Abstract
By 2014 approximately 2.2 million km2 (~43%) of Brazil’s Legal Amazonia region had been incorporated into an extensive network of 718 protected areas, which are comprised by 372 indigenous lands, 313 federal, state and municipal (county) conservation units, and 33 Maroon territories (Quilombos). Although protected areas occupy vast expanses in Amazonia, their importance as carbon reserves needs to be better understood. In this study, we estimate the total carbon in 2014 held in protected areas in Brazil’s “Legal Amazonia” and “Amazonia biome” regions, and the carbon loss in the portions of these protected areas that were cleared by 2014. In 2014, a total of 33.4 Pg C or 57.0% of all carbon stored in Legal Amazonia was held in protected areas and 32.7 Pg C or 58.5% of all the carbon stored in the Amazonia biome was held in protected areas. By 2014, carbon lost due to clearing in protected areas in Legal Amazonia and the Amazonia biome totaled, respectively, 0.787 (or 2.3%) and 0.702 (or 2.1%) Pg C if one assumes that previously each protected area was entirely covered by native vegetation. If the protection of these areas is effective, about half of the carbon in Brazilian Amazonia will be maintained. Carbon in protected areas has strategic value for environmental conservation and for mitigation of climate change because these areas are under lower risk of being emitted to the atmosphere than carbon stored in vegetation located outside of protected areas, although the effectiveness of protected areas varies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Amazonia is a large and dynamic reservoir of carbon that holds about 20% of the total carbon contained in the world’s terrestrial vegetation (Malhi et al. 2006; Houghton 2007; Saatchi et al. 2007, 2011; Baccini et al. 2012). However, in the Brazilian portion of Amazonia a substantial amount of the carbon that was originally stored has already been lost due to clearing of forest and non-forest vegetation, the area cleared totaling at least 960,000 km2 (Hansen et al. 2010, 2013; Brazil, IBAMA 2015; Nogueira et al. 2015; Brazil, INPE 2016). Additional carbon has been lost to degradation of standing forest, mapped to be around 92,000 km2 from 1997 to 2013, after excluding degraded areas that were subsequently converted to deforestation (Brazil, INPE 2014). Almost all of the carbon that is “lost” from the vegetation is emitted as greenhouse gases such as carbon dioxide (CO2) and methane (CH4) (Fearnside 1997, 2000). This occurs whether biomass is oxidized via combustion or decay (Barbosa and Fearnside 1996). When biomass is burned a small percentage is left in the soil as charcoal (see Fearnside 2016). When standing forest is degraded, a small part of the carbon in decaying biomass is incorporated into the soil as organic matter (Barros and Fearnside 2016), although these additions to the soil carbon pool can eventually be expected to be released.
Brazil holds roughly two thirds of Amazonia as a whole, regardless of how the region and its Brazilian portion are defined. Two sub-national regions in Brazil are used for different policies and laws related to Amazonia: “Legal Amazonia” and the “Amazonia biome.” “Legal Amazonia” was decreed in 1953 based on geopolitical criteria and roughly one-quarter of this region was originally occupied by savannas and other non-forest vegetation, while the “Amazonia biome” was defined in 2004 by the Brazilian Institute of Geography and Statistics (IBGE) as the area where the predominant original vegetation was Amazonian forest (although it contains a variety of enclaves of other vegetation types). The Amazonia biome is entirely contained within Legal Amazonia (with the exception of a minuscule area in the state of Maranhão); when the distinction between the two regions is not important, we use the term “Brazilian Amazonia.” Information for both Legal Amazonia and the Amazonia biome is needed for policy relevance in Brazil because each of these officially defined geographical areas serves as the basis for a distinct set of policies and government activities.
In Brazil’s “Legal Amazonia” region the original carbon stock of 71.8 Pg C (1 Pg = 1015 g = 1 gigaton) had been reduced to 58.6 Pg C (a reduction of 18.3%) due to clearing of around 967,000 km2 of primary forest and non-forest vegetation by 2014 (Cleared areas from: Brazil, IBAMA 2015; Brazil, INPE 2016; Biomass from: Nogueira et al. 2015). The same analysis shows that the “Amazonia biome” had its original carbon stock of 67.2 Pg C reduced to 56.0 Pg C (a reduction of 16.7%) by around 673,000 km2 of clearing. The term “by” a given year is used in this paper to mean cumulative deforestation or carbon loss from the “pre-modern” status (~1970) up to and including the official deforestation estimate for the year in question, the deforested areas being based on satellite imagery taken in July or August (dry season in most of Amazonia). The “pre-modern” status refers to the condition at the time of the RADAMBRASIL surveys (Brazil, Projeto RADAMBRASIL 1973–1983).
In response to the changes undergone by Brazil’s Amazon forest, large natural or semi-natural areas have been delimited as “protected areas” to maintain endangered species and territories with high biodiversity (“hotspots”) to halt the spread of deforestation and to protect traditional human populations. By 2014, around 2.2 million km2 of Legal Amazonia (43%) had been delimited as some type of protected area (Brazil, MMA 2014, 2015a; Brazil, FUNAI 2015a; Brazil, INCRA 2015a). Under Brazilian legislation, protected areas are essentially composed of “conservation units” and territories traditionally occupied by Indigenous peoples or by Quilombo communities (Maroon territories). Quilombos are formed by descendants of fugitive slaves or by those with presumed African ancestry related to resistance to the historical oppression of slavery in Brazil (Brazil, Decree n° 4887, 20 November 2003; Brazil, PNAP 2006; Matos 2006). Conservation units are classified under Brazil’s National System of Conservation Units (SNUC) into 12 categories, which are divided into “strictly protected” units (fully protected reserves) and “sustainable-use” units (limited-use reserves) (Brazil, SNUC 2000). Conservation units are defined as “strictly protected” when they are intended solely for conservation of biological diversity, with resource exploitation and human occupation being prohibited except for scientific research and restricted educational visitation (e.g., biological reserves, national parks, ecological stations, etc.). Conservation units are considered to be for “sustainable use” when they seek to reconcile conservation with the sustainable use of natural resources. Continued habitation by traditional non-indigenous human populations is allowed, but with restrictions on the use of natural resources (e.g., extractive reserves, national forests, sustainable development reserves, etc.) (Brazil, SNUC 2000). In the case of indigenous lands and Maroon territories, inclusion of human residents is inherent to the purpose of these areas in ensuring the survival and cultural protection of traditional populations, but this is presumed to occur with low-impact use of natural resources by the resident populations (Arruda 1999; Brazil, PNAP 2006; Nepstad et al. 2006).
