern Italy. Trees 7:367–375. https://doi.org/10.1007/s00468-003-0247-1
Sefrova H (2002) Phyllonorycter issikii (Kumata, 1963) – bionomics, ecological impact and spread in Europe (Lepidoptera, Gracillariidae). Acta univ. agric. et silvic. Mendel Brun 2:99–104. https://doi.org/10.13140/2.1.4972.0320
Sefrova H (2002) Phyllonorycter robiniella (Clemens, 1859) – egg, larvae, bionomics and its spread in Europe (Lepidoptera, Gracillariidae). Acta Univ Agric Et Silvic Mendel Brun 3:7–12
Shvydenko IM, Stankevych SV, Zabrodina IV, Bulat AG, Pozniakova SI, Goroshko VV, Hordiiashchenko AYu, Matsyura AV (2021) Diversity and distribution of leaf mining insects in deciduous tree plantations. A Rev Ukrainian J Ecol 11(1):399–408
Spencer DM, Spencer JFT, Defigueroa L, Heluane H (1992) Yeasts associated with rotting citrus fruits in Tucuman Argentina. Mycol Res 96:891–892. https://doi.org/10.1016/S0953-7562(09)81038-1
Stankevych SV, Biletskyj YEM, Zabrodina IV, Yevtushenko MD, Baidyk HV, Lezhenina IP, Filatov MO, Sirous LYA, Yushchuk DD, Stankevych SV, Vasylieva YV, Golovan LV, Zabrodina IV, Lutytska NV, Nakonechna YO, Molchanova OA, Chupryna YY, Zhukova LV (2019) Chronicle of insect pests’ massive reproduction. Ukrainian J Ecol 9(1):262–274
Stankevych SV, YeM B, Zabrodina IV, Yevtushenko MD, Dolya MM, Lezhenina IP, Baidyk HV, Filatov MO, Sirous LA, Melenti VO, Molchanova OA, Zhukova LV, Golovan LV, Polozhenets VM, Nemerytska LV, Klymenko IV (2020) Cycle populations dynamics of harmful insects. Ukrainian J Ecol 10(3):147–161
Tóth R, Nosek J, Mora-Montes HM, Gabaldon T, Bliss JM, Nosanchuk JD, Turner SA, Butler G, Vágvölgyi C, Gácser A (2019) Candida parapsilosis: from genes to the bedside. ASM J Clin Microbiol Rev 32(2):e00111-e118. https://doi.org/10.1128/CMR.00111-18
Vadkertiová R, Molnárová J, Vránová D, Sláviková E (2012) Yeasts and yeast-like organisms associated with fruits and blossoms of different fruit trees. Can J Microbiol 58(12):1344–1352. https://doi.org/10.1139/cjm-2012-0468
van Asbeck EC, Clemons KV, Stevens DA (2009) Candida parapsilosis: a review of its epidemiology, pathogenesis, clinical aspects, typing and antimicrobial susceptibility. Crit Rev Microbiol 35:283–309. https://doi.org/10.3109/10408410903213393
Vannette RL, Fukami T (2016) Nectar microbes can reduce secondary metabolites in nectar and alter effects on nectar consumption by pollinators. Ecology 97:1410–1419. https://doi.org/10.1890/15-0858.1
Wagner D, DeFoliart L, Doak P, Schneiderheinze J (2008) Impact of epidermal leaf mining by the aspen leaf miner (Phyllocnistis populiella) on the grow, physiology, and leaf longevity of quaking aspen. Oecologia 157(2):259–267. https://doi.org/10.1007/s00442-008-1067-1
Funding
The study was funded by the Russian Ministry of Science and Higher Education (agreement no. 075–15-2021–1051) and as a part of the Scientific Project of the State Order of the Government of Russian Federation to Lomonosov Moscow State University (no. 121040800174–6). The study was carried out on the scientific equipment of the Collective Usage Center “I.I. Mechnikov NIIVS”, Moscow, Russia, with the financial support of the project by the Russian Federation represented by the Ministry of Science of Russia, Agreement No. 075-15-2021-676 dated 28.07.2021.
Author information
Authors and Affiliations
Contributions
A.V. Kachalkin: conceptualization; data curation; visualization; writing original draft; funding acquisition; supervision. A.M. Glushakova: conceptualization; data curation; formal analysis; methodology; visualization; writing original draft.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics statement
The study was approved by the Ethics Committee of Lomonosov Moscow State University.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Glushakova, A., Kachalkin, A. Yeasts associated with mines on tree leaves in the urban areas. Int Microbiol 26, 1113–1121 (2023). https://doi.org/10.1007/s10123-023-00370-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10123-023-00370-0