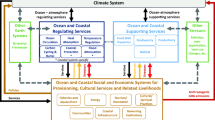
Abstract
Marine social-ecological conditions in the Arctic are rapidly changing. With many transboundary issues, such as shifting ranges of fisheries, biodiversity loss, sea ice retreat, economic development and pollution, greater pan-Arctic assessment and co-management are necessary. We adapted the Ocean Health Index (OHI) to compile pan-Arctic data and evaluate ocean health for nine regions above the Arctic Circle to assess the extent to which pan-Arctic assessment is possible and identify broad social-ecological trends. While the quality and availability of data varied, we assessed and scored nine OHI goals, including the pressures and resilience measures acting upon them. Our results show the Arctic is sustainably delivering a range of benefits to people, but with room for improvement in all goals, particularly tourism, fisheries, and protected places. Successful management of biological resources and short-term positive impacts on biodiversity in response to climate change underlie these high goal scores. The OHI assesses the past and near-term future but does not account for medium- and long-term future risks associated with climate change, highlighting the need for ongoing monitoring, dynamic management, and strong action to mitigate its anticipated effects. A general increase in and standardisation of monitoring is urgently needed in the Arctic. Unified assessments, such as this one, can support national comparisons, data quality assessments, and discussions on the targeting of limited monitoring capabilities at the most pressing and urgent transboundary management challenges, which is a priority for achieving successful Arctic stewardship.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Arctic ecosystems are experiencing profound physical, ecological, and social changes, driven largely by a warming climate and increasing economic development (Hovelsrud et al. 2011; Wassmann et al. 2011). There is a need to establish a baseline of the biophysical and socio-economic dimensions of ocean ecosystems across the Arctic, which can then be used to assess the consequences of future change (IOC/UNESCO 2010). Such baseline assessments are necessary to support strategic and evidence-based decisions for conservation and economic investment through ecosystem-based management (Elliott 2014). Tools, such as the Ocean Health Index (Halpern et al. 2012), can help provide a framework for collating and analysing a wide breadth of baseline data to facilitate management (Borja et al. 2016).
Each Arctic state above the Arctic Circle (Russia, Canada, USA, Norway, Denmark (Greenland)) responds to and manages its Arctic areas through its own national governance system. However, many issues are transboundary in nature, requiring co-management and collaboration (Van Pelt et al. 2017). For example, Arctic ecosystems support globally significant fisheries, with many species already undergoing range shifts and changes in abundance, growth, and phenology (Wassmann et al. 2011; Pinsky et al. 2018). Also, shipping through the Northern Sea Route is increasing annually (Northern Sea Route Information Office 2013), with future projections signalling exponential increases (Smith and Stephenson 2013). Yet, while there is some international cooperation through bodies such as the Arctic Council, there have been few legally binding commitments across nations to collectively and systematically manage the challenges facing the Arctic marine areas. Such examples are limited to international agreements on oil spill preparedness, search and rescue at sea, and the Oslo Declaration preventing fishing in the currently ice-covered central Arctic Ocean (Baker and Yeager 2015; Molenaar 2015). A review by Protection of the Arctic Marine Environment (2013), an Arctic Council working group, found that there was a need for further coordination across institutions (e.g. monitoring conducted on a polar-wide basis using consistent methods, with central data storage) and further cooperation and knowledge-sharing between Arctic countries and institutions. The current disconnect in monitoring and policy at the pan-Arctic scale “limits the ability to efficiently make effective management decisions” (CAFF 2014). Ultimately, it recognised that there is a need to amend existing instruments or develop new ones to strengthen governance for the conservation and sustainable use of the Arctic marine environment.
The Ocean Health Index (OHI) is a tailorable marine assessment framework to comprehensively and quantitatively evaluate ocean health (Halpern et al. 2012; ohi-science.org). It is increasingly being used to help guide thinking around marine management, particularly in data-limited areas, by providing a structure to analyse data availability (Lowndes et al. 2015). We performed an OHI assessment for the Arctic to bring together disparate data and establish an initial baseline of social-ecological conditions, with a focus on highlighting areas of potential concern (both geographically and by goal), exposing data uncertainties, and highlighting potential interactions between marine management goals and short- and long-term outcomes. We discuss our results in the context of future management and decision-making in the pan-Arctic region. Like the OHI (Lowndes et al. 2017), the Arctic OHI (AOHI) is a flexible framework with accompanying open software and can be iteratively improved over time as better data becomes available or stakeholder values are more comprehensively included.
Methods
Pan-Arctic region
Many different definitions of the Arctic exist, with boundaries defined by physical delineations (e.g. climate), latitude, extent of continuous permafrost or sea ice, treeline, or geopolitical borders (Maher 2007). Indeed, spatial delineations of the Arctic even differ between Arctic Council Working Groups (Koivurova 2010). We used exclusive economic zone (EEZ) boundaries that fall above the Arctic Circle as the primary filter for developing an Arctic OHI (AOHI) that can feed into national and international monitoring and policy efforts across the Arctic, with the exception of Southern Greenland which was included because data reported for Greenland often included this area (Fig. 1; Table S1). We excluded the high seas regions because we chose to focus on comparing national Arctic EEZs for management potential. We call our case study region the pan-Arctic area.
We further subdivided the Norwegian, Greenland, and Canadian EEZs based on defined management areas and scales of data reporting. We could not subdivide Russia’s Arctic region, despite it being the largest of all countries, due to Russia’s marine governance structure, which is managed centrally and thus limited data was available at sub-national scale for many goals.
Index calculations
The OHI framework and methods are detailed extensively in the literature and public domains (Halpern et al. 2012, 2015a, b, 2017; ohi-science.org); here, we present a brief overview and focus on changes and new approaches instituted for the AOHI. The OHI is based on assessing the status of an area against a set of goals, which represent key values and benefits people hold and want from a healthy ocean (Table 1). A healthy ocean is defined as one that sustainably delivers a range of benefits to people now and in the future (Halpern et al. 2012). A key design element of the OHI is that it can be adapted to fit different scales and incorporate different values and benefits into its goals, depending on the location and aim of the assessment (Lowndes et al. 2015); for example, OHI assessors determine the number of goals to be evaluated depending on the region of interest and the aspects of ocean health relevant to that region. Numerous OHI assessments have been completed all over the world (Halpern et al. 2013; Elfes et al. 2014; Selig et al. 2015; Longo et al. 2017; Daigle et al. 2017), with many more in progress. Relative to the global assessment, localised assessments are able to take advantage of higher-resolution data, more locally relevant reference points, and goals adapted to local values (Daigle et al. 2017). Where localised data are unavailable, regional assessments can use existing country-level data from the global OHI.
The OHI is calculated by combining individual indicators via a structured framework designed to measure progress towards optimal sustainable delivery of each of the goals (four of which are further subdivided into sub-goals). For the AOHI, we assess 9 of 10 goals in the global OHI, with carbon storage not assessed due to lack of data for calculating a meaningful indicator (Table 1). Each goal and sub-goal are measured on a scale of 0–100, with 100 being the highest possible score. Each goal score, Gi, is calculated as the average of the current status, xi, and the likely future status, \( {\widehat{x}}_{\mathrm{i},\mathrm{F}} \):
Current status measures the most recent year’s performance relative to a reference point of the highest sustainable performance for that goal. Likely, future status captures the near-term (5 years from current status) future performance for the goal based on recent trend in status, (Ti, calculated as the slope in the change of the status score of the previous 5 years), pressures that can threaten the delivery of each goal pi), and resilience factors (ri) which can mitigate these pressures.
