Abstract
Low-level laser irradiation (LLLI) shows effects in orthodontic pain relief and periodontal inflammation control. The aim of this article is to investigate the analgesic and inflammation-modulatory effects of low-level laser irradiation among orthodontic patients with compromised periodontium. A randomised controlled trial with split-mouth design was conducted in 27 adults with treated and controlled chronic periodontitis over 6 months. One side of the dental arch underwent repeated treatment under a 940-nm diode laser (EZlase; Biolase Technology Inc.) with a beam size of 2.8 cm2 for 60 seconds at 8.6 J/cm2, whilst the other side received pseudo-laser treatment. Laser irradiation was applied repeatedly for 8 times during the first 6 weeks after bracket bonding and monthly thereafter until the end of orthodontic treatment. Subjective pain (assessed by visual analogue scale in pain diary and by chairside archwire activation), periodontal status (assessed by periodontal clinical parameters), cytokines in gingival crevicular fluid (interleukin 1β, prostaglandin E2, substance P) and periodontopathic bacteria (Porphyromonas gingivalis and Treponema denticola) in supragingival plaque were assessed. The intensity of pain was lower on the laser-irradiated side at multiple follow-up visits (P < 0.05). The pain subsided 1 day earlier on the laser side, with a lower peak value during the first week after initial archwire placement (P < 0.05). The laser side exhibited a smaller reduction in bite force during the first month (mean difference = 3.17, 95% CI: 2.36–3.98, P < 0.05 at 1-week interval; mean difference = 3.09, 95% CI: 1.87–4.32, P < 0.05 at 1-month interval). A smaller increase was observed in the plaque index scores on the laser side at 1-month (mean difference = 0.19, 95% CI: 0.13–0.24, P < 0.05) and in the gingival index scores at the 3-month follow-up visit (mean difference = 0.18, 95% CI: 0.14–0.21, P < 0.05). Laser irradiation inhibited the elevation of interleukin-1β, prostaglandin E2 and substance P levels during the first month (P < 0.05). However, no intergroup difference was detected in the bacteria levels. Low-level laser irradiation exhibits benefits in pain relief and inflammation control during the early stage of adjunctive orthodontic treatment in periodontally compromised individuals.
Similar content being viewed by others
Introduction
Tooth displacement associated with compromised periodontal support requires adjunctive orthodontic treatment. As a key constituent of the team approach to periodontal disease, orthodontic treatment is beneficial for preserving and restoring deteriorating dentition (1, 2). However, tooth movement with compromised periodontal tissues poses great challenges to orthodontists (3). Firstly, pain and discomfort are common complaints from patients who are undergoing orthodontic treatment, which can result in a weakened masticatory function and a poorer oral health-related quality of life (4, 5). Secondly, inflammation control is a key issue to consider in orthodontic treatment. On one hand, uncontrolled inflammation is detrimental to periodontal health and tooth movement, possibly leading to gingival recession, attachment loss, bone resorption or root resorption (2, 6). On the other hand, the nature of fixed appliances commonly helps dental plaque aggravation, especially if insufficient oral hygiene is maintained due to pain and discomfort (5). Therefore, other approaches to facilitate pain management and inflammation control, in addition to strict oral hygiene instruction and regular periodontal maintenance, are likely to ensure safe and efficient tooth movement in patients with reduced periodontal support.
Low-level laser irradiation (LLLI) is a type of laser therapy with low-energy outputs to keep the temperature of the treated tissue below 36.5 °C or normal body temperature (7,8,9). Owing to its nonthermal bio-stimulatory effects, LLLI has been reported as an effective adjunct to nonsurgical periodontal treatment for inflammation control and tissue reparation in patients with periodontitis (10). In addition, interests in the application of LLLI in modern orthodontics have recently grown because less painful and more efficient tooth movement can be anticipated (11, 12). Very few adverse effects have been observed thus far in the dental application of LLLI.
However, the application of LLLI as part of a joint orthodontic-periodontal treatment strategy has not been studied. A randomised controlled trial was thus conducted to investigate the effects of LLLI in the orthodontic treatment of patients with controlled periodontitis on subjective pain (primary outcome) and periodontal inflammation (secondary outcome) over a 6-month period. To eliminate any bias, we initially hypothesised that pain perceptions and periodontal inflammation status did not differ between patients with controlled chronic periodontitis who are receiving orthodontic treatment in conjunction with LLLI and those who are undergoing orthodontic treatment without the incorporation of LLLI.