Although some protected areas that have been legally created are not effectively under protection (e.g., they lack agrarian “regularization,” elaboration and implementation of a management plan, basic infrastructure and hiring personnel), these areas are considered to be one of the most straightforward and reliable means of reducing deforestation and forest degradation (Bruner et al. 2001; Andam et al. 2008). Protected areas would be especially effective at preventing deforestation fires close to roads, where burning is known to be most likely (Laurance et al. 2002; Ferreira et al. 2005; Adeney et al. 2009; Barber et al. 2014). In addition to reducing deforestation at the present time, protected areas remain to reduce deforestation in the future, which is generally not provided by government programs for repression and surveillance of deforestation (Dutschke 2007; Fearnside 2008; Nepstad et al. 2014). Protected areas have a potential role in preventing the passing of climatic tipping points in the Amazon (Walker et al. 2009). Avoiding these regional climatic changes is essential because they would irreversibly affect ecosystem patterns (especially in drier ecosystems), even over a considerable range of deforestation scenarios (Nepstad et al. 2006; Dudley et al. 2010; Ricketts et al. 2010). Protected areas play a major role in avoiding emissions from deforestation and from degradation resulting from logging and fire (Fearnside 2008).
Carbon stocks currently held in Amazonia’s protected areas are under lower risk of being emitted to the atmosphere from deforestation than is carbon stored in vegetation located outside of protected areas. Thus, besides the intrinsic environmental service of each non-emitted carbon atom in a protected area, which is the same as the benefit of any non-emitted carbon atom stored outside of protected areas, there will be an additional value for carbon stored in protected areas reflecting the effectiveness of legal protection in reducing the probability of future emission (Nolte et al. 2013). Carbon stored in protected areas in locations at higher risk or pressure for deforestation has greater value than carbon in remote areas; this is because pressure for carbon emission is immediate, unlike carbon stored in remote protected areas (Fearnside 2008). Carbon loss inside protected areas, as compared to that in the surrounding area, may be used as a measure of reserve effectiveness in preventing deforestation or forest degradation.
The effectiveness of protected areas in preventing deforestation, as indicated by comparing percentages of clearing inside these areas with clearing in a 10-km buffer around them, varies depending on the type, size and administrative level of the area, provided comparisons are within an area with comparable exposure to deforestation. In a study of 80 protected areas in the southwestern portion of the arc of deforestation, Vitel et al. (2009) found that large areas protect better than small ones, that indigenous lands protect better than other categories, and that federal-level conservation units protect better than state-level ones. Sustainable development units had somewhat better results than integral protection units, a counter-intuitive result that is probably explained by the finding by Nolte et al. (2013) in a study of 292 protected areas throughout Legal Amazonia showing that location near or far from deforestation frontiers overshadows any effect from protected-area type. Nolte et al. found that all protected-area types avoid deforestation and that indigenous lands and strictly protected areas are particularly effective in locations with high deforestation pressure.
In spite of the importance of protected areas as carbon reserves (Walker et al. 2014), an aspect that undermines evaluations of environmental benefits of protected areas is lack of consistent estimates of carbon storage. Few estimates are available (e.g., Campbell et al. 2008a, 2008b; Walker et al. 2014); especially lacking are estimates that are based on representative sampling of Brazilian Amazonia as a whole and that include carbon stored in non-forest vegetation. In addition, estimates are lacking of the amounts of carbon lost in protected areas distributed throughout the Amazon that consider the categories of human-use restriction and the level of degradation that has already occurred.
We stress that deforestation is not the only threat to carbon stocks in Amazonian protected areas. Climate change also threatens these stocks, and this impact can be manifest through increasing levels of degradation as the region’s climate regime moves to one characterized by a greater frequency of extreme droughts (e.g., Marengo and Espinoza 2016; Zemp et al. 2017). These can kill trees directly by surpassing their tolerance for hydraulic stress and for higher temperatures. These stresses are intertwined, as plants require more water at higher temperatures. Based on projected annual mean temperatures from a variety of climate models, Feeley et al. (2016) found that by 2050 almost all protected areas in Brazilian Amazonia will have “disappearing climates” (as defined by Williams et al. 2007), meaning that, assuming there is no migration of species between protected areas (migration is unlikely for trees on a time scale of decades), many of the present species would not survive. A positive effect of future increases in atmospheric CO2 is a reduction in the water requirements of trees that makes them more resilient to drought stress. However, higher CO2 would exacerbate an already apparent increase in lianas, which are favored both by higher CO2 and by dryer climate (Fearnside 2013; Laurance et al. 2014a, b). Another essential factor is forest fires, which will also be favored by the changed climate. Fires kill trees even if CO2 is abundant.
In the present study, we will estimate (i) the amount of carbon stored in vegetation in protected areas in Legal Amazonia and the Amazonia biome from a recently published biomass map (Nogueira et al. 2015), including carbon storage in both forested and non-forested vegetation formations. Among the uses for this information is its relevance to efforts to reward avoided greenhouse-gas emissions through REDD+ (Reducing Emissions from Deforestation and forest Degradation) projects. The information is also relevant to decisions on creating new protected areas and on reinforcing those that already exist. We used biomass mapping free of any clearing or non-natural disturbances to estimate (ii) the amount of the carbon reduction caused by clearing in protected areas with various degrees of conservation and land-use restriction. Finally, we use the same biomass dataset and methodological approach to evaluate (iii) how much of the carbon remaining in 2014 in Legal Amazonia and the Amazonia biome was in protected areas, and we discuss the implications of these results for management strategies.