Following Halpern et al. (2012), β represents a weighting factor of 0.67, giving trend twice the importance compared to pressure and resilience terms, reflecting the better indication of near-term trajectory that trend provides. Resilience not only measures policies or international conventions to which regions are or are not party but also includes ecological and socio-economic resilience. For climate change-related pressures, resilience layers were set as zero for the AOHI, as no resilience measures adequately offset the pressures of climate change in the Arctic (Bennett et al. 2015). See Tables S2 and S3 for pressure and resilience matrices.
We calculated the overall regional AOHI scores as an equally weighted average of goal scores because, given the heterogeneity of people and environments across the Arctic, determining weightings would be a substantial undertaking and outside the scope of this study. Furthermore, we focus on results comparing goals and regions to avoid focus on higher aggregation and weightings. Weightings could be altered in the future based on stakeholder consultations (Halpern et al. 2014; Daigle et al. 2017). Finally, the overall AOHI score was derived using an area-weighted mean of scores for each region within the assessed area. Below, we describe the data and methods used to calculate AOHI scores. All original data, scripts used for processing, final data layers, and goal models are open access and freely available online at https://github.com/OHI-Science/arc. The AOHI was calculated using the R package “ohicore” (Ocean Health Index 2016).
Arctic Ocean Health Index goal calculations
Given the scale and heterogeneity of the region and to facilitate comparisons across these scales, most goal models and reference points were not changed from methods used in global assessments (Halpern et al. 2017), except for two new goals, marine mammal harvest and artisanal needs, which were adapted natural products and artisanal opportunities goals to better suit the Arctic region (Table 1). An overview and key details on all goals are provided below (with expanded detail in Supplementary materials where necessary); greater detail is provided for the two goals that were adapted for this assessment. Data sources are listed in Table 1, and a full list of data layers can be found in Table S4.
The AOHI focuses on the Arctic region, including partial coastlines of several nations. This scale results in added complexity because many data sources are reported at national-level resolution, and so cannot be directly used. For example, data for the entire USA are not representative of the North Arctic Alaskan coast, as they represent all US regions. Furthermore, the large study area and heterogeneity of the region made obtaining data challenging; when local data were not available or not comparable across all Arctic regions, we often used global spatial data refined to the Arctic region (detailed below). In tailoring the assessment to the Arctic, we were able to replace or adapt 74% of the data layers (n = 81) to be specific to the Arctic. The unchanged 26% mainly consisted of resilience scores for national-level factors, such as whether each country was a signatory to the Convention on International Trade of Endangered Species (CITES).
Due to difficulties and gaps in monitoring the Arctic, pan-Arctic datasets are likely to contain many uncertainties or errors which could affect the results of the AOHI. It is beyond the scope of this work to fully assess and account for these possible sources of uncertainty (Burgass et al. 2017). As an example to help illustrate and understand how uncertainty might affect results, we recalculated the fisheries sub-goal of food provision using a different source dataset for fisheries catch.
Food provision
This goal intends to capture whether seafood provisioning potential is sustainably maximised in each region, through both wild harvest (fisheries sub-goal) and cultivation (mariculture sub-goal). The two sub-goals are combined to give an overall score for food provision, weighted by their tonnage contributions.
Fisheries
The fisheries sub-goal is based on the amount of wild-caught seafood that is sustainably caught within the study area. We used data for this goal taken from the Sea Around Us Project (www.seaaroundus.org), which reconstructs catch data and spatially distributes catch across the world at half-degree resolution (Watson et al. 2004; Pauly and Zeller 2016). In line with (Halpern et al. 2015b), we used catch data to calculate B/BMSYFootnote 1 as a measure of stock status when stock assessments were not available and penalised scores when taxa were not reported at species level to highlight a potential lack of adequate species-level management. Finally, we calculated overall status as the mean of the stock status scores, weighted by the average overall catch in that area, across the time series (see Supplementary materials). We also used an alternative fisheries dataset, from Watson (2017) to test the sensitivity of results to the data used. These data are spatially disaggregated catch data at 0.5° cells, similarly presented to the Seas Around Us dataset. As such, we processed the data in a similar manner and ran this through the AOHI to see how scores might change.
Mariculture
The mariculture sub-goal assesses the sustainability and production of ocean-farmed seafood. Mariculture currently only occurs in Norway and northwest Russia, which were the only two regions to include this sub-goal. We estimated sustainability of production based on Trujillo (2008), as has been done in other OHI studies (see Supplementary materials). The goal model calculates status as the mariculture yield multiplied by sustainability coefficient and normalised by coastal population, which is necessary for undertaking aquaculture. Arctic aquaculture is limited and a global reference point would not be appropriate, so a regional reference was set as the 95th percentile of the top performing region, Norway. While this currently only includes two regions, the expansion of aquaculture in to new regions in the future means the same reference can be used for repeat assessments.
Clean waters
The objective for the clean waters goal is to maintain the ocean free of contamination, pathogens, and anthropogenic nutrient enrichment, for both recreation and environmental health. This goal used four types of pollution data: trash (marine plastics), chemical (runoff, shipping and ports), pathogen (sewage waste), and nutrients (land-based inputs). We refined each of these global data layers (Halpern et al. 2015a, b) at the 1-km2 raster level to only include areas within the AOHI and scaled each raw pollution data layer from 0 to 1, with 1 indicating the highest level of global pollution (Halpern et al. 2015a). We calculated goal status for each region by determining mean rescaled score for each pollution type, subtracting the mean rescaled pollution scores from 1, and combining the four scores using a geometric mean.
Coastal livelihoods and economies
This goal tracks the number and quality of jobs and the amount of revenue produced from marine-related industries and sectors through two sub-goals, livelihoods and economies. A score of 100 reflects productive coastal economies that avoid the loss of ocean-dependent livelihoods while maximising livelihood quality.
Livelihoods
This sub-goal describes livelihood quantity and quality for people living on the coast. The livelihoods sub-goal includes two equally weighted sub-components, the number of jobs, which is a proxy for livelihood quantity, and the per capita average annual wages, which is a proxy for job quality. We obtained job and wage data for marine sectors from within each region at as fine a scale as possible (Tables S7 and S8) and then aggregated it by region (e.g. Arctic Alaska data aggregated from Northwest Arctic Borough and North Slope Borough statistics). Wages were then adjusted using purchasing power parity (PPP), which enables direct comparison between nations and across years. As per Halpern et al. (2017), the reference point for jobs is a temporal comparison using a 5-year moving reference value, in which a score of 100 indicates that the number of marine jobs in a given area has not declined relative to 5 years previously. Similarly, for wages, a score of 100 means the adjusted wage has not declined relative to the highest average annual wage observed across all reporting units 5 years previously.
Economies
The economies sub-goal captures the economic value associated with marine industries based on reported revenue from marine sectors. We obtained revenue data for marine sectors across the Arctic (see Table S9). Values were adjusted by PPP, and as for livelihoods, the reference value was a moving target temporal comparison. A score of 100 indicates that revenue has not decreased compared to its value 5 years previous.
Sense of place
The sense of place goal aims to capture the desire to preserve areas and species that contribute to peoples’ connection to the oceans. This connection might arise from socio-cultural values which local communities have for traditions tied to the existence of these places or species, or from the existence of species or locations that are iconic to a wider public, though they may never be experienced directly.
