Abstract
Duodenogastroesophageal reflux (DGER) following esophagectomy or gastrectomy can cause severe esophagitis, which impairs patients’ quality of life and increases the risk of esophageal carcinogenesis. It is sometimes resistant to medical treatment, and surgical treatment is considered effective in such cases. However, an optimal operative procedure for medical treatment-resistant reflux esophagitis (RE) after proximal gastrectomy (PG) with esophagogastrostomy (EG) has not yet been established. We performed the right gastroepiploic vessels-preserving antrectomy and Roux-en-Y biliary diversion in a 70-year-old man with medical treatment-resistant severe esophagitis caused by DGER following PG with EG for esophagogastric junction cancer. The postoperative course was uneventful, and esophagogastroduodenoscopy performed on the 19th postoperative day showed marked improvement in the esophageal erosions. The patient reported symptomatic relief. The right gastroepiploic vessels-preserving antrectomy and Roux-en-Y biliary diversion were considered safe and feasible for medical treatment-resistant RE following PG with EG.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Reflux esophagitis (RE) is a major postoperative complication following proximal gastrectomy (PG) with esophagogastrostomy (EG) that significantly impairs patients’ quality of life. According to evidence-based clinical practice guidelines for gastroesophageal reflux disease in the Japanese Society of Gastroenterology [1], surgical treatment can be effective for medical treatment-resistant RE after esophagectomy or gastrectomy. However, there have been few detailed reports on surgical treatment for RE after PG with EG. Here, we report a case of severe RE following PG with EG that was successfully treated with the right gastroepiploic vessels-preserving antrectomy and Roux-en-Y biliary diversion.
Case report
A 70-year-old man was referred to our institution due to severe heart burn and weight loss; the patient’s body mass index (BMI) decreased from 24.4 to 14.6 kg/m2 in a year. He had undergone open PG with EG for esophagogastric junction cancer at another hospital approximately a year before his first visit to our hospital. According to the operative record, EG was performed using a 25-mm circular stapler, and anterior 180-degree partial fundoplication was added for anti-reflux. Esophagogastroduodenoscopy (EGD) performed 2 months after the operation revealed RE, and oral intake was limited because of reflux symptoms. Medical treatment using a gastric acid secretion suppressor, prokinetic agent, protease inhibitor, and gastric mucosal protectant was not effective enough to relieve the symptoms. At the time of the first visit to our hospital, the patient had severe malnutrition with geriatric nutritional risk index (GNRI) [2] of 68.9 (major nutrition-related risk).
EGD performed at our institution demonstrated severe RE (modified Los Angeles classification [3]: Grade D) with bile pigmentation in almost the entire esophageal mucosa (Fig. 1a). Bile reflux gastritis was also observed (Fig. 1b). The esophagogastric anastomosis was widely patent (Fig. 1a), and computed tomography (CT) revealed that it was located in the lower mediastinum (Fig. 1c). Component analysis of the esophageal fluid revealed highly concentrated duodenal contents, including bilirubin, bile acid, trypsin, lipase, pancreatic phospholipase A2, and elastase 1 (Table 1). An upper gastrointestinal series showed gastroesophageal reflux in the upright position. Twenty-four-hour esophageal multichannel intraluminal impedance and pH (MII-pH) monitoring under a potassium-competitive acid blocker (P-CAB; vonoprazan 20 mg) demonstrated frequent weakly acidic and weakly alkaline reflux events (Table 2). The total fraction time at a pH below 4.0 was 0%. High-resolution manometry indicated absent contractility in Chicago classification version 4.0 (Fig. 1d) [4]. Considering the findings of the preoperative examinations, antrectomy and Roux-en-Y biliary diversion were thought to be the best surgical regimen to treat esophagitis caused by the combined reflux of gastric and duodenal juices in this patient.