Materials and methods
Protected areas in legal Amazonia and the Amazonia biome
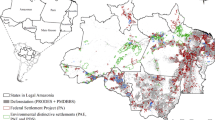
In the present study, we analyzed 718 protected areas in Legal Amazonia created from 1959 to 2014, which cover (without overlapping) 2.18 million km2, or about 43% of the region. Of this total, 600 are located in the Amazonia biome, where these protected areas cover 2.03 million km2 or about 48% of the biome. Vector maps of Legal Amazonia and the Amazonia biome were used to select protected areas that have over 50% of their area inside the boundaries of these regions (Brazil, MMA 2015b). “Vector maps” are composed of areas (polygons) delimited by lines, for example representing the boundary of a protected area, as distinguished from “raster maps” that have information assigned to squares (cells) on a grid. Altogether, without deducting occasional overlapping between protected areas of different types, this study included 372 Indigenous lands (totaling 1.111 million km2), 313 conservation units (totaling 1.172 million km2) and 33 Maroon territories (Quilombos) (totaling 10,212 km2) (Table 1), which were listed by April 2015 in the National Register of Conservation Units (CNUC), National Indian Foundation (FUNAI) and National Institute for Agrarian Reform (INCRA) datasets (Brazil, Decree n° 4887, 20 November 2003, Brazil, FUNAI 2015a; Brazil, INCRA 2015a; Brazil, MMA 2015b). The spatially referenced database of protected areas was organized and analyzed using ArcGIS® software by ESRI (2017). Information on Esri® software is available at http://www.esri.com.
This study did not include all Maroon territories in Legal Amazonia or in the Amazonia biome because many of these territories are still undergoing the process of recognition or certification. In the present study, we analyzed only the 33 Maroon territories for which land titles had been emitted and for which vector maps were available on INCRA’s website (see Brazil, INCRA 2015a, b; Fundação Cultural Palmares 2015). Similarly, the present study included only Private Reserves of Natural Heritage (RPPNs) for which geographical information was available from the Brazilian Ministry of Environment (Brazil, MMA 2015b).
Estimates of carbon loss and remaining stock in protected areas
The carbon stored in each protected area was estimated using algebraic maps in ArcGIS software by overlaying the vector map of protected areas in Legal Amazonia and in the Amazonia biome on the biomass maps for these regions available in Nogueira et al. (2015). The same map was used to estimate the total amount of carbon loss caused by clearing in protected areas. Carbon was calculated from biomass (oven-dry weight) using the average carbon content of 48.5% in forest near Manaus measured by da Silva (2007). The map was based on reconstructing the original vegetation types at a scale of 1:250,000, with each vegetation type assigned its corresponding average dry biomass per hectare, which includes both live and dead vegetation components above- and below ground (i.e., roots of trees, but excluding the soil carbon) (Nogueira et al. 2015). Biomass values were obtained from 2317 1-ha plots sampled in forest as well as from 553 (1-ha each) plots in “contact zones” (ecotones) between two or more vegetation types and 1277 sub-plots of varied size obtained from 39 studies conducted in non-forest vegetation (Nogueira et al. 2008a, 2015). Biomass was estimated mainly from wood volume (n = 2860 plots), for which data were obtained from RADAMBRASIL inventories. Allometric equations were used to estimate biomass in only 10 plots (1 ha each) in forest located in southwestern Amazonia and in some plots or sub-plots covered by vegetation classified as non-forest (see Nogueira et al. 2008a, b, 2015).
In the plots inventoried by RADAMBRASIL, wood volume of all tree boles with DBH ≥ 31.8 cm was estimated (Nogueira et al. 2008a). Wood volume of tree boles with DBH ≥ 10 cm and <31.8 cm were added using a volume expansion factor (VEF). Based on the estimated wood volume of all trees with DBH ≥ 10 cm, biomass was calculated using the average wood density estimated for each plot after weighting by the proportional composition of the species (Nogueira et al. 2007). Other biomass pools were added based on their proportional contribution relative to the above-ground biomass of live trees with DBH ≥ 10 cm. These additional components include seedlings, small trees (DBH < 10 cm), dead trees (fallen and standing), roots of trees (but excluding the soil carbon), aboveground necromass, lianas, and palms (Nogueira et al. 2008a; Table 1).
The result is biomass mapped for the whole of Legal Amazonia or for the Amazonia biome prior to the great increases in disturbance that began in the 1970s; this is denoted as “pre-modern biomass” (Nogueira et al. 2015). These pre-modern biomass maps were used to estimate the “original” carbon stock in each protected area in Legal Amazonia and in the Amazonia biome, free of any degradation caused by non-indigenous human use or by non-natural disturbances. This “pre-modern” stock is considered in this study to be equivalent to the “original” stock of the protected areas. This also serves as an indication of a baseline representing the stock when the protected areas were created. The carbon maps from other studies (e.g., Saatchi et al. 2011; Baccini et al. 2012) were not used to estimate carbon reduction in protected areas for two reasons: (1) because they did not report carbon free of degradation (and therefore would underestimate pre-modern biomass) and (2) because they did not provide carbon estimates for the whole of Legal Amazonia.
Carbon that had been held in the areas cleared by 2014 was estimated, thus allowing estimates of the gross reduction of carbon storage in protected areas by clear cutting (i.e., complete removal of forest or non-forest cover). This reduction was estimated for all categories of protected area. Reabsorption of carbon by secondary vegetation in areas abandoned after cutting or after use was not included in the carbon reduction estimate. Carbon held in the remaining vegetation in 2014 was estimated without considering additional losses in these areas from such processes as selective logging, illegal opening of roads, and forest fires. The area occupied by the original and remaining vegetation was calculated using a coordinate system in the Albers equal-area conic projection. These areas were then multiplied by the mean carbon per hectare.