Iconic species
Iconic marine species fell into three categories: (1) those that are considered globally iconic were taken from the WWF Global Priority and Flagship species list and are selected for all regions in which they occur, (2) those that were considered regionally iconic were taken from CAFF (2010) who identified species that were of wide relevance to indigenous or local people across the Arctic, and (3) those that are locally iconic for cultural or social reasons, which may differ between regions. Locally iconic species were selected based on a review of grey literature to help synthesise and identify which species were culturally important for each region. Determining what is iconic is ultimately subjective, but methods such as those used by Daigle et al. (2017), who defined iconic species as those that appear on Canadian coins in their Canadian OHI assessment, or Roll et al. (2016) who investigated cultural importance of reptiles through internet interest, show that more systematic methods are possible. Iconic species selected for each region can be found in Table S9. The average conservation status of these iconic species (from the International Union for Conservation of Nature (IUCN) Red List) was converted to a numerical score (see Table S13), with the reference point equal to having all these species listed as least concern. Given the plethora of species that could potentially be considered iconic across the Arctic, a more systematic and locally driven selection process would be beneficial in the future.
Protected places
We altered the name of this goal slightly to focus on “protected places,” rather than “lasting special places” found in other OHI assessments, although it is assessed in the same way. This name change was due to the lack of information related to what areas might be considered special in the Arctic, and the fact that many of these areas might be unsuitable for protection given their cultural importance as fishing and hunting grounds. We scored this goal by comparing the amount of protected area within 3 nautical miles offshore and 1 km inland, as defined by the World Database on Protected Areas (WDPA), compared to a reference point of 30% of the total area protected.
Coastal protection
This goal assesses the amount of protection provided by marine and coastal habitats against erosion to coastal areas. In the global assessment, saltmarsh, coral, mangrove, and sea ice habitats are all assessed, while in the Arctic the main habitat offering coastal protection benefits is sea ice. In the AOHI, we assessed the current amount of shoreline sea ice (averaged over the previous 3 years to help reduce the impact of natural variation) compared to the reference condition (average sea ice extent between 1979 and 2000).
Marine mammal harvest
This goal is analogous to the natural products goal that is assessed globally, in that it aims to measure the sustainable harvest of non-food marine resources—in this case the harvesting of marine mammals for furs, ivory, and other resources, which is an important activity commercially and culturally across the Arctic (Hovelsrud et al. 2008). While marine mammals are also eaten, this is not a sole reason for their exploitation and thus, we do not count marine mammal harvests under the food provision goal. Accurate and repeated measures of sub-population sizes are not available for many Arctic marine mammals, which are actively hunted, making construction of a meaningful indicator challenging (Laidre et al. 2015). We therefore only considered species for which either a quota (assumed to be sustainable) or potential biological removal (PBR) rate was available (Table S12); these tended to be pinniped species which haul out of the water, making population estimates easier. Several whale species are hunted by Arctic communities under aboriginal quotas issued by the International Whaling Commission (IWC). These are issued over a 5-year period, rather than annually, and the current quotas are operational until 2018, at which point they can be properly assessed for under- and overharvest. We do not include these whale species in this iteration of the AOHI, but assessing a wider array of species than just pinnipeds would provide a more comprehensive understanding of the status of marine mammal harvest across the Arctic. We calculated the goal status as the ratio of current harvest compared to the current reference point (quota or PBR), similar to the natural products goal for the Southern Ocean OHI assessment (Longo et al. 2017).
A Catch per Catch Limit score (C/CL) was initially calculated to determine landings relative to the quota or PBR (catch limit) for each region and year:
These values were then converted to a stock status score (S′), which ranges from 0 to 1 (Fig. 2) and penalised for over- and under-harvesting (although over-harvesting is more harshly penalised). The lowest value that can be obtained when the catch is lower than the catch limit (i.e. under-harvest) is 0.25 as under-harvesting can be beneficial to rebuild populations. A buffer range of 0.9 to 1.1 was established around a C/CL score of 1.0 to account for uncertainty and fully reward regions aiming to meet quotas. If a region contains more than a single species of hunted marine mammal, then scores were averaged across species.
How stock status score was generated from the Catch per Catch Limit score (Eq. (3)) for marine mammal harvest
Biodiversity
The Biodiversity goal captures the preservation of biodiversity for its aesthetic, existence, and supporting service values into the future. Biodiversity is measured through two proxy sub-goals, habitats, and species. Monitoring biodiversity on a pan-Arctic scale until recently has been disjointed and non-standardised, meaning we relied heavily on global data. With the launch of the Circumpolar Biodiversity Monitoring Plan, this is a goal which hopefully can be improved in the future (M.J. Gill, et al. 2011).
Habitats
Soft-bottom habitat and sea ice were the only habitats for which data exist across the entire assessment region, signalling a requirement for greater monitoring of Arctic marine habitats. Habitat condition for soft bottom subtidal habitat was estimated using a proxy based on the intensity of trawl fishing relative to soft bottom subtidal habitat area (see Supplementary materials for details). Sea ice condition was estimated by comparing the average extent of all current sea ice edge (averaged over the previous 3 years), compared to the reference point of the 1979–2000 average extent.
Species
Species status data come from the IUCN Red List, which assesses entire taxonomic groups in categories of threatened status. Thus, the reference point for this goal is to have all assessed species in the region with an extinction risk status of least concern, scaled so that a score of zero is reached when 75% of species are extinct (following Halpern et al. 2012). Species distributions were determined using IUCN (IUCN 2017) and Aquamaps (Kaschner et al. 2015) species range maps. Species scores (Table S13) were averaged for each 0.5° cell, and then cell scores were averaged for each region (adjusting for the area of the raster cell and number of species present within the cell).
Artisanal needs
This goal was altered from the original “Artisanal Fishing Opportunities” following the approach taken by Daigle et al. (2017), who changed the goal to a more Canadian-centric approach. As such, this goal assesses what is required from the ocean to allow people to hunt and fish artisanally. We recognised three broad themes to assess this:
-
1)
Shoreline sea ice extent—fluctuating and/or diminishing shoreline sea ice can physically restrict access for artisanal hunters and fishers and can shift species distributions, making them harder to track (Laidler et al. 2009; Huntington et al. 2016).
-
2)
Extinction risk of artisanally targeted marine mammals—marine mammals are widely hunted across the Arctic for their furs, ivory, and as a food source (Hovelsrud et al. 2008).
-
3)
Sustainability of artisanally targeted fish stocks—to ensure that artisanal fishers have healthy fish stocks to harvest into the future (Zeller et al. 2011).
Shoreline sea ice scores (h) were calculated in the same way as the coastal protection goal, comparing the current condition of the previous 3 years (Cc) with a reference point of average extent 1979–2000 (Cr) so that:
The less shoreline sea ice, the lower the score.
Extinction risk of targeted marine mammals (xmm) was calculated in the same way as the iconic species sub-goal, but only included marine mammals that are artisanally targeted in each region (Table S12). The reference point was to have all targeted marine mammals at “least concern” status:
where for each IUCN threat category i, Si is the number of assessed species and wi is the status value (Table S13). Sustainability of artisanally targeted fish stocks (xart) was calculated as per the food provision goal but included only those species listed as artisanally targeted in the Sea Around Us Project data and with no under-harvesting penalty applied:
where i is an individual taxon and n is the total number of taxa in the reported artisanal catch for each region throughout the time-series, and C is the average catch, since the first non-null record, for each taxon within each region. Stock status scores (SS) are derived from B/BMSY values—where for B/BMSY < 0.95 (1.0–5% buffer), status declines with direct proportionality to the rate of decline of B with respect to BMSY. No under-harvesting penalty was applied so any B/BMSY score > 0.95, received a SS of 1.
The status for this goal is an average of scores for each of the sub-components:
Norway’s score included only artisanal fish stocks, as artisanal marine mammal hunting is not practiced and sea ice is not plentiful or used for fishing.