Materials and methods
Study design
This study was a double-blind randomised controlled trial with a split-mouth design. Pairs of displaced single-rooted teeth with controlled periodontitis located on both sides of the maxillary and/or mandibular arches were randomly assigned to either the laser group (low-level laser irradiation) or the placebo group (pseudo-laser treatment). Block randomisation in groups of six was applied. The randomised schedule was generated by a statistician and sealed in sequentially numbered, opaque envelopes to protect the allocation sequence. The patients were not aware of the treatment allocation due to the identical settings of LLLI and the placebo laser, except for the actual irradiation. The clinical operator was blinded, and a designated assistant took charge of setting the laser parameters for each treatment. The outcome assessor was also blinded, as s/he was not included in the randomisation process, participant recruitment or clinical treatment. The protocol for this study was approved by the Institutional Review Board of the University of Hong Kong/Hospital Authority Hong Kong West Cluster (HKU/HA HKW IRB, Ref. No. UW 12-049). The study was also registered on the HKU Clinical Trial Register (HKUCTR-1683) and the US National Institutes of Health Clinical Trial Register (Ref. No. NCT02352038).
Selection of participants
Ethnic Chinese patients with controlled chronic periodontitis were recruited from Prince Philip Dental Hospital with the understanding and written consent of each participant and according to the Declaration of Helsinki (version 2008). Patients were included according to the following criteria:
- (1)
Adult patients (over 30 years of age), male or female
- (2)
Systemically healthy
- (3)
No medications (antibiotic and/or anti-inflammatory) in the previous month
- (4)
Previous chronic periodontitis (moderate to severe) with at least two pairs of clinically matched displaced single-rooted teeth on contralateral hemi-arches with the following characteristics:
(4a) A pocket depth of more than 5 mm, clinical attachment loss of more than 3 mm and radiographic signs of bone loss before nonsurgical periodontal treatment
(4b) A pocket depth of less than 4 mm and/or periodontal status identified as stable for at least 3 months after nonsurgical periodontal treatment
The exclusion criteria were as follows:
- (1)
Patients with aggressive periodontitis
- (2)
Patients with a smoking habit
- (3)
Patients who were pregnant
- (4)
Patients with ongoing or previous orthodontic treatment
- (5)
Teeth treated with periodontal surgery
Sample size estimation
Based on our previous systematic review (12), with a standard deviation of 5 (13), to detect a difference of 3 in the pain score between the laser and placebo semi-arches (14), a sample size of 27 would be required. The significance level was set at 5% and the test power at 80%. A dropout rate of 10% over 6 months was assumed (G*Power 3.1.7 version).
Orthodontic treatment
Adjunctive orthodontic treatment of all patients in the study was carried out by the same clinical operator. Preadjusted appliances with 0.022 × 0.028-inch2 brackets/buccal tubes were bonded on the maxillary and/or mandibular dental arches, with 0.014-inch thermal nickel-titanium (NiTi) as the initial archwire for the first 2 months followed by 0.016-inch thermal NiTi for the third month.
Low-level laser irradiation
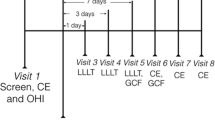
The teeth on one half of the dental arch were randomly allocated to the test side and received low-level laser irradiation using a GaAlAs laser with a wavelength of 940 nm (EZlase; Biolase Technology Inc., Irvine, CA). The irradiation was delivered by a quadrant-sized probe that covered the region from the central incisor to the first molar on the test side. The output power was confirmed at 800 mW with a power metre (OPHIR Nova II Power Metre, Ophir-Spiricon, LLC, UT, USA). The laser probe was first targeted at the level of the bracket and the buccal gingival margin and was then shifted to the level of the buccal alveolar mucosa covering the root area; both at 1 cm. With the duration of each irradiation set to 30 seconds, the energy density delivered in a single application was calculated to be 8.6 J/cm2. A repeated regimen was adopted for laser therapy throughout orthodontic treatment (Fig. 1). The patient, the clinical operator and the designated assistant all wore laser-protective eyewear according to standard safety rules.
Outcome assessment
The timeline for outcome assessment is presented in Fig. 1.