Cumulative clearing of vegetation by 2014 in each of the protected areas in Legal Amazonia and the Amazonia biome was obtained from maps available from the Project for Monitoring Amazon Deforestation (PRODES), which identifies areas as clear-cut if greater than 6.25 ha in area (Brazil, INPE 2016). Cumulative deforestation by 2014 was fully computed for all states in Legal Amazonia. PRODES deforestation monitoring uses images, mainly from the Landsat 5, 7, or 8 satellites (other satellites can be used, such as CBERS 2B, UK2-DMC and ResourceSat) with spatial resolution of 30 m, but resampled to 60 m in the maps provided on INPE’s website (Brazil, INPE 2013). In the case of protected areas in the states of Mato Grosso, Maranhão, and Tocantins, areas cleared by 2010 (polygons >2 ha in area) were also identified in non-forest vegetation types (mainly cerrado) monitored by the Deforestation Monitoring in Brazilian Biomes Project (PMDBBS) (Brazil, IBAMA 2015). The PMDBBS monitoring used Landsat-5 TM with a resolution of 30 m and CBERS 2B–CCD with a resolution of 20 m (Brazil, IBAMA 2011, 2015). Cumulative clearing of vegetation by 2014 for each protected area is given in the Online Resources (Table S1), including estimates of areas mapped as deforested but that overlap with watercourses.
Results
Carbon stocks in protected areas in 2014
In 2014 a total of 33.4 Pg of carbon was held in all of the protected areas in Legal Amazonia and 32.7 Pg C in the Amazonia biome, with average stocks in the vegetation of 158.9 and 166.0 Mg C ha−1, respectively (Table S2). The amount of carbon held in these protected areas was equivalent to 57.0% of the total carbon stock in native vegetation in Legal Amazonia in 2014 and 58.5% of the total carbon in the Amazonia biome, when calculated using the same biomass dataset and methodological approaches. Both in Legal Amazonia and the Amazonia biome, most of the protected carbon in 2014 was held in indigenous lands and in federal and state conservation units, which together stored approximately 99% of the total carbon stock in all protected areas analyzed in these regions, without discounting any overlaps between different types of protected areas (Tables S3 and S4). There were substantially lower amounts of carbon in municipal (county) conservation units and in Maroon territories, although the mean stock of carbon per hectare in the vegetation remaining in 2014 was high in all of the protected-area types analyzed (Tables S3 and S4). Carbon densities and protected-area types are shown in Fig. 1.
More carbon is stored in sustainable-use conservation units than in integral protection units, both in Legal Amazonia and in the Amazonia biome, although in the sustainable-use units the magnitude of carbon loss is greater and average carbon loss per hectare is generally smaller, the only exception being state-level conservation units with the “strictly protected” regime in the Amazonia biome (Tables S5 and S6). Among the categories of conservation units in Legal Amazonia and the Amazonia biome (Fig. S1 in the Online Resources), most carbon is stored in national forests (15.7% or 2.8 Pg C) and state forests (12.9% or 2.3 Pg C), federal, state and municipal parks (~23.5% or 4.2 Pg C) and in extractive reserves (11.5% or 2.0 Pg C). The total remaining carbon stock and mean remaining carbon per hectare in 2014 for each protected area are given in the Online Resources (Tables S7–S9).
Carbon stock loss in protected areas
Considering all protected areas without any overlapping, there was a total carbon stock loss of 0.787 Pg C (or 2.3%) in protected areas in Legal Amazonia by 2014 and 0.702 Pg C (or 2.1%) in the Amazonia biome, assuming that previously each protected area was entirely covered with native vegetation without any degradation caused by human use or by non-natural disturbances (Table S2). Considering protected areas situated in Legal Amazonia, loss of vegetation with the highest carbon density occurred in federal conservation units, while municipal conservation units had the lowest per-hectare carbon loss (Table S3). If only the protected areas situated in the Amazonia biome are analyzed, high per-hectare carbon loss (>160 Mg C ha−1) are found in all categories of protected area (Fig. 1; Table S4).
Among the types of protected areas analyzed, the largest percentage of carbon loss occurred in the Maroon territories, both in Legal Amazonia and in the Amazonia biome, where about 10% of the carbon stock in the native vegetation was lost, usually in areas with the highest carbon density per hectare (Tables S3 and S4). However, the largest absolute amounts of carbon stock loss occurred in conservation units, mainly in state conservation units (Tables S3 and S4). Among the conservation-unit categories in Legal Amazonia, carbon loss occurred mainly in environmental protection areas (APAs) (60.2% or 0.333 Pg C), in extractive reserves (13.8% or 0.076 Pg C) and in national and state forests (12.1% or 0.067 Pg C). When protected areas in the Amazonia biome are analyzed, carbon loss occurred similarly in APAs (57.6% or 0.290 Pg C), in extractive reserves (14.5% or 0.073 Pg C) and in federal and state forests (13.2% or 0.067 Pg C). Considering all protected areas, the largest carbon reductions occurred in protected areas located in critical regions undergoing deforestation. Total carbon stock loss and mean carbon loss per hectare estimated for each protected area are shown in the Online Resources (Tables S7–S9).