Tourism and recreation
A healthy ocean should provide tourism and recreation opportunities for people to enjoy. This goal uses employment in tourism as a proxy for the number of people engaged in tourism and recreation across the Arctic. As such, it should respond dynamically to the number of people actively seeking tourist opportunities in each region, because if tourism increases or decreases then the number of jobs needed to service this sector should respond similarly. The number of tourism jobs are converted to percentage of employment in tourism to adjust for population size differences and multiplied by a sustainability coefficient drawn from the Travel and Tourism Competitiveness Report, which assesses countries for their overall quality, future potential, and long-term sustainability of tourism (Crotti and Mashri 2015). For a region-wide comparison, the reference point is set as the 90th percentile of the best performing region across all years, to account for outliers.
Results
Overall, the Arctic within EEZs scored 78 out of 100 (Fig. 3). Scores varied substantially across assessed regions, from 65 in Jan Mayen to 87 in Svalbard (Fig. 3; Fig. S1). Averaged across the Arctic, three goals scored 90 or above: biodiversity (95), livelihoods and economies (93), and clean waters (90). Marine mammal harvest scored 88 and artisanal needs 81. Coastal protection scored 79 and sense of place scored 77, but there was disparity between the sub-goals, with iconic species scoring 85 and protected places scoring 68. Food provision scored 67 overall, while its sub-goals of mariculture and fisheries scored 36 and 68, respectively. Tourism and recreation was the lowest scoring goal (33).
Overall, species-related goals (species sub-goal of biodiversity and iconic species sub-goal of sense of place) scored highest across all regions but still show substantial room for improvement, with 82% of the 401 marine species assessed by the IUCN in the study region considered to be least concern, 5% near threatened, and 13% threatened (vulnerable, endangered, or critically endangered). Habitat-related goals (coastal protection goal and habitat sub-goal of biodiversity) presented mixed results. For soft-bottomed habitat, large areas remain free from disturbance of commercial fishing. For example, Arctic Alaska’s northern coast is designated a Fishery Control Zone, with no commercial fishing activity allowed. As such, habitat scores were higher in regions, which were more remote and under less fishing pressure (Russian Arctic, Greenland, Canada, Arctic Alaska). The Barents and Norwegian Seas are subject to trawling for key target species (Buhl-Mortensen et al. 2016), meaning Arctic Norway and Jan Mayen received lower scores for soft-bottom habitat (81 and 63, respectively). Sea ice-related goals (sea ice edge extent within the habitat sub-goal, shoreline sea ice extent for coastal protection) are also high with the exception of Arctic Norway and Svalbard, which are lower because the Barents Sea has experienced some of the most significant warming and variable sea ice conditions on the planet (Sato et al. 2014; Eriksen et al. 2017; Onarheim and Årthun 2017).
The clean waters goal scored well for all regions; this reflects the low population density of the Arctic in general and few sources of pollution. Norway scored the lowest (87), which was largely driven by a much higher chemical pollution score than other regions and reflects both the higher population density along the coastline and prominent shipping routes.
The protected places sub-goal of sense of place varied widely across regions, with both very high and very low scores. Although there has been an expansion of Arctic protected areas over the last 50 years, across the CAFF area, only 4.2% of marine areas are protected, with terrestrial areas garnering a much higher 20.2% protection, showing that there is room for improvement in this area (CAFF and PAME 2017).
Marine mammal harvest scored high across all regions, except Arctic Norway and Jan Mayen, which scored lower primarily due to a significant under-harvest of seals compared to their quotas. Significantly, the Canadian commercial seal hunt occurs largely outside of the AOHI study area and so was not included. Marine mammals were also considered in the artisanal needs goal, which also included sustainability of artisanal fish stocks and extent of shoreline sea ice. Scores for this goal were generally high, in part because the fisheries component of the goal did not penalise underfishing and many artisanal stocks appear healthy.
Fisheries scores were between 50 and 75, with the exception of West Greenland (87), Arctic Norway (87), and Jan Mayen (11). High scores for Arctic Norway and West Greenland align with landings primarily being from Marine Stewardship Council (MSC) certified fisheries. The Jan Mayen score potentially reveals issues with the spatial distribution of the catch data from SAUP. For this region, 96% of landings are classified as “marine fish unidentified”—which is heavily penalised for poor taxonomic reporting in the OHI. Unidentified landings for Arctic Norway and Svalbard (part of the same management area) are much lower at 4 and 13%, respectively, meaning the methods for distributing catch data for this region may be causing unfair penalty.
Running the Watson (2017) dataset through the AOHI reduced the overall Index Fisheries score by 13 (68 to 55); however, overall AOHI scores declined by just one point from 78 to 77 when using this alternative fisheries dataset.
From a regional perspective, Watson’s data resulted in a dramatic improvement of Jan Mayen’s Fisheries score, from 11 with the SAUP data to 59 with Watson’s (Fig. 4). East Greenland also saw an improvement using Watson’s data (54 to 62). However, all other regions saw a decrease in scores. Arctic Alaska, Nunavut, Canadian Beaufort, and Svalbard all had scores reduced by less than 10, yet Arctic Russia (20), Arctic Norway (28), and West Greenland (34) all had large decreases. All regions except West Greenland (83 down to 79) and Jan Mayen (65 up to 75) showed a decrease in overall OHI scores of 2 or less.
Discussion
The Arctic is a globally unique and important geography for biophysical, cultural, and economic reasons; yet, its management is disjointed and resources for monitoring are limited. We have made the first attempt to piece together disparate datasets across the pan-Arctic area to quantitatively assess ocean health from a human-centric perspective. Despite challenges, we have shown that such studies are possible and provide an initial baseline of current pan-Arctic social-ecological conditions using freely available data and sharing the AOHI framework and open code for future iteration and improvement (Lowndes et al. 2017). Given the Arctic is rapidly changing, baselines need to be established and systems continually evaluated in order to inform management (Hussey et al. 2016). We discuss the context of our results below and highlight spatial patterns of interest.
Spatial patterns and management considerations
Relatively high scores for species-related goals are likely due to rebuilding of once heavily exploited whale and pinniped populations, absence of large commercial fishing fleets in many parts of the Arctic, and increased productivity from climate change being beneficial to many fish species, at least in the short term (Mcrae et al. 2012). Our assessment falls largely in line with the Arctic Species Trend Index (ASTI), which found an increase in Arctic marine vertebrates from 1970 to 2005 (Eamer et al. 2012). While the AOHI reflects the fact that in the short-term climate change may be having a potentially beneficial effect for many species, it does not account for long term risks to marine biodiversity. Arctic species often have particularly narrow temperature ranges and are highly susceptible to invasions. Projections indicate that the Arctic could be at high risk of invasive species and localised extinctions, highlighting the need for ongoing monitoring and dynamic and predictive management (Cheung et al. 2009; Eamer et al. 2013; Garciá Molinos et al. 2016).
Arctic sea ice is critical for climate regulation, coastal protection, and as habitat to a range of species that live in or around the ice or use it for life history events, such as reproduction, moulting, or resting (Laidre et al. 2015). It is therefore also important to people who depend on sea ice-associated species as resources and for cultural reasons. Sea ice dynamics in the Arctic are being altered by warming at twice the global mean rate, and some projections suggest an ice-free summer by 2040 (AMAP 2017). Therefore, measuring these changes is an important component of the AOHI (Overland and Wang 2013). High sea-ice related scores (habitat and coastal protection) are in keeping with the global OHI assessment, which found reduced sea ice scores in sub-Arctic countries (Lithuania, Sweden, Finland, Norway (which includes Svalbard), Estonia, Latvia), but not yet at higher latitudes (Halpern et al. 2017). While the data we used considers temporal and spatial extent, it does not consider depth of sea ice, which may be important to sea ice-associated biodiversity (Kovacs et al. 2011). Further work could also include representing the shifting seasonality of sea ice (Haine and Martin 2017).