Subjective pain assessment
The pain intensity was assessed using serial 100-mm visual analogue scales both as self-reported in a pain diary within the first week and as an immediate response to archwire ligation at the chairside during the first 3 months of orthodontic treatment. A score of 0 at the left end of the scale indicated ‘no pain’, and 100 at the right end indicated ‘the most severe pain’. Each patient was asked to place a mark on the linear scale to record their pain intensity separately for the left and right sides. The mark was measured in millimetres with a ruler from the left end. The patients were advised to avoid analgesic drugs during the study process. The patients were also asked to complete a series of self-explanatory questions to elicit the time course of pain perception.
Measurement of maximum voluntary bite force
After activation of the archwire, the maximum voluntary bite force was measured with an occlusal force gauge (GM10, Nagano Keiki, Japan) during the first 3 months of orthodontic treatment. Measurements were taken bilaterally on each target tooth with its opposing antagonist and were averaged to obtain the respective maximum voluntary bite force for each group.
Periodontal clinical examination and supragingival bacteria analysis
Periodontal clinical parameters were recorded by one blinded examiner with a periodontal probe (CPU 15 UNC; Hu-Friedy, Chicago, IL) at six sites for each target tooth. The following parameters were recorded: plaque index (PI), gingival index (GI), bleeding on probing (BOP), probing pocket depth (PPD), gingival recession (GR) and clinical attachment loss (CAL). Pre-calibration was performed at baseline on 10% of the sample size (3 patients) with a weighted Kappa value of 0.75–0.80 for all periodontal clinical indices, indicating good intra-examiner reliability. The mean values of both groups were calculated from the measurements of all target teeth in the corresponding group.
Supragingival plaque samples were collected with sterile probes from the area surrounding the bracket and buccal cervical region. Bacteria DNA were extracted from the clinical samples using QIAamp DNA Mini kits (Qiagen, CA). The bacterial loads of Porphyromonas gingivalis (P. gingivalis) and Treponema denticola (T. denticola) were analysed with a StepOnePlus real-time PCR system (Life Technologies Inc., Grand Island, NY, USA) with the TaqMan® Gene Expression Assays (Applied Biosystems, Foster City, CA, USA) (Appendix 1). The absolute bacteria counts were calculated and expressed as the number of cells per sample. The proportions of each species tested were calculated by dividing the absolute bacterial counts by the total bacterial counts.
Analysis of cytokines in gingival crevicular fluid
Gingival crevicular fluid (GCF) samples were taken from all target teeth in both groups at multiple time-points. After supragingival plaque was gently removed, GCF was collected from the mesiobuccal and distobuccal sites with prefabricated paper strips (Periopaper; Oraflow Inc., New York, NY, USA), which were inserted into the pockets until resistance was felt and held in place for 30 seconds. They were immediately transferred to separate microcentrifuge tubes containing phosphate-buffered saline solution. Enzyme-linked immunosorbent assay (ELISA) was performed to assess the levels of interleukin 1β (IL-1ß), prostaglandin E2 (PGE2) and substance P (SP) using commercially available human ELISA kits (R&D Systems Inc., Minneapolis, MN, USA). The concentration of total proteins in each sample was assessed with a Pierce BCA Protein Assay Kit (Pierce Biotechnology, Rockford, IL, USA) according to the manufacturer’s instructions. The level of each biomarker was expressed in pg/μg of total protein in the GCF.
Statistical analysis
For data with a nonnormal distribution, Friedman’s two-way analysis was performed for intragroup comparisons among various time-points and the Wilcoxon signed rank test for intergroup comparisons. For data with a normal distribution, repeated measures ANOVA was performed for intragroup comparisons, and paired samples t-tests were used for intergroup comparisons. All statistical analyses were performed using SPSS software version 23.0 (SPSS Inc., Chicago, IL, USA), and the significance level (α) was set at 0.05 for two-tailed tests.
Results
Characteristics of the study participants
Thirty-four patients with controlled chronic periodontitis were referred for assessment of eligibility. Six patients did not meet the inclusion criteria, and one patient refused to participate in the study. Thus, 27 patients (22 female) between 31 and 60 years (mean, 47 years; SD, 8 years) with a total of 198 single-rooted and displaced target teeth were enrolled in this study. The participants were presented with mild to moderate crowding or spacing. The majority underwent non-extraction orthodontic treatment (N = 19, 70.4%). The remainders (N = 8, 29.6%) each had 1 to 3 teeth (12 teeth in total: 3 maxillary incisors, 6 mandibular incisors and 3 maxillary premolars) extracted before treatment, due to crowding and/or poor periodontal prognosis. No adverse events were reported during the study process. A flow chart of the study’s progression is shown in Fig. 2. No significant difference was found between the laser group and the placebo group for any assessment, indicating that the two sides of the mouth were comparable at the baseline (Appendix 2).