Discussion
Other maps of Amazonian biomass have been developed by Saatchi et al. (2007, 2011) and Baccini et al. (2012). Mitchard et al. (2014) compared the spatial distribution of biomass in these maps, as well as that of Nogueira et al. (2008a), which is an earlier version of the forest vegetation portion of the map used in this study, and found that these maps do not agree. Detailed comparisons of the data underlying these biomass maps show that the map used in the present study is based on many more ground plots than are the other studies (Fearnside 2016). For the forest portion of Brazilian Amazonia, the map used in the present study (Nogueira et al. 2015) is based on 1-ha plots at 2317 distinct locations (and an additional 553 1-ha plots in “contact zones” (ecotones) between rainforest and seasonal forest, savanna or campinarana), while that of Saatchi et al. (2007) is based on plots at 53 distinct locations of which 28 sampled ≥1 ha and Saatchi et al. (2011) is based on plots at 96 distinct locations of which 63 sampled ≥1 ha. The Baccini et al. (2012) estimate in Brazilian Amazonia is based on an undisclosed number out of a set of 283 0.16-ha plots distributed throughout the world’s tropical forests. The variance of per-hectare biomass in tropical forest increases rapidly as plot size decreases below 1 ha (Clark and Clark 2000). Even in studies with many carefully measured 1-ha plots in close proximity in the same forest type the variance is substantial. In forests near Manaus, Laurance et al. (1999) found 65 plots in the Biological Dynamics of Forest Fragments Project to have a mean aboveground live biomass of trees, including palms (based on trees >10 cm DBH with a 12% correction for small trees) of 356 ± 47 Mg ha−1 with a coefficient of variation (CV) of 13.2%, while in 72 plots in the Ducke Reserve, de Castilho et al. (2006) found a mean aboveground live biomass for trees, including palms, of 327.8 ± 41.9 Mg ha−1 with a CV of 12.8% (based on all trees ≥30 cm DBH in each 1-ha plot, trees 10–29.9 cm DBH in a 0.5 ha subplot, and trees 1–9.9 cm DBH in a 0.1 ha subplot). For samples at widely separated locations the variance is substantially greater, making widely distributed sampling locations a priority for reducing uncertainty in regional biomass estimates (Fearnside 2016). Note that the RADAMBRASIL dataset used in the present study is much more widely and evenly distributed than are the data underlying other studies (Fearnside 2016).
Information is needed on Amazon forest biomass in a form that can be used in calculating the benefits of avoiding deforestation and degradation in specific territorial units, such as protected areas. A first step is the information presented here on the “original” (pre-modern) biomass of the forests that presently remain standing at these locations (Tables S7–S9). The role that these protected areas play as a bulwark against regional and global climate change faces a wide variety of threats from degradation and from outright deforestation.
Conclusions
In addition to their value for the conservation of biological diversity, protected areas in Brazilian Amazonia are valuable because they hold very large amounts of carbon. Protected areas account for most of the carbon that remained stored in the vegetation of Brazil’s Legal Amazonia and Amazonia biome regions in 2014. This carbon is especially valuable because it is in areas that are under legal protection; protection reduces the risk of future emission, although these areas have varying levels of permitted access and use of natural resources.
Historically, protected areas have been established following criteria for biological conservation and for maintaining traditional human populations, but changes in global climate make it necessary to incorporate maintenance of carbon stocks as one of the ecosystem services that justifies creating and maintaining these areas. This highlights the importance of carbon estimates in protected areas as a tool for defining development policies for the Amazon region.
References
Adeney JM, Christensen NL, Pimm SL, Freckleton RP (2009) Reserves Protect against Deforestation Fires in the Amazon. PLoS ONE 4(4):e5014
Andam KS, Ferraro PJ, Pfaff A, Sanchez-Azofeifa GA, Robalino JA (2008) Measuring the effectiveness of protected area networks in reducing deforestation. Proc Nat Acad Sci USA 105:16089–16094. doi:10.1073/pnas.0800437105
Arruda R (1999) Populações tradicionais e a proteção dos recursos naturais em unidades de conservação. Revista Ambiente & Sociedade 2(5):79–92. doi:10.1590/S1414-753X1999000200007
Baccini A, Goetz SJ, Walker WS, Laporte NT, Sun M, Sulla-Menashe D, Hackler J, Beck PSA, Dubayah R, Friedl MA, Samanta S, Houghton RA (2012) Estimated carbon dioxide emissions from tropical deforestation improved by carbon-density maps. Nat Clim Chang 2:182–185. doi:10.1038/nclimate1354
Barber CP, Cochrane MA, Souza CM Jr, Laurance WF (2014) Roads, deforestation, and the mitigating effect of protected areas. Biol Conserv 177:203–209. doi:10.1016/j.biocon.2014.07.004
Barbosa RI, Fearnside PM (1996) Pasture burning in Amazonia: dynamics of residual biomass and the storage and release of aboveground carbon. Journal of Geophysical Research (Atmospheres) 101(D20):25,847–25,857. doi:10.1029/96JD02090