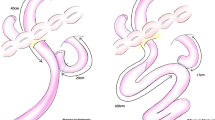
Preoperative findings. a, b Esophagogastroduodenoscopy (EGD) showing severe esophageal erosion and erythematous gastric mucosa with bile reflux. Bile pigmentation was observed in almost the entire esophageal mucosa, and the esophagogastric anastomosis was widely patent (white arrow). There was a pocket in the remnant stomach created by the procedure in the primary operation (black arrow). c Computed tomography (CT) demonstrating esophagogastric anastomosis located in the lower mediastinum (yellow arrow). d Absent contractility observed using high-resolution manometry
A laparotomy was performed with a midline incision from the xiphoid process to the umbilicus. The remnant stomach was mobilized, as shown by the three-dimensional image reconstructed from the preoperative CT using SYNAPSE VINCENT (FUJIFILM Holdings Corporation, Tokyo, Japan) (Fig. 2a). The right gastroepiploic vessels were carefully separated from the antrum and pylorus to preserve the blood flow of the remaining stomach, while the infrapyloric and right gastric arteries were ligated and divided. The duodenum and stomach were transected using a linear stapler, and antrectomy was completed (Fig. 2b). For transection of the stomach, a stapler was fired across the staple line at the anterior wall of the lower gastric body, which was created by closing the entry hole of a circular stapler in the primary operation. Blood supply to the remaining stomach from the right gastroepiploic artery was confirmed using the Medical Imaging Projection System (Mitaka Kohki Co., Ltd., Tokyo, Japan) (Fig. 2c) [5]. The jejunum was transected 20 cm distal to the ligament of Treitz, and Roux-en-Y reconstruction with a 50-cm Roux limb was performed (both gastrojejunostomy and jejunojejunostomy were performed using a 45-mm linear stapler) (Fig. 2d, e). Jejunostomy was performed for supplementary nutrition in case the postoperative oral intake was inadequate. The operative time was 277 min, and blood loss was 50 mL.
Surgical findings. a Preoperative three-dimensional computed tomography (CT) reconstruction; RGEA: right gastroepiploic artery; *staple line of the esophagogastric anastomosis; †staple line of the proximal edge of the remnant stomach; ‡staple line for closure of the circular stapler entry hole. b The right gastroepiploic vessels were preserved (white arrows). The stomach was transected on the dashed line. c Medical Imaging Projection System showed blood supply from the right gastroepiploic artery (white arrows) to the remaining stomach (asterisk). d Roux-en-Y reconstruction with a 50-cm Roux limb was performed. e Schema of the completion image of the right gastroepiploic vessels-preserving antrectomy and Roux-en-Y biliary diversion; RGEA: right gastroepiploic artery; *anterior 180-degree partial fundoplication performed in the primary operation; †staple line created by closing the circular stapler entry hole in the primary operation
The postoperative course was uneventful, and oral intake increased as the reflux symptoms improved. All medications, except P-CAB (vonoprazan 20 mg), were discontinued after the operation. EGD performed on the 19th postoperative day showed marked improvement in esophagitis (modified Los Angeles classification: Grade M) and gastritis (Fig. 3a, b). The duodenal contents were no longer detected in the esophageal fluid (Table 1). The patient was discharged on the 20th postoperative day. The esophageal reflux symptom score in the PGSAS-37 questionnaires [6] a month after the operation significantly improved with a postoperative score of 2.0 from 5.25 pre-surgery. Twenty-four-hour esophageal MII-pH monitoring under P-CAB (vonoprazan 20 mg) performed 2 months after the operation showed a significant decrease in weakly alkaline reflux events and an increase in acidic and weakly acidic reflux events (Table 2). The total fraction time at a pH below 4.0 was 1.3%. The improvement in RE was maintained as seen on EGD performed at 3 months after the operation, and P-CAB was replaced with a proton-pump inhibitor (PPI; lansoprazole 30 mg). The dose of lansoprazole was reduced to 15 mg after 3 months. Oral intake increased to fulfill nutritional requirements without enteral nutrition. The BMI and GNRI increased from 14.6 to 19.3 kg/m2 and from 68.9 (major nutrition-related risk) to 94.3 (low nutrition-related risk), respectively, at 6 months after the operation.
Discussion
PG is indicated for esophagogastric junction and upper-third gastric cancers. EG is a commonly performed reconstruction method after PG, and various anti-reflux techniques have been developed to prevent postoperative RE [7,8,9,10]. Although the development of EG using a linear stapler has decreased the incidence of postoperative RE compared to a circular stapler [11], EG still carries a high risk of postoperative RE.