Results of subjective pain perception
As shown in Table 1, pain intensity increased from 1 hour after placement of the initial archwire and peaked at 24 hours before it gradually decreased to the baseline level thereafter in both groups (P < 0.01). A significantly higher score was found on the placebo side at both 6-hour (P = 0.03) and 24-hour (P = 0.02) intervals. Patients felt significantly greater maximum pain intensity on the placebo side (P = 0.02) over the 7 days after placement of the initial archwire. The pain subsided 1 day earlier on the laser-irradiated side after the orthodontic treatment began (P = 0.03) (Appendix 3). As a response to the chairside archwire ligation, significantly less pain was reported on the laser side 1 day (P = 0.04) and 1 week after the placement of the 0.014-inch thermal NiTi (P = 0.03) and 1 week after the placement of the 0.016-inch thermal NiTi (P = 0.02) (Table2). The bite force decreased dramatically over the first 3 months of orthodontic treatment (P < 0.01), followed by a trend of recovery (Table 3). Significantly smaller reductions were found on the laser side at both 1-week and 1-month intervals (P = 0.02 at 1 week and P = 0.04 at 1 month).
Results of periodontal indices and supragingival bacteria load
Generally speaking, the periodontal status remained stable throughout the 6 months of orthodontic treatment (Table 4). The placebo group had a greater accumulation of plaque at the gingival margin than the laser group, and statistical significance was shown at the 1-month (P < 0.01) and 3-month follow-up (P = 0.03) visits. In terms of the level of change from the baseline value, the intergroup difference was found to be significant at the 1-month follow-up visit (P = 0.02). The intergroup difference in the amount of increase was detectable at a significant level at the 3-month follow-up visit (P = 0.01). When the measured results of the groups were compared, a higher BOP was found in the placebo group, which was significant at the 1-month follow-up visit (P = 0.03). No significant intragroup or intergroup differences were detected for PPD, GR or CAL at any time-point.
The log10-transformed bacterial loads and proportions of P. gingivalis and T. denticola are displayed in Appendix 4. The bacterial load of T. denticola within both groups increased significantly at the 1-month follow-up visit (P < 0.01). An increased proportion of P. gingivalis for the placebo group in comparison with the baseline (P = 0.02) was seen at the 1-day follow-up visit. No significant intergroup differences were found for the absolute load or proportions of either species.
Results of GCF levels of biomarkers
The levels of PGE2 and SP in the GCF were found to increase sharply during the first day after initial force activation (P = 0.01 for PGE2, P < 0.01for SP) and to decline thereafter (Table 5). The laser group showed significantly less increase in the GCF levels of IL-1β than the placebo group at 1 day (P = 0.01) and 1 month (P = 0.03) after the placement of the initial archwire. Significantly less increase was seen in the GCF levels of PGE2 within the laser group than in the placebo group at the 1-day (P = 0.02) and 1-week (P < 0.01) intervals. The increase in the GCF levels of SP was found to be more well controlled in the laser group at the 1-day interval (P < 0.01).
Discussion
This is the first RCT to investigate the effects of LLLI on the management of orthodontic pain (primary outcome) and periodontal inflammation (secondary outcome) in the orthodontic treatment of periodontally compromised patients. Comprehensive outcome assessment was carried out to test the hypothesis. Objective analyses of GCF biomarkers were included to supplement and validate the subjective assessment of pain perception. In addition to periodontal clinical indices, GCF biomarkers and the supragingival bacteria load were analysed to provide evidence of LLLI’s effect on the modulation of periodontal inflammation. Overall, this study provides promising findings regarding the analgesic and anti-inflammatory effects of LLLI over the 6-month orthodontic treatment. Therefore, the null hypothesis of this study is rejected or at least partially rejected.
Study design and subject characteristics
Orthodontic pain is a highly subjective symptom with wide inter-subject variation (12). Moreover, the response of periodontal tissue to inflammation and mechanical forces has been shown to be affected by various host factors (15, 16). Therefore, a split-mouth design was adopted in this study.