Barros HS, Fearnside PM (2016) Soil carbon stock changes due to edge effects in central Amazon forest fragments. For Ecol Manag 379:30–36. doi:10.1016/j.foreco.2016.08.002
Brazil, FUNAI (Fundação Nacional do Índio) (2015a) Download de dados geográficos. Terra Indígena (Regularizada, Homologada, Declarada, Delimitada e Área em Estudo). FUNAI, Brasília, DF, Brazil. http://www.funai.gov.br/index.php/shape. Accessed 24 April 2015
Brazil, FUNAI (Fundação Nacional do Índio) (2015b) Modalidades de Terras Indígenas. FUNAI, Brasília, DF, Brazil. http://www.funai.gov.br/index.php/indios-no-brasil/terras-indigenas. Accessed 12 May 2015
Brazil, IBAMA (Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis) (2011) Monitoring Deforestation of Brazilian Biomes Project (PMDBBS), Monitoramento do Bioma Cerrado 2009–2010. IBAMA, Brasília, DF, Brazil. http://siscom.ibama.gov.br/monitorabiomas/cerrado/index.htm. Accessed 04 October 2013
Brazil, IBAMA (Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis) (2015) Monitoring Deforestation of Brazilian Biomes Project (PMDBBS). IBAMA, Brasília, DF, Brazil. http://siscom.ibama.gov.br/monitorabiomas/cerrado/index.htm. Accessed 12 July 2015
Brazil, INCRA (Instituto Nacional de Colonização e Reforma Agrária) (2015a) Download de dados geográficos. INCRA, Brasília, DF, Brazil. http://acervofundiario.incra.gov.br/i3geo/datadownload.htm. Accessed 14 April 2015
Brazil, INCRA (Instituto Nacional de Colonização e Reforma Agrária) (2015b) Quilombolas. INCRA, Brasília, DF, Brazil. http://www.incra.gov.br/quilombolas. Accessed 11 May 2015
Brazil, INPE (Instituto Nacional de Pesquisas Espaciais) (2013) Metodologia para o Cálculo da Taxa Anual de Desmatamento na Amazônia Legal. Coordenadoria Geral de Observação da Terra Programa Amazônia (Projeto PRODES), INPE, São José dos Campos, SP, Brazil. http://www.obt.inpe.br/prodes/metodologia_TaxaProdes.pdf. Accessed 16 June 2016
Brazil, INPE (Instituto Nacional de Pesquisas Espaciais) (2014) Sistema DEGRAD. INPE, São José dos Campos, SP, Brazil. http://www.obt.inpe.br/degrad. Accessed 11 Mar 2017
Brazil, INPE (Instituto Nacional de Pesquisas Espaciais) (2016) Projeto de Monitoramento do Desmatamento na Amazônia Legal (PRODES). INPE, São José dos Campos, SP, Brazil. http://www.dpi.inpe.br/prodesdigital/prodes.php. Accessed 1 March 2016
Brazil, MMA (Ministério do Meio Ambiente) (2014) Cadastro Nacional de Unidades de Conservação (CNUC), Tabela consolidada das Unidades de Conservação. Atualizada em 27 October 2014. MMA, Brasília, DF, Brazil. http://www.mma.gov.br/images/arquivo/80112/CNUC_Categorias_Out14.pdf. Accessed 31 January 2015
Brazil, MMA (Ministério do Meio Ambiente) (2015a) Cadastro Nacional de Unidades de Conservação. MMA, Brasília, DF, Brazil. http://www.mma.gov.br/areas-protegidas/cadastro-nacional-de-ucs/mapas. Accessed 31 January 2015
Brazil, MMA (Ministério do Meio Ambiente) (2015b) Download de dados geográficos, MMA, Brasília, DF, Brazil. http://mapas.mma.gov.br/i3geo/datadownload.htm. Accessed 17 April 2015
Brazil, PNAP (Plano Nacional Estratégico para Áreas Protegidas) (2006) Plano Nacional Estratégico para Áreas Protegidas (PNAP), Decreto n° 5758 de 13 de abril de 2006, Ministério do Meio Ambiente (MMA), Brasília, DF, Brazil. http://www.mma.gov.br/estruturas/205/_arquivos/planonacionaareasprotegidas_205.pdf. Accessed 16 June 2016
Brazil, Projeto RADAMBRASIL (1973–1983) Levantamento de recursos naturais. Ministério das Minas e Energia, Departamento Nacional de Produção Mineral, Rio de Janeiro, Brazil, 36 vols
Brazil, SNUC (Sistema Nacional de Unidades de Conservação) (2000) Sistema Nacional de Unidades de Conservação (SNUC), Lei no. 9985 de 18 de julho de 2000. Serviço Brasileiro Florestal (SBF), Ministério do Meio Ambiente (MMA), Brasília, DF, Brazil. http://www.mma.gov.br/images/arquivos/areas_protegidas/snuc/Livro%20SNUC%20PNAP.pdf. Accessed 16 June 2016
Bruner AG, Gullison RE, Rice RE, da Fonseca GAB (2001) Effectiveness of parks in protecting tropical biodiversity. Science 291:125–128. doi:10.1126/science.291.5501.125
Campbell A, Kapos V, Lysenko I, Scharlemann JPW, Dickson B, Gibbs HK, Hansen M, Miles L (2008a) Carbon emissions from forest loss in protected areas. UNEP World Conservation Monitoring Centre, Cambridge
Campbell A, Miles L, Lysenko I, Hughes A, Gibbs H (2008b) Carbon storage in protected areas: technical report. UNEP World Conservation Monitoring Centre, Cambridge
Clark DB, Clark DA (2000) Landscape-scale variation in forest structure and biomass in a tropical rain forest. For Ecol Manag 137:185–198. doi:10.1016/S0378-1127(99)00327-8
da Silva RP (2007) Alometria, estoque e dinâmica da biomassa de florestas primárias e secundárias na região de Manaus (AM). PhD thesis, Instituto Nacional de Pesquisas da Amazônia (INPA), Manaus, AM, Brazil
de Castilho CV, Magnusson WE, de Araújo RNO, Luizão RCC, Luizão FJ, Lima AP, Higuchi N (2006) Variation in aboveground tree live biomass in a central Amazonian forest: effects of soil and topography. For Ecol Manag 234:85–96. doi:10.1016/j.foreco.2006.06.024
Dudley N, Stolton S, Belokurov A, Krueger L, Lopoukhine N, MacKinnon K, Sandwith T, Sekhran N (2010) Natural Solutions: protected areas helping people cope with climate change. A report funded and commissioned by IUCN-WCPA, TNC, UNDP, WCS, The World Bank and WWF, Gland, Switzerland, Washington, DC & New York, USA