In the present case, duodenogastroesophageal reflux (DGER), which is the regurgitation of the gastric and duodenal juices into the esophagus, caused severe RE. Although proximal gastrectomy should preserve the function of the pylorus to prevent duodenogastric reflux, when EG is performed in the lower mediastinum, DGER may occur due to negative pressure in the thoracic cavity. Usui et al. [12] reported that the risk of DGER significantly increased in patients after esophagectomy and esophagogastric tube reconstruction when the esophagogastric anastomosis was located lower in the mediastinum, especially below the bottom of the aortic arch. DGER causes adenocarcinoma in the esophagus through esophageal mucosal injury [13, 14]. Therefore, DGER must be treated appropriately and carefully to prevent esophageal carcinogenesis. However, an optimal operative procedure for medical treatment-resistant esophagitis caused by DGER after PG with EG has not yet been established.
The gold standard operation for RE is total or partial fundoplication; however, it cannot be simply applied to patients with RE after PG with EG. This is because the anti-reflux effect of wrapping the remnant stomach around the lower esophagus is uncertain, as it failed in the primary operation of the present case. Completion total gastrectomy with esophagojejunostomy is associated with a high risk of anastomotic leakage due to severe esophageal erosion and technical difficulties of intramediastinal anastomosis at a high location. Subtotal esophagectomy with cervical anastomosis is too invasive as a revision surgery, especially for patients with a poor general condition, as in this geriatric patient. Compared to these operative procedures, antrectomy and Roux-en-Y biliary diversion are technically easy and minimally invasive; therefore, they are applicable for most patients regardless of their general condition. As a similar surgical strategy for RE after EG, D’Journo et al. [15] reported excellent results of the right gastroepiploic vessels-preserving antrectomy and Roux-en-Y biliary diversion for RE following subtotal esophagectomy and gastric reconstruction. In this case series, the esophagogastric anastomosis was located high in the neck or upper mediastinum. Although it was located in the lower mediastinum in the present case, a marked improvement in RE was obtained in a very short period after the operation.
This revisional approach, consisting of Roux-en-Y biliary diversion combined with antrectomy to control gastric acid secretion, had its limitations. Indeed, 24-h esophageal MII-pH monitoring at 2 months postoperatively showed that the alkaline reflux events decreased as planned, but the acid reflux events unexpectedly increased probably due to the decreased neutralization of gastric contents by the duodenal fluid. Therefore, it was not possible to confirm how much gastric acid secretion was actually reduced. However, we consider that antrectomy was worthwhile, given that the esophageal erosions eventually improved and the symptoms were relieved; these outcomes were further maintained when the patient was switched from P-CAB to PPI and the PPI dose was reduced.
In conclusion, the right gastroepiploic vessels-preserving antrectomy and Roux-en-Y biliary diversion were considered safe and feasible for medical treatment-resistant RE following PG with EG.
References
Evidence-based clinical practice guidelines for gastroesophageal reflux disease (GERD) 2021. 3rd ed. pp. 114–115.
Bouillanne O, Morineau G, Dupont C, Coulombel I, Vincent JP, Nicolis I, et al. Geriatric nutritional risk index: a new index for evaluating at-risk elderly medical patients. Am J Clin Nutr. 2005;82(4):777–83. https://doi.org/10.1093/ajcn/82.4.777.
Hoshihara Y, Hashimoto M. Endoscopic classification of reflux esophagitis (in Japanese). Nihon Rinsho. 2000;58(9):1808–12.
Yadlapati R, Kahrilas PJ, Fox MR, Bredenoord AJ, Gyawali CP, Roman S, et al. Esophageal motility disorders on high-resolution manometry: Chicago classification version 4.0. Neurogastroenterol Motil. 2021;33(1):e14058. https://doi.org/10.1111/nmo.14058.
Nishino H, Hatano E, Seo S, Nitta T, Saito T, Nakamura M, et al. Real-time navigation for liver surgery using projection mapping with indocyanine green fluorescence: development of the novel medical imaging projection system. Ann Surg. 2018;267(6):1134–40. https://doi.org/10.1097/SLA.0000000000002172.
Nakada K, Ikeda M, Takahashi M, Kinami S, Yoshida M, Uenosono Y, et al. Characteristics and clinical relevance of postgastrectomy syndrome assessment scale (PGSAS)-45: newly developed integrated questionnaires for assessment of living status and quality of life in postgastrectomy patients. Gastric Cancer. 2015;18(1):147–58. https://doi.org/10.1007/s10120-014-0344-4.