Most of the study participants were female (22, or 81.5%), perhaps because women have greater awareness of dental appearance and dental treatment demand is higher among women (17, 18). About two thirds of the study participants were middle-aged and elderly adults older than 45 years (mean age, 47 years; SD, 8 years), possibly because of the clinical course of periodontitis. All measurements showed no intergroup differences at the baseline, which indicates that the laser and placebo groups on the two sides of the dental arches were generally comparable (Appendix 1).
Orthodontic and low-level laser treatment
Orthodontic treatment plays an adjunctive role in the interdisciplinary treatment of periodontitis, so non-extraction fixed appliance therapy was carried out in most patients. Light force was applied on an approximate number of teeth on both sides of dental arch with small standardised thermal NiTi wires for the first 3 months (10, 12). The 940-nm wavelength was reported to be beneficial for orthodontic pain management, periodontal inflammation control and bone remodelling in both in vitro and in vivo studies (19,20,21). The energy density of each irradiation was confirmed to be 8.6 J/cm2 and 4 J/tooth, which also conformed to the recommended dosage of 0.5 to 10 J/site for orthodontic pain relief and 1 to 10 J/ cm2 for periodontal inflammation control (10, 12). A multiple application regimen was established throughout the treatment process to exert long-term bio-modulatory effects on the periodontal tissue.
Subjective pain assessment
In both experimental groups, the self-reported pain over 7 days followed a similar pattern of change, with the most severe pain appearing at the 24-hour interval, which is consistent with most previous findings (22, 23). It was determined via intergroup comparison that the laser-irradiated side experienced less pain than the placebo side. However, the difference was only significant at the 6-hour and 24-hour intervals and marginally significant at the 3-day interval. It was also reported that LLLI showed an obvious analgesic effect during the first 3 days and not thereafter (24, 25). The natural time course of pain may contribute to the reduction in the differences between the two experimental groups (22). LLLI was also found to reduce the maximum pain intensity over 7 days, which supports the findings of other studies (26, 27). Laser irradiation was found to bring the endpoint of pain forward by 1 day when compared with the placebo side. The same phenomenon was also observed by some scholars who found a longer duration of pain on the placebo side over 7 days (26, 28). LLLI significantly reduced the pain level triggered by chairside archwire ligation during the first month after the placement of 0.014-inch archwire and during the first week after the placement of 0.016-inch archwire (9-week interval). Although nonsteroidal anti-inflammatory drugs (NSAIDs) have shown confirmed analgesic effects on orthodontic pain, their negative effects on periodontal tissue remodelling and tooth movement rate have limited their clinical application (22, 29). In contrast to NSAIDs, which mainly inhibit the cyclooxygenase enzyme system responsible for the production of prostaglandins, LLLI achieves its analgesic effects by attenuating the local inflammatory response, with no adverse effects on periodontal tissues (30).
Many studies have reported biting and chewing as the activities most negatively affected by orthodontic pain (4, 31, 32). Pain serves as a modifying factor that limits the maximum bite force due to reflex mechanisms (4, 33). Therefore, the maximum voluntary bite force of anterior occluding pairs was measured to serve as another indicator of subjective perception of pain. During the first 3 months of treatment, a remarkable and continuous drop occurred in the magnitude of bite force followed by a tendency to recover at the 3-month follow-up visit. Neuromuscular adaptation to mechanical loading on the periodontal tissue may account for this finding (34, 35). When compared with placebo treatment, LLLI was shown to prevent reduction of the bite force, especially during the first month after initial archwire placement. Likewise, a significantly lower level of chewing pain was reported on the laser-treated side than on the placebo side for all 7 days after separator placement (21). Another study suggested that LLLI was the most effective strategy to control pain when chewing, biting and fitting front and back teeth over 7 days and that it produced effects comparable to those of ibuprofen (36). These findings indirectly support the results of our study and reinforce the assumption that LLLI may have an analgesic effect, which is manifested by protecting the biting performance from deteriorating.
Periodontal clinical status and supragingival bacteria load
Based on the 6-month follow-up visit, the periodontal status of periodontally compromised patients who underwent orthodontic tooth movement remained quite stable, with no significant changes in PPD, BOP, CAL or GR and with a slight yet significant increase in PI and GI. This finding is in agreement with the mainstream of current evidence (37,38,39). Despite regular oral hygiene reinforcement and periodontal maintenance, the accumulated supragingival plaque after bracket placement contributed to an increased level of gingival inflammation, although the alteration was not considered clinically significant. This finding was consistent with the detected elevation in the proportion of P. gingivalis over that of total bacteria after the placement of orthodontic appliances within the placebo group and in the absolute count of T. denticola at the 1-month interval for both groups. When the periodontal indices were compared between groups, the laser-irradiated group had more favourable results, with significantly less supragingival plaque and milder gingival inflammation at 1-month and 3-month follow-up visits. LLLI appears as to be a promising adjunctive approach to mechanical debridement in the control of periodontal inflammation and wound healing for patients with chronic periodontitis (10). Although no intergroup difference was found in the quantity of P. gingivalis or T. denticola, it cannot be denied that the lower pain level perceived on the laser-irradiated side favoured the maintenance of oral hygiene during the early stage of tooth movement.