The livelihoods and economies goals do not currently consider informal economies or subsistence livelihoods, which are prevalent in many regions across the Arctic (Larsen et al. 2015). We were unable to find data to support the development of an indicator reflecting these less formal elements of the economy at the pan-Arctic scale, particularly as the informal economy varies widely between regions (Schmidt et al. 2015). Furthermore, many indigenous communities view economic development differently; while many wish to maintain traditional lifestyles, many communities are keen to mitigate high levels of poverty, ill health, and food security issues through full-time employment and the benefits that economic development can bring (Stewart et al. 2011; McCauley et al. 2016). These issues are inherently local, making it challenging to find a meaningful reference point at the pan-Arctic scale. Gaining a better contextualised understanding of how people conceptualise the elements of wellbeing within each region might yield information allowing relative change in wellbeing to be compared (Woodhouse et al. 2015).
Arctic tourism above the Arctic Circle is largely dominated by cruise ship tourism, which has grown markedly since 2008, particularly for Svalbard where tourism employment is high relative to the permanent population (Viken 2011). However, numbers remain far below more accessible sub-Arctic areas (Maher 2017), indicating that demand may be present and there is much room for growth, as indicated by the AOHI scores. The future of tourism in the Arctic is unclear but has the potential for significant social, economic, and ecological impacts, both positive and negative (Stewart et al. 2015). There are already concerns of exceeding carrying capacity in countries such as Iceland, which has seen a six-fold increase in tourism since 2008 (Maher 2017). Balancing the economic benefits of tourism while maintaining the environmental and cultural sense of place that makes tourism attractive is a difficult undertaking. Setting out a shared vision for Arctic tourism and developing infrastructure in areas that will have positive social impacts and minimise negative environmental impacts should be a priority for Arctic nations.
The marine mammal harvest goal indicates that marine mammals with population data are being harvested sustainably across the Arctic, showing that sustainable management of these species is possible if supported by scientific research. For example, although controversial, the management of the Canadian harp seal hunt can be considered a conservation success, with the number of individuals rising from a low of 1.1 million in the early 1970s to over seven million today. This supports the importance of robust monitoring and evidence-based quotas (Hammill et al. 2015). However, the analysis of marine mammal harvest was restricted by the need for information relating to both landings of marine mammals and viable population estimates leading to quotas or PBR estimates, which unfortunately excluded most marine mammals in each region. Abundance and trend data for Arctic marine mammals is poor or largely absent, which makes quantifying the sustainability of harvests difficult. Obtaining population estimates for new metrics from sources, such as indigenous knowledge or the Circumpolar Biodiversity Monitoring Plan (Gill et al. 2011), should be a priority in aiding management of marine mammals, particularly in the face of climate change (Laidre et al., 2015; Gill et al. 2011). Ideally, establishing metrics and quotas would be a systematic process that would enhance co-management of marine mammal species locally and at a pan-Arctic scale.
The food provision scores reward areas with the highest levels of sustainable catch, showing the benefits of well managed and productive fisheries. Arctic fisheries are an increasingly controversial topic; seemingly offering large potential for food provision and economic benefits, but with high ecological risk (Lam et al. 2016). The USA has recognised this risk by closing a large proportion of its Arctic EEZ to industrial fishing, and all the Arctic countries have signed an agreement to prohibit fishing in the Central Arctic Ocean. Fisheries in the Barents and Norwegian Seas have been recovering from previous exploitation, improved management, and beneficial effects of climate change, which have led to a threefold increase in spawning stock biomass in the last 15 years (Dalpadado et al. 2014; Grønnevet 2016). Using a different dataset revealed changes in scores, showing that data selection can be a critical component driving scores. While our study often did not have the luxury of multiple datasets, exploring two Fisheries goal datasets revealed potential issues in each, showing that this type of sensitivity analysis would be useful in similar studies. The increased score for Jan Mayen with Watson’s data shows that key differences exist in the spatial disaggregation of the catch data in this area; the higher SAUP scores for Norway and West Greenland, where commercial fisheries operate mainly under MSC certification, align with what we would expect to see.
Implications for future pan-Arctic management
Our AOHI assessment provides a starting point for consideration of pan-Arctic social-ecological dynamics, which like other composite indicators can be iteratively improved over time as more and better data become available and dynamics are better understood (Burgass et al. 2017). Understanding current limitations and how social-ecological systems are changing is necessary for effective management (Harris et al. 2017). Many of the goals within the AOHI are transboundary in nature and require co-management (biodiversity, marine mammal harvest, coastal protection, tourism and recreation, clean waters, and fisheries). Consideration of these interlinkages is critical for management; the heterogeneity of the Arctic means that system dynamics are important not only between different goals but also across regions and localities. For example, co-management of many marine mammal species, such as walrus and polar bears across national borders and between indigenous groups, has been successful in ensuring sustainability and maintaining human wellbeing (Laidre et al. 2015).
The AOHI provides a snapshot in time of the current status of the Arctic, as well as an indication of the near-term future state. The biggest driver of change in the Arctic is climate change. Although it is included in our assessment and its effects are already being felt, these will be most noticeable over the medium and long terms (Bennett et al. 2015). Subsequent AOHI assessments will be required to track the impacts of climate change across the region. As the Arctic “opens up,” the opportunities for economic development will become even more numerous for oil and gas extraction, tourism, shipping, and infrastructure. Understanding the risks that climate change poses to the health of the ocean and the wellbeing of the people who depend on ocean resources will require multi-faceted modelling, with a strong emphasis on social science (Ford et al. 2015). Given the sensitivities of people and environments in the Arctic, pan-Arctic assessments, such as ours, can help inform decision-making on strategies for investment to minimise social-ecological risk and maximise benefits across the region. Pan-Arctic plans for environmental protection and sustainable development would limit ad hoc developments, which could otherwise pose severe risks to unique ecological communities or areas of biodiversity. Similarly, given the heterogeneity of the region and in order to protect and restore the full range of biodiversity across the Arctic, coordinating efforts across large scales is required to ensure an ecologically coherent network of protected areas (Harris et al. 2017).
A key area of uncertainty for marine management in the Arctic is the potential for tipping points or thresholds, which can be classified as periods of rapid, non-linear (Serrao-Neumann et al. 2016). Given the range and scale of pressures on the Arctic, particularly climate change, these tipping points may well be crossed even in the short-term, which would compromise the predictions of the AOHI. Late action to halt or reverse a tipping point is highly ineffective compared with early identification and preservation of system resilience (Selkoe et al. 2015). Through our assessment, we provide data for a range of different physical, biological, and social data layers that can be accessed from raw data through to aggregated final scores of the AOHI to inform system-wide management. Monitoring of these layers should be focused towards identifying potential tipping points in order that pre-emptive action can be taken.
Ultimately, threats, such as climate change, go beyond pan-Arctic governance and will require global mitigation in reducing greenhouse gas emissions. Despite climate change being a huge risk to the Arctic, the disconnection of communities, authorities, and governments at the pan-Arctic scale prohibits a clear and united message. This work provides foundational datasets, which can be of use for both pan-Arctic assessment and local-decision-making for Arctic futures. Ensuring a participatory process and inclusion of the full range of stakeholders which is often vital in ensuring evaluation of management strategies or alternative futures is appropriate and useful (Dichmont and Fulton 2017). Promoting pan-Arctic monitoring, management, and decision-making, joined with a bottom-up approach of case studies and storytelling, could help position the Arctic as a bellwether for climate change and help create increasingly ambitious, robust, and equitable climate policy at the global scale. A data-driven approach, such as the OHI, allows the quantification and clear communication of broad results to a range of stakeholders both inside and outside the Arctic. It can therefore help communicate complex issues and include more stakeholders through transparency and open web-based tools. However, when being used for management purposes, the data and models must be interpreted carefully as with any scientific output. As such, we have been transparent with the data that has been used and their limitations such that these can be factored in to any future use of the AOHI.