Kamikawa Y, Kobayashi T, Ueyama S, Kambara T, Baba T, Fujii T. A reconstruction technique in oesophagogastrostomy after proximal gastrectomy (in Japanese). Shujutsu. 1998;10:1477–83.
Hosoda K, Yamashita K, Moriya H, Washio M, Mieno H, Ema A, et al. Esophagogastric junction cancer successfully treated by laparoscopic proximal gastrectomy and lower esophagectomy with intrathoracic double-flap technique: A case report. Asian J Endosc Surg. 2018;11(2):160–4. https://doi.org/10.1111/ases.12419.
Omori T, Yamamoto K, Yanagimoto Y, Shinno N, Sugimura K, Takahashi H, et al. A novel valvuloplastic esophagogastrostomy technique for laparoscopic transhiatal lower esophagectomy and proximal gastrectomy for Siewert type II esophagogastric junction carcinoma – the tri double-flap hybrid method. J Gastrointest Surg. 2021;25(1):16–27. https://doi.org/10.1007/s11605-020-04547-0.
Hosogi H, Sakaguchi M, Yagi D, Onishi R, Hashimoto Y, Sakai Y, et al. Side-overlap esophagogastric tube (SO-EG) reconstruction after minimally invasive Ivor Lewis esophagectomy or laparoscopic proximal gastrectomy for cancer of the esophagogastric junction. Langenbecks Arch Surg. 2022;407(2):861–9. https://doi.org/10.1007/s00423-021-02377-5.
Sugita H, Sakuramoto S, Oya S, Fujiwara N, Miyawaki Y, Satoh H, et al. Linear stapler anastomosis for esophagogastrostomy in laparoscopic proximal gastrectomy reduce reflux esophagitis. Langenbecks Arch Surg. 2021;406(8):2709–16. https://doi.org/10.1007/s00423-021-02250-5.
Usui H, Fukuya M, Itatsu K, Miyata K, Miyahara R, Funasaka K, et al. The impact of the location of esophagogastrostomy on acid and duodenogastroesophageal reflux after transthoracic esophagectomy with gastric tube reconstruction and intrathoracic esophagogastrostomy. World J Surg. 2018;42(2):599–605. https://doi.org/10.1007/s00423-021-02377-5.
Theisen J, Peters JH, Fein M, Hughes M, Hagen JA, Demeester SR, et al. The mutagenic potential of duodenoesophageal reflux. Ann Surg. 2005;241(1):63–8. https://doi.org/10.1097/01.sla.0000150072.55037.e3.
Song S, Guha S, Liu K, Buttar NS, Bresalier RS. COX-2 induction by unconjugated bile acids involves reactive oxygen species-mediated signalling pathways in Barrett’s oesophagus and oesophageal adenocarcinoma. Gut. 2007;56(11):1512–21. https://doi.org/10.1136/gut.2007.121244.
D’Journo XB, Martin J, Gaboury L, Ferraro P, Duranceau A. Roux-en-Y diversion for intractable reflux after esophagectomy. Ann Thorac Surg. 2008;86(5):1646–52. https://doi.org/10.1016/j.athoracsur.2008.06.054.
Acknowledgements
We would like to thank Editage (www.editage.com) for English language editing.
Funding
None.
Author information
Authors and Affiliations
Contributions
YH wrote the manuscript. TN, TK, YK, YI, YK, TT, and HS participated in patient treatment. HS, a professor at our department, reviewed the literature. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Informed consent for publication
Informed consent was obtained from the patient for the publication of this report and the use of accompanying images.
Statement of human and animal rights
This study was conducted in accordance with the Ethical Principles for Medical Researching Involving Human Subjects, as outlined in The Declaration of Helsinki.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hojo, Y., Nakamura, T., Kumamoto, T. et al. Marked improvement of severe reflux esophagitis following proximal gastrectomy with esophagogastrostomy by the right gastroepiploic vessels-preserving antrectomy and Roux-en-Y biliary diversion. Gastric Cancer 25, 1117–1122 (2022). https://doi.org/10.1007/s10120-022-01316-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10120-022-01316-7