Dutschke M (2007) CDM forestry and the ultimate objective of the climate convention. Mitig Adapt Strateg Glob Chang 12(2):275–302. doi:10.1007/s11027-005-9013-6
ESRI (Environmental Systems Research Institute) (2017) ArcGIS 10.0. GIS software, Desktop. ESRI, Redlands, CA, USA. http://www.esri.com. Accessed 3 April 2017
Fearnside PM (1997) Greenhouse gases from deforestation in Brazilian Amazonia: net committed emissions. Clim Chang 35(3):321–360. doi:10.1023/A:1005336724350
Fearnside PM (2000) Greenhouse gas emissions from land-use change in Brazil’s Amazon region. In: Lal R, Kimble JM, Stewart BA (eds) Global climate change and tropical ecosystems: advances in soil science. CRC Press, Boca Raton, pp 231–249
Fearnside PM (2008) Amazon forest maintenance as a source of environmental services. Annals of the Brazilian Acad of Sci 80(1):101–114. doi:10.1590/S0001-37652008000100006
Fearnside PM (2013) Vines, CO2 and Amazon forest dieback. Nature Online Comment. doi:10.1038/ nature11882
Fearnside PM (2016) Brazil’s Amazonian forest carbon: the key to Southern Amazonia’s significance for global climate. Regional Environ Change. doi:10.1007/s10113-016-1007-2
Feeley KJ, Silman MR, Franklin J (2016) Disappearing climates will limit the efficacy of Amazonian protected areas. Divers Distrib 22(11):1081–1084. doi:10.1111/ddi.12475
Ferreira LV, Venticinque E, Almeida S (2005) O desmatamento na Amazônia e a importância das áreas protegidas. Estudos Avançados 19(53):157–166. doi:10.1590/S0103-40142005000100010
Fundação Cultural Palmares (2015) Fundação Cultural Palmares, Comunidades Quilombolas. http://www.palmares.gov.br/?page_id=88. Accessed 2 June 2015
Hansen MC, Potapov PV, Moore R, Hancher M, Turubanova SA, Tyukavina A, Thau D, Stehman SV, Goetz SJ, Loveland TR, Kommareddy A, Egorov A, Chini L, Justice CO, Townshend JRG (2013) High-resolution global maps of 21st-century forest cover change. Science 342:850–853. doi:10.1126/science.1244693
Hansen MC, Stehman SV, Potapov PV (2010) Quantification of global gross forest cover loss. Proc Nat Acad Sciences USA 107:8650–8655. doi:10.1073/pnas.0912668107
Houghton RA (2007) Balancing the global carbon budget. Annu Rev Earth Planet Sci 35:313–347. doi:10.1146/annurev.earth.35.031306.140057
Laurance WF, Fearnside PM, Laurance SG, Delamonica P, Lovejoy TE, Rankin-de Merona JM, Chambers JQ, Gascon C (1999) Relationship between soils and Amazon forest biomass: a landscape-scale study. For Ecol Manag 118(1–3):127–138. Doi:10.1016/S0378-1127(98)00494-0
Laurance WF, Albernaz AKM, Schroth G, Fearnside PM, Bergen S, Venticinque EM, da Costa C (2002) Predictors of deforestation in the Brazilian Amazon. J Biogeogr 29:737–748. doi:10.1046/j.1365-2699.2002.00721.x
Laurance WF, Andrade AS, Magrach A, Camargo JLC, Campbell M, Fearnside PM, Edwards W, Valsko JJ, Lovejoy TE, Laurance SG (2014a) Apparent environmental synergism drives the dynamics of Amazonian forest fragments. Ecology 95(11):3018–3026. doi:10.1890/14-0330.1
Laurance WF, Andrade AS, Magrach A, Camargo JLC, Valsko JJ, Campbell M, Fearnside PM, Edwards W, Lovejoy TE, Laurance SG (2014b) Long-term changes in liana abundance and forest dynamics in undisturbed Amazonian forests. Ecology 95(6):1604–1611. doi:10.1890/13-1571.1
Malhi Y, Wood D, Baker TR, Wright J, Phillips OL, Cochrane T, Meir P, Chave J, Almeida S, Arroyo L, Higuchi N, Killeen TJ, Laurance SG, Laurance WF, Lewis SL, Monteagudo A, Neill DA, Vargas PN, Pitman NCA, Quesada CA, Salomão R, Silva JNM, Lezama AT, Terborgh J, Martínez RV, Vinceti B (2006) The regional variation of aboveground live biomass in old-growth Amazonian forests. Glob Chang Biol 12:1–32. doi:10.1111/j.1365-2486.2006.01120.x
Marengo JA, Espinoza JC (2016) Extreme seasonal droughts and floods in Amazonia: causes, trends and impacts. Int J Climatol 36:1033–1050. doi:10.1002/joc.4420
Matos H (2006) Remanescentes das comunidades dos quilombos: Memória do cativeiro e políticas de reparação no Brasil. Revista USP 68:104–111. http://www.revistas.usp.br/revusp/article/view/13486/15304 Accessed 5 July 2017
Mitchard ETA, Feldpausch TR, Brienen RJW, Lopez-Gonzalez G, Monteagudo A, Baker TR, Lewis SL, Lloyd J, Quesada CA, Gloor M, ter Steege H, Meir P, Alvarez E, Araujo-Murakami A, Aragão LEOC, Arroyo L, Aymard G, Banki O, Bonal D, Brown S, Brown FI, Cerón CE, Chama Moscoso V, Chave J, Comiskey JA, Cornejo F, Corrales Medina M, Da Costa L, Costa FRC, Di Fiore A, Domingues TF, Erwin TL, Frederickson T, Higuchi N, Honorio Coronado EN, Killeen TJ, Laurance WF, Levis C, Magnusson WE, Marimon BS, Marimon Junior BH, Mendoza Polo I, Mishra P, Nascimento MT, Neill D, Núñez Vargas MP, Palacios WA, Parada A, Pardo Molina G, Peña-Claros M, Pitman N, Peres CA, Poorter L, Prieto A, Ramirez-Angulo H, Restrepo Correa Z, Roopsind A, Roucoux KH, Rudas A, Salomão RP, Schietti J, Silveira M, de Souza PF, Steininger MK, Stropp J, Terborgh J, Thomas R, Toledo M, Torres-Lezama A, van Andel TR, van der Heijden GMF, Vieira ICG, Vieira S, Vilanova-Torre E, Vos VA, Wang O, Zartman CE, Malhi Y, Phillips OL, (2014) Markedly divergent estimates of Amazon forest carbon density from ground plots and satellites. Glob Ecol Biogeogr 23(8):935-946. doi:10.1111/geb.12168