The OHI goals are intended to provide a broad comparative framework and to encourage thinking about what we consider to be a “healthy” system and how far we are from that state in relation to a range of human-defined functions and goals. It can be updated and improved iteratively over time should new data become available or should new social considerations need to be factored in. This will be particularly important for risks, such as ocean acidification and climate change, which has the potential to alter the structure and biodiversity of ecosystems (and subsequently impact people), but the long-term effects of which are not well understood (Lam et al. 2016). Likewise, while oil and gas extraction is not included here (as with other OHI assessments), due to it being intrinsically unsustainable, it is undoubtedly of huge economic and social importance to some Arctic areas and therefore stakeholders may wish to include it in future assessments.
Conclusions
Our AOHI is a first step towards measuring the status of the ocean across the high pan-Arctic area. In general, we found the Arctic to be sustainably delivering a range of benefits to people, with room for improvement in all goals, but particularly in sustainable tourism, mariculture, fisheries, and protected places. Biodiversity-focussed goals presented encouraging scores, showing how improved ecosystem management through recovering fisheries and sustainable marine mammal exploitation, were having a positive effect. However, the assessment was constrained by limitations in pan-Arctic data, in particular the disjointed and non-comparable nature of data from different Arctic regions. While validating a composite index is a difficult undertaking, the process of its formulation and understanding where conceptual and data uncertainties are located is inevitably crucial for informing management. Obtaining comparable data from across the Arctic to minimise these uncertainties is a priority for informing robust pan-Arctic stewardship; such efforts should be targeted towards the most pressing and urgent transboundary management challenges, such as fisheries, biodiversity, and economic development (shipping, tourism, extractive activities; Tesar et al., 2016).
Notes
For a particular fish stock, the ratio of observed biomass (B) to the biomass that would provide maximum sustainable yield (BMSY). When B/BMSY = 1, then biomass equals BMSY. If B/BMSY falls below 1, biomass is too low to provide maximum sustainable yield.
References
AMAP (2017) Snow, water, ice and permafrost in the Arctic (SWIPA) 2017. Oslo, Norway
Baker B, Yeager B (2015) Coordinated ocean stewardship in the Arctic: needs , challenges and possible models for an Arctic ocean coordinating agreement. Transnatl Environ Law 4:359–394. https://doi.org/10.1017/S204710251500019910.1017/S2047102515000151
Bennett JR, Shaw JD, Terauds A, Smol JP, Aerts R, Bergstrom DM, Blais JM, Cheung WW, Chown SL, Lea M-A, Nielsen UN, Pauly D, Reimer KJ, Riddle MJ, Snape I, Stark JS, Tulloch VJ, Possingham HP (2015) Polar lessons learned: long-term management based on shared threats in Arctic and Antarctic environments. Front Ecol Environ 13:316–324. https://doi.org/10.1890/140315
Borja Á, Elliott M, Andersen JH, Berg T, Carstensen J, Halpern BS, Heiskanen A-S, Korpinen S, Lowndes JSS, Martin G, Rodriguez-Ezpeleta N (2016) Overview of integrative assessment of marine systems: the ecosystem approach in practice. Front Mar Sci 3:68. https://doi.org/10.3389/fmars.2016.00020
Buhl-Mortensen L, Ellingsen KE, Buhl-Mortensen P, Skaar KL, Gonzalez-Mirelis G (2016) Trawling disturbance on megabenthos and sediment in the Barents Sea: chronic effects on density, diversity, and composition. ICES J Mar Sci J du Cons 73:i98–i114. https://doi.org/10.1093/icesjms/fsv200
Burgass MJ, Halpern BS, Nicholson E, Milner-Gulland EJ (2017) Navigating uncertainty in environmental composite indicators. Ecol Indic 75:268–278. https://doi.org/10.1016/j.ecolind.2016.12.034
CAFF (2010) Arctic biodiversity trends 2010—selected indicators of change. CAFF International Secretariat, Akureyri
CAFF (2014) Marine ecosystem monitoring. http://www.caff.is/marine. Accessed 19 Nov 2014
CAFF, PAME (2017) Arctic protected areas: indicator report 2017. Akureyi, Iceland
Cavalieri DJ, Parkinson CL, Gloersen P, Zwally H (2015) Sea ice concentrations from Nimbus-7 SMMR and DMSP SMM/I-SSMIS passive microwave data. 1979-2015. NASA NSIDC DAAC, Boulder. https://doi.org/10.5067/8GQ8LZQVL0VL
Cheung WWL, Lam VWY, Sarmiento JL, Kearney K, Watson R, Pauly D (2009) Projecting global marine biodiversity impacts under climate change scenarios. Fish Fish 10:235c251. https://doi.org/10.1111/j.1467-2979.2008.00315.x
Crotti R, Mashri T (2015) The Travel & Tourism Competitiveness Report 2015. Geneva: World Economic Forum
Daigle RM, Archambault P, Halpern BS, Stewart Lowndes JS, Côté IM (2017) Incorporating public priorities in the Ocean Health Index: Canada as a case study. PLoS One 12:e0178044. https://doi.org/10.1371/journal.pone.0178044
Dalpadado P, Arrigo KR, Hjøllo SS, Rey F, Ingvaldsen RB, Sperfeld E, Van Dijken GL, Stige LC, Olsen A, Ottersen G (2014) Productivity in the Barents Sea—response to recent climate variability. PLoS One 9:e95273. https://doi.org/10.1371/journal.pone.0095273
Dichmont CM, Fulton EA (2017) Fisheries science and participatory management strategy evaluation: eliciting objectives, visions and systems models. In: Bunnefeld N, Nicholson E, Milner-Gulland EJ (eds) Decision-making in conservation and natural resource management. Cambridge University Press, Cambridge, UK, p 275
Eamer J, Russell DE, McRae L, Böhm M, Deinet S. Collen B, Gill MJ (2012) The Arctic Species Trend Index 2011: Update of the ASTI, an in‐depth look at marine species and development of spatial analysis techniques. CAFF Assessment Series No. 9. Conservation of Arctic Flora and Fauna, Iceland
Eamer J, Donaldson GM, Gaston AJ, Kosobokova KN, Lárusson KF, Melnikov IA, Reist JD, Richardson E, Staples L, von Quillfeldt CH (2013) Life linked to ice: A guide to sea‐ice‐associated biodiversity in this time of rapid change. CAFF Assessment Series No. 10. Conservation of Arctic Flora and Fauna, Iceland
Elfes CT, Longo C, Halpern BS, Hardy D, Scarborough C, Best BD, Pinheiro T, Dutra GF (2014) A regional-scale ocean health index for Brazil. PLoS One 9:1–11. https://doi.org/10.1371/journal.pone.0092589
Elliott M (2014) Integrated marine science and management: wading through the morass. Mar Pollut Bull 86:1–4. https://doi.org/10.1016/j.marpolbul.2014.07.026