GCF biomarkers associated with pain and inflammation level
Among the various cytokines in the GCF, the variations in IL-1β, PGE2 and SP are the most widely studied and have been identified as having an independent association with orthodontic pain (40). The levels of PGE2 and SP in the GCF peaked at the 24-hour interval in the placebo group, followed by a steady recovery over the following 2 months. This finding is corroborated by studies in adolescents and young adults, in whom an increase in the levels of many inflammation mediators was found to be most pronounced at the 24-hour interval (40). The fluctuation of GCF biomarkers coincided with the change pattern of subjective pain intensity, which suggests that a neuro-immune interaction occurs in the periodontal tissue during orthodontic tooth movement (41). The GCF levels of all three cytokines at various time-points showed less increase in the laser-irradiated group, especially during the first month. Bicakci was among the scholars who investigated GCF pain-related biomarkers in LLLI-aided orthodontic treatment. A significantly lower level of PGE2 was reported in the LLLI-treated group 1 hour and 24 hours after separator placement (13). This study identified the inhibitory effect of LLLI on the increase of three pain-related cytokines in the GCF, which suggests that laser treatment had analgesic effects from the biochemical perspective on periodontal patients who underwent fixed-appliance therapy.
The LLLI-treated side exhibited less plaque accumulation and gingival inflammation than the placebo side during the first month, which coincides with its better control of the upregulation of IL-1β, PGE2 and SP levels in GCF. IL-1β, PGE2 and SP have been found to be critical in gingival inflammation and alveolar bone resorption (42, 43). This favourable effect on inflammation control was also reported in another study, which showed that LLLI could prevent substantial increases in the GCF levels of IL-1β in the short term and improve the periodontal clinical parameters at the intermediate follow-up in lingual orthodontic patients (44). The first immediate effect of LLLI on the reduction of periodontal tissue may be on neurogenic acute inflammation, which is manifested by regulation of pain, vasodilation, chemotactic activity and cytokine release. It may then have a longer-lasting effect on chronic inflammation, as characterised by accelerated tissue repair (collagen synthesis, angiogenesis and growth factor release), so that the host-bacteria-force equilibrium can be maintained and tooth movement can occur safely and effectively (30, 45).
Limitations of this study and future directions for study
The characteristics of our study subjects hindered investigation of gender and age differences in the treatment response to orthodontic tooth movement in conjunction with LLLI. However, this study sample may to some extent represent the general patient population who seek orthodontic treatment after having controlled chronic periodontitis. Although it is impossible to analyse the existence and extent of any potential carry-across effect, the results of studies with a split-mouth design appear to be comparable with those generated by a parallel design (12). This also gives rise to the strict eligibility criteria in participant recruitment, which results in this study’s limited yet still adequate sample size. Studies with a larger sample size or multiple centres can be carried out to validate the findings of this study and the possible effects of gender and age. The results of this study are based on short- and intermediate-term observations. Therefore, long-term follow-up is anticipated to provide further verification of LLLI’s merits in anti-inflammatory biostimulation. Besides, the subjective assessment of pain by VAS is another limitation of this study, which explains why were pain-related cytokines in GCF also substantiated for LLLI’s effect on pain relief.