Nepstad D, McGrath D, Stickler C, Alencar A, Azevedo A, Swette B, Bezerra T, DiGiano M, Shimada J, da Motta RS, Armijo E, Castello L, Brando P, Hansen MC, McGrath-Horn M, Carvalho O, Hess L (2014) Slowing Amazon deforestation through public policy and interventions in beef and soy supply chains. Science 344:1118–1123. doi:10.1126/science.1248525
Nepstad D, Schwartzman S, Bamberger B, Santilli M, Ray D, Schlesinger P, Lefebvre P, Alencar A, Prinz E, Fiske G, Rolla A (2006) Inhibition of Amazon deforestation and fire by parks and indigenous lands. Conserv Biol 20(1):65–73. doi:10.1111/j.1523-1739.2006.00351.x
Nogueira EM, Fearnside PM, Nelson BW, França MB (2007) Wood density in forests of Brazil’s ‘arc of deforestation’: implications for biomass and flux of carbon from land-use change in Amazonia. Forest Ecol and Manage 248:119–135. doi:10.1016/j.foreco.2007.04.047
Nogueira EM, Fearnside PM, Nelson BW, Barbosa RI, Keizer EWH (2008a) Estimates of forest biomass in the Brazilian Amazon: new allometric equations and adjustments to biomass from wood-volume inventories. For Ecol Manag 256:1853–1867. doi:10.1016/j.foreco.2008.07.022
Nogueira EM, Nelson BW, Fearnside PM, França MB, de Oliveira ACA (2008b) Tree height in Brazil’s ‘arc of deforestation’: shorter trees in south and southwest Amazonia imply lower biomass. For Ecol Manag 255:2963–2972. doi:10.1016/j.foreco.2008.02.002
Nogueira EM, Yanai AM, Fonseca FOR, Fearnside PM (2015) Carbon stock loss from deforestation through 2013 in Brazilian Amazonia. Glob Chang Biol 21(3):1271–1292. doi:10.1111/gcb.12798
Nolte C, Agrawal A, Silvius KM, Soares-Filho BS (2013) Governance regime and location influence avoided deforestation success of protected areas in the Brazilian Amazon. Proc Nat Acad Sci USA 110(13):4956–4961. doi:10.1073/pnas.1214786110
Ricketts TH, Soares-Filho B, da Fonseca GAB, Nepstad D, Pfaff A, Petsonk A, Anderson A, Boucher D, Cattaneo A, Conte M, Creighton K, Linden L, Maretti C, Moutinho P, Ullman R, Victurine R (2010) Indigenous lands, protected areas, and slowing climate change. PLoS Biol 8(3):e1000331. doi:10.1371/journal.pbio.1000331
Saatchi SS, Harris NL, Brown S, Lefsky M, Mitchard ETA, Salas W, Zutta BR, Buermann W, Lewis SL, Hagen S, Petrova S, White L, Silman M, Morel A (2011) Benchmark map of forest carbon stocks in tropical regions across three continents. Proc Nat Acad Sci USA 108(24):9899–9904. doi:10.1073/pnas.1019576108
Saatchi SS, Houghton RA, Alvalá RCS, Soares JV, Yu Y (2007) Distribution of aboveground live biomass in the Amazon basin. Glob Chang Biol 13:816–837. doi:10.1111/j.1365-2486.2007.01323.x
Vitel CSMN, Fearnside PM, Graça PMLA (2009) Análise da inibição do desmatamento pelas áreas protegidas na parte Sudoeste do Arco de desmatamento. In: Epiphanio JCN, Galvão LS (eds) Anais XIV Simpósio Brasileiro de Sensoriamento Remoto, Natal, Brasil 2009. Instituto Nacional de Pesquisas Espaciais (INPE), São José dos Campos, pp 6377–6384. http://marte.sid.inpe.br/col/dpi.inpe.br/sbsr%4080/2008/11.13.14.42/doc/6377-6384.pdf
Walker R, Moore NJ, Arima E, Perz S, Simmons C, Caldas M, Vergara D, Bohrer C (2009) Protecting the Amazon with protected areas. Proc Nat Acad Sci USA 106:10582–10586. doi:10.1073/pnas.0806059106
Walker W, Baccini A, Schwartzman S, Ríos S, Oliveira-Miranda MA, Augusto C, Ruiz MR, Arrasco CS, Ricardo B, Smith R, Meyer C, Jintiach JC, Campos EV (2014) Forest carbon in Amazonia: the unrecognized contribution of indigenous territories and protected natural areas. Carbon Manage 5:479–485. doi:10.1080/17583004.2014.990680
Williams JW, Jackson ST, Kutzbach JE (2007) Projected distributions of novel and disappearing climates by 2100 AD. Proc Nat Acad Sci USA 104:5738–5742. doi:10.1073/pnas.0606292104
Zemp DC, Schleussner C-F, HMJ B, Hirota M, Montade V, Sampaio G, Staal A, Wang-Erlandsson L, Rammig A (2017) Self-amplified Amazon forest loss due to vegetation-atmosphere feedbacks. Nat Commun 8:art. 14681. doi:10.1038/ncomms14681
Acknowledgments
We thank the National Institute for Research in Amazonia (INPA: PRJ15.125), the National Institute of Science and Technology for the Environmental Services of Amazonia (INCT-SERVAMB), the National Council for Scientific and Technological Development (CNPq/PCI Program Proc. 304130/2013-3 and 301183/2015-5; CNPq: Proc. 304020/2010-9; 573810/2008-7), the Foundation for Support of Research in Amazonas State (FAPEAM: Proc. 708565), and the National Postdoctoral Program (PNPD/CAPES: Proc. n° 028176/2009-41) for financial and logistical support. This article is a contribution of the Brazilian Research Network on Global Climate Change, FINEP/ Rede CLIMA Grant No. 01.13.0353-00. Three reviewers contributed valuable comments.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor:Wolfgang Cramer.
Electronic supplementary material
ESM 1
(PDF 778 kb).
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Nogueira, E.M., Yanai, A.M., de Vasconcelos, S.S. et al. Carbon stocks and losses to deforestation in protected areas in Brazilian Amazonia. Reg Environ Change 18, 261–270 (2018). https://doi.org/10.1007/s10113-017-1198-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10113-017-1198-1