Eriksen E, Rune H, Gjøs H, Primicerio R (2017) Spatial and temporal changes in the Barents Sea pelagic compartment during the recent warming. Prog Oceanogr 151:206–226. https://doi.org/10.1016/j.pocean.2016.12.009
Ford JD, McDowell G, Pearce T (2015) The adaptation challenge in the Arctic. Nat Clim Chang 5:1046–1053. https://doi.org/10.1038/nclimate2723
Garciá Molinos J, Halpern BS, Schoeman DS, Brown CJ, Kiessling W, Moore PJ, Pandolfi JM, Poloczanska ES, Richardson AJ, Burrows MT (2016) Climate velocity and the future global redistribution of marine biodiversity. Nat Clim Chang 6:83–88. https://doi.org/10.1038/nclimate2769
Gill MJ, Crane K, Hindrum R, Arneberg P, Bysveen I, Denisenko NV, Gofman V, Grant-Friedman A, Gudmundsson G, Hopcroft RR, Iken K, Labansen A, Liubina OS, Melnikov IA, Moore SE, Reist JD, Sirenko BI, Stow J, Ugarte F, Vongrave D, Watkins J (2011) Arctic marine biodiversity monitoring plan (CBMP-MARINE PLAN). CAFF International Secretariat, Akureyri
Grønnevet L (2016) The joint Russian-Norwegian governance of the Barents Sea LME fisheries. Environ Dev 17:296–309. https://doi.org/10.1016/j.envdev.2015.07.006
Haine TWN, Martin T (2017) The Arctic-Subarctic sea ice system is entering a seasonal regime: implications for future Arctic amplification. Sci Rep 7:4618. https://doi.org/10.1038/s41598-017-04573-0
Halpern BS, Longo C, Hardy D, McLeod KL, Samhouri JF, Katona SK, Kleisner K, Lester SE, O’Leary J, Ranelletti M, Rosenberg AA, Scarborough C, Selig ER, Best BD, Brumbaugh DR, Chapin FS, Crowder LB, Daly KL, Doney SC, Elfes C, Fogarty MJ, Gaines SD, Jacobsen KI, Karrer LB, Leslie HM, Neeley E, Pauly D, Polasky S, Ris B, St Martin K, Stone GS, Sumaila UR, Zeller D (2012) An index to assess the health and benefits of the global ocean. Nature 488:615–620. https://doi.org/10.1038/nature11397
Halpern BS, Longo C, McLeod KL, Cooke R, Fischhoff B, Samhouri JF, Scarborough C (2013) Elicited preferences for components of ocean health in the California Current. Mar Policy 42:68–73. https://doi.org/10.1016/j.marpol.2013.01.019
Halpern BS, Longo C, Scarborough C, Hardy D, Best BD, Doney SC, Katona SK, McLeod KL, Rosenberg AA, Samhouri JF (2014) Assessing the health of the U.S. west coast with a regional-scale application of the Ocean Health Index. PLoS One 9:e98995. https://doi.org/10.1371/journal.pone.0098995
Halpern BS, Frazier M, Potapenko J, Casey KS, Koenig K, Longo C, Lowndes JS, Rockwood RC, Selig ER, Selkoe KA, Walbridge S (2015a) Spatial and temporal changes in cumulative human impacts on the world’s ocean. Nat Commun 6:7615. https://doi.org/10.1038/ncomms8615
Halpern BS, Longo C, Lowndes JSS, Best BD, Frazier M, Katona SK, Kleisner KM, Rosenberg AA, Scarborough C, Selig ER (2015b) Patterns and emerging trends in global ocean health. PLoS One 10:e0117863. https://doi.org/10.1371/journal.pone.0117863
Halpern BS, Frazier M, Afflerbach J, O’Hara C, Katona S, Stewart Lowndes JS, Jiang N, Pacheco E, Scarborough C, Polsenberg J (2017) Drivers and implications of change in global ocean health over the past five years. PLoS One 12:1–23. https://doi.org/10.1371/journal.pone.0178267
Hammill MO, Stenson GB, Doniol-Valcroze T, Mosnier A (2015) Conservation of northwest Atlantic harp seals: past success, future uncertainty? Biol Conserv 192:181–191. https://doi.org/10.1016/j.biocon.2015.09.016
Harris PT, Macmillan-lawler M, Kullerud L, Rice JC (2017) Arctic marine conservation is not prepared for the coming melt. ICES J Mar Sci 1–11 . doi: https://doi.org/10.1093/icesjms/fsx173
Hovelsrud GK, Mckenna M, Huntington HP (2008) Marine mammal harvests and other interactions with humans. Ecol Appl 18:135–147. https://doi.org/10.1890/06-0843.1
Hovelsrud GK, Poppel B, Van Oort B, Reist JD (2011) Arctic societies, cultures, and peoples in a changing cryosphere. Ambio 40:100–110. https://doi.org/10.1007/s13280-011-0219-4
Huntington HP, Quakenbush LT, Nelson M (2016) Effects of changing sea ice on marine mammals and subsistence hunters in northern Alaska from traditional knowledge interviews. Biol Lett 12:20160198. https://doi.org/10.1098/rsbl.2016.0198
Hussey NE, Harcourt RG, Auger-Methe M (2016) Now is the time to protect the Arctic. Science 354(80):1243–1243. https://doi.org/10.1126/science.aah6603
IOC/UNESCO (2010) Why monitor the Arctic Ocean?
IUCN (2017) IUCN Red List of threatened species. Version 2017-2. www.iucnredlist.org
Kaschner K, Rius-Barile J, Kesner-Reyes K, Garilao C, Kullander S, Rees T, Froese R (2015) AquaMaps: predicted range maps for aquatic species. Available: http://www.aquamaps.org. Accessed 17 Nov 2014
Koivurova T (2010) Limits and possibilities of the Arctic Council in a rapidly changing scene of Arctic governance. Polar Rec (Gr Brit) 46:146–156. https://doi.org/10.1017/S0032247409008365
Kovacs KM, Lydersen C, Overland JE, Moore SE (2011) Impacts of changing sea-ice conditions on Arctic marine mammals. Mar Biodivers 41:181–194. https://doi.org/10.1007/s12526-010-0061-0
Laidler GJ, Ford JD, Gough WA, Ikummaq T, Gagnon AS, Kowal S, Qrunnut K, Irngaut C (2009) Travelling and hunting in a changing Arctic: assessing Inuit vulnerability to sea ice change in Igloolik, Nunavut. Clim Chang 94:363–397. https://doi.org/10.1007/s10584-008-9512-z
Laidre KL, Stern H, Kovacs KM, Lowry L, Moore SE, Regehr EV, Ferguson SH, Wiig Ø, Boveng P, Angliss RP, Born EW, Litovka D, Quakenbush L, Lydersen C, Vongraven D, Ugarte F (2015) Arctic marine mammal population status, sea ice habitat loss, and conservation recommendations for the 21st century. Conserv Biol 00:1–14. https://doi.org/10.1111/cobi.12474
Lam VWY, Cheung WWL, Sumaila UR (2016) Marine capture fisheries in the Arctic: winners or losers under climate change and ocean acidification? Fish Fish 17:335–357. https://doi.org/10.1111/faf.12106
Larsen JN, Schweitzer P, Petrov A (2015) Arctic social indicators. https://doi.org/10.6027/TN2014-568
Longo CS, Frazier M, Doney SC, Rheuban JE, Halpern BS (2017) Using the Ocean Health Index to identify opportunities and challenges to improving Southern Ocean ecosystem health. Front Mar Sci 4:1–18. https://doi.org/10.3389/fmars.2017.00020
Lowndes JSS, Pacheco EJ, Best BD, Scarborough C, Longo C, Katona SK, Halpern BS (2015) Best practices for assessing ocean health in multiple contexts using tailorable frameworks. PeerJ 3:e1503. https://doi.org/10.7717/peerj.1503
Lowndes JSS, Best BD, Scarborough C, Afflerbach JC, Frazier MR, O’Hara CC, Jiang N, Halpern BS (2017) Our path to better science in less time using open data science tools. Nat Ecol Evol 1:0160. https://doi.org/10.1038/s41559-017-0160
Maher PT (2007) Arctic tourism: a complex system of visitors, communities, and environments. Polar Geogr 30:1–5. https://doi.org/10.1080/10889370701666507