References
Re S, Corrente G, Abundo R, Cardaropoli D (2000) Orthodontic treatment in periodontally compromised patients: 12-year report. The International journal of periodontics & restorative dentistry20:31–39
Jin LJHKDJ. Periodontic-orthodontic interactions—rationale, sequence and clinical implications. 2007
Gkantidis N, Christou P, NJJoor T (2010) The orthodontic–periodontic interrelationship in integrated treatment challenges: a systematic review. 37:377–390
Goldreich H, Gazit E, Lieberman MA, JDJAJoO R (1994) The effect of pain from orthodontic arch wire adjustment on masseter muscle electromyographic activity. Orthopedics D 106:365–370
Liu Z, McGrath C, UJAJoO H (2011) Changes in oral health-related quality of life during fixed orthodontic appliance therapy: an 18-month prospective longitudinal study. Orthopedics D 139:214–219
Rabie ABM, Yang Y, Wong RW. Treatment of periodontally compromised patients. , 2010
Kert J, Rose L. Clinical laser therapy : low level laser therapy [Veksoe, Denmark?]: Scandinavian Medical Laser Technology, 1989
Aoki A, Mizutani K, Schwarz F et al (2015) Periodontal and peri-implant wound healing following laser therapy. Periodontology 200068:217–269
Borzabadi-Farahani A, Cronshaw M (2017) Lasers in orthodontics. Lasers in Dentistry—Current Concepts: Springer:247–271
Ren C, McGrath C, Jin L, Zhang C, Yang Y (2017) The effectiveness of low-level laser therapy as an adjunct to non-surgical periodontal treatment: a meta-analysis. J Periodontal Res52:8–20
Ge MK, He WL, Chen J et al (2015) Efficacy of low-level laser therapy for accelerating tooth movement during orthodontic treatment: a systematic review and meta-analysis. Lasers Med Sci30:1609–1618
Ren C, McGrath C, Yang Y (2015) The effectiveness of low-level diode laser therapy on orthodontic pain management: a systematic review and meta-analysis. Lasers Med Sci30:1881–1893
Bicakci AA, Kocoglu-Altan B, Toker H, Mutaf I, Sumer Z (2012) Efficiency of low-level laser therapy in reducing pain induced by orthodontic forces. Photomed Laser Surg30:460–465
Dominguez A, Velasquez SA (2013) Effect of low-level laser therapy on pain following activation of orthodontic final archwires: a randomized controlled clinical trial. Photomed Laser Surg31:36–40
DJJop G (2008) Cytokines that promote periodontal tissue destruction. 79:1585–1591
Antoun JS, Mei L, Gibbs K, MJP F (2017) Effect of orthodontic treatment on the periodontal tissues. 74:140–157
Hassel A, Wegener I, Rolko C, IJIdj N (2008) Self-rating of satisfaction with dental appearance in an elderly German population. 58:98–102
Tin-Oo MM, Saddki N, NJBoh H (2011) Factors influencing patient satisfaction with dental appearance and treatments they desire to improve aesthetics. 11:6
Lui J, Corbet E, LJJopr J (2011) Combined photodynamic and low-level laser therapies as an adjunct to nonsurgical treatment of chronic periodontitis. 46:89–96
Hakki SS, SBJLims B (2012) Effects of different setting of diode laser on the mRNA expression of growth factors and type I collagen of human gingival fibroblasts. 27:325–331
Qamruddin I, Alam MK, Fida M, AGJAJoO K (2016) Effect of a single dose of low-level laser therapy on spontaneous and chewing pain caused by elastomeric separators. Orthopedics D 149:62–66
Krishnan V (2007) Orthodontic pain: from causes to management--a review. Eur J Orthod29:170–179
Polat Ö (2007) Pain and discomfort after orthodontic appointments. Seminars in Orthodontics: Elsevier:292–300
Eslamian L, Borzabadi-Farahani A, Hassanzadeh-Azhiri A, Badiee MR, RJLims F (2014) The effect of 810-nm low-level laser therapy on pain caused by orthodontic elastomeric separators. 29:559–564
Fujiyama K, Deguchi T, Murakami T, Fujii A, Kushima K, TJTAO T-Y (2008) Clinical effect of CO2 laser in reducing pain in orthodontics. 78:299–303
Tortamano A, Lenzi DC, ACSS H et al (2009) Low-level laser therapy for pain caused by placement of the first orthodontic archwire: a randomized clinical trial. 136:662–667
Domínguez A, SAJP V (2013) Effect of low-level laser therapy on pain following activation of orthodontic final archwires: a randomized controlled clinical trial. surgery l 31:36–40
Marini I, Bartolucci ML, Bortolotti F, Innocenti G, Gatto MR, GAJLims B (2015) The effect of diode superpulsed low-level laser therapy on experimental orthodontic pain caused by elastomeric separators: a randomized controlled clinical trial. 30:35–41
Long H, Wang Y, Jian F, Liao L-N, Yang X (2016) Lai W-LJIjoos. Current advances in orthodontic pain 8:67
Bjordal JM, Johnson MI, Iversen V, Aimbire F, Lopes-Martins RABJP (2006) Low-level laser therapy in acute pain: a systematic review of possible mechanisms of action and clinical effects in randomized placebo-controlled trials. Therapy L 24:158–168
Michelotti A, Farella M, RJTEJoO M (1999) Sensory and motor changes of the human jaw muscles during induced orthodontic pain. 21:397–404
Farzanegan F, Zebarjad SM, Alizadeh S, FJAJoO A (2012) Pain reduction after initial archwire placement in orthodontic patients: a randomized clinical trial. Orthopedics D 141:169–173
Bakke M. Bite force and occlusion Seminars in orthodontics: Elsevier, 2006: 120-126.