Maher PT (2017) Tourism futures in the Arctic. In: Latola K, Savela H (eds) The interconnected Arctic—UArctic Congress 2016. Springer International Publishing, Cham, pp 213–220
McCauley D, Heffron R, Pavlenko M, Rehner R, Holmes R (2016) Energy justice in the Arctic: implications for energy infrastructural development in the Arctic. Energy Res Soc Sci 16:141–146. https://doi.org/10.1016/j.erss.2016.03.019
Mcrae L, Deinet S, Gill M, Collen B (2012) Tracking trends in Arctic marine populations. CAFF assessment series no. 7. Iceland
Molenaar EJ (2015) The Oslo declaration on high seas fishing in the Central Arctic Ocean. In: Heininen L, Exner-Pirot H, Plouffe J (eds) . Northern Research Forum, Akureyri, pp 427–431
Northern Sea Route Information Office (2013) Final statistics figures for transit navigation on the NSR in 2013. In: North. Sea Route Inf. Off. http://www.arctic-lio.com/node/209. Accessed 17 Nov 2014
Ocean Health Index (2016) ohicore version: v2016.1 core Ocean Health Index functions, [downloaded 08/12/2016]. https://github.com/OHI-Science/ohicore
Onarheim IH, Årthun M (2017) Toward an ice-free Barents Sea. Geophys Res Lett 44:8387–8395. https://doi.org/10.1002/2017GL074304
Overland JE, Wang M (2013) When will the summer Arctic be nearly sea ice free? Geophys Res Lett 40:2097–2101. https://doi.org/10.1002/grl.50316
Pauly D, Zeller D (2016) Catch reconstructions reveal that global marine fisheries catches are higher than reported and declining. Nat Commun 7:10244. https://doi.org/10.1038/ncomms10244
Pinsky ML, Reygondeau G, Caddell R, Palacios-Abrantes J, Spijkers J, Cheung WWL (2018) Preparing ocean governance for species on the move. Science 360(80):1189–1191. https://doi.org/10.1126/science.aat2360
Protection of the Arctic Marine Environment (2013) The Arctic Ocean Review Project PHASE II report, 2011–2013
Roll U, Mittermeier JC, Diaz GI, Novosolov M, Feldman A, Itescu Y, Meiri S, Grenyer R (2016) Using Wikipedia page views to explore the cultural importance of global reptiles. Biol Conserv 204:42–50. https://doi.org/10.1016/j.biocon.2016.03.037
Sato K, Inoue J, Watanabe M (2014) Influence of the Gulf Stream on the Barents Sea ice retreat and Eurasian coldness during early winter. Environ Res Lett 9:084009. https://doi.org/10.1088/1748-9326/9/8/084009
Schmidt JI, Aanesen M, Klokov KB, Khrutschev S, Hausner VH (2015) Demographic and economic disparities among Arctic regions. Polar Geogr 0513:1–20. https://doi.org/10.1080/1088937X.2015.1065926
Selig ER, Frazier M, O’Leary JK, Jupiter SD, Halpern BS, Longo C, Kleisner KL, Sivo L, Ranelletti M (2015) Measuring indicators of ocean health for an island nation: the Ocean Health Index for Fiji. Ecosyst Serv 16:403–412. https://doi.org/10.1016/j.ecoser.2014.11.007
Selkoe KA, Blenckner T, Caldwell MR, Crowder LB, Erickson AL, Essington TE, Estes JA, Fujita RM, Halpern BS, Hunsicker ME, Kappel CV, Kelly RP, Kittinger JN, Levin PS, Lynham JM, Mach ME, Martone RG, Mease LA, Salomon AK, Samhouri JF, Scarborough C, Stier AC, White C, Zedler J (2015) Principles for managing marine ecosystems prone to tipping points. Ecosyst Heal Sustain 1:art17. https://doi.org/10.1890/EHS14-0024.1
Serrao-Neumann S, Davidson JL, Baldwin CL, Dedekorkut-Howes A, Ellison JC, Holbrook NJ, Howes M, Jacobson C, Morgan EA (2016) Marine governance to avoid tipping points: can we adapt the adaptability envelope? Mar Policy 65:56–67. https://doi.org/10.1016/j.marpol.2015.12.007
Smith LC, Stephenson SR (2013) New Trans-Arctic shipping routes navigable by midcentury. Proc Natl Acad Sci U S A 110:E1191–E1195. https://doi.org/10.1073/pnas.1214212110
Stewart EJ, Dawson J, Draper D (2011) Cruise tourism and residents in Arctic Canada: development of a resident attitude typology. J Hosp Tour Manag 18:95–106. https://doi.org/10.1375/jhtm.18.1.95
Stewart E, Dawson J, Johnston M (2015) Risks and opportunities associated with change in the cruise tourism sector: community perspectives from Arctic Canada. Polar J 5:403–427. https://doi.org/10.1080/2154896X.2015.1082283
Tesar C, Dubois M, Shestakov A (2016) Toward strategic, coherent, policy-relevant arctic science. Science 353(80):1368–1370. https://doi.org/10.1126/science.aai8198
Trujillo P (2008) Using a mariculture sustainability index to rank the performance of 53 countries. In: Alder J, Pauly D (eds) The performance of 53 countries in managing marine resources. Vancouver, Fisheries Centre Research Reports Fisheries Centre, University of British Columbia
Van Pelt TI, Huntington HP, Romanenko OV, Mueter FJ (2017) The missing middle: central Arctic Ocean gaps in fishery research and science coordination. Mar Policy 85:79–86. https://doi.org/10.1016/j.marpol.2017.08.008
Viken A (2011) Tourism, research, and governance on Svalbard: a symbiotic relationship. Polar Rec (Gr Brit) 47:335–347. https://doi.org/10.1017/S0032247410000604
Wassmann P, Duarte C, Agustí S, Sejr MK (2011) Footprints of climate change in the Arctic marine ecosystem. Glob Chang Biol 17:1235–1249. https://doi.org/10.1111/j.1365-2486.2010.02311.x
Watson RA (2017) A database of global marine commercial, small-scale, illegal and unreported fisheries catch 1950–2014. Sci Data 4:1–9. https://doi.org/10.1038/sdata.2017.39
Watson R, Kitchingman A, Gelchu A, Pauly D (2004) Mapping global fisheries: sharpening our focus. Fish Fish 5:168–177. https://doi.org/10.1111/j.1467-2979.2004.00142.x
Woodhouse E, Homewood KM, Beauchamp E, Clements T, McCabe JT, Wilkie D, Milner-Gulland EJ (2015) Guiding principles for evaluating the impacts of conservation interventions on human well-being. Philos Trans R Soc B Biol Sci 370:20150103. https://doi.org/10.1098/rstb.2015.0103
Zeller D, Booth S, Pakhomov E, Swartz W, Pauly D (2011) Arctic fisheries catches in Russia, USA, and Canada: baselines for neglected ecosystems. Polar Biol 34:955–973. https://doi.org/10.1007/s00300-010-0952-3
Funding
This work was supported by a Kristian Gerhard Jebsen Scholarship awarded through the Grantham Institute for Climate Change and the Environment, Imperial College London.
The authors would like to thank the three anonymous reviewers whose comments greatly improved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: Kathleen Hermans
Electronic supplementary material
ESM 1
(DOCX 460 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Burgass, M.J., Milner-Gulland, E.J., Stewart Lowndes, J.S. et al. A pan-Arctic assessment of the status of marine social-ecological systems. Reg Environ Change 19, 293–308 (2019). https://doi.org/10.1007/s10113-018-1395-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10113-018-1395-6