Winocur E, Davidov I, Gazit E, Brosh T, ADJTAO V (2007) Centric slide, bite force and muscle tenderness changes over 6 months following fixed orthodontic treatment. 77:254–259
Alomari SA, ESJAoj AA (2012) Occlusal bite force changes during 6 months of orthodontic treatment with fixed appliances. 28:–197
Bayani S, Rostami S, Ahrari F, Saeedipouya I (2016) A randomized clinical trial comparing the efficacy of bite wafer and low level laser therapy in reducing pain following initial arch wire placement. Laser therapy25:121–129
Boyd RL, Leggott PJ, Quinn RS, Eakle WS, Chambers D (1989) Periodontal implications of orthodontic treatment in adults with reduced or normal periodontal tissues versus those of adolescents. American journal of orthodontics and dentofacial orthopedics: official publication of the American Association of Orthodontists, its constituent societies, and the American Board of Orthodontics96:191–198
Paolantonio M, Festa F, di Placido G, D’Attilio M, Catamo G, Piccolomini R (1999) Site-specific subgingival colonization by Actinobacillus actinomycetemcomitans in orthodontic patients. American journal of orthodontics and dentofacial orthopedics: official publication of the American Association of Orthodontists, its constituent societies, and the American Board of Orthodontics115:423–428
Naranjo AA, Trivino ML, Jaramillo A, Betancourth M, Botero JE (2006) Changes in the subgingival microbiota and periodontal parameters before and 3 months after bracket placement. American journal of orthodontics and dentofacial orthopedics: official publication of the American Association of Orthodontists, its constituent societies, and the American Board of Orthodontics130:275.e217–275.e222
Ren Y, Vissink A (2008) Cytokines in crevicular fluid and orthodontic tooth movement. Eur J Oral Sci116:89–97
Krishnan V, ZJJodr D (2009) On a path to unfolding the biological mechanisms of orthodontic tooth movement. 88:597–608
Lee HJ, Kang IK, Chung CP, Choi SM (1995) The subgingival microflora and gingival crevicular fluid cytokines in refractory periodontitis. J Clin Periodontol22:885–890
Yamaguchi M, Yoshii M, Kasai K (2006) Relationship between substance P and interleukin-1beta in gingival crevicular fluid during orthodontic tooth movement in adults. Eur J Orthod28:241–246
Abellan R, Gomez C, Oteo MD, Scuzzo G, Palma JC (2016) Short- and medium-term effects of low-level laser therapy on periodontal status in lingual orthodontic patients. Photomed Laser Surg34:284–290
Kasai K, Yuching Chou M, Yamaguchi M (2015) Molecular effects of low-energy laser irradiation during orthodontic tooth movement. Semin Orthod21:203–209
Acknowledgments
The authors would like to acknowledge Ms. Amy Yim, Ms. Cammie So and Ms. Jessica Fu for their contributions in patient management and study coordination. Our appreciation goes to Ms. Samantha Li for assistance in statistical analysis and Ms. Cindy Yeung for help in questionnaire development and delivering. Many thanks to Ms. Becky Cheung, Ms. Joyce Yau and Mr. Alan Wong for their technical support in biochemical analysis of samples. Mr. Edmund Choi’s technical support in the laser application is appreciated.
Funding
This study was supported by the Health and Medical Research Fund (HMRF 01121056) from Food and Health Bureau, the government of the Hong Kong SAR.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ren, C., McGrath, C., Gu, M. et al. Low-level laser-aided orthodontic treatment of periodontally compromised patients: a randomised controlled trial. Lasers Med Sci 35, 729–739 (2020). https://doi.org/10.1007/s10103-019-02923-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-019-02